3.1. Characterization of the Biological Activity of Endophytic Bacteria
For the application of microorganisms both to enhance plant growth and to protect them from adverse factors, a crucial step is the selection of strains possessing a set of beneficial properties. In the first stage of the research, the agronomically valuable properties of five endophytic bacterial strains were investigated (
Table 1).
One of the well-known mechanisms for improving and regulating plant growth by microorganisms is their ability to synthesize various phytohormones. The stimulation of plant growth resulting from the application of microorganisms is predominantly associated with their ability to synthesize auxins, primarily IAA [
29]. All examined bacteria demonstrated the ability to produce IAA (
Table 1), except for the
Bacillus simplex B9 strain. The highest concentration of IAA was found in the
Pseudomonas putida D7 strain (
Table 1). The amount of produced IAA varied between 45.2 and 69.2 μg mL
−1 depending on the strain, which is similar to or significantly higher than that observed in other endophytic bacterial strains [
30,
31].
The next criterion for assessing the biological activity of the strains was the evaluation of their resistance to adverse environmental factors.
Among the adverse factors of biotic nature, phytopathogenic microflora plays a key role. Infections of agricultural crops caused by pathogenic fungi are among the most widespread and harmful, as they not only reduce the quantity of the harvest but also significantly degrade its quality due to the accumulation of mycotoxins [
