Role of Wnt Signaling During In-Vitro Bovine Blastocyst Development and Maturation in Synergism with PPARδ Signaling
Abstract
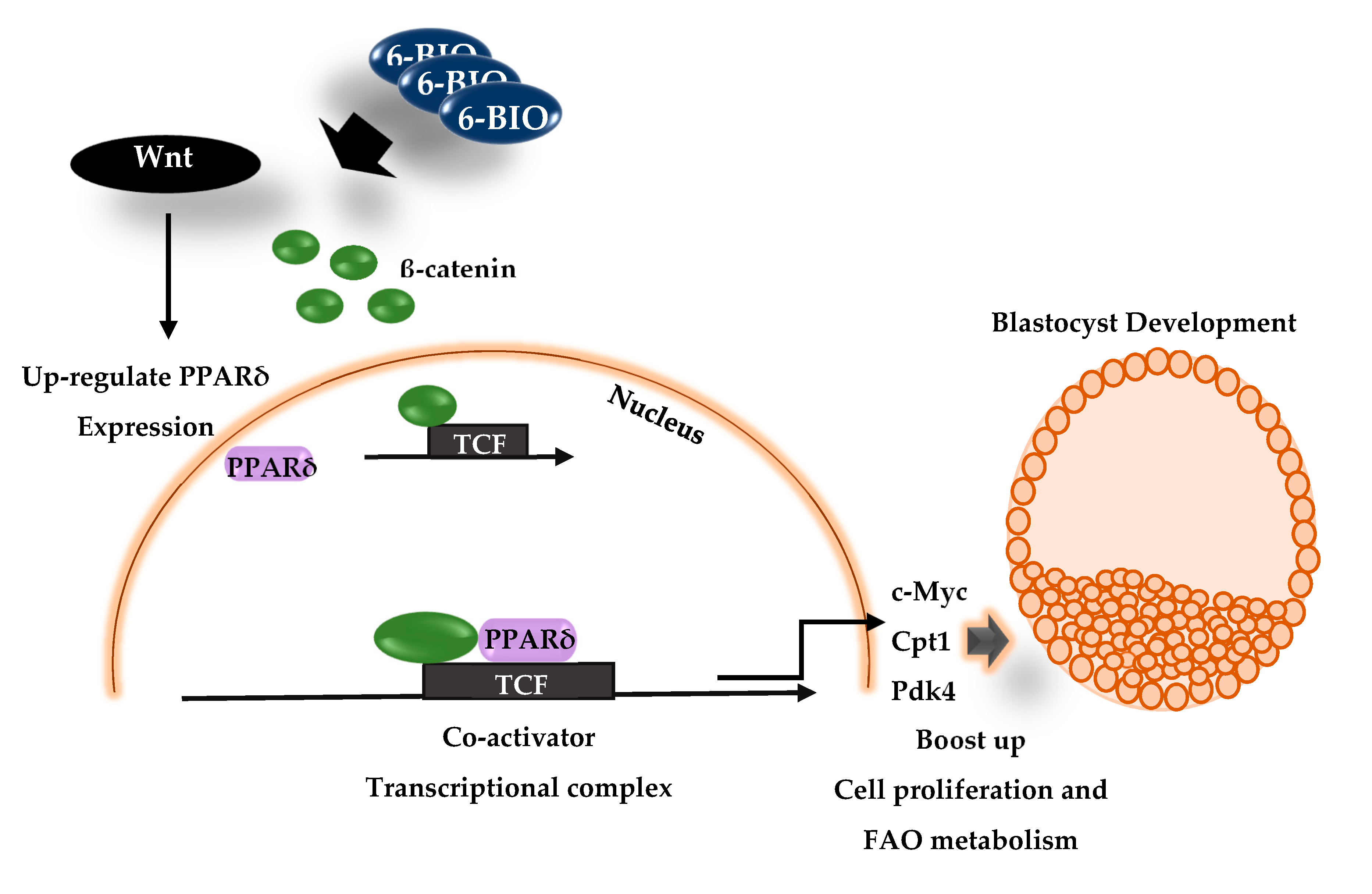
:1. Introduction
2. Materials and Methods
2.1. Drug Treatment for Wnt Stimulation
2.2. Drug Treatment for PPARδ Inhibition
2.3. Aspiration of Oocyte and In-Vitro Maturation
2.4. In-Vitro Fertilization and In-Vitro Culture of Embryos
2.5. RNA Extraction and Quantitative Real Time PCR (q-RT-PCR) Analysis
2.6. Immunofluorescence Analysis
2.7. Quantification of Lipid Content by Fluorescent Probe Nile Red
2.8. Invasion Assay
2.9. Statistical Analysis
3. Results
3.1. Augmentation of Canonical Wnt Activity via 6-Bio Treatment Enhances Bovine BL Development and Hatching
3.2. Effect of 6-Bio Treatment on Proliferation and Lineage Specification during BL Development
3.3. Exogenous Stimulation of Canonical Wnt Activity Induces PPARδ Signaling during Bovine Embryonic Development
3.4. Wnt Regulate BL Development by Sustaining Proliferative Signaling via Modulating PPARδ Expression
3.5. Wnt Promote BL Development and Hatching via Regulating FAO Metabolism through PPARδ Signaling
3.6. Canonical Wnt Activation in Co-ordination with PPARδ Reduces Lipid Content and Enhances BL Implantation Potential
3.7. Exogenous Stimulation of Canonical Wnt Activity Promote Co-localization of PPARδ with β-Catenin during Bovine Embryonic Development
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Parks, J.C.; McCallie, B.R.; Janesch, A.M.; Schoolcraft, W.B.; Katz-Jaffe, M.G. Blastocyst gene expression correlates with implantation potential. Fertil. Steril. 2011, 95, 1367–1372. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yan, J. Update of Wnt signaling in implantation and decidualization. Reprod. Med. Biol. 2016, 15, 95–105. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, B.T.; Tamai, K.; He, X. Wnt/beta-catenin signaling: Components, mechanisms, and diseases. Dev. Cell 2009, 17, 9–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Logan, C.Y.; Nusse, R. The Wnt signaling pathway in development and disease. Annu. Rev. Cell Dev. Biol. 2004, 20, 781–810. [Google Scholar] [CrossRef] [Green Version]
- Clevers, H.; Nusse, R. Wnt/beta-catenin signaling and disease. Cell 2012, 149, 1192–1205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Amerongen, R.; Nusse, R. Towards an integrated view of Wnt signaling in development. Development 2009, 136, 3205–3214. [Google Scholar] [CrossRef] [Green Version]
- Komiya, Y.; Habas, R. Wnt signal transduction pathways. Organogenesis 2008, 4, 68–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Denicol, A.C.; Dobbs, K.B.; McLean, K.M.; Carambula, S.F.; Loureiro, B.; Hansen, P.J. Canonical WNT signaling regulates development of bovine embryos to the blastocyst stage. Sci. Rep. 2013, 3, 1266. [Google Scholar] [CrossRef] [Green Version]
