Opposite Nuclear Dynamics of Two FRH-Dominated Frequency Proteins Orchestrate Non-Rhythmic Conidiation in Beauveria bassiana
Abstract
:1. Introduction
2. Materials and Methods
2.1. Identification and Bioinformatic Analysis of FRQ and FRH Proteins in B. bassiana
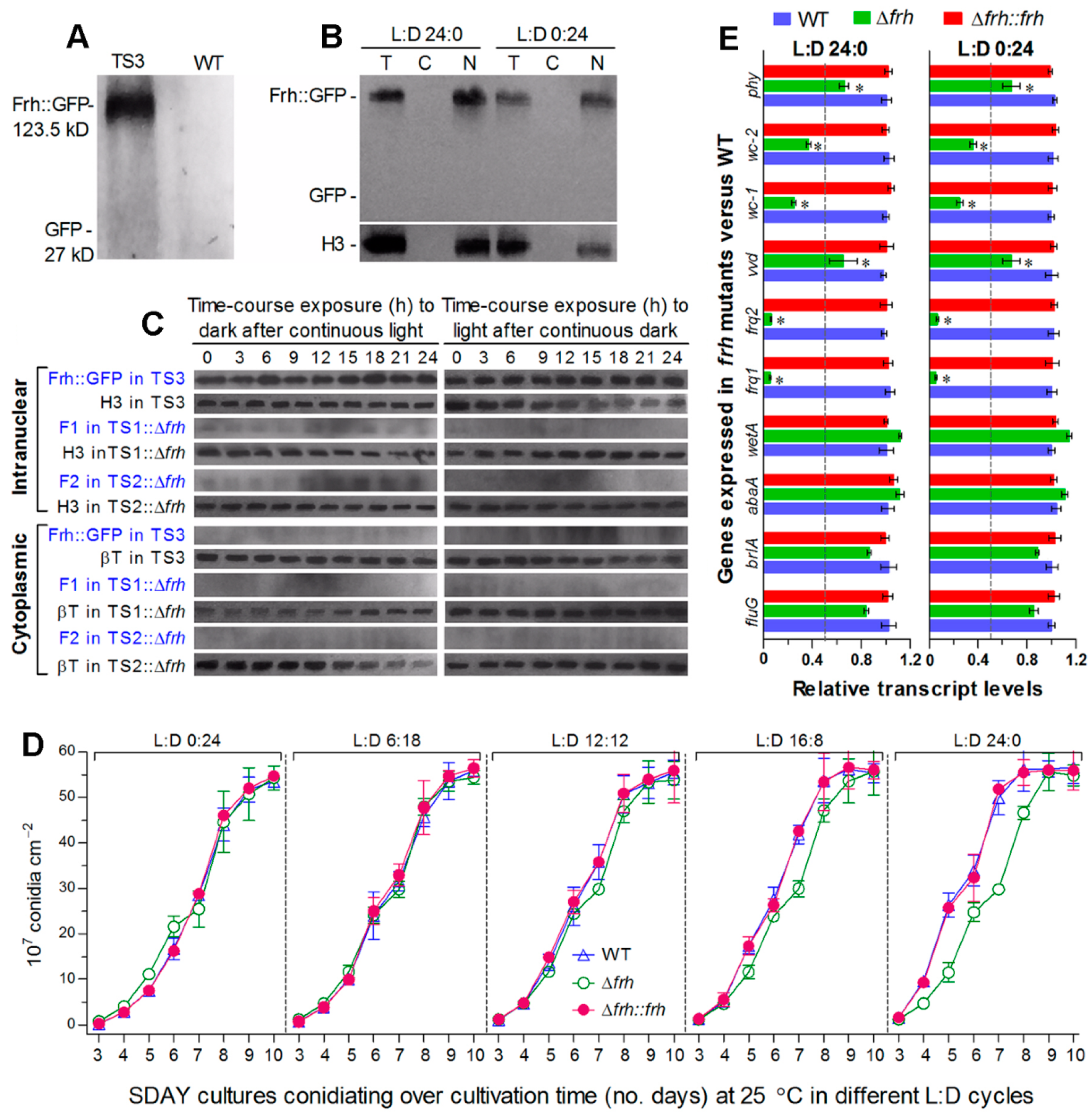
2.2. Generation of frq1, frq2, and frh Mutants
Subcellular Localization of Frq1, Frq2, and FRH
2.3. Time-Course Western Blot Experiments
2.4. Assessment of Conidiation Capacity in Response to Daylight Length
2.5. Transcriptional Profiling
3. Results
3.1. Sequence Features of Frq1, Frq2, and FRH in B. bassiana
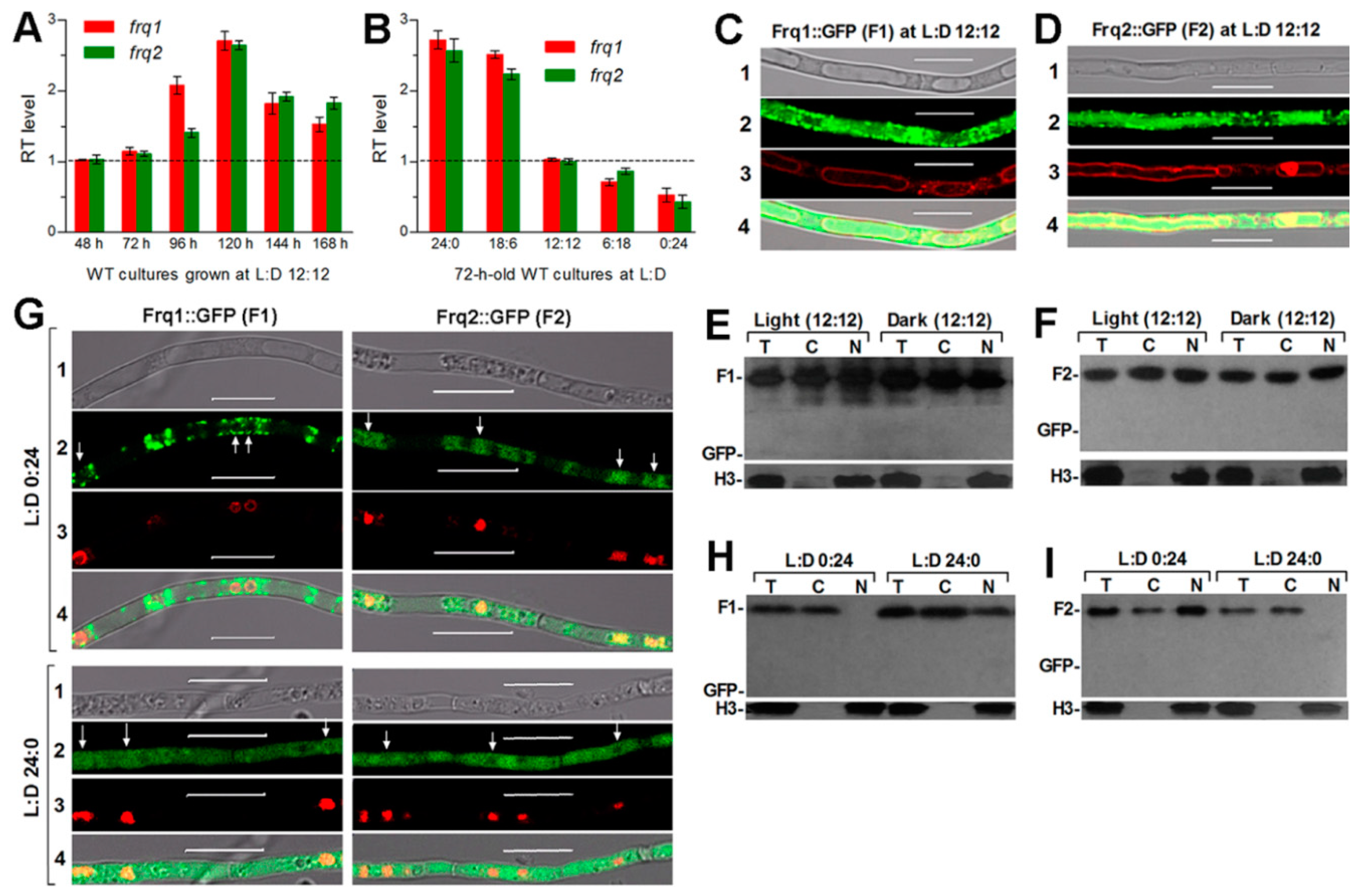
3.2. Expression of Frq1 and Frq2 Is Mutually Independent
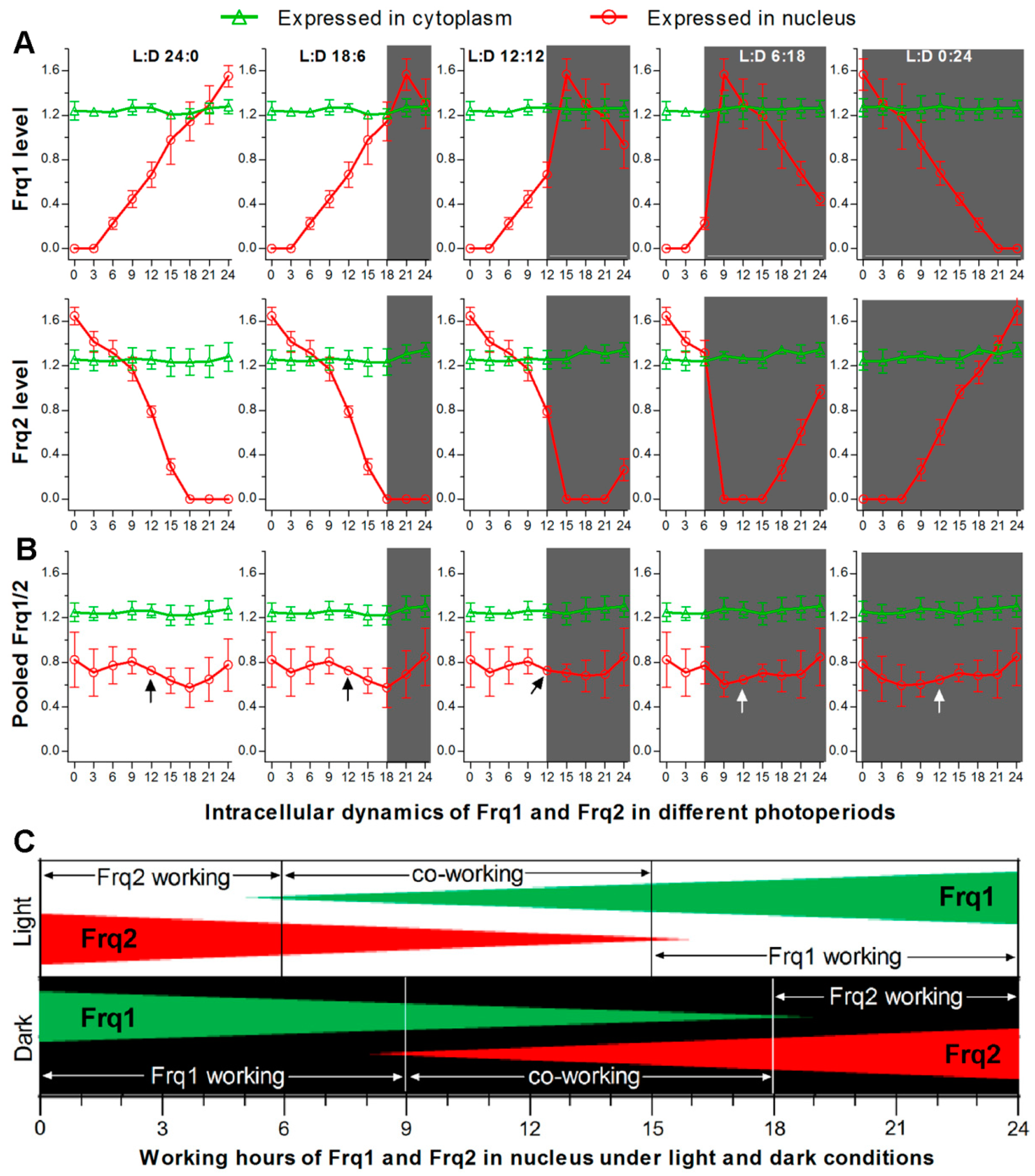
3.3. Frq1 and Frq2 Show Light-Dependent Opposite Dynamics in Nucleus and Light-Independent High-Level Stability in Cytoplasm
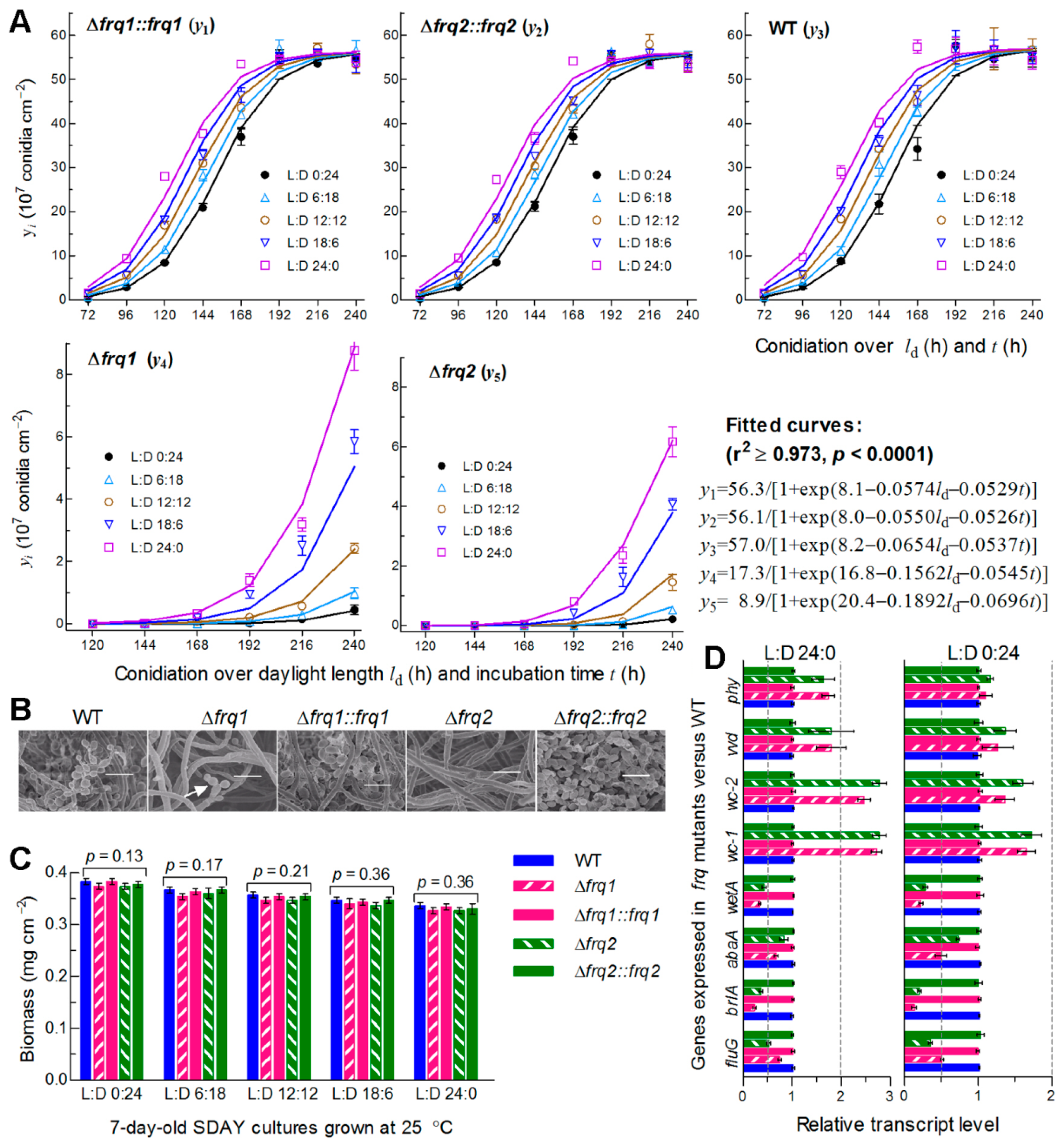
3.4. Frq1 and Frq2 Are Essential for Rapid Conidiation in Different Photoperiods
3.5. FRH Is Required for Nuclear Dynamics and Cytoplasmic Stability of Frq1 and Frq2
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Otamendi, A.; Espeso, E.A.; Etxebeste, O. Identification and characterization of Aspergillus nidulans mutants impaired in asexual development under phosphate stress. Cells 2019, 8, 1520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Etxebeste, O.; Garzia, A.; Espeso, E.A.; Ugalde, U. Aspergillus nidulans asexual development: Making the most of cellular modules. Trends Microbiol. 2010, 18, 569–576. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.S.; Yu, J.H. Genetic control of asexual sporulation in filamentous fungi. Curr. Opin. Microbiol. 2012, 15, 669–677. [Google Scholar] [CrossRef] [PubMed]
- De Faria, M.R.; Wraight, S.P. Mycoinsecticides and mycoacaricides: A comprehensive list with worldwide coverage and international classification of formulation types. Biol. Control 2007, 43, 237–256. [Google Scholar] [CrossRef]
