3.1. Microbial Survival Rate in Compost
The method of counting we used can obtain approximate data. This is particularly relevant for bacteria. For micromycetes, it is easier to obtain a more accurate result using this technique due to the visibility. In this work, it was sufficient to know that, after introducing a concrete microorganism, there was generally no titer decrease. However, some confirmation that this occurs due to the survival of the introduced microorganisms was shown by using molecular biological methods on B. amyloliquefaciens example.
The initial titers of
B. subtilis,
B. amyloliquefaciens, and
P. aeruginosa introduced to the compost were 4.5 × 10
8, 3.5 × 10
8, and 6.0 × 10
8 CFU g
−1 (mL
−1), respectively. After 14 days of incubating the C7 variants at 28 °C, the bacterial counts were 1.6 × 10
9, 1.2 × 10
10 and 8.0 × 10
9 CFU g
−1 (mL
−1) for those with introduced
B. subtilis (C7B1),
B. amyloliquefaciens (C7B2), and
P. aeruginosa (C7P), respectively. We found that after 14 days of incubating C36 variants at 28 °C, the bacterial titer decrease was not significant: 1.7 × 10
7, 1.8 × 10
7 and 2.1 × 10
8 CFU g
−1 for variants with
B. subtilis (C36B1),
B. amyloliquefaciens (C36B2), and
P. aeruginosa (C36P), respectively (
Figure S1).
A. corrugatus showed a slight decrease in the titer after incubation for 14 days at 28 °C, from 5.5 × 10
5 to 3.0 × 10
4 CFU g
−1 and 1.0 × 10
4 CFU ml
−1 for C36A and C7A, respectively (
Figure S2). Therefore, all inoculated microorganisms survived in composts with 36% dry matter content. In the C7 compost,
A. corrugatus also survived, and meanwhile, the bacterial titer increased. The results are shown in
Table 3.
The survival rate of the
B. amyloliquefaciens introduced to the compost was evaluated via molecular biology methods using qPCR (
Table 4).
The total number of prokaryotes in the control variants (without introducing microorganisms) decreased after 14 days of incubation; in the case of variant C36L14, this was more intensive than for C7L14: three orders of magnitude vs. one order of magnitude (
Table 4), respectively. This shares a common trend with the results obtained using classical microbiological analysis methods (
Table 3). Adding LB nutrient media likely had a stimulating effect on the autochthonous microbiota of these composts, after which, competition for nutrients increased. At the same time, the variants with the compost suspension (C7) initially contained more easily available soluble compounds than the variants with solid compost (C36). Thus, in the latter case, the competition between microorganisms was more intense, which caused a greater decrease in the number of prokaryotes.
Because of the introduction, the number of microorganisms in all types of compost generally did not decrease after 14 days of incubation, which agrees with the results of microbial abundance analysis based on the seeding on nutrient media.
The results of the analysis carried out with primers specific to
B. amyloliquefaciens show that the microorganism survived after introducing to both 36% and 7% dry matter composts. At the same time, in both the C36C14 and C7C14 variants, a drop in the number of holA gene copies by three orders of magnitude was observed after 14 days of incubation (
Table 4). The differences in the results obtained using molecular biological and classical microbiological methods are because the former were more specific to a concrete microorganism, while the latter were characterized by some overestimation, which was influenced by the diversity of the compost microbiota, in particular, initially including the autochthonous species
B. amyloliquefaciens.
3.3. Changes in the Suppressive Capacity of Composts after Introducing Antagonistic Microorganisms
Among the three phytopathogenic organisms, the most expressive suppressive activity in the compost was against
C. rosea. The best variant was the compost with 36% dry matter and an introduced antagonist composition (C36C), with an MGI value of 91.7% (
Figure 2a,
Table 7). We assume that the fungus
A. corrugatus made a significant contribution to the suppression of
C. rosea because its individual introduction was also highly effective in C36A (MGI value, 91.4%), which was not significantly different from variant C36C (
p > 0.05).
Among the bacterial agents, B. amyloliquefaciens is of interest, and its introduction was effective in both C36 and C7 (MGIs of 89.5 and 90.5%, respectively). In C36, these bacteria have slightly less influence than the best two variants described above; based on our statistical analysis, it was in the intermediate group between the lowest and highest results in all ranges of the C7 and C36 variants. C7 was even slightly more effective than variant C7C with the introduction of a microbial composition and it statistically differed from all variants with individual culture inoculants (p < 0.05).
B. subtilis and P. aeruginosa showed no similar effects. The MGIs for C36B1 and C7B1 were a few percentages lower than controls C36W and C7W, respectively, but were not significant (p > 0.05, one group with controls). There was a slight increase for C36P (87.1% inhibition), while C7P had a smaller effect than the control (53.3%). However, as with B. subtilis variants, in terms of statistics, it was in one group of results with the controls.