- Tribulo, P.; Leao, B.; Lehloenya, K.C.; Mingoti, G.Z.; Hansen, P.J. Consequences of endogenous and exogenous WNT signaling for development of the preimplantation bovine embryo. Biol. Reprod. 2017, 96, 1129–1141. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.T.; Piotrowska, K.; Ciemerych, M.A.; Milenkovic, L.; Scott, M.P.; Davis, R.W.; Zernicka-Goetz, M. A genome-wide study of gene activity reveals developmental signaling pathways in the preimplantation mouse embryo. Dev. Cell 2004, 6, 133–144. [Google Scholar] [CrossRef] [Green Version]
- Lloyd, S.; Fleming, T.P.; Collins, J.E. Expression of Wnt genes during mouse preimplantation development. Gene Expr. Patterns 2003, 3, 309–312. [Google Scholar] [CrossRef]
- Kemp, C.; Willems, E.; Abdo, S.; Lambiv, L.; Leyns, L. Expression of all Wnt genes and their secreted antagonists during mouse blastocyst and postimplantation development. Dev. Dyn. 2005, 233, 1064–1075. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Tranguch, S.; Jia, X.; Zhang, H.; Das, S.K.; Dey, S.K.; Kuo, C.J.; Wang, H. Inactivation of nuclear Wnt-beta-catenin signaling limits blastocyst competency for implantation. Development 2008, 135, 717–727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, J.C.; Wun, W.S.; Goldsby, J.S.; Wun, I.C.; Noorhasan, D.; Wu, K.K. Stimulation of embryo hatching and implantation by prostacyclin and peroxisome proliferator-activated receptor delta activation: Implication in IVF. Hum. Reprod. 2007, 22, 807–814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, J.; Lu, W.F.; Liang, S.; Choi, J.W.; Kim, N.H.; Cui, X.S. Peroxisome proliferator-activated receptor delta improves porcine blastocyst hatching via the regulation of fatty acid oxidation. Theriogenology 2017, 90, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Blitek, A.; Szymanska, M. Regulation of expression and role of peroxisome proliferator-activated receptors (PPARs) in luminal epithelial and stromal cells of the porcine endometrium. Theriogenology 2019, 127, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, A.; Yokoyama, Y.; Umemoto, M.; Futagami, M.; Sakamoto, T.; Bing, X.; Mizunuma, H. Clinical implication of expression of cyclooxygenase-2 and peroxisome proliferator activated-receptor gamma in epithelial ovarian tumours. Br. J. Cancer 2004, 91, 633–638. [Google Scholar] [CrossRef] [Green Version]
- Jansson, E.A.; Are, A.; Greicius, G.; Kuo, I.C.; Kelly, D.; Arulampalam, V.; Pettersson, S. The Wnt/beta-catenin signaling pathway targets PPARgamma activity in colon cancer cells. Proc. Natl. Acad. Sci. USA 2005, 102, 1460–1465. [Google Scholar] [CrossRef] [Green Version]
- Hwang, I.; Kim, J.; Jeong, S. beta-Catenin and peroxisome proliferator-activated receptor-delta coordinate dynamic chromatin loops for the transcription of vascular endothelial growth factor A gene in colon cancer cells. J. Biol. Chem. 2012, 287, 41364–41373. [Google Scholar] [CrossRef] [Green Version]