- Wang, C.S.; Feng, M.G. Advances in fundamental and applied studies in China of fungal biocontrol agents for use against arthropod pests. Biol. Control 2014, 68, 128–135. [Google Scholar] [CrossRef]
- Feng, M.G.; Poprawski, T.J.; Khachatourians, G.G. Production, formulation and application of the entomopathogenic fungus Beauveria bassiana for insect control: Current status. Biocontrol Sci. Technol. 1994, 4, 3–34. [Google Scholar] [CrossRef]
- Zhang, A.X.; Mouhoumed, A.Z.; Tong, S.M.; Ying, S.H.; Feng, M.G. BrlA and AbaA govern virulence- required dimorphic switch, conidiation and pathogenicity in a fungal insect pathogen. mSystems 2019, 4, e00140-19. [Google Scholar] [CrossRef] [Green Version]
- Li, F.; Shi, H.Q.; Ying, S.H.; Feng, M.G. WetA and VosA are distinct regulators of conidiation capacity, conidial quality, and biological control potential of a fungal insect pathogen. Appl. Microbiol. Biotechnol. 2015, 99, 10069–10081. [Google Scholar] [CrossRef]
- Tong, S.M.; Zhang, A.X.; Guo, C.T.; Ying, S.H.; Feng, M.G. Daylight length-dependent translocation of VIVID photoreceptor in cells and its essential role in conidiation and virulence of Beauveria bassiana. Environ. Microbiol. 2018, 20, 169–185. [Google Scholar] [CrossRef]
- Zhu, J.; Ying, S.H.; Feng, M.G. The Na+/H+ antiporter Nhx1 controls vacuolar fusion indispensible for the life cycle in vitro and in vivo of a fungal insect pathogen. Environ. Microbiol. 2016, 18, 3884–3895. [Google Scholar] [CrossRef]
- Chu, Z.J.; Sun, H.H.; Zhu, X.G.; Ying, S.H.; Feng, M.G. Discovery of a new intravacuolar protein required for the autophagy, development and virulence of Beauveria bassiana. Environ. Microbiol. 2017, 19, 2806–2818. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q.; Wang, J.J.; Fu, B.; Ying, S.H.; Feng, M.G. Gcn5-dependent histone H3 acetylation and gene activity is required for the asexual development and virulence of Beauveria bassiana. Environ. Microbiol. 2018, 20, 1484–1497. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.J.; Cai, Q.; Qiu, L.; Ying, S.H.; Feng, M.G. The histone acetyltransferase Mst2 sustains the biological control potential of a fungal insect pathogen through transcriptional regulation. Appl. Microbiol. Biotechnol. 