Granulocyte Colony Stimulating Factor (GCSF) Can Attenuate Neuropathic Pain by Suppressing Monocyte Chemoattractant Protein-1 (MCP-1) Expression, through Upregulating the Early MicroRNA-122 Expression in the Dorsal Root Ganglia
Abstract
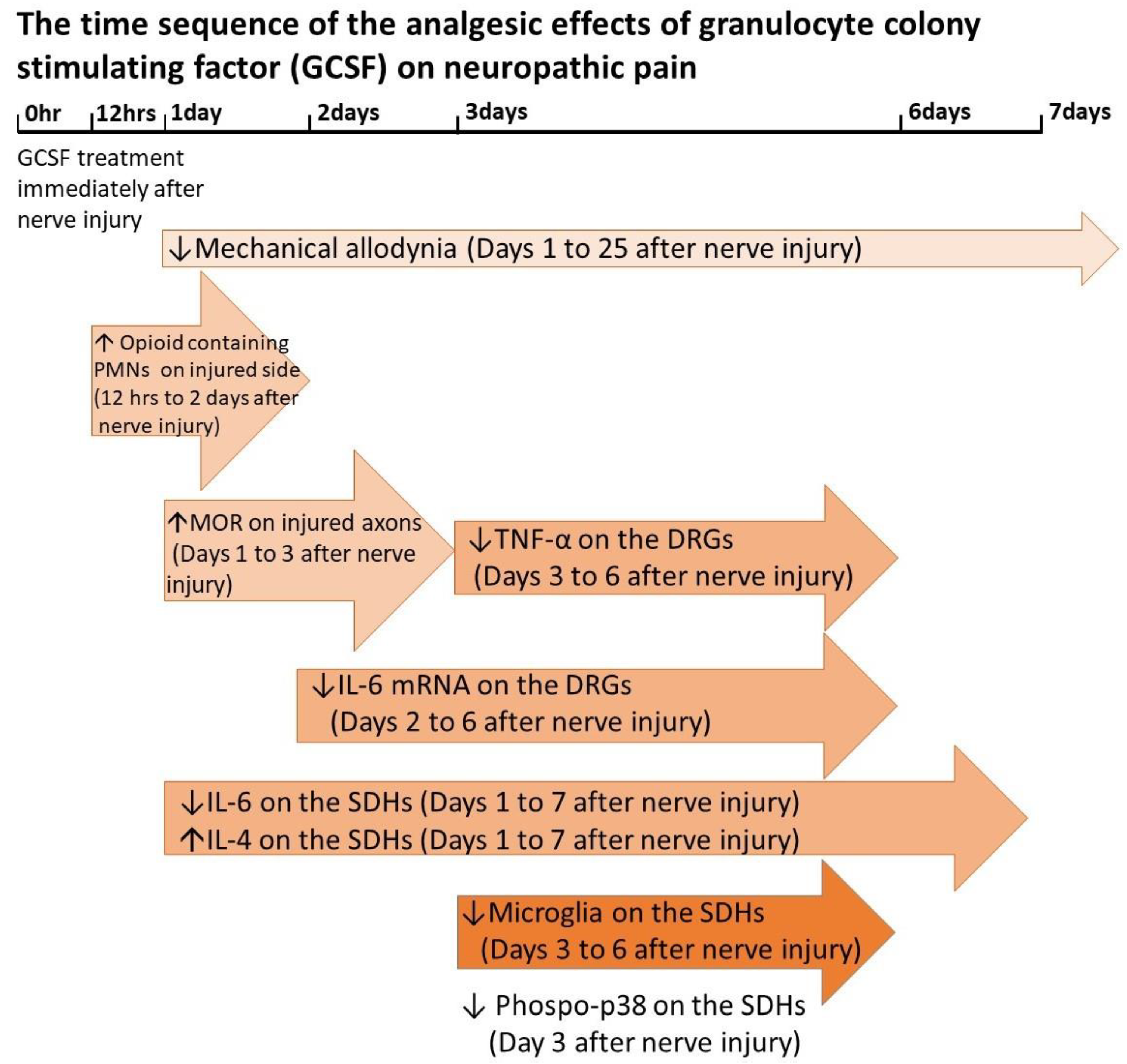
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Surgical Procedure and GCSF Treatment Protocol
2.3. Behavioral Tests for Mechanical Allodynia
2.4. MicroRNA Isolation and Purification
2.5. nCounter Data Analysis
2.6. Enzyme-Linked Immunosorbent Assay (ELISA)
2.7. Immunohistochemistry (IHC) Studies
2.8. Statistical Analysis
3. Results
3.1. Single Early Systemic GCSF Treatment Alleviated Mechanical Allodynia in CCI Rats
3.2. GCSF Upregulated MicroRNA-122 Expression in the DRGs of CCI Rats on the 1st Day after Nerve Injury
3.3. GCSF Decreased MCP-1 Expression in the DRGs of CCI Rats on the 7th Day after Nerve Injury
4. Discussion
4.1. GCSF Upregulated MicroRNA-122 Expression in the DRGs of CCI Rats at the Early Phase after Nerve Injury
4.2. MicroRNA-122 Can Suppress the Pro-Inflammatory Cytokines and MCP-1 Expressions in an Ex Vivo Study
4.3. MCP-1 in the DRGs and SDHs Plays an Important Role to Promote Neuropathic Pain Formation
4.4. GCSF Suppressed MCP-1 Expression in the DRGs to Attenuate Neuropathic Pain
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Chao, P.K.; Lu, K.T.; Lee, Y.L.; Chen, J.C.; Wang, H.L.; Yang, Y.L.; Cheng, M.Y.; Liao, M.F.; Ro, L.S. Early systemic granulocyte-colony stimulating factor treatment attenuates neuropathic pain after peripheral nerve injury. PLoS ONE 2012, 7, e43680. [Google Scholar] [CrossRef] [PubMed]
- Liao, M.F.; Yeh, S.R.; Lo, A.L.; Chao, P.K.; Lee, Y.L.; Hung, Y.H.; Lu, K.T.; Ro, L.S. An early granulocyte colony-stimulating factor treatment attenuates neuropathic pain through activation of mu opioid receptors on the injured nerve. Sci. Rep. 2016, 6, 25490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koda, M.; Furuya, T.; Kato, K.; Mannoji, C.; Hashimoto, M.; Inada, T.; Kamiya, K.; Ota, M.; Maki, S.; Okawa, A.; et al. Delayed granulocyte colony-stimulating factor treatment in rats attenuates mechanical allodynia induced by chronic constriction injury of the sciatic nerve. Spine 2014, 39, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Koda, M.; Takahashi, H.; Sakuma, T.; Inada, T.; Kamiya, K.; Ota, M.; Maki, S.; Okawa, A.; Takahashi, K.; et al. Granulocyte colony-stimulating factor attenuates spinal cord injury-induced mechanical allodynia in adult rats. J. Neurol. Sci. 2015, 355, 79–83. [Google Scholar] [CrossRef]