32]. One of the positive effects of bacteria on crops is their ability to protect plants from phytopathogens through direct and indirect mechanisms [
33].
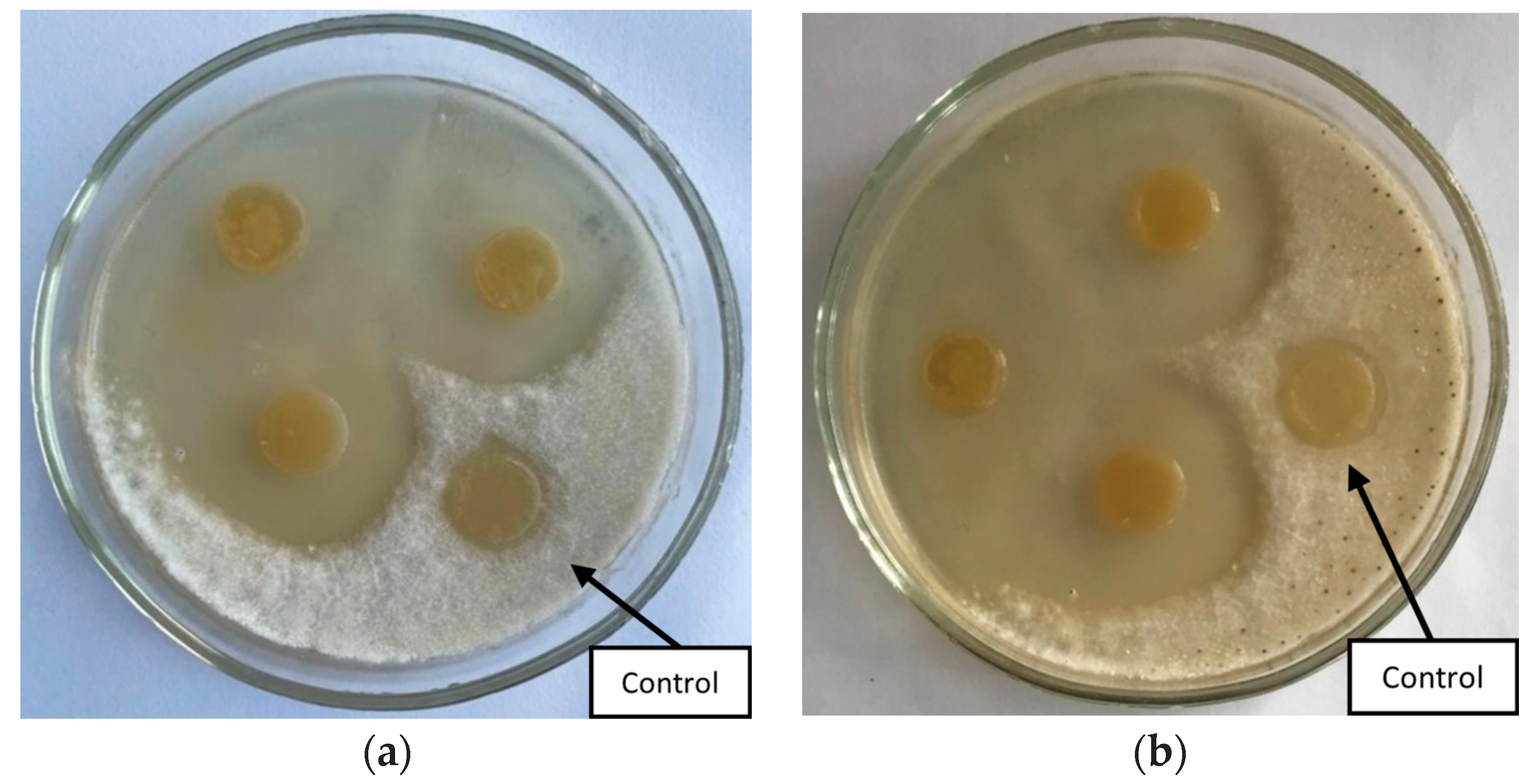
The study of the antagonistic activity of bacterial strains against
Fusarium solani and
Fusarium oxysporum showed that three out of five strains inhibit the growth of phytopathogens (
Figure 1). The zones of growth suppression ranged from 1.8 to 3.0 cm (
Table 1).
Salinization of soils is one of the most crucial abiotic stress factors that negatively impact plant privity [
34]. The application of salt-tolerant growth-promoting bacteria may contribute to stress alleviation and enhance the resilience of crops grown in saline soils [
34].
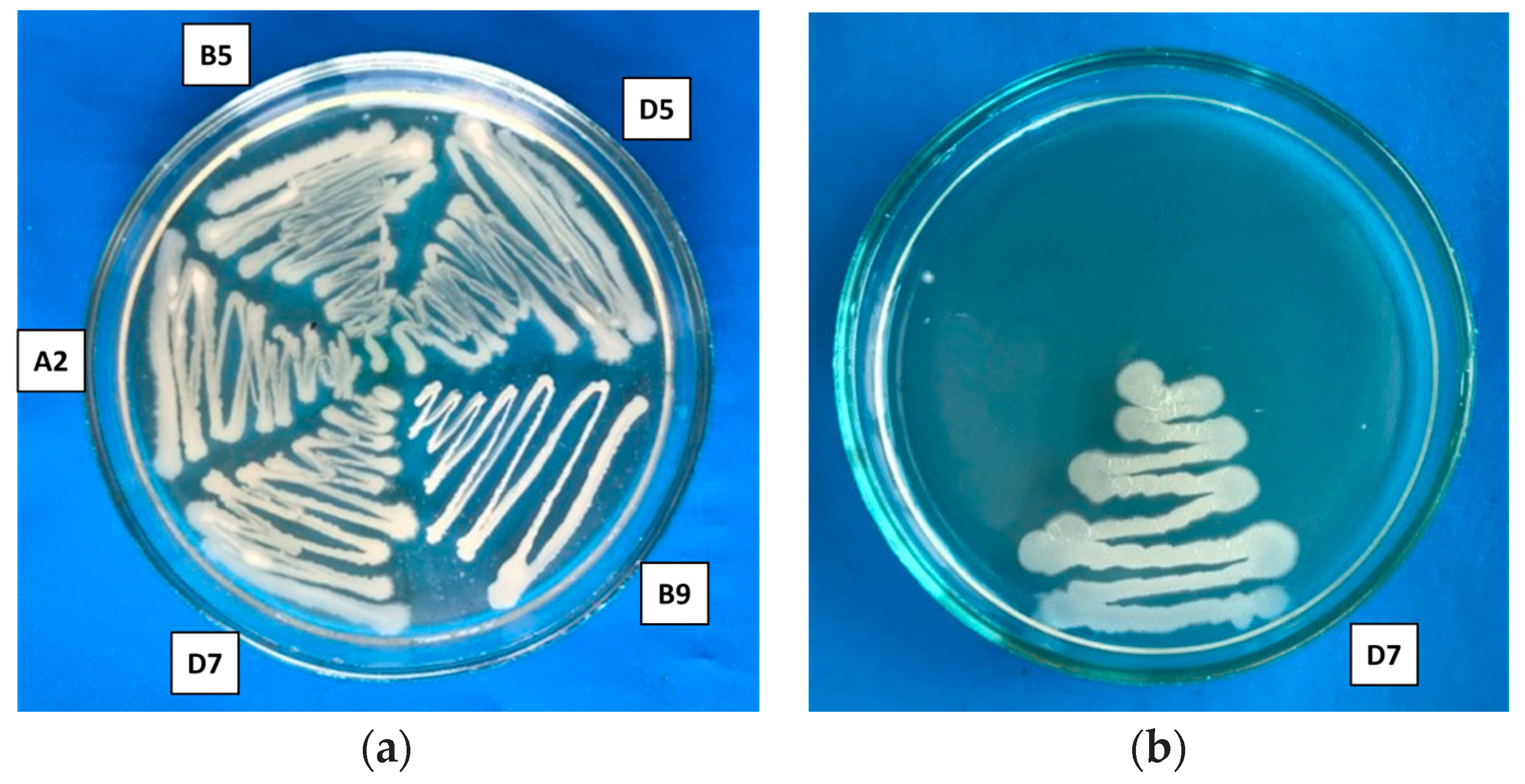
In the study of halotolerance, it was shown that all strains were resistant to a salt concentration of 5%, and one strain,
Pseudomonas putida D7, demonstrated the ability to grow in a medium with 15% NaCl (
Table 1,
Figure 2). According to the classification, the investigated strains are moderately halophilic, exhibiting optimal growth at NaCl concentrations ranging from 3% to 15% (~0.5–2.7 M). Halophilic bacteria have several advantages compared to other microorganisms, as they possess high metabolic activity, allowing them to grow in extreme conditions and produce a variety of valuable biologically active compounds, including those with antimicrobial properties [
35].
PHA is a class of polyesters of various hydroxyalkanoic acids, which are synthesized by many Gram-positive and Gram-negative bacteria and accumulate intracellularly [
