Cellular and Developmental Biology of TRPM7 Channel-Kinase: Implicated Roles in Cancer
Abstract
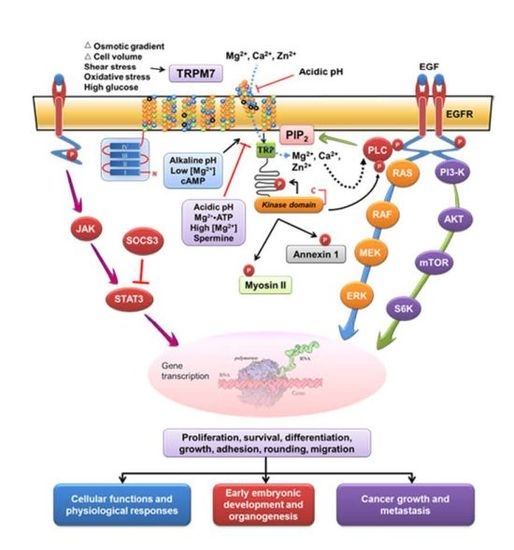
:1. Introduction
2. Structure and Functions of TRPM7 Channel-Kinase
2.1. Structure of TRPM7

2.2. Biochemical and Electrophysiological Functions of TRPM7
2.3. Molecular Determinants of the Functions of TRPM7 Channel-Kinase
| Mutations of Amino Acids | Effect on TRPM7 Function | Reference |
|---|---|---|
| Channel Pore Forming Segment (Human, aa. 1036–1056) | ||
| m E1047Q (Glu→Gln) or h E1047A (Glu→Ala) | Loss of channel permeability to Mg2+ and Ca2+. | [50,53] |
| m E1052Q (Glu→Gln) | Decreased Mg2+ and Ca2+ binding, and reduced Mg2+ and Ca2+ currents. | [50] |
| h E1052A (Glu→Ala) | Decreased Mg2+ and Ca2+ binding, and reduced Mg2+ and Ca2+ currents. Partial reduction of proton conductivity. | [26,53] |
| h D1054A (Asp→Ala) | Loss of proton conductivity. | [26] |
| h D1059A (Asp→Ala) | Partial reduction of proton conductivity. | [26] |
| Serine/Threonine Rich Region (Human, aa. 1380–1596) | ||
| h T1482I (Thr→Ile) (a natural variant) (autophosphorylation site) | Increased sensitivity of channel to Mg2+-mediated suppression, and decreased current even at reduced [Mg2+]. | [51,54] |
| m D1510 (Asp) | Caspase-mediated cleavage at Asp-1510 resulted in up-regulated channel activity. | [48] |
| m S1511A (Ser→Ala) (autophosphorylation site) | No change in Ca2+ influx or sensitivity to Mg2+-mediated inhibition. | [38] |
| m S1567A (Ser→Ala) (autophosphorylation site) | No change in Ca2+ influx or sensitivity to Mg2+-mediated inhibition. | [38] |
| m Y1553F (Tyr→Phe) | ~75% of wild-type kinase activity. | [21] |
| m Y1553L (Tyr→Leu) | ~50% of wild-type kinase activity. | |
| m Y1553A (Tyr→Ala) | ~35% of wild-type kinase activity. | |
| m R1558A (Arg→Ala) | ~15% of wild-type kinase activity. | [21] |
| Kinase Domain (Human, aa. 1597–1824) | ||
| m R1622L (Arg→Leu) (binding of PO43− of ATP) | <1% of wild-type kinase activity. | [38] |
| h K1648R (Lys→Arg) or (phosphotransfer activity) | Diminished kinase activity. No change in channel activation in response to decreased free Mg2+ or Mg·ATP. Attenuated suppression of channel activity in response to free Mg2+ or Mg·ATP. | [11] |
| m K1646R (Lys→Arg) (phosphotransfer activity) | <1% of wild-type kinase activity. | [38] |
| m K1727A (Lys→Ala) | <1% of wild-type kinase activity. | [38] |
| m N1731V (Asn→Val) (binding of PO43− of ATP) | <1% of wild-type kinase activity. | [38] |
| m E1760A (Glu→Ala) | ~15% of wild-type kinase activity. | [21] |
| m D1765N (Asp→Asn) or m D1765A (Asp→Ala) | <1% of wild-type kinase activity. | [38] |
| m Q1767N (Gln→Asn) or m Q1767A (Gln→Ala) (metal binding) | <1% of wild-type kinase activity. | [38] |
| m T1774S (Thr→Ser) | <1% of wild-type kinase activity. | [38] |
| m T1774A (Thr→Ala) (binding of PO43− of ATP) | ~6% of wild-type kinase activity. | |
| m D1775A (Asp→Ala) (metal binding) | <1% of wild-type kinase activity. | [38] |
| m N1795A (Asn→Ala) (binding of peptide substrate) | ~2% of wild-type kinase activity. | [38] |
| h G1799D (Gly→Asp) (phosphotransfer activity) | Diminished kinase activity. No change in channel activation in response to decreased free Mg2+ or Mg·ATP. Attenuated suppression of channel activity in response to free Mg2+ or Mg·ATP. | [11] |
3. TRPM7 Channel-Kinase in Normal Cellular Functions and Embryonic Development
3.1. TRPM7 in Cellular Processes and Physiological Functions
| Cell Type | Functional Roles of TRPM7 Channel-Kinase | References |
|---|---|---|
| Lymphocytes | - Required for Mg2+-dependent viability and proliferation of chicken B lymphocytes (DT-40). - Required for proliferation involving phosphoinositide 3-kinase. - Required for differentiation. - Required for survival of T lymphocytes by preventing Fas-induced apoptosis. | [9,11,48,55] |
| Neurons | - Oxidative stress activates TRPM7, which mediates anoxic death in human neurons; suppression of TRPM7 prevents anoxic neuronal death. - Facilitates fusion of cholinergic vesicle with plasma membrane and neurotransmitter release in cholinergic synaptic vesicles. | [23,56,57,58,59] |
| Interstitial cells of Cajal | - Required for pacemaker activity of mouse duodenum. - Expressed in the interstitial cells of Cajal of human colon and small intestine and involved in the generation of the slow waves. | [60,61] |
| Melanoblasts | - Required for survival of melanophores in zebrafish larvae. | [62,63] |
| Vascular smooth muscle cells | - Functional TRPM7 channels translocate to plasma membrane in response to fluid flow. - Angiotensin II promotes proliferation of VSMCs in ascending aorta by increasing TRPM7 protein via Ca2+-influx-mediated activation of the Pyk2-ERK1/2-Elk-1 pathway. | [64,65] |
| Osteoblasts | - Required for platelet-derived growth factor-induced proliferation and migration of human osteoblast MG-63 cells. | [66,67] |
| Cervical epithelia | - Required for volume regulation as TRPM7-like currents activated by osmotic swelling-induced mechanical stretch of human cervical cancer HeLa cells. | [68] |
| Mast cells | - Required for survival of human lung mast cells and human mast cell lines (LAD2, HMC-1). | [69] |