2018, 102, 1343–1355. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q.; Tong, S.M.; Shao, W.; Ying, S.H.; Feng, M.G. Pleiotropic effects of the histone deacetylase Hos2 linked to H4-K16 deacetylation, H3-K56 acetylation and H2A-S129 phosphorylation in Beauveria bassiana. Cell. Microbiol. 2018, 20, e12839. [Google Scholar] [CrossRef]
- Cai, Q.; Wang, Z.K.; Shao, W.; Ying, S.H.; Feng, M.G. Essential role of Rpd3-dependent lysine modification in the growth, development and virulence of Beauveria bassiana. Environ. Microbiol. 2018, 20, 1590–1606. [Google Scholar] [CrossRef]
- Wang, Z.K.; Cai, Q.; Tong, S.M.; Ying, S.H.; Feng, M.G. C-terminal Ser/Thr residues are vital for the regulatory role of Ste7 in the asexual cycle and virulence of Beauveria bassiana. Appl. Microbiol. Biotechnol. 2018, 102, 6973–6986. [Google Scholar] [CrossRef]
- Dunlap, J.C. Molecular bases for circadian clocks. Cell 1999, 96, 271–290. [Google Scholar] [CrossRef] [Green Version]
- Baker, C.L.; Loros, J.J.; Dunlap, J.C. The circadian clock of Neurospora crassa. FEMS Microbiol. Rev. 2012, 36, 95–110. [Google Scholar] [CrossRef] [Green Version]
- Hurley, J.M.; Loros, J.J.; Dunlap, J.C. Circadian oscillators: Around the transcription-translation feedback loop and on to output. Trends Biochem. Sci. 2016, 41, 834–846. [Google Scholar] [CrossRef] [Green Version]
- He, Q.; Cheng, P.; Yang, Y.H.; Wang, L.X.; Gardner, K.H.; Liu, Y. White collar-1, a DNA binding transcription factor and a light sensor. Science 2002, 297, 840–843. [Google Scholar] [CrossRef]
- Froehlich, A.C.; Liu, Y.; Loros, J.J.; Dunlap, J.C. White Collar-1, a circadian blue light photoreceptor, binding to the frequency promoter. Science 2002, 297, 815–819. [Google Scholar] [CrossRef] [PubMed]