33]. In the present study, the strains
Ps. flavescens D5 and
B. aerophillus A2 demonstrated the ability to produce PHA (
Table 1).
3.3. Biosynthesis of Microbial Exopolysaccharides and Their Rheological Properties
In addition to plant-beneficial microorganisms, such ingredients of seed coating as binders that help to release a suitable amount of plant-beneficial microorganisms in physiologic conditions and ensure the adherence and cohesion of the material on the seed surface and keep the ingredients active are used [
37,
39,
40]. The microbial polymer solution should be water-soluble with a low viscosity for complete atomization of the liquid onto seeds [
40].
Earlier, we isolated strains
Aureobasidium pullulans C7 [
41] and
Bacillus thuringiensis C8, which showed the ability to biosynthesize exopolysaccharide (EPS). The
A. pullulans C7 strain synthesized 12.53 ± 0.48 g L
−1 exoglycan on the 4th day of fermentation, and the yield coefficient for biomass was 349.02% (
Table 3). This indicates that in this medium the substrate is utilized to a greater extent for the formation of EPS than for the formation of cell mass. The amount of polysaccharide accumulated by the studied strain is comparable with the data of other researchers [
42,
43].
The strain
B. thuringiensis C8 produced 3.97 g L
−1 of exoglycan (
Table 3). The yield coefficient for bacterial biomass indicates the potential of this strain as a producer of EPS. The ability of strains of the genus
Bacillus, including
B. thuringiensis, to produce EPS is confirmed in the works of other researchers [
44,
45].