- Sethi, J.K.; Vidal-Puig, A. Wnt signalling and the control of cellular metabolism. Biochem. J. 2010, 427, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Aparicio, I.M.; Garcia-Herreros, M.; Fair, T.; Lonergan, P. Identification and regulation of glycogen synthase kinase-3 during bovine embryo development. Reproduction 2010, 140, 83–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sidrat, T.; Kong, R.; Khan, A.A.; Idrees, M.; Xu, L.; Sheikh, M.E.; Joo, M.D.; Lee, K.L.; Kong, I.K. Difference in Developmental Kinetics of Y-Specific Monoclonal Antibody Sorted Male and Female In Vitro Produced Bovine Embryos. Int. J. Mol. Sci. 2020, 21, 244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sen Roy, S.; Seshagiri, P.B. Expression and function of cyclooxygenase-2 is necessary for hamster blastocyst hatching. Mol. Hum. Reprod. 2013, 19, 838–851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, J.C.; Wun, W.S.; Goldsby, J.S.; Matijevic-Aleksic, N.; Wu, K.K. Cyclooxygenase-2-derived endogenous prostacyclin enhances mouse embryo hatching. Hum. Reprod. 2004, 19, 2900–2906. [Google Scholar] [CrossRef] [Green Version]
- Dovizio, M.; Alberti, S.; Sacco, A.; Guillem-Llobat, P.; Schiavone, S.; Maier, T.J.; Steinhilber, D.; Patrignani, P. Novel insights into the regulation of cyclooxygenase-2 expression by platelet-cancer cell cross-talk. Biochem. Soc. Trans. 2015, 43, 707–714. [Google Scholar] [CrossRef] [Green Version]
- Peters, J.M.; Gonzalez, F.J.; Muller, R. Establishing the Role of PPARbeta/delta in Carcinogenesis. Trends Endocrinol. Metab. 2015, 26, 595–607. [Google Scholar] [CrossRef] [Green Version]
- Stephen, R.L.; Gustafsson, M.C.; Jarvis, M.; Tatoud, R.; Marshall, B.R.; Knight, D.; Ehrenborg, E.; Harris, A.L.; Wolf, C.R.; Palmer, C.N. Activation of peroxisome proliferator-activated receptor delta stimulates the proliferation of human breast and prostate cancer cell lines. Cancer Res. 2004, 64, 3162–3170. [Google Scholar] [CrossRef] [Green Version]
- Zuo, X.; Peng, Z.; Moussalli, M.J.; Morris, J.S.; Broaddus, R.R.; Fischer, S.M.; Shureiqi, I. Targeted genetic disruption of peroxisome proliferator-activated receptor-delta and colonic tumorigenesis. J. Natl. Cancer Inst. 2009, 101, 762–767. [Google Scholar] [CrossRef] [Green Version]
- Montag, M.; Koll, B.; Holmes, P.; van der, V. Significance of the number of embryonic cells and the state of the zona pellucida for hatching of mouse blastocysts in vitro versus in vivo. Biol. Reprod. 2000, 62, 1738–1744. [Google Scholar] [CrossRef]
- Dunning, K.R.; Anastasi, M.R.; Zhang, V.J.; Russell, D.L.; Robker, R.L. Regulation of fatty acid oxidation in mouse cumulus-oocyte complexes during maturation and modulation by PPAR agonists. PLoS ONE 2014, 9, e87327. [Google Scholar] [CrossRef] [Green Version]