- Yamazaki, M.; Sakuma, T.; Kato, K.; Furuya, T.; Koda, M. Granulocyte colony-stimulating factor reduced neuropathic pain associated with thoracic compression myelopathy: Report of two cases. J. Spinal Cord Med. 2013, 36, 40–43. [Google Scholar] [CrossRef] [Green Version]
- Kato, K.; Yamazaki, M.; Okawa, A.; Furuya, T.; Sakuma, T.; Takahashi, H.; Kamiya, K.; Inada, T.; Takahashi, K.; Koda, M. Intravenous administration of granulocyte colony-stimulating factor for treating neuropathic pain associated with compression myelopathy: A phase I and IIa clinical trial. Eur. Spine J. 2013, 22, 197–204. [Google Scholar] [CrossRef] [Green Version]
- Baez, A.; Martin-Antonio, B.; Piruat, J.I.; Prats, C.; Alvarez-Laderas, I.; Barbado, M.V.; Carmona, M.; Urbano-Ispizua, A.; Perez-Simon, J.A. Granulocyte colony-stimulating factor produces long-term changes in gene and microRNA expression profiles in CD34+ cells from healthy donors. Haematologica 2014, 99, 243–251. [Google Scholar] [CrossRef] [Green Version]
- He, L.; Hannon, G.J. MicroRNAs: Small RNAs with a big role in gene regulation. Nat. Rev. Genet. 2004, 5, 522–531. [Google Scholar] [CrossRef]
- Niederberger, E.; Kynast, K.; Lotsch, J.; Geisslinger, G. MicroRNAs as new players in the pain game. Pain 2011, 152, 1455–1458. [Google Scholar] [CrossRef]
- Kress, M.; Huttenhofer, A.; Landry, M.; Kuner, R.; Favereaux, A.; Greenberg, D.; Bednarik, J.; Heppenstall, P.; Kronenberg, F.; Malcangio, M.; et al. microRNAs in nociceptive circuits as predictors of future clinical applications. Front. Mol. Neurosci. 2013, 6, 33. [Google Scholar] [CrossRef] [Green Version]
- Elramah, S.; Landry, M.; Favereaux, A. MicroRNAs regulate neuronal plasticity and are involved in pain mechanisms. Front. Cell Neurosci. 2014, 8, 31. [Google Scholar] [CrossRef] [Green Version]
- Bali, K.K.; Kuner, R. Noncoding RNAs: Key molecules in understanding and treating pain. Trends Mol. Med. 2014, 20, 437–448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andersen, H.H.; Duroux, M.; Gazerani, P. MicroRNAs as modulators and biomarkers of inflammatory and neuropathic pain conditions. Neurobiol. Dis. 2014, 71, 159–168. [Google Scholar] [CrossRef] [Green Version]
- Lopez-Gonzalez, M.J.; Landry, M.; Favereaux, A. MicroRNA and chronic pain: From mechanisms to therapeutic potential. Pharmacol. Ther. 2017, 180, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.B.; Zhu, Y.; Chen, B.L.; Song, G.; Peng, M.S.; Hu, H.Y.; Zheng, Y.L.; Chen, C.C.; Yang, J.Z.; Chen, P.J.; et al. Network and pathway-based analysis of microRNA role in neuropathic pain in rat models. J. Cell Mol. Med. 2019, 23, 4534–4544. [Google Scholar] [CrossRef] [PubMed]
- Kynast, K.L.; Russe, O.Q.; Moser, C.V.; Geisslinger, G.; Niederberger, E. Modulation of central nervous system-specific microRNA-124a alters the inflammatory response in the formalin test in mice. Pain 2013, 154, 368–376. [Google Scholar] [CrossRef]