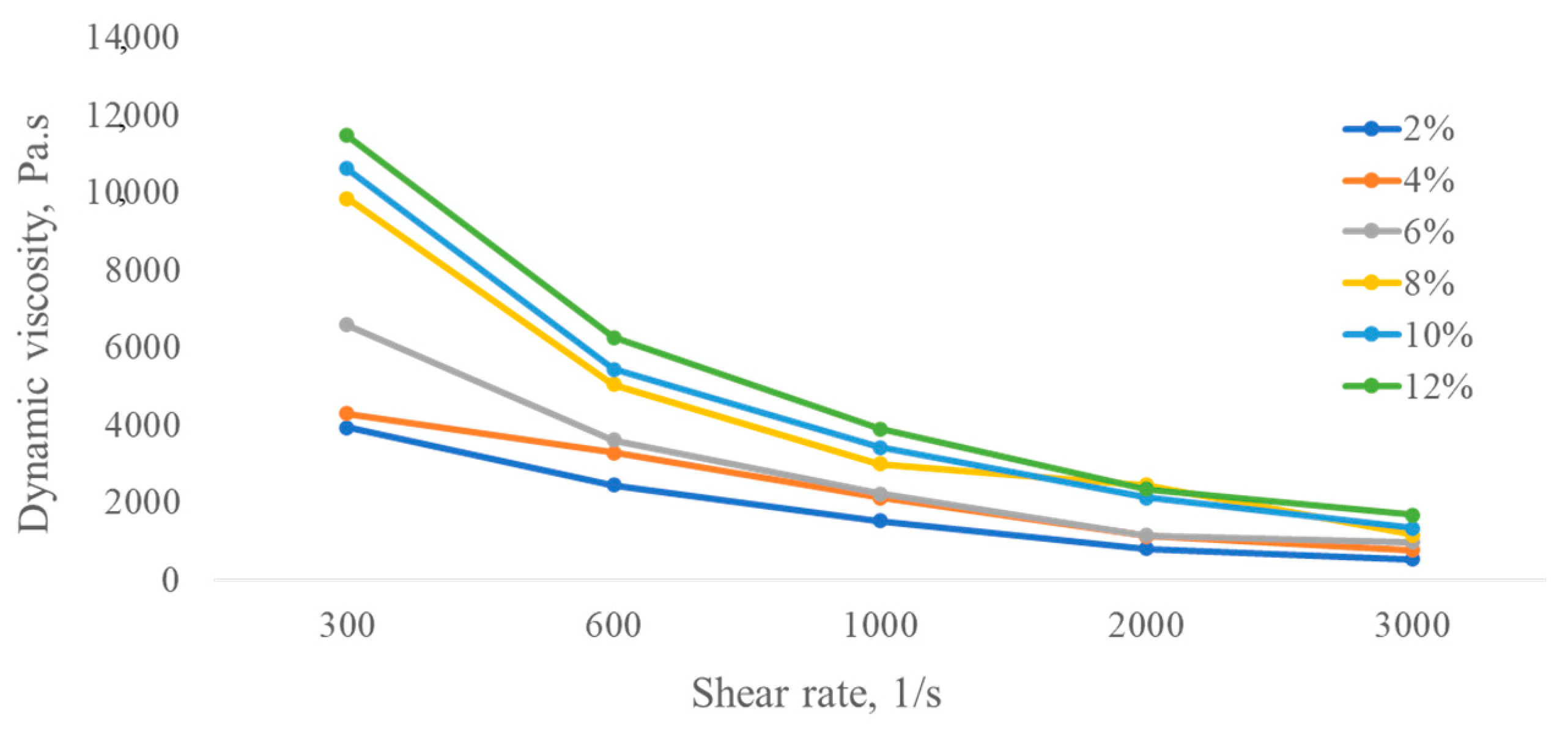
Further, measurements of dynamic viscosity were made for polymer solutions obtained by cultivation of A. pullulans C7 and B.thuringiensis C8.
The dynamic viscosity of each concentration solution produced by
B. thuringiensis C8 obviously decreased with the increase in shear rate (
Figure 3), showing a shear-thinning behavior, which means that this kind of polysaccharide solution belongs to non-Newtonian fluid (or pseudoplastic flow behavior). Solutions of polymers in water and at the same concentrations can sometimes have oppositional behaviors, i.e., Newtonian or non-Newtonian fluids, depending on their structures, molecular weights, and polymer microbial producers [
46,
47].
It was also noted that the viscosity increased with increasing polymer solution concentration; however, at lower concentrations, the rheological measurements became erratic. The shear-thinning phenomenon could be due to the rate of formation of new entanglements lower than the externally imposed disruption rate with an increase in shear rate. Another distinguishing feature is that the microbial solution showed comparatively high viscosity rates at all dilute concentrations.
The measurement of the dynamic viscosity of the polymer solution obtained by the cultivation of
A. pullulans C7 showed the dependence on dynamic viscosity at the shear rate at different concentrations ranging from 2% to 12% (
w/
v) at 25 °C (
Figure 4). The shear rate increased with increasing polymer concentration, thereby demonstrating that viscosity strongly depends on concentration.