- De Paula, R.M.; Lewis, Z.A.; Greene, A.V.; Seo, K.S.; Morgan, L.W.; Vitalini, M.W.; Bennett, L.; Gomer, R.H.; Bell-Pedersen, D. Two circadian timing circuits in Neurospora crassa cells share components and regulate distinct rhythmic processes. J. Biol. Rhythm. 2006, 21, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.; He, Q.; Wang, L.; Liu, Y. Regulation of the Neurospora circadian clock by an RNA helicase. Gene Dev. 2005, 19, 234–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, C.; Loros, J.J.; Dunlap, J.C. Nuclear localization is required for function of the essential clock protein FRQ. EMBO J. 1998, 17, 1228–1235. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Garceau, N.Y.; Loros, J.J.; Dunlap, J.C. Thermally regulated translational control of FRQ mediates aspects of temperature responses in the Neurospora circadian clock. Cell 1997, 89, 477–486. [Google Scholar] [CrossRef] [Green Version]
- Colot, H.V.; Loros, J.J.; Dunlap, J.C. Temperature-modulated alternative splicing and promoter use in the circadian clock gene frequency. Mol. Biol. Cell 2005, 12, 5563–5571. [Google Scholar] [CrossRef] [Green Version]
- Diernfellner, A.; Colot, H.V.; Dintsis, O.; Loros, J.J.; Dunlap, J.C.; Brunner, M. Long and short isoforms of Neurospora clock protein FRQ support temperature-compensated circadian rhythms. FEBS Lett. 2007, 581, 5759–5764. [Google Scholar] [CrossRef] [Green Version]
- Diernfellner, A.C.; Querfurth, C.; Salazar, C.; Hofer, T.; Brunner, M. Phosphorylation modulates rapid nucleocytoplasmic shuttling and cytoplasmic accumulation of Neurospora clock protein FRQ on a circadian time scale. Gene Dev. 2009, 23, 2192–2200. [Google Scholar] [CrossRef] [Green Version]
- Crosthwaite, S.K.; Dunlap, J.C.; Loros, J.J. Neurospora wc-1 and wc-2: Transcription, photoresponses, and the origins of circadian rhythmicity. Science 1997, 276, 763–769. [Google Scholar] [CrossRef]
- Froehlich, A.C.; Loros, J.J.; Dunlap, J.C. Rhythmic binding of a WHITE COLLAR-containing complex to the frequency promoter is inhibited by FREQUENCY. Proc. Natl. Acad. Sci. USA 2003, 100, 5914–5919. [Google Scholar] [CrossRef] [Green Version]