The compost without introducing of microorganisms was effective against this phytopathogen by itself, and the addition of LB nutrient medium had no influence on the suppressive potential (no significant difference compared with the control, p > 0.05) while diluting the compost (suspension variants, C7) caused a significant reduction (p < 0.05) in the inhibition efficiency (MGIs for C36W, C36L, C7W, and C7L: 86.3, 86.0, 58.4, and 54.4%, respectively).
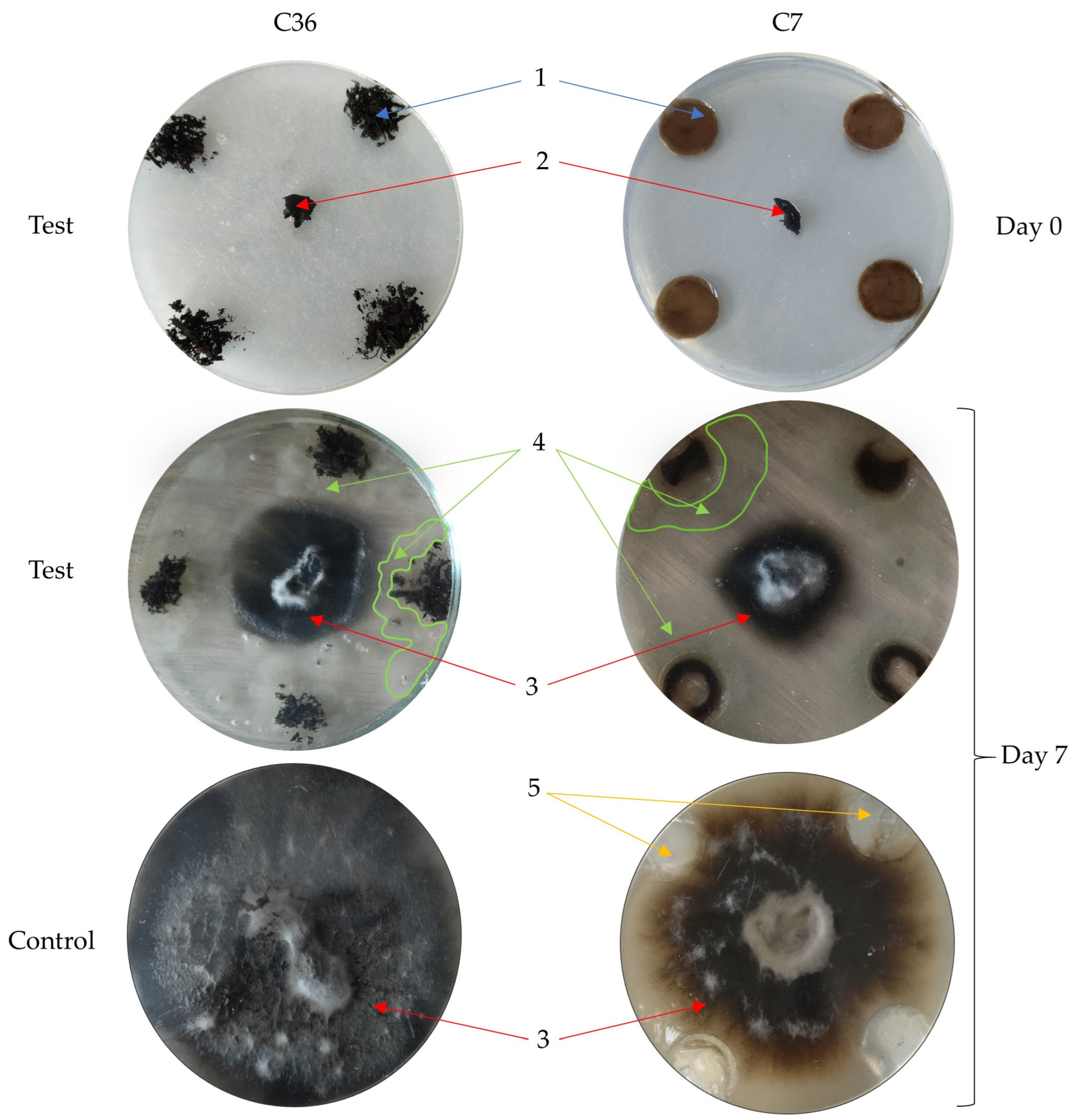
Thus,
C. rosea supersession likely depends largely on the antagonistic activity of specific compost fungal autochthonous species and their abundance in the environment. Given the results in