| Fibroblasts | - Membrane tension activates TRPM7 channels and Ca2+ flickers, directing migration in human embryonic lung fibroblasts. - Transforming growth factor-β increased expression of TRPM7 in human atrial fibroblasts associated with myofibroblast differentiation and fibrogenesis in atrial fibrillation. | [24,70] |
| Vascular endothelia | - Silencing TRPM7 promotes growth/proliferation and nitric oxide production viaERK in human umbilical vein vascular endothelial cells (HUVECs). - Silencing TRPM7 inhibits growth and migration of human microvascular endothelial cells (HMEC) but stimulates growth of HUVECs, partly because of impaired phosphorylation of ERK in HMEC. - Inhibition of TRPM7 leads to increased cell growth and migration in HUVECs. - TRPM7 contributes to hyperglycemia-induced injury of HUVECs. | [71,72,73] |
| Bone marrow derived mesenchymal stem cells | - Required for survival of mouse bone marrow-derived mesenchymal stem cells; expression increased during osteogenesis suggesting its involvement in differentiation. | [74] |
| Embryonic stem cells | - Kinase domain, but not kinase activity, is required for proliferation of mouse embryonic stem cells. | [29] |
| Pancreatic epithelia | - Required for proliferation, cell cycle progression, and growth involving Mg2+ and Soc3a in exocrine pancreatic epithelia of zebrafish larvae. | [75] |
| Hepatic stellate cells | - Required for survival by preventing TRAIL-induce apoptosis. - Regulates platelet-derived growth factor-BB-induced proliferation via PI3K and ERK in a rat hepatic stellate cell line (HSC-T6). - Required for activation and proliferation of HSCs by preventing endoplasmic reticulum stress-mediated apoptosis. | [76,77,78] |
| Atrial myocytes | - TRPM7-like current was recorded in human atrial myocytes, and expression of TRPM7 is up-regulated in atria with atrial fibrillation or membrane rupture. | [79,80] |
| Kidney cells | - TRPM7 contributes to elevated level of reactive oxygen species that leads to cell rounding mediated by the p38 MAPK/JNK-dependent activation of the Ca2+-dependent protease calpain in the immortalized human embryonic kidney cells (HEK 293), and during ischemia reperfusion in the mouse transplanted kidney. | [81,82,83] |
| Adipocytes | - Required for proliferation and differentiation of 3T3-L1 pre-adipocytes. | [84] |
| Prostate epithelia | - Increased Ca2+ to Mg2+ ratio in human prostate epithelia enhances. TRPM7-mediated currents and promotes cellular entry of Ca2+, leading to increase in cell proliferation. | [85] |

3.2. TRPM7 Channel-Kinase in Early Embryonic Development and Organogenesis
| Developmental Processes | Mutant Phenotypes | Functional Roles | References |
|---|---|---|---|
| Embryogenesis | - Early embryonic lethality between E 6.5 and E7.5 in mouse. | - Required for intestinal absorption of Mg2+ and whole body magnesium homeostasis. | [29,86] |
| Gastrulation | - Defects in cell polarization and alignment during convergent extension in Xenopus. | - TRPM7 channel but not the kinase domain required for regulating polarized cell movements during gastrulation involving Mg2+ via non-canonical Wnt signaling and modulation of the small GTPase Rac levels. | [87] |
| Melanogenesis | - Skin hypopigmentation in zebrafish larvae. | - Required for survival of melanophores in zebrafish larvae. - Loss-of-function mutation in Trpm7 leads to cell death of melanophores that is dependent on melanin synthesis. | [62,63,75,88,89,90,91] |
| Skeletogenesis | - Skeletal deformities in zebrafish with accelerated endochondral ossification and delayed intra-membranous ossification. - Dwarf zebrafish adults. | - Not reported. | [62] |
| Thymopoiesis | - Selective deletion of Trpm7 in T-cell lineage accelerates thymic involution in mouse. | - Required for differentiation and maintenance of thymic epithelia. - Required for STAT3 activity in thymic medullary cells. | [86] |
| Nervous system | - Defects in touch-response in zebrafish larvae. - Paralysis of hind legs of mouse with deletion of Trpm7 in committed neural crest progenitors; loss of large-diameter sensory neurons in lumbar dorsal root ganglion of mouse embryos depleted of TRPM7. | - Possibly required for synaptic release of neurotransmitters between sensory neurons and interneurons in zebrafish larvae. - Required for development of neural crest progenitors into dorsal root ganglion sensory neurons in mouse. - Required for differentiation or function of dopaminergic neurons in zebrafish larvae. | [59,92,93] |
| Nephrogenesis | - Nephrolithiasis in zebrafish larvae. - Defect formation of kidney with relatively few glomeruli and large renal cysts in mouse. | - Required for homeostasis of whole body Mg2+ and Ca2+ in zebrafish involving stanniocalcin 1 and fibroblast growth factor 23. | [92,94] |
| Exocrine pancreatic organogenesis | - Relatively small pancreas with immature acini and hypomorphic ducts in zebrafish larvae. | - Required for pancreatic epithelial proliferation and growth, which are sensitive to Mg2+ in extracellular medium and involving Socs3a. | [75,90,91] |
4. Expression and Roles of TRPM7 in Cancer
4.1. Oncologic Roles of TRPM7
| Cancer | Expression | Functional roles of TRPM7 | References |
|---|---|---|---|