- Cheng, P.; Yang, Y.; Heintzen, C.; Liu, Y. Coiled-coil domain-mediated FRQ-FRQ interaction is essential for its circadian clock function in Neurospora. EMBO J. 2001, 20, 101–108. [Google Scholar] [CrossRef] [Green Version]
- Cheng, P.; Yang, Y.; Liu, Y. Interlocked feedback loops contribute to the robustness of the Neurospora circadian clock. Proc. Natl. Acad. Sci. USA 2001, 98, 7408–7413. [Google Scholar] [CrossRef] [Green Version]
- Aronson, B.D.; Johnson, K.A.; Loros, J.J.; Dunlap, J.C. Negative feedback defining a circadian clock: Autoregulation of the clock gene frequency. Science 1994, 263, 1578–1584. [Google Scholar] [CrossRef]
- Denault, D.L.; Loros, J.J.; Dunlap, J.C. WC-2 mediates WC-1–FRQ interaction within the PAS protein-linked circadian feedback loop of Neurospora. EMBO J. 2001, 20, 109–117. [Google Scholar] [CrossRef] [Green Version]
- Hong, C.I.; Ruoff, P.; Loros, J.J.; Dunlap, J.C. Closing the circadian negative feedback loop: FRQ-dependent clearance of WC-1 from the nucleus. Gene Dev. 2008, 22, 3196–3204. [Google Scholar] [CrossRef] [Green Version]
- Baker, C.L.; Kettenbach, A.N.; Loros, J.J.; Gerber, S.A.; Dunlap, J.C. Quantitative proteomics reveals a dynamic interactome and phase-specific phosphorylation in the Neurospora circadian clock. Mol. Cell 2009, 34, 354–363. [Google Scholar] [CrossRef] [Green Version]
- He, Q.; Cha, J.; Lee, H.C.; Yang, Y.; Liu, Y. CKI and CKII mediate the FREQUENCY-dependent phosphorylation of the WHITE COLLAR complex to close the Neurospora circadian negative feedback loop. Gene Dev. 2006, 20, 2552–2565. [Google Scholar] [CrossRef] [Green Version]
- Schafmeier, T.; Haase, A.; Kaldi, K.; Scholz, J.; Fuchs, M.; Brunner, M. Transcriptional feedback of Neurospora circadian clock gene by phosphorylation-dependent inactivation of its transcription factor. Cell 2005, 122, 235–246. [Google Scholar] [CrossRef] [Green Version]