Figure 2a (4) and
Table 7, we can see the role of fungus in inhibiting
C. rosea mycelial growth.
B. amyloliquefaciens is also an effective biocontrol agent.
Antagonist inoculation increased the suppressive potential of the initial composts best (i.e., compared with the control) against
P. solitum, despite its generally low suppression efficiency compared with the other two phytopathogens (
Table 7). Among the C36 variants, the microbial composition (MGI, 84.0%; control MGI, 11.1%) and
B. subtilis (83.4%) were the best inoculants (statistically significant differences compared with other C36 variants,
p < 0.05, and were included in the same group of results). The fungal contribution was also recorded (C36A—MGI, 77.2%, a statistically close group of results with the best two variants). Treatment with
P. aeruginosa provided suppression of fungal mycelial growth by 59.0%.
Using LB nutrient medium instead of water in the C7L variant did not enhance the suppression of the phytopathogen (the MGIs were at the same levels for C7W and C7L, p > 0.05). However, the LB components in C36 had some impact in terms of suppressive capacity (a significant difference between the C36W and C36L variants, p < 0.05). This may be directly or indirectly due to stimulating the autochthonous microbiota of the compost and specific changes in synthesis of some metabolites effective against P. solitum. Given the values showing the influence of individual culture introductions, B. subtilis actively inhibits the mycelium growth of P. solitum, as described above. The nutrient medium components (when LB was introduced to the C36L variant instead of water) likely stimulated these autochthonous bacteria, as they are often dominant in compost communities. Thus, this is an effective way to repress this fungus.
The best mycelial growth inhibition (MGI 89.9%) was obtained by introducing a microbial composition to compost suspension C7C (significantly significant differences compared with all ranges of the C7 and C36 variants,
p < 0.05, but statistically close to the results for the C36C and C36B1;
Figure 2b).
Individually introducing B. subtilis and P. aeruginosa cultures to the compost suspension was ineffective against P. solitum (MGIs for C7B1 and C7P, 0.0%), and there was no statistically significant difference compared with controls C7W and C7L (p > 0.05). Using B. amyloliquefaciens (C7B2) and A. corrugatus (C7A) caused the suppression of the phytopathogen by 58.3% and 67.1%, respectively, and significantly differed from all C7 variants (p < 0.05).
Generalizing these results, P. solitum is sufficiently resistant to the influence of compost, and its mycelial growth is more difficult to inhibit, its reduction mostly depends on the presence and concentration of antagonists in the medium. The Bacillus genus made a significant contribution when introduced to both C36 and C7, and A. corrugatus made a significant contribution when introduced to the compost suspension.
In evaluating the biocontrol potential of the analyzed variants against
Alternaria alternata, it should be noted that the suppression increases caused by introducing microorganisms to C36 (compared with the control) were less intense than for the other phytopathogens. However, inoculating the compost suspensions (C7 variants) with microorganisms was more effective than inoculating the C36 variants given the increases in suppression (
Table 7,
Figure 2b).
The LB nutrient medium components did not improve the antagonistic activity, and in the C7L variant, the result was lower (MGI 26.5%) than that of the C7W control variant (41.4%) (p < 0.05).
For
A. alternata, the most successful inoculum was the composition of all four microorganisms, which, when introduced to both C36 and C7, achieved a mycelial growth inhibition rate of 81.9% (
Figure 2c). At the same time, variant C7A was also highly effective; the estimated rate of mycelial inhibition was 81.0%. However, statistically, the MGI values for all C36 variants were at approximately the same level, in some cases slightly higher (C36P) or slightly lower (C36B1, C36B2) than the control (in a range of 71.1–76.7%) (
Figure S8), but this was not significant (
p > 0.05, one group of results). As noted for the compost suspension variants, the differences in variants with introduced microorganisms compared with controls C7W and C7L were greater; however, among all the biocontrol agents (including microbial composition), the MGI was in about the same range of values (75.1–77.0%,
p > 0.05, one group).
In general, when using the antagonist composition, the mycelium growth inhibition values of the three phytopathogens were in a relatively narrow range of 81.9–91.7% for both types of compost (C36 and C7). The standard deviation did not exceed 8%, which may reflect a lower degree of scatter in the data. All of this may indicate more stable results in comparison with the use of single cultures.
Considering all the analysis results, the values obtained after applying composts with 36% dry matter were more consistent and higher. To some extent, this may be due to the way microorganisms grow when introduced to a solid or liquid medium. In the latter case, the microorganism takes longer to adapt to conditions; in the former case, the environment is closer to its native one, which leads it to rapidly achieve the maximum rate of biomass accumulation [
65,
66].