| Pancreatic adenocarcinoma | - Increased in human pancreatic adenocarcinoma tissues and cell lines. - Increased in chronic pancreatitis, pancreatic intra-epithelial neoplasms | - Required for cellular proliferation and cell cycle progression involving Mg2+. - Required for preventing replicative senescence. - Required for cell migration involving Mg2+. - Required for cell invasion. | [75,95,96,97,98] |
| Breast carcinoma | - Over-expression in human breast carcinoma tissues and cell lines. - Increased expression in infiltrating ductal carcinoma with microcalcifications - Somatic mutation T720S (Thr→Ser) in a breast infiltrating ductal carcinoma | - Required for cancer cell proliferation in vitro. - Required for cancer cell migration in vitro and tumor metastasis in a mouse xenograft model. - Waixenicin A, TRPM7 blocker, inhibits growth and survival of breast cancer cells MCF-7. - TRPM7 involved in estrogen receptor-negative metastatic breast cancer cells migration through kinase domain. - Involved in ginsenoside Rd-induced apoptosis in cells. - Involved in epithelial mesenchymal transition. - TRPM7 mediates migration and invasion of breast cancer cells (MDA-MB-435) involving phosphorylation of Src and MAPK. | [99,100,101,102,103,104,105,106,107] |
| Gastric carcinoma | - Expressed in human gastric adenocarcinoma cell lines (AGS, MKN-1, MKN-45, SNU-1, SNU-484) - Somatic mutation M830V (Met→Val) in gastric adenocarcinoma | - Required for cell survival involving Mg2+. - Waixenicin A, TRPM7 blocker, inhibits growth andsurvival of gastric cancer cells AGS. - Involved in ginsenoside Rd-induced apoptosis AGS cells. | [105,107,108,109,110] |
| Head and neck Carcinoma | - Expressed in FaDu cells and SCC-25 cells. - High expression in 5-8F cells, low expression in 6-10B cells. | - Required for cell growth and proliferation. - Required for migration of nasopharyngeal carcinomacells (5-8F and 6-10B). - Proliferation of FaDu hypopharyngeal squamous cells (FaDu) inhibited by midazolam that targets TRPM7. | [111,112,113] |
| Retinoblastoma | - Existence in 5-8F cells | - Required for cell proliferation. - Required for 5-8F cell migration. | [114] |
| Melanoma | - Expressed in cell lines | - Not reported. | [63,115] |
| Lung carcinoma | - Expressed in A549 cells | - Required for migration of A549 cells. | [116] |
| Erythroleukemia | - TRPM7-like currents in cell lines. | - Not reported. | [117] |
| Colon cancer | -TRPM7 (Thr1482Ile) polymorphism | - TRPM7 (Thr1482Ile) polymorphism associated with elevated risk of both adenomatous and hyperplastic polyps. - Individuals with TRPM7 (Thr1482Ile) polymorphism with a high Ca:Mg ratio intake in diet at a relatively high risk of developing adenoma and hyperplastic polyps. | [54] |
| Leukemia | - Not reported. | - Waixenicin inhibits and T cell leukemia (Jurkat T lymphocytes) and rat basophilic leukemia cells (RBL1) through blocking TRPM7 channel activity. | [118] |
| Neuroblastoma | - Not reported. | - In mouse neuroblastoma cells (N1E-115), TRPM7 promotes formation of Ca2+ sparking and invadosome by affecting actomyosin contractility independent from Ca2+ influx. | [119] |
| Ovarian carcinoma | - Somatic mutation S406C (Ser→Cys) in ovarian serous carcinoma | - Not reported. | [107] |
| Prostate cancer | - Expressed in human prostate cancer cell line DU145 | - Increased Ca2+ to Mg2+ ratio in prostate cancer cells enhances TRPM7-mediated currents and promotes cellular entry of Ca2+, leading to increase in cell proliferation. | [85] |
4.2. Potential Role of TRPM7 as a Cancer Biomarker and Therapeutic Target
5. Conclusion and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Venkatachalam, K.; Montell, C. TRP channels. Annu. Rev. Biochem. 2007, 76, 387–417. [Google Scholar] [CrossRef]
- Clapham, D.E. TRP channels as cellular sensors. Nature 2003, 426, 517–524. [Google Scholar] [CrossRef]
- Freichel, M.; Flockerzi, V. Biological functions of TRPs unravelled by spontaneous mutations and transgenic animals. Biochem. Soc. Trans. 2007, 35, 120–123. [Google Scholar] [CrossRef]
- Minke, B.; Cook, B. TRP channel proteins and signal transduction. Physiol. Rev. 2002, 82, 429–472. [Google Scholar]
- Nilius, B.; Owsianik, G. The transient receptor potential family of ion channels. Genome Biol. 2011, 12. [Google Scholar] [CrossRef]
- Yee, N.S. Ion channels. In Encyclopedia of Cancer; Schwab, M., Ed.; Springer: New York, NY, USA, 2012. [Google Scholar]
- Fleig, A.; Penner, R. The TRPM ion channel subfamily: Molecular, biophysical and functional features. Trends Pharmacol. Sci. 2004, 25, 633–639. [Google Scholar] [CrossRef]
- Mederos y Schnitzler, M.; Waring, J.; Gudermann, T.; Chubanov, V. Evolutionary determinants of divergent calcium selectivity of TRPM channels. FASEB J. 2008, 22, 1540–1551. [Google Scholar] [CrossRef]
- Nadler, M.J.; Hermosura, M.C.; Inabe, K.; Perraud, A.L.; Zhu, Q.; Stokes, A.J.; Kurosaki, T.; Kinet, J.P.; Penner, R.; Scharenberg, A.M.; et al. LTRPC7 is a Mg.ATP-regulated divalent cation channel required for cell viability. Nature 2001, 411, 590–595. [Google Scholar] [CrossRef]
- Runnels, L.W.; Yue, L.; Clapham, D.E. TRP-PLIK, a bifunctional protein with kinase and ion channel activities. Science 2001, 291, 1043–1047. [Google Scholar] [CrossRef]