- Ni, J.; Gao, Y.; Gong, S.; Guo, S.; Hisamitsu, T.; Jiang, X. Regulation of mu-opioid type 1 receptors by microRNA134 in dorsal root ganglion neurons following peripheral inflammation. Eur. J. Pain 2013, 17, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liu, F.; Wei, M.; Qiu, Y.; Ma, C.; Shen, L.; Huang, Y. Chronic constriction injury-induced microRNA-146a-5p alleviates neuropathic pain through suppression of IRAK1/TRAF6 signaling pathway. J. Neuroinflamm. 2018, 15, 179. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Shen, L.; Ma, C.; Huang, Y. Differential expression of miRNAs in the nervous system of a rat model of bilateral sciatic nerve chronic constriction injury. Int. J. Mol. Med. 2013, 32, 219–226. [Google Scholar] [CrossRef]
- Sakai, A.; Suzuki, H. Nerve injury-induced upregulation of miR-21 in the primary sensory neurons contributes to neuropathic pain in rats. Biochem. Biophys. Res. Commun. 2013, 435, 176–181. [Google Scholar] [CrossRef]
- Chen, H.P.; Zhou, W.; Kang, L.M.; Yan, H.; Zhang, L.; Xu, B.H.; Cai, W.H. Intrathecal miR-96 inhibits Nav1.3 expression and alleviates neuropathic pain in rat following chronic construction injury. Neurochem. Res. 2014, 39, 76–83. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, H.; Zi, T. Overexpression of microRNA-141 relieves chronic constriction injury-induced neuropathic pain via targeting high-mobility group box 1. Int. J. Mol. Med. 2015, 36, 1433–1439. [Google Scholar] [CrossRef] [Green Version]
- Sun, W.; Zhang, L.; Li, R. Overexpression of miR-206 ameliorates chronic constriction injury-induced neuropathic pain in rats via the MEK/ERK pathway by targeting brain-derived neurotrophic factor. Neurosci. Lett. 2017, 646, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.N.; Yuan, Y.T.; Ye, D.; Kang, L.M.; Wen, J.; Chen, H.P. MiR-183-5p Alleviates Chronic Constriction Injury-Induced Neuropathic Pain Through Inhibition of TREK-1. Neurochem. Res. 2018, 43, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Brandenburger, T.; Johannsen, L.; Prassek, V.; Kuebart, A.; Raile, J.; Wohlfromm, S.; Kohrer, K.; Huhn, R.; Hollmann, M.W.; Hermanns, H. MiR-34a is differentially expressed in dorsal root ganglia in a rat model of chronic neuropathic pain. Neurosci. Lett. 2019, 708, 134365. [Google Scholar] [CrossRef] [PubMed]
- Sakai, A.; Saitow, F.; Maruyama, M.; Miyake, N.; Miyake, K.; Shimada, T.; Okada, T.; Suzuki, H. MicroRNA cluster miR-17-92 regulates multiple functionally related voltage-gated potassium channels in chronic neuropathic pain. Nat. Commun. 2017, 8, 16079. [Google Scholar] [CrossRef] [PubMed]