- Schafmeier, T.; Diernfellner, A.; Schafer, A.; Dintsis, O.; Neiss, A.; Brunner, M. Circadian activity and abundance rhythms of the Neurospora clock transcription factor WCC associated with rapid nucleo-cytoplasmic shuttling. Gene Dev. 2008, 22, 3397–3402. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.; Kettenbach, A.N.; Zhou, X.Y.; Loros, J.J.; Dunlap, J.C. The phospho-code determining circadian feedback loop closure and output in Neurospora. Mol. Cell 2019, 74, 771–784. [Google Scholar] [CrossRef]
- Guo, J.H.; Cheng, P.; Liu, Y. Functional significance of FRH in regulating the phosphorylation and stability of Neurospora circadian clock protein FRQ. J. Biol. Chem. 2010, 285, 11508–11515. [Google Scholar] [CrossRef] [Green Version]
- Cha, J.; Yuan, H.Y.; Liu, Y. Regulation of the activity and cellular localization of the circadian clock protein FRQ. J. Biol. Chem. 2011, 286, 11469–11478. [Google Scholar] [CrossRef] [Green Version]
- Shi, M.; Collett, M.; Loros, J.J.; Dunlap, J.C. FRQ-interacting RNA helicase mediates negative and positive feedback in the Neurospora circadian clock. Genetics 2010, 184, 351–361. [Google Scholar] [CrossRef] [Green Version]
- Hurley, J.M.; Larrondo, L.F.; Loros, J.J.; Dunlap, J.C. Conserved RNA helicase FRH acts nonenzymatically to support the intrinsically disordered Neurospora clock protein FRQ. Mol. Cell 2013, 52, 832–843. [Google Scholar] [CrossRef] [Green Version]
- Lauinger, L.; Diernfellner, A.; Falk, S.; Brunner, M. The RNA helicase FRH is an ATP-dependent regulator of CK1a in the circadian clock of Neurospora crassa. Nat. Commun. 2014, 5, 3598. [Google Scholar] [CrossRef] [Green Version]
- Conrad, K.S.; Hurley, J.M.; Widom, J.; Ringelberg, C.S.; Loros, J.J.; Dunlap, J.C.; Crane, B.R. Structure of the frequency-interacting RNA helicase: A protein interaction hub for the circadian clock. EMBO J. 2016, 35, 1707–1719. [Google Scholar] [CrossRef] [Green Version]
- Greene, A.V.; Keller, N.; Haas, H.; Bell-Pedersen, D. A circadian oscillator in Aspergillus spp. regulates daily development and gene expression. Eukaryot. Cell 2003, 2, 231–237. [Google Scholar] [CrossRef] [Green Version]