- Schmitz, C.; Perraud, A.L.; Johnson, C.O.; Inabe, K.; Smith, M.K.; Penner, R.; Kurosaki, T.; Fleig, A.; Scharenberg, A.M. Regulation of vertebrate cellular Mg2+ homeostasis by TRPM7. Cell 2003, 114, 191–200. [Google Scholar] [CrossRef]
- Kunert-Keil, C.; Bisping, F.; Kruger, J.; Brinkmeier, H. Tissue-specific expression of TRP channel genes in the mouse and its variation in three different mouse strains. BMC Genomics 2006, 7. [Google Scholar] [CrossRef]
- Fonfria, E.; Murdock, P.R.; Cusdin, F.S.; Benham, C.D.; Kelsell, R.E.; McNulty, S. Tissue distribution profiles of the human TRPM cation channel family. J. Recept. Signal Transduct. Res. 2006, 26, 159–178. [Google Scholar] [CrossRef]
- Ramsey, I.S.; Delling, M.; Clapham, D.E. An introduction to TRP channels. Annu. Rev. Physiol. 2006, 68, 619–647. [Google Scholar] [CrossRef]
- Kraft, R.; Harteneck, C. The mammalian melastatin-related transient receptor potential cation channels: An overview. Pflügers Arch. 2005, 451, 204–211. [Google Scholar] [CrossRef]
- Fujiwara, Y.; Minor, D.L., Jr. X-ray crystal structure of a TRPM assembly domain reveals an antiparallel four-stranded coiled-coil. J. Mol. Biol. 2008, 383, 854–870. [Google Scholar] [CrossRef]
- Chubanov, V.; Waldegger, S.; Mederos y Schnitzler, M.; Vitzthum, H.; Sassen, M.C.; Seyberth, H.W.; Konrad, M.; Gudermann, T. Disruption of TRPM6/ TRPM7 complex formation by a mutation in the TRPM6 gene causes hypomagnesemia with secondary hypocalcemia. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 2894–2899. [Google Scholar] [CrossRef]
- Schmitz, C.; Dorovkov, M.V.; Zhao, X.; Davenport, B.J.; Ryazanov, A.G.; Perraud, A.L. The channel kinases TRPM6 and TRPM7 are functionally nonredundant. J. Biol. Chem. 2005, 280, 37763–37771. [Google Scholar]
- Li, M.; Jiang, J.; Yue, L. Functional characterization of homo- and heteromeric channel kinases TRPM6 and TRPM7. J. Gen. Physiol. 2006, 127, 525–537. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Matsushita, M.; Nairn, A.C.; Kuriyan, J. Crystal structure of the atypical protein kinase domain of a TRP channel with phosphotransferase activity. Mol. Cell 2001, 7, 1047–1057. [Google Scholar] [CrossRef]
- Crawley, S.W.; Cote, G.P. Identification of dimer interactions required for the catalytic activity of the TRPM7 alpha-kinase domain. Biochem. J. 2009, 420, 115–122. [Google Scholar] [CrossRef]
- Monteilh-Zoller, M.K.; Hermosura, M.C.; Nadler, M.J.; Scharenberg, A.M.; Penner, R.; Fleig, A. TRPM7 provides an ion channel mechanism for cellular entry of trace metal ions. J. Gen. Physiol. 2003, 121, 49–60. [Google Scholar]
- Aarts, M.; Iihara, K.; Wei, W.L.; Xiong, Z.G.; Arundine, M.; Cerwinski, W.; MacDonald, J.F.; Tymianski, M. A key role for TRPM7 channels in anoxic neuronal death. Cell 2003, 115, 863–877. [Google Scholar] [CrossRef]
- Wei, C.; Wang, X.; Chen, M.; Ouyang, K.; Song, L.S.; Cheng, H. Calcium flickers steer cell migration. Nature 2009, 457, 901–905. [Google Scholar] [CrossRef]
- Jiang, J.; Li, M.; Yue, L. Potentiation of TRPM7 inward currents by protons. J. Gen. Physiol. 2005, 126, 137–150. [Google Scholar] [CrossRef]
- Numata, T.; Okada, Y. Proton conductivity through the human TRPM7 channel and its molecular determinants. J. Biol. Chem. 2008, 283, 15097–15103. [Google Scholar] [CrossRef]
- Schlingmann, K.P.; Weber, S.; Peters, M.; Niemann Nejsum, L.; Vitzthum, H.; Klingel, K.; Kratz, M.; Haddad, E.; Ristoff, E.; Dinour, D.; et al. Hypomagnesemia with secondary hypocalcemia is caused by mutations in TRPM6, a new member of the TRPM gene family. Nat. Genet. 2002, 31, 166–170. [Google Scholar] [CrossRef]
- Schlingmann, K.P.; Waldegger, S.; Konrad, M.; Chubanov, V.; Gudermann, T. TRPM6 and TRPM7-gatekeepers of human magnesium metabolism. Biochem. Biophys. Acta 2007, 1772, 813–821. [Google Scholar]
- Ryazanova, L.V.; Rondon, L.J.; Zierler, S.; Hu, Z.; Galli, J.; Yamaguchi, T.P.; Mazur, A.; Fleig, A.; Ryazanov, A.G. TRPM7 is essential for Mg2+ homeostasis in mammals. Nat. Commun. 2010, 1. [Google Scholar] [CrossRef]
- Walder, R.Y.; Landau, D.; Meyer, P.; Shalev, H.; Tsolia, M.; Borochowitz, Z.; Boettger, M.B.; Beck, G.E.; Englehardt, R.K.; Carmi, R.; et al. Mutation of TRPM6 causes familial hypomagnesemia with secondary hypocalcemia. Nat. Genet. 2002, 31, 171–174. [Google Scholar] [CrossRef]
- Takezawa, R.; Schmitz, C.; Demeuse, P.; Scharenberg, A.M.; Penner, R.; Fleig, A. Receptor-mediated regulation of the TRPM7 channel through its endogenous protein kinase domain. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 6009–6014. [Google Scholar] [CrossRef]
- Runnels, L.W.; Yue, L.; Clapham, D.E. The TRPM7 channel is inactivated by PIP(2) hydrolysis. Nat. Cell Biol. 2002, 4, 329–336. [Google Scholar]
- Langeslag, M.; Clark, K.; Moolenaar, W.H.; van Leeuwen, F.N.; Jalink, K. Activation of TRPM7 channels by phospholipase c-coupled receptor agonists. J. Biol. Chem. 2007, 282, 232–239. [Google Scholar] [CrossRef]
- Kozak, J.A.; Matsushita, M.; Nairn, A.C.; Cahalan, M.D. Charge screening by internal pH and polyvalent cations as a mechanism for activation, inhibition, and rundown of TRPM7/mic channels. J. Gen. Physiol. 2005, 126, 499–514. [Google Scholar] [CrossRef]
- Chokshi, R.; Matsushita, M.; Kozak, J.A. Detailed examination of Mg2+ and pH sensitivity of human TRPM7 channels. Am J Physiol Cell Physiol 2012, 302, C1004–C1011. [Google Scholar] [CrossRef]