At lower concentrations (<8.0%,
w/
v), viscosity virtually remained unchanged with increasing shear rate, thus suggesting that the pullulan aqueous solution exhibits Newtonian flow behavior. It is known that Newtonian fluid viscosity is constant no matter the shear rate or applied shear stress experienced by the fluid. However, with increasing concentration to 8%
w/
v, the flow behavior was changed to pseudoplastic. Such flow behavior of pullulan can happen because of the separation of exopolysaccharides from each other or the alignment of them with the shear field and thereby a decrease in viscosity up to an approximately constant value [
48]. Also, it is known that the viscosity is dependent on the structure and concentration of the polymer, its molecular weight and distribution, the conformation of macromolecules in the solution and its interaction with solvents, the type of intermolecular and intramolecular aggregation, and the flexibility of the chains with temperature. A similar change in flow behavior with increasing concentration was reported for pullulan and other polysaccharides [
49,
50].
The data obtained allow us to suggest the microbial polymer, pullulan, produced by A. pullulans C7 as a potential seed coating binder due to rheological characteristics. It is already known as an excellent film former and is functional for a variety of applications, including for use as an adhesive, binder, and thickener to modify or maintain the texture of food.
It has also been reported that pullulan has considerable mechanical strength and other functional properties such as adhesiveness, film and fiber formability, and enzymatically mediated degradability [
51]. High flexibility and a lack of crystallinity provide pullulan with the capacity to form thin layers, electrospun nanofibers, nanoparticles, flexible coatings, stand-alone films, and three-dimensional objects [
51,
52]. Due to its peculiar characteristics, pullulan is extensively used in different sectors, the three main realms of application pertaining to the pharmaceutical, biomedical, and food fields.
3.4. The Use of Various Antifungal Formulations for Seed Treatments in Pot Experiments
Seed coating is a method that involves applying exogenous materials to the surface of seeds to enhance their properties and/or deliver active components (such as plant growth regulators, nutrients, and microbial inoculants). This process can protect seeds from phytopathogens, increase germination rates, improve plant resistance to stress factors, and enhance overall plant growth [
37,
52,
53,
54].
In the next stage of the research, various antifungal formulations for seed coating were developed, and their impact on barley growth under phytopathogenic conditions was assessed (