Scheuerell and Mahaffee (2004) [
67] and Deepthi and Reddy (2013) [
68] reported that the suppressive activity of composts may be more dependent on their various characteristics than on the number of antagonists in the medium. However, it is worth noting that variants C36W and C36L, without microbial inoculation, showed minimal suppressive activity against
P. solitum, while C7W and C7L did not inhibit the fungus (
Figure S7). Suppressing this phytopathogen likely depends greatly on the number of antagonists in the compost. Since there are known cases of resistance to some chemical fungicides in this species [
37] and based on the results of the analysis—which show that the growth of
P. solitum mycelium is less affected by inhibition than
C. rosea and
A. alternata, a larger amount of microbial fungicide compounds is probably needed to provide the proper level of biocontrol. Interestingly, a similar trend associated with the concentration of biocontrol agents in the medium was also shown for
C. rosea.
It is worth noting that no strong differences were found when inoculants were used to inhibit the growth of
A. alternata. Both in individual cultures and in microbial combination cases, suppression was observed at approximately the same level, with a nonsignificant increase for the combination. The introduction enhanced the suppressive potential of the initial compost suspensions (C7 variants), a feature that is difficult to observe in solid compost (C36 variants). Pane et al. (2012) [
50] reported significant variability in suppressing
A. alternata with compost tea, and this was independent of the type of extractant used to prepare the teas (in other words, it did not depend on the abiotic factor or certain additives). Deepthi and Reddy (2013) [
68] noted that introducing microorganisms in combination can even reduce suppressive activity against
A. alternata. However, conversely, the authors note that sometimes individual isolates and composts exposed under alternative conditions can be more effective in suppressing this phytopathogen. This can have a more significant effect than increasing the number of antagonistic microorganisms in the substrate. Ramírez-Cariño et al. (2020) [
14] noted that individual treatments with antagonists from the
Bacillus and
Trichoderma genera provided the same
A. alternata biocontrol effect as a combined application. Thus, suppressing
A. alternata may be greatly influenced by abiotic factors that affect the autochthonous microbiota of composts.
Our results were varied for the three species of fungal pathogens. Indeed, heterogeneity in the effect of different antagonists on phytopathogens has been found by other researchers. Daami-Remadi et al. (2012) [
69] showed that
Aspergillus, Penicillium, and
Trichoderma genera isolated from low-dry-matter compost inhibited two species of
Pythium in different ways. All fungi achieved 100% suppression of
P. aphanidermatum, but for
P. ultimum, the results ranged from 15 to 96%. De Corato et al. (2018) [
15] observed significant differences in the inhibition of pathogens by composts ranging from 25 to 80% overall when examining a wide range of phytopathogens.
Pane et al. (2012) [
50] suggested that the antagonism may experience an antibiotic-like effect without physical antagonist–pathogen interactions, which was indicated by the presence of distinct growth inhibition zones. Zouari et al. (2020) [
27] focused on the idea that the specificity of cell wall degradation enzyme synthesis (proteolytic, lipolytic, chitinolytic potential) of antagonists isolated from compost extracts greatly influences and inhibits fungal pathogens. Therefore, concrete fungal suppression can be caused by differences in the native autochthonous microbiota of composts and the suppressive potential of one or more communities. Considering the above aspects, variation in some environmental conditions may result in the succession of the microbial community and, accordingly, changes in the suppressive properties of the substrate. This can also include changes in the production of metabolites by compost-native microbiota after further inoculation with individual microorganisms. In our case, as shown in
Figure 2, there were variants with both a distinct zone of inhibition (an antibiotic-like effect), and other interactions between antagonists and pathogens were observed (
Figure S8). This may indicate the presence of several suppression mechanisms at once. The major contribution of certain mechanisms and the specifics producing highly effective metabolites in relation to concrete phytopathogens should be examined in detail. We studied the possibility of generally enhancing the suppressive properties of composts by introducing various additional inoculants. These issues are of interest and need to be investigated in more depth.
Therefore, forming communities approximating natural ones is a promising process, as well as deeper studies on changes in the mechanisms of production highly effective metabolites against phytopathogens using new composition. However, as can be seen from the above description, a large number of reports on bacterial–fungal interactions are based on the Bacillus, Pseudomonas, and Trichoderma genera; therefore, studying these aspects regarding the role of Aspergillus fungi is of interest, as they are commonly found in compost and the rhizosphere and (as shown in our study) significantly contribute to phytopathogen suppression. The data obtained in this research have the potential for further in-depth study because they only indirectly show the effect of introducing A. corrugatus, in association with Pseudomonas and Bacillus bacteria, to compost without a deeper investigation of the antagonism mechanism.