- Yu, H.; Zhang, Z.; Lis, A.; Penner, R.; Fleig, A. TRPM7 is regulated by halides through its kinase domain. Cell Mol. Life Sci. 2013, 70, 2757–2771. [Google Scholar] [CrossRef]
- Hofmann, T.; Schafer, S.; Linseisen, M.; Sytik, L.; Gudermann, T.; Chubanov, V. Activation of TRPM7 channels by small molecules under physiological conditions. Pflügers Arch. 2014. [Google Scholar] [CrossRef]
- Matsushita, M.; Kozak, J.A.; Shimizu, Y.; McLachlin, D.T.; Yamaguchi, H.; Wei, F.Y.; Tomizawa, K.; Matsui, H.; Chait, B.T.; Cahalan, M.D.; et al. Channel function is dissociated from the intrinsic kinase activity and autophosphorylation of TRPM7/chak1. J. Biol. Chem. 2005, 280, 20793–20803. [Google Scholar] [CrossRef]
- Clark, K.; Middelbeek, J.; Morrice, N.A.; Figdor, C.G.; Lasonder, E.; van Leeuwen, F.N. Massive autophosphorylation of the ser/thr-rich domain controls protein kinase activity of TRPM6 and TRPM7. PloS One 2008, 3, e1876. [Google Scholar]
- Kim, T.Y.; Shin, S.K.; Song, M.Y.; Lee, J.E.; Park, K.S. Identification of the phosphorylation sites on intact TRPM7 channels from mammalian cells. Biochem. Biophys. Res. Commun. 2012, 417, 1030–1034. [Google Scholar] [CrossRef]
- Dorovkov, M.V.; Ryazanov, A.G. Phosphorylation of annexin I by TRPM7 channel-kinase. J. Biol. Chem. 2004, 279, 50643–50646. [Google Scholar] [CrossRef]
- Clark, K.; Langeslag, M.; van Leeuwen, B.; Ran, L.; Ryazanov, A.G.; Figdor, C.G.; Moolenaar, W.H.; Jalink, K.; van Leeuwen, F.N. TRPM7, a novel regulator of actomyosin contractility and cell adhesion. EMBO J. 2006, 25, 290–301. [Google Scholar] [CrossRef]
- Clark, K.; Middelbeek, J.; Lasonder, E.; Dulyaninova, N.G.; Morrice, N.A.; Ryazanov, A.G.; Bresnick, A.R.; Figdor, C.G.; van Leeuwen, F.N. TRPM7 regulates myosin IIa filament stability and protein localization by heavy chain phosphorylation. J. Mol. Biol. 2008, 378, 790–803. [Google Scholar] [CrossRef]
- Deason-Towne, F.; Perraud, A.L.; Schmitz, C. Identification of ser/thr phosphorylation sites in the c2-domain of phospholipase c gamma2 (PLCγ2) using TRPM7-kinase. Cell. Signal. 2012, 24, 2070–2075. [Google Scholar] [CrossRef]
- Perraud, A.L.; Zhao, X.; Ryazanov, A.G.; Schmitz, C. The channel-kinase TRPM7 regulates phosphorylation of the translational factor eEF2 via eEF2-k. Cell. Signal. 2011, 23, 586–593. [Google Scholar] [CrossRef]
- Ryazanova, L.V.; Dorovkov, M.V.; Ansari, A.; Ryazanov, A.G. Characterization of the protein kinase activity of TRPM7/chak1, a protein kinase fused to the transient receptor potential ion channel. J. Biol. Chem. 2004, 279, 3708–3716. [Google Scholar]
- Demeuse, P.; Penner, R.; Fleig, A. TRPM7 channel is regulated by magnesium nucleotides via its kinase domain. J. Gen. Physiol. 2006, 127, 421–434. [Google Scholar] [CrossRef]
- Desai, B.N.; Krapivinsky, G.; Navarro, B.; Krapivinsky, L.; Carter, B.C.; Febvay, S.; Delling, M.; Penumaka, A.; Ramsey, I.S.; Manasian, Y.; et al. Cleavage of TRPM7 releases the kinase domain from the ion channel and regulates its participation in fas-induced apoptosis. Dev. Cell 2012, 22, 1149–1162. [Google Scholar] [CrossRef]
- Krapivinsky, G.; Krapivinsky, L.; Manasian, Y.; Clapham, D.E. The TRPM7 chanzyme is cleaved to release a chromatin-modifying kinase. Cell 2014, 157, 1061–1072. [Google Scholar] [CrossRef]
- Li, M.; Du, J.; Jiang, J.; Ratzan, W.; Su, L.T.; Runnels, L.W.; Yue, L. Molecular determinants of Mg2+ and Ca2+ permeability and pH sensitivity in TRPM6 and TRPM7. J. Biol. Chem. 2007, 282, 25817–25830. [Google Scholar] [CrossRef]
- Hermosura, M.C.; Nayakanti, H.; Dorovkov, M.V.; Calderon, F.R.; Ryazanov, A.G.; Haymer, D.S.; Garruto, R.M. A TRPM7 variant shows altered sensitivity to magnesium that may contribute to the pathogenesis of two guamanian neurodegenerative disorders. Proc. Natl. Acad. Sci. U. S. A. 2005, 102, 11510–11515. [Google Scholar] [CrossRef]
- UniProt, C. Activities at the universal protein resource (uniprot). Nucleic Acids Res. 2014, 42, D191–D198. [Google Scholar] [CrossRef]
- Numata, T.; Okada, Y. Molecular determinants of sensitivity and conductivity of human TRPM7 to Mg2+ and Ca2+. Channels (Austin) 2008, 2, 283–286. [Google Scholar] [CrossRef]
- Dai, Q.; Shrubsole, M.J.; Ness, R.M.; Schlundt, D.; Cai, Q.; Smalley, W.E.; Li, M.; Shyr, Y.; Zheng, W. The relation of magnesium and calcium intakes and a genetic polymorphism in the magnesium transporter to colorectal neoplasia risk. Am. J. Clin. Nutr. 2007, 86, 743–751. [Google Scholar]
- Sahni, J.; Scharenberg, A.M. TRPM7 ion channels are required for sustained phosphoinositide 3-kinase signaling in lymphocytes. Cell MeTable 2008, 8, 84–93. [Google Scholar] [CrossRef]
- Aarts, M.M.; Tymianski, M. TRPMs and neuronal cell death. Pflügers Arch. 2005, 451, 243–249. [Google Scholar] [CrossRef]
- Krapivinsky, G.; Mochida, S.; Krapivinsky, L.; Cibulsky, S.M.; Clapham, D.E. The TRPM7 ion channel functions in cholinergic synaptic vesicles and affects transmitter release. Neuron 2006, 52, 485–496. [Google Scholar] [CrossRef]
- Brauchi, S.; Krapivinsky, G.; Krapivinsky, L.; Clapham, D.E. TRPM7 facilitates cholinergic vesicle fusion with the plasma membrane. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 8304–8308. [Google Scholar] [CrossRef]