Figure 5).
As active components, a microbial inoculant consisting of a suspension of four compatible strains was used:
Ps. flavescens D5,
B. aerophilus A2,
S. proteamaculans B5, and
Ps. putida D7. As polymer components, PHA produced by the strain
Ps. flavescens D5, and pullulan, produced by the yeast strain
A. pullulans C7, were used. PHA was included in the mixture due to its antifungal properties, as previously identified in earlier studies [
15,
16].
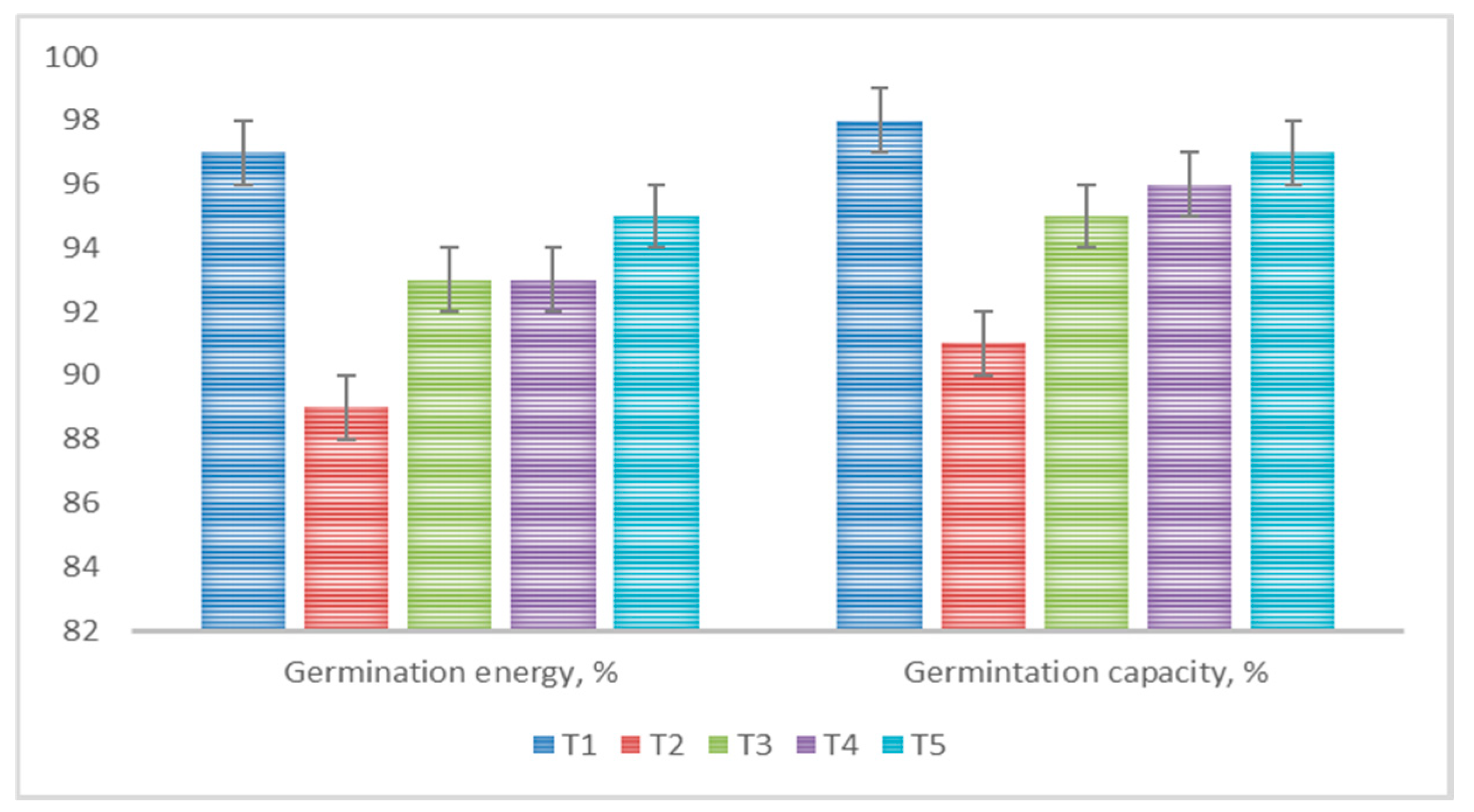
Uniform seed emergence and early crop development are crucial aspects for achieving high crop yields. Seed coating is an effective method that improves seed-sowing qualities and activates the internal resources of the seed material [
52]. In the conducted research, pre-sowing seed treatment, in most cases, enhanced their germination energy and germination capacity. The greatest effect was observed when applying a bacterial suspension in combination with a polymer mixture, where germination energy and germination capacity reached 95% and 97%, respectively (
Figure 6).
The pre-sowing treatment of seeds demonstrated a pronounced growth-stimulating effect on barley plants, as evidenced by a significant (
p < 0.05) increase in morphometric indicators (
Table 4).
The barley’s response varied depending on the type of treatment. Root length is a crucial morphometric indicator as roots are in contact with soil and soil microflora, absorbing water with mineral compounds. The greatest root elongation was observed in variant T5 with the application of a bacterial suspension and a polymer mixture (1.6 times), followed by treatment T3, where root elongation was noted at 1.5 times. Stem length is also a significant characteristic when assessing the plant’s response to different pre-sowing seed treatments. Treated variants showed an increase in stem length by 20–53%. The greatest increase in stem length was observed in variants T3 and T5 (
Figure 7). It is shown that the stem mass of treated plants was more than 1.5–1.8 times greater, and root mass was 1.1–1.6 times greater compared to the untreated control (
Table 4).
In the conducted research, the observed stimulating effect on the growth parameters of barley can be attributed to several reasons. One of the mechanisms of the positive influence on plants is the ability of the strains included in the composition to produce the phytohormone IAA, which regulates cell division and elongation, their proliferation and differentiation, as well as the development of vascular tissues and apical dominance [
55]. Another mechanism for improving morphometric plant parameters under conditions of phytopathogenic stress is the biocontrol properties of strains and the protective role of biopolymers.