- Kang, H.J.; Hwang, S.J.; Yoon, J.A.; Jun, J.H.; Lim, H.J.; Yoon, T.K.; Song, H. Activation of peroxisome proliferators-activated receptor delta (PPARdelta) promotes blastocyst hatching in mice. Mol. Hum. Reprod. 2011, 17, 653–660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clevers, H. Wnt/beta-catenin signaling in development and disease. Cell 2006, 127, 469–480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kemler, R.; Hierholzer, A.; Kanzler, B.; Kupping, S.; Hansen, K.; Taketo, M.M.; de Vries, W.N.; Knowles, B.B.; Solter, D. Stabilization of beta-catenin in the mouse zygote leads to premature epithelial to mesenchymal transition in the epiblast. Development 2004, 131, 5817–5824. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Madeja, Z.E.; Hryniewicz, K.; Orsztynowicz, M.; Pawlak, P.; Perkowska, A. WNT/beta-catenin signaling affects cell lineage and pluripotency-specific gene expression in bovine blastocysts: Prospects for bovine embryonic stem cell derivation. Stem Cells Dev. 2015, 24, 2437–2454. [Google Scholar] [CrossRef]
- de Jaime-Soguero, A.; Abreu de Oliveira, W.A.; Lluis, F. The Pleiotropic Effects of the Canonical Wnt Pathway in Early Development and Pluripotency. Genes 2018, 9, 93. [Google Scholar] [CrossRef] [Green Version]
- Blauwkamp, T.A.; Nigam, S.; Ardehali, R.; Weissman, I.L.; Nusse, R. Endogenous Wnt signalling in human embryonic stem cells generates an equilibrium of distinct lineage-specified progenitors. Nat. Commun. 2012, 3, 1070. [Google Scholar] [CrossRef]
- Zhang, X.; Peterson, K.A.; Liu, X.S.; McMahon, A.P.; Ohba, S. Gene regulatory networks mediating canonical Wnt signal-directed control of pluripotency and differentiation in embryo stem cells. Stem Cells 2013, 31, 2667–2679. [Google Scholar] [CrossRef] [Green Version]
- Takao, Y.; Yokota, T.; Koide, H. Beta-catenin up-regulates Nanog expression through interaction with Oct-3/4 in embryonic stem cells. Biochem. Biophys. Res. Commun. 2007, 353, 699–705. [Google Scholar] [CrossRef] [Green Version]
- Madeja, Z.E.; Sosnowski, J.; Hryniewicz, K.; Warzych, E.; Pawlak, P.; Rozwadowska, N.; Plusa, B.; Lechniak, D. Changes in sub-cellular localisation of trophoblast and inner cell mass specific transcription factors during bovine preimplantation development. BMC Dev. Biol. 2013, 13, 32. [Google Scholar] [CrossRef] [Green Version]
- Ralston, A.; Cox, B.J.; Nishioka, N.; Sasaki, H.; Chea, E.; Rugg-Gunn, P.; Guo, G.; Robson, P.; Draper, J.S.; Rossant, J. Gata3 regulates trophoblast development downstream of Tead4 and in parallel to Cdx2. Development 2010, 137, 395–403. [Google Scholar] [CrossRef] [Green Version]