- Low, S.E.; Amburgey, K.; Horstick, E.; Linsley, J.; Sprague, S.M.; Cui, W.W.; Zhou, W.B.; Hirata, H.; Saint-Amant, L.; Hume, R.I.; et al. TRPM7 is required within zebrafish sensory neurons for the activation of touch-evoked escape behaviors. J. Neurosci. 2011, 31, 11633–11644. [Google Scholar] [CrossRef]
- Kim, B.J.; Lim, H.H.; Yang, D.K.; Jun, J.Y.; Chang, I.Y.; Park, C.S.; So, I.; Stanfield, P.R.; Kim, K.W. Melastatin-type transient receptor potential channel 7 is required for intestinal pacemaking activity. Gastroenterology 2005, 129, 1504–1517. [Google Scholar] [CrossRef]
- Kim, B.J.; Park, K.J.; Kim, H.W.; Choi, S.; Jun, J.Y.; Chang, I.Y.; Jeon, J.H.; So, I.; Kim, S.J. Identification of TRPM7 channels in human intestinal interstitial cells of Cajal. World J. Gastroenterol. 2009, 15, 5799–5804. [Google Scholar] [CrossRef]
- Elizondo, M.R.; Arduini, B.L.; Paulsen, J.; MacDonald, E.L.; Sabel, J.L.; Henion, P.D.; Cornell, R.A.; Parichy, D.M. Defective skeletogenesis with kidney stone formation in dwarf zebrafish mutant for trpm7. Curr. Biol. 2005, 15, 667–671. [Google Scholar] [CrossRef]
- McNeill, M.S.; Paulsen, J.; Bonde, G.; Burnight, E.; Hsu, M.Y.; Cornell, R.A. Cell death of melanophores in zebrafish trpm7 mutant embryos depends on melanin synthesis. J. Invest. Dermatol. 2007, 127, 2020–2030. [Google Scholar] [CrossRef]
- Oancea, E.; Wolfe, J.T.; Clapham, D.E. Functional TRPM7 channels accumulate at the plasma membrane in response to fluid flow. Circ. Res. 2006, 98, 245–253. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, M.; Fan, X.H.; Chen, J.H.; Guan, Y.Y.; Tang, Y.B. Upregulation of TRPM7 channels by angiotensin II triggers phenotypic switching of vascular smooth muscle cells of ascending aorta. Circ. Res. 2012, 111, 1137–1146. [Google Scholar] [CrossRef]
- Abed, E.; Moreau, R. Importance of melastatin-like transient receptor potential 7 and cations (magnesium, calcium) in human osteoblast-like cell proliferation. Cell Prolif. 2007, 40, 849–865. [Google Scholar] [CrossRef] [Green Version]
- Abed, E.; Moreau, R. Importance of melastatin-like transient receptor potential 7 and magnesium in the stimulation of osteoblast proliferation and migration by platelet-derived growth factor. Am. J. Physiol. Cell Physiol. 2009, 297, C360–C368. [Google Scholar] [CrossRef]
- Numata, T.; Shimizu, T.; Okada, Y. Direct mechano-stress sensitivity of TRPM7 channel. Cell. Physiol. Biochem. 2007, 19, 1–8. [Google Scholar] [CrossRef]
- Wykes, R.C.; Lee, M.; Duffy, S.M.; Yang, W.; Seward, E.P.; Bradding, P. Functional transient receptor potential melastatin 7 channels are critical for human mast cell survival. J. Immunol. 2007, 179, 4045–4052. [Google Scholar] [CrossRef]
- Du, J.; Xie, J.; Zhang, Z.; Tsujikawa, H.; Fusco, D.; Silverman, D.; Liang, B.; Yue, L. TRPM7-mediated Ca2+ signals confer fibrogenesis in human atrial fibrillation. Circ. Res. 2010, 106, 992–1003. [Google Scholar] [CrossRef]
- Baldoli, E.; Castiglioni, S.; Maier, J.A. Regulation and function of TRPM7 in human endothelial cells: TRPM7 as a potential novel regulator of endothelial function. PLoS One 2013, 8, e59891. [Google Scholar] [CrossRef]
- Baldoli, E.; Maier, J.A. Silencing TRPM7 mimics the effects of magnesium deficiency in human microvascular endothelial cells. Angiogenesis 2012, 15, 47–57. [Google Scholar] [CrossRef]
- Inoue, K.; Xiong, Z.G. Silencing TRPM7 promotes growth/proliferation and nitric oxide production of vascular endothelial cells via the ERK pathway. Cardiovasc Res 2009, 83, 547–557. [Google Scholar] [CrossRef]
- Cheng, H.; Feng, J.M.; Figueiredo, M.L.; Zhang, H.J.; Nelson, P.L.; Marigo, V.; Beck, A. Transient receptor potential melastatin type 7 channel is critical for the survival of bone marrow derived mesenchymal stem cells. Stem Cells Dev. 2010, 19, 1393–1403. [Google Scholar] [CrossRef]
- Yee, N.S.; Zhou, W.; Liang, I.C. Transient receptor potential ion channel TRPM7 regulates exocrine pancreatic epithelial proliferation by Mg2+-sensitive Socs3a signaling in development and cancer. Dis. Mod. Mech. 2011, 4, 240–254. [Google Scholar] [CrossRef]
- Liu, H.; Li, J.; Huang, Y.; Huang, C. Inhibition of transient receptor potential melastain 7 channel increases HSCs apoptosis induced by trail. Life Sci. 2012, 90, 612–618. [Google Scholar] [CrossRef]
- Fang, L.; Zhan, S.; Huang, C.; Cheng, X.; Lv, X.; Si, H.; Li, J. TRPM7 channel regulates PGDF-BB-induced proliferation of hepatic stellate cells via PI3K and ERK pathways. Toxicol. Appl. Pharmacol. 2013, 272, 713–725. [Google Scholar] [CrossRef]
- Zhu, Y.; Men, R.; Wen, M.; Hu, X.; Liu, X.; Yang, L. Blockage of TRPM7 channel induces hepatic stellate cell death through endoplasmic reticulum stress-mediated apoptosis. Life Sci. 2014, 94, 37–44. [Google Scholar] [CrossRef]
- Macianskiene, R.; Martisiene, I.; Zablockaite, D.; Gendviliene, V. Characterization of Mg2+-regulated TRPM7-like current in human atrial myocytes. J. Biomed. Sci. 2012, 19. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Sun, H.Y.; Chen, K.H.; Du, X.L.; Liu, B.; Cheng, L.C.; Li, X.; Jin, M.W.; Li, G.R. Evidence for functional expression of TRPM7 channels in human atrial myocytes. Basic Res. Cardiol. 2012, 107. [Google Scholar] [CrossRef]