- Hevia, M.A.; Canessa, P.; Mueller-Esparza, H.; Larrondo, L.F. A circadian oscillator in the fungus Botrytis cinerea regulates virulence when infecting Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2015, 112, 8744–8749. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Singh, P.; Park, J.; Park, S.; Friedman, A.; Zheng, T.; Lee, Y.-H.; Lee, K. Genetic and molecular characterization of a blue light photoreceptor MGWC-1 in Magnaporthe oryzae. Fungal Genet. Biol. 2011, 48, 400–407. [Google Scholar] [CrossRef]
- Salichos, L.; Rokas, A. The diversity and evolution of circadian clock proteins in fungi. Mycologia 2010, 102, 269–278. [Google Scholar] [CrossRef] [Green Version]
- Xiao, G.H.; Ying, S.H.; Zheng, P.; Wang, Z.L.; Zhang, S.W.; Xie, X.Q.; Shang, Y.F.; Zheng, H.J.; Zhou, Y.; St. Leger, R.J.; et al. Genomic perspectives on the evolution of fungal entomopathogenicity in Beauveria bassiana. Sci. Rep. 2012, 2, 483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, L.; Wang, J.J.; Chu, Z.J.; Ying, S.H.; Feng, M.G. Phytochrome controls conidiation in response to red/far-red light and daylight length and regulates multi-stress tolerance in Beauveria bassiana. Environ. Microbiol. 2014, 16, 2316–2328. [Google Scholar] [CrossRef] [PubMed]
- Ying, S.H.; Feng, M.G.; Keyhani, N.O. Use of uridine auxotrophy (ura3) for markerless transformation of the mycoinsecticide Beauveria bassiana. Appl. Microbiol. Biotechnol. 2013, 97, 3017–3025. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Z.K.; Sun, H.H.; Ying, S.H.; Feng, M.G. Characterization of the Hog1 MAPK pathway in the entomopathogenic fungus Beauveria bassiana. Environ. Microbiol. 2017, 19, 1808–1821. [Google Scholar] [CrossRef]
- Zhang, L.B.; Tang, L.; Ying, S.H.; Feng, M.G. Two eisosome proteins play opposite roles in autophagic control and sustain cell integrity, function and pathogenicity in Beauveria bassiana. Environ. Microbiol. 2017, 19, 2037–2052. [Google Scholar] [CrossRef]
- Xie, X.Q.; Guan, Y.; Ying, S.H.; Feng, M.G. Differentiated functions of Ras1 and Ras2 proteins in regulating the germination, growth, conidiation, multi-stress tolerance and virulence of Beauveria bassiana. Environ. Microbiol. 2013, 15, 447–462. [Google Scholar] [CrossRef]