The state of the photosynthetic apparatus is an indicator of the physiological condition of plants. One of the primary characteristics of photosynthetic activity is the content of chlorophyll pigments [
56]. In previous studies, fluorescence visualization analysis of chlorophyll was applied to assess the condition of the plant photosynthetic system under the influence of biotic [
56,
57,
58] and abiotic [
59,
60,
61] stress. High chlorophyll content may indicate potentially high agricultural productivity [
56].
In the present study, under conditions of biotic stress induced by the phytopathogen
F. oxysporum, the total chlorophyll content in barley leaves decreased by 2.7 times compared to the indicator for plants grown under normal conditions, reaching 1.03 ± 0.03 mg g
−1 (
Table 5). This likely indicates changes in the pigment–protein complexes of light-harvesting antennae and reaction centers of photosystems. Seed treatment had a positive effect on the photosynthetic activity of barley under phytopathogenic stress. This positive effect was to increase the content of chlorophyll
a in leaves by 1.4–2.1 times, chlorophyll
b by 2–2.4 times, and the total content of chlorophyll (a + b) by 1.6–2.2 times. The maximum effect was achieved in variant T5 with the application of a bacterial suspension and a polymer mixture (
Table 5). The observed differences in pigment content may be associated with the production of certain compounds by the studied bacteria, influencing the biosynthesis and/or degradation processes of chlorophylls, as well as creating more favorable growth conditions for plants under stress.
It is known that stress factors lead to a disruption in the balance between the generation of reactive oxygen species (ROS) and their neutralization. Among the essential mechanisms of plant tolerance mediated by bacteria is the involvement of these microorganisms in detoxifying ROS through the modulation of the natural antioxidant defense systems of plants—both non-enzymatic (proline, ascorbic acid, glutathione, cysteine, flavonoids, carotenoids, and tocopherol) and enzymatic (superoxide dismutase, peroxidase, catalase, ascorbate peroxidase, guaiacol peroxidase, and glutathione reductase), all components of which are in complex functional interaction [
62,
63].
The increase in proline content is one of the characteristic responses of plants to various types of stress, including biotic stress, providing the first stage of plant adaptation. Proline serves multiple functions, including the regulation of cytosolic acidity, minimization of lipid peroxidation by scavenging free radicals, and stabilization of subcellular components and structures (proteins and membranes) [
64]. A higher level of proline in barley leaves was observed when plants were grown in soil with an elevated infectious background compared to untreated plants in sterile soil (
Table 5). In the untreated variant under phytopathogenic stress, the proline concentration was 1.7 mg g
−1, exceeding this indicator in plants grown in favorable conditions by 1.8 times. In treated plants, the proline content was lower. The most noticeable decrease in proline was observed in the variant with simultaneous seed coating in a bacterial suspension and a polymer mixture (
Table 5). The obtained results indicate a reduction in the stress experienced by plants due to the pre-sowing seed treatment. Similar to our findings, a reduction in proline levels in various plant species under the influence of microbial treatment has been demonstrated in several studies [
65,
66].
In the conducted studies, an increase in the activity of antioxidant enzymes was observed when untreated plants were grown under conditions of phytopathogenic stress compared to plants grown in stress-free conditions (
Table 6). The obtained data indicate that in response to the action of stress factors, there is an activation of the plant’s defense system.
The pre-sowing seed treatment (T3–T5) led to an increase in catalase activity by 1.3–1.5 times under stress conditions. For ascorbate peroxidase and guaiacol peroxidase, an increase in enzyme activity was observed under phytopathogenic stress in the seed treatment with the bacterial suspension and polymer mixture (T5)—by 2.4 times and 2.7 times, respectively (
Table 6). Similarly to the obtained data, previous studies have reported an increase in the activity of antioxidant enzymes in plants when inoculated with bacteria as one of the defense mechanisms of plants when grown under stressful conditions [
67,
68,
69].
Thus, it was shown that when seeds were treated with the T5 composition, plant growth parameters (weight and length of roots) significantly increased compared to the T3 variant with a bacterial suspension. In addition, the use of the T5 composition contributed more to the attenuation of plant stress caused by phytopathogens compared to the use of microorganisms only (
Table 5 and
Table 6). This indicates that the addition of biopolymers to formulations for seed treatments enhances microbe-induced plant tolerance to phytopathogens.