- Su, L.T.; Chen, H.C.; Gonzalez-Pagan, O.; Overton, J.D.; Xie, J.; Yue, L.; Runnels, L.W. TRPM7 activates m-calpain by stress-dependent stimulation of p38 MAPK and c-Jun n-terminal kinase. J. Mol. Biol. 2010, 396, 858–869. [Google Scholar] [CrossRef]
- Meng, Z.; Cao, R.; Wang, Y.; Cao, H.; Liu, T.; Yang, Z.; Wang, X. Suppression of renal TRPM7 may alleviate kidney injury in the renal transplantation. World J. Urol. 2013. [Google Scholar] [CrossRef]
- Chen, H.C.; Su, L.T.; Gonzalez-Pagan, O.; Overton, J.D.; Runnels, L.W. A key role for Mg2+ in TRPM7's control of ros levels during cell stress. Biochem. J. 2012, 445, 441–448. [Google Scholar] [CrossRef]
- Chen, K.H.; Xu, X.H.; Liu, Y.; Hu, Y.; Jin, M.W.; Li, G.R. TRPM7 channels regulate proliferation and adipogenesis in 3T3-L1 preadipocytes. J. Cell. Physiol. 2014, 229, 60–67. [Google Scholar]
- Sun, Y.; Selvaraj, S.; Varma, A.; Derry, S.; Sahmoun, A.E.; Singh, B.B. Increase in serum Ca2+/Mg2+ ratio promotes proliferation of prostate cancer cells by activating TRPM7 channels. J. Biol. Chem. 2013, 288, 255–263. [Google Scholar] [CrossRef]
- Jin, J.; Desai, B.N.; Navarro, B.; Donovan, A.; Andrews, N.C.; Clapham, D.E. Deletion of TRPM7 disrupts embryonic development and thymopoiesis without altering Mg2+ homeostasis. Science 2008, 322, 756–760. [Google Scholar] [CrossRef]
- Liu, W.; Su, L.T.; Khadka, D.K.; Mezzacappa, C.; Komiya, Y.; Sato, A.; Habas, R.; Runnels, L.W. TRPM7 regulates gastrulation during vertebrate embryogenesis. Dev. Biol. 2011, 350, 348–357. [Google Scholar] [CrossRef]
- Cornell, R.A.; Yemm, E.; Bonde, G.; Li, W.; d'Alencon, C.; Wegman, L.; Eisen, J.; Zahs, A. Touchtone promotes survival of embryonic melanophores in zebrafish. Mech. Dev. 2004, 121, 1365–1376. [Google Scholar] [CrossRef]
- Arduini, B.L.; Henion, P.D. Melanophore sublineage-specific requirement for zebrafish touchtone during neural crest development. Mech. Dev. 2004, 121, 1353–1364. [Google Scholar] [CrossRef]
- Yee, N.S.; Lorent, K.; Pack, M. Exocrine pancreas development in zebrafish. Dev. Biol. 2005, 284, 84–101. [Google Scholar] [CrossRef]
- Yee, N.S.; Kazi, A.A.; Yee, R.K. Translating discovery in zebrafish pancreatic development to human pancreatic cancer: Biomarkers, targets, pathogenesis, and therapeutics. Zebrafish 2013, 10, 132–146. [Google Scholar] [CrossRef]
- Jin, J.; Wu, L.J.; Jun, J.; Cheng, X.P.; Xu, H.X.; Andrews, N.C.; Clapham, D.E. The channel kinase, TRPM7, is required for early embryonic development. Proc. Natl. Acad. Sci. U. S. A. 2012, 109, E225–E233. [Google Scholar]
- Decker, A.R.; McNeill, M.S.; Lambert, A.M.; Overton, J.D.; Chen, Y.C.; Lorca, R.A.; Johnson, N.A.; Brockerhoff, S.E.; Mohapatra, D.P.; Macarthur, H.; et al. Abnormal differentiation of dopaminergic neurons in zebrafish trpm7 mutant larvae impairs development of the motor pattern. Dev. Biol. 2014, 386, 428–439. [Google Scholar] [CrossRef]
- Elizondo, M.R.; Budi, E.H.; Parichy, D.M. trpm7 regulation of in vivo cation homeostasis and kidney function involves stanniocalcin 1 and fgf23. Endocrinology 2010, 151, 5700–5709. [Google Scholar] [CrossRef]
- Rybarczyk, P.; Gautier, M.; Hague, F.; Dhennin-Duthille, I.; Chatelain, D.; Kerr-Conte, J.; Pattou, F.; Regimbeau, J.M.; Sevestre, H.; Ouadid-Ahidouch, H. Transient receptor potential melastatin-related 7 channel is overexpressed in human pancreatic ductal adenocarcinomas and regulates human pancreatic cancer cell migration. Int. J. Cancer 2012, 131, E851–E861. [Google Scholar] [CrossRef]
- Yee, N.S.; Yee, R.K. Ion channels as novel pancreatic cancer biomarkers and targets. In New Advances on Disease Biomarkers and Molecular Targets in Biomedicine; Lee, N.P.-Y., Ed.; Springer: New York, NY, USA, 2013. [Google Scholar]
- Yee, N.S.; Chan, A.S.; Yee, J.D.; Yee, R.K. TRPM7 and TRPM8 ion channels in pancreatic adenocarcinoma: Potential roles as cancer biomarkers and targets. Scientifica 2012, 2012, 415158:1–415158:8. [Google Scholar]
- Yee, N.S.; Zhou, W.; Lee, M.; Yee, R.K. Targeted silencing of TRPM7 ion channel induces replicative senescence and produces enhanced cytotoxicity with gemcitabine in pancreatic adenocarcinoma. Cancer Lett. 2012, 318, 99–105. [Google Scholar] [CrossRef]
- Middelbeek, J.; Kuipers, A.J.; Henneman, L.; Visser, D.; Eidhof, I.; van Horssen, R.; Wieringa, B.; Canisius, S.V.; Zwart, W.; Wessels, L.F.; et al. TRPM7 is required for breast tumor cell metastasis. Cancer Res. 2012, 72, 4250–4261. [Google Scholar] [CrossRef]
- Guilbert, A.; Gautier, M.; Dhennin-Duthille, I.; Haren, N.; Sevestre, H.; Ouadid-Ahidouch, H. Evidence that TRPM7 is required for breast cancer cell proliferation. Am. J. Physiol. Cell Physiol. 2009, 297, C493–C502. [Google Scholar] [CrossRef]
- Guilbert, A.; Gautier, M.; Dhennin-Duthille, I.; Rybarczyk, P.; Sahni, J.; Sevestre, H.; Scharenberg, A.M.; Ouadid-Ahidouch, H. Transient receptor potential melastatin 7 is involved in oestrogen receptor-negative metastatic breast cancer cells migration through its kinase domain. Eur. J. Cancer 2013, 49, 3694–3707. [Google Scholar] [CrossRef]
- Mandavilli, S.; Singh, B.B.; Sahmoun, A.E. Serum calcium levels, TRPM7, TRPC1, microcalcifications, and breast cancer using breast imaging reporting and data system scores. Breast Cancer (London) 2012, 2013, 1–7. [Google Scholar]