- Yagi, R.; Kohn, M.J.; Karavanova, I.; Kaneko, K.J.; Vullhorst, D.; DePamphilis, M.L.; Buonanno, A. Transcription factor TEAD4 specifies the trophectoderm lineage at the beginning of mammalian development. Development 2007, 134, 3827–3836. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfeffer, P.L.; Pearton, D.J. Trophoblast development. Reproduction 2012, 143, 231–246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vallee, A.; Lecarpentier, Y. Crosstalk Between Peroxisome Proliferator-Activated Receptor Gamma and the Canonical WNT/beta-Catenin Pathway in Chronic Inflammation and Oxidative Stress During Carcinogenesis. Front. Immunol. 2018, 9, 745. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lim, H.; Gupta, R.A.; Ma, W.G.; Paria, B.C.; Moller, D.E.; Morrow, J.D.; DuBois, R.N.; Trzaskos, J.M.; Dey, S.K. Cyclo-oxygenase-2-derived prostacyclin mediates embryo implantation in the mouse via PPARdelta. Genes Dev. 1999, 13, 1561–1574. [Google Scholar] [CrossRef]
- Oey, N.A.; Ruiter, J.P.; Attie-Bitach, T.; Ijlst, L.; Wanders, R.J.; Wijburg, F.A. Fatty acid oxidation in the human fetus: Implications for fetal and adult disease. J. Inherit. Metab. Dis. 2006, 29, 71–75. [Google Scholar] [CrossRef]












| Groups (nM) | Oocytes, n | Speculated Zygotes, n | Cleaved Embryos, % | Total Blastocyst, % | Hatched Blastocyst, % |
|---|---|---|---|---|---|
| Control | 340 | 315 | 245 (77.8 ± 0.5) b | 93 (29.6 ± 0.5) b | 41 (44.9 ± 1.8) b |
| 6-Bio (200) | 295 | 277 | 217 (78.3 ± 0.6) b | 94 (33.9 ± 0.9) c | 53 (55.9 ± 3.4) c |
| 6-Bio (300) | 313 | 290 | 227 (78.3 ± 0.3) b | 99 (34.1 ± 1.1) c | 56 (55.9 ± 3.4) c |
| 6-Bio (400) | 346 | 321 | 254 (78.9 ± 0.4) b | 121 (37.8 ± 0.5) d | 78 (64.3 ± 1.2) c |
| 6-Bio (500) | 165 | 151 | 83 (55.0 ± 1.7) a | 31 (20.8 ± 1.3) a | 7 (22.5 ± 3.2) a |
| Groups | Oocytes, n | Speculated Zygotes, n | Cleaved Embryos, % | Total Blastocyst, % | Hatched Blastocyst, % |
|---|---|---|---|---|---|
| Control | 502 | 481 | 377 (78.5 ± 0.2) c | 140 (29.2 ± 0.5 ) c | 63 (45.0 ± 0.9) c |
| 6-Bio (400 nM) | 521 | 496 | 387 (78.2 ± 0.3) c | 188 (37.8 ± 1.3) d | 115 (61.1 ± 1.6) d |
| Gsk3787 (15 µM) | 453 | 430 | 323 (75.0 ± 0.4) a | 69 (16.1 ± 0.6) a | 5 (7.2 ± 2.5) a |
| 6-Bio + Gsk3787 | 521 | 494 | 378 (76.7 ± 0.4) b | 120 (24.3 ± 0.4 ) b | 43 (35.5 ± 1.2) b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sidrat, T.; Khan, A.A.; Idrees, M.; Joo, M.-D.; Xu, L.; Lee, K.-L.; Kong, I.-K. Role of Wnt Signaling During In-Vitro Bovine Blastocyst Development and Maturation in Synergism with PPARδ Signaling. Cells 2020, 9, 923. https://doi.org/10.3390/cells9040923
Sidrat T, Khan AA, Idrees M, Joo M-D, Xu L, Lee K-L, Kong I-K. Role of Wnt Signaling During In-Vitro Bovine Blastocyst Development and Maturation in Synergism with PPARδ Signaling. Cells. 2020; 9(4):923. https://doi.org/10.3390/cells9040923
Chicago/Turabian StyleSidrat, Tabinda, Abdul Aziz Khan, Muhammad Idrees, Myeong-Don Joo, Lianguang Xu, Kyeong-Lim Lee, and Il-Keun Kong. 2020. "Role of Wnt Signaling During In-Vitro Bovine Blastocyst Development and Maturation in Synergism with PPARδ Signaling" Cells 9, no. 4: 923. https://doi.org/10.3390/cells9040923