- Ying, S.H.; Feng, M.G. Novel blastospore-based transformation system for integration of phosphinothricin resistance and green fluorescence protein genes into Beauveria bassiana. Appl. Microbiol. Biotechnol. 2006, 72, 206–210. [Google Scholar] [CrossRef]
- Tong, S.M.; Wang, D.Y.; Gao, B.J.; Ying, S.H.; Feng, M.G. The DUF1996 and WSC domain-containing protein Wsc1I acts as a novel sensor of stress cues for Hog1 activation in Beauveria bassiana. Cell. Microbiol. 2019, 21, e13100. [Google Scholar] [CrossRef]
- Seo, J.A.; Guan, Y.; Yu, J.H. FluG-dependent asexual development in Aspergillus nidulans occurs via derepression. Genetics 2006, 172, 1535–1544. [Google Scholar] [CrossRef] [Green Version]
- Diernfellner, A.C.; Schafmeier, T. Phosphorylations: Making the Neurospora crassa circadian clock tick. FEBS Lett. 2011, 585, 1461. [Google Scholar] [CrossRef] [Green Version]
- Larrondo, L.F.; Olivares-Yanez, C.; Baker, C.L.; Loros, J.J.; Dunlap, J.C. Decoupling circadian clock protein turnover from circadian period determination. Science 2015, 347, 6221. [Google Scholar] [CrossRef] [Green Version]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tong, S.-M.; Wang, D.-Y.; Cai, Q.; Ying, S.-H.; Feng, M.-G. Opposite Nuclear Dynamics of Two FRH-Dominated Frequency Proteins Orchestrate Non-Rhythmic Conidiation in Beauveria bassiana. Cells 2020, 9, 626. https://doi.org/10.3390/cells9030626
Tong S-M, Wang D-Y, Cai Q, Ying S-H, Feng M-G. Opposite Nuclear Dynamics of Two FRH-Dominated Frequency Proteins Orchestrate Non-Rhythmic Conidiation in Beauveria bassiana. Cells. 2020; 9(3):626. https://doi.org/10.3390/cells9030626
Chicago/Turabian StyleTong, Sen-Miao, Ding-Yi Wang, Qing Cai, Sheng-Hua Ying, and Ming-Guang Feng. 2020. "Opposite Nuclear Dynamics of Two FRH-Dominated Frequency Proteins Orchestrate Non-Rhythmic Conidiation in Beauveria bassiana" Cells 9, no. 3: 626. https://doi.org/10.3390/cells9030626