- Meng, X.; Cai, C.; Wu, J.; Cai, S.; Ye, C.; Chen, H.; Yang, Z.; Zeng, H.; Shen, Q.; Zou, F. TRPM7 mediates breast cancer cell migration and invasion through the MAPK pathway. Cancer Lett. 2013, 333, 96–102. [Google Scholar] [CrossRef]
- Kim, B.J. Involvement of melastatin type transient receptor potential 7 channels in ginsenoside rd-induced apoptosis in gastric and breast cancer cells. J. Gins. Res. 2013, 37, 201–209. [Google Scholar] [CrossRef]
- Kim, B.J.; Nam, J.H.; Kwon, Y.K.; So, I.; Kim, S.J. The role of waixenicin A as transient receptor potential melastatin 7 blocker. Basic Clin. Pharmacol. Toxicol. 2013, 112, 83–89. [Google Scholar] [CrossRef]
- Davis, F.M.; Azimi, I.; Faville, R.A.; Peters, A.A.; Jalink, K.; Putney, J.W., Jr.; Goodhill, G.J.; Thompson, E.W.; Roberts-Thomson, S.J.; Monteith, G.R. Induction of epithelial-mesenchymal transition (EMT) in breast cancer cells is calcium signal dependent. Oncogene 2014, 33, 2307–2316. [Google Scholar] [CrossRef]
- Greenman, C.; Stephens, P.; Smith, R.; Dalgliesh, G.L.; Hunter, C.; Bignell, G.; Davies, H.; Teague, J.; Butler, A.; Stevens, C.; et al. Patterns of somatic mutation in human cancer genomes. Nature 2007, 446, 153–158. [Google Scholar] [CrossRef]
- Kim, B.J.; Kim, S.Y.; Lee, S.; Jeon, J.H.; Matsui, H.; Kwon, Y.K.; Kim, S.J.; So, I. The role of transient receptor potential channel blockers in human gastric cancer cell viability. Can. J. Physiol. Pharmacol. 2012, 90, 175–186. [Google Scholar] [CrossRef]
- Kim, B.J.; Nah, S.Y.; Jeon, J.H.; So, I.; Kim, S.J. Transient receptor potential melastatin 7 channels are involved in ginsenoside Rg3-induced apoptosis in gastric cancer cells. Basic Clin. Pharmacol. Toxicol. 2011, 109, 233–239. [Google Scholar] [CrossRef]
- Kim, B.J.; Park, E.J.; Lee, J.H.; Jeon, J.H.; Kim, S.J.; So, I. Suppression of transient receptor potential melastatin 7 channel induces cell death in gastric cancer. Cancer Sci. 2008, 99, 2502–2509. [Google Scholar] [CrossRef]
- Chen, J.P.; Luan, Y.; You, C.X.; Chen, X.H.; Luo, R.C.; Li, R. TRPM7 regulates the migration of human nasopharyngeal carcinoma cell by mediating Ca2+ influx. Cell Calcium 2010, 47, 425–432. [Google Scholar] [CrossRef]
- Jiang, J.; Li, M.H.; Inoue, K.; Chu, X.P.; Seeds, J.; Xiong, Z.G. Transient receptor potential melastatin 7-like current in human head and neck carcinoma cells: Role in cell proliferation. Cancer Res. 2007, 67, 10929–10938. [Google Scholar] [CrossRef]
- Dou, Y.; Li, Y.; Chen, J.; Wu, S.; Xiao, X.; Xie, S.; Tang, L.; Yan, M.; Wang, Y.; Lin, J.; et al. Inhibition of cancer cell proliferation by midazolam by targeting transient receptor potential melastatin 7. Oncol. Lett. 2013, 5, 1010–1016. [Google Scholar]
- Hanano, T.; Hara, Y.; Shi, J.; Morita, H.; Umebayashi, C.; Mori, E.; Sumimoto, H.; Ito, Y.; Mori, Y.; Inoue, R. Involvement of TRPM7 in cell growth as a spontaneously activated Ca2+ entry pathway in human retinoblastoma cells. J. Pharmacol. Sci. 2004, 95, 403–419. [Google Scholar] [CrossRef]
- Guo, H.; Carlson, J.A.; Slominski, A. Role of TRPM in melanocytes and melanoma. Exp. Dermatol. 2012, 21, 650–654. [Google Scholar] [CrossRef]
- Gao, H.; Chen, X.; Du, X.; Guan, B.; Liu, Y.; Zhang, H. EGF enhances the migration of cancer cells by up-regulation of TRPM7. Cell Calcium 2011, 50, 559–568. [Google Scholar] [CrossRef]
- Mason, M.J.; Schaffner, C.; Floto, R.A.; Teo, Q.A. Constitutive expression of a Mg2+-inhibited K+ current and a TRPM7-like current in human erythroleukemia cells. Am. J. Physiol. Cell. Physiol. 2012, 302, C853–C867. [Google Scholar] [CrossRef]
- Zierler, S.; Yao, G.; Zhang, Z.; Kuo, W.C.; Porzgen, P.; Penner, R.; Horgen, F.D.; Fleig, A. Waixenicin A inhibits cell proliferation through magnesium-dependent block of transient receptor potential melastatin 7 (TRPM7) channels. J. Biol. Chem. 2011, 286, 39328–39335. [Google Scholar]
- Visser, D.; Langeslag, M.; Kedziora, K.M.; Klarenbeek, J.; Kamermans, A.; Horgen, F.D.; Fleig, A.; van Leeuwen, F.N.; Jalink, K. TRPM7 triggers Ca2+ sparks and invadosome formation in neuroblastoma cells. Cell Calcium 2013, 54, 404–415. [Google Scholar] [CrossRef]
- Yee, N.S.; Li, Q.; Kazi, A.A.; Yang, Z.; Berg, A.; Kimball, S.R.; Yee, R.K.; Pennsylvania State University, Hershey, PA, USA. 2014.
- Wolf, F.I.; Trapani, V. Magnesium and its transporters in cancer: A novel paradigm in tumour development. Clin. Sci. (Lond) 2012, 123, 417–427. [Google Scholar] [CrossRef]
- Yee, N.S.; Li, Q.; Kazi, A.A.; Yee, R.K.; Pennsylvania State University, Hershey, PA, USA. 2014.
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yee, N.S.; Kazi, A.A.; Yee, R.K. Cellular and Developmental Biology of TRPM7 Channel-Kinase: Implicated Roles in Cancer. Cells 2014, 3, 751-777. https://doi.org/10.3390/cells3030751
Yee NS, Kazi AA, Yee RK. Cellular and Developmental Biology of TRPM7 Channel-Kinase: Implicated Roles in Cancer. Cells. 2014; 3(3):751-777. https://doi.org/10.3390/cells3030751
Chicago/Turabian StyleYee, Nelson S., Abid A. Kazi, and Rosemary K. Yee. 2014. "Cellular and Developmental Biology of TRPM7 Channel-Kinase: Implicated Roles in Cancer" Cells 3, no. 3: 751-777. https://doi.org/10.3390/cells3030751