- Willemen, H.L.; Huo, X.J.; Mao-Ying, Q.L.; Zijlstra, J.; Heijnen, C.J.; Kavelaars, A. MicroRNA-124 as a novel treatment for persistent hyperalgesia. J. Neuroinflamm. 2012, 9, 143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cai, W.; Zhao, Q.; Shao, J.; Zhang, J.; Li, L.; Ren, X.; Su, S.; Bai, Q.; Li, M.; Chen, X.; et al. MicroRNA-182 Alleviates Neuropathic Pain by Regulating Nav1.7 Following Spared Nerve Injury in Rats. Sci. Rep. 2018, 8, 16750. [Google Scholar] [CrossRef] [Green Version]
- Geiss, G.K.; Bumgarner, R.E.; Birditt, B.; Dahl, T.; Dowidar, N.; Dunaway, D.L.; Fell, H.P.; Ferree, S.; George, R.D.; Grogan, T.; et al. Direct multiplexed measurement of gene expression with color-coded probe pairs. Nat. Biotechnol. 2008, 26, 317–325. [Google Scholar] [CrossRef]
- Jopling, C. Liver-specific microRNA-122: Biogenesis and function. RNA Biol. 2012, 9, 137–142. [Google Scholar] [CrossRef] [Green Version]
- Vliegenthart, A.D.B.; Berends, C.; Potter, C.M.J.; Kersaudy-Kerhoas, M.; Dear, J.W. MicroRNA-122 can be measured in capillary blood which facilitates point-of-care testing for drug-induced liver injury. Br. J. Clin. Pharmacol. 2017, 83, 2027–2033. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.L.; Lu, X.L.; Yan, C.Y.; Wan, Q.L.; Cheng, G.C.; Li, Y.M. Circulating miR-122-5p as a potential novel biomarker for diagnosis of acute myocardial infarction. Int. J. Clin. Exp. Pathol. 2015, 8, 16014–16019. [Google Scholar] [PubMed]
- Liu, D.Z.; Jickling, G.C.; Ander, B.P.; Hull, H.; Zhan, X.; Cox, C.; Shroff, N.; Dykstra-Aiello, C.; Stamova, B.; Sharp, F.R. Elevating microRNA-122 in blood improves outcomes after temporary middle cerebral artery occlusion in rats. J. Cereb. Blood Flow Metab. 2016, 36, 1374–1383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, M.; Kanda, T.; Sasaki, R.; Haga, Y.; Jiang, X.; Wu, S.; Nakamoto, S.; Yokosuka, O. MicroRNA-122 Inhibits the Production of Inflammatory Cytokines by Targeting the PKR Activator PACT in Human Hepatic Stellate Cells. PLoS ONE 2015, 10, e0144295. [Google Scholar] [CrossRef] [Green Version]
- Bennett, G.J.; Xie, Y.K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 1988, 33, 87–107. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Guo, N.; Peng, G.; Wang, X.; Han, M.; Raincrow, J.; Chiu, C.H.; Coolen, L.M.; Wenthold, R.J.; Zhao, Z.Q.; et al. Role of SIP30 in the development and maintenance of peripheral nerve injury-induced neuropathic pain. Pain 2009, 146, 130–140. [Google Scholar] [CrossRef] [Green Version]
- Kiguchi, N.; Kobayashi, Y.; Kishioka, S. Chemokines and cytokines in neuroinflammation leading to neuropathic pain. Curr. Opin. Pharmacol. 2012, 12, 55–61. [Google Scholar] [CrossRef]
- Miller, R.J.; Jung, H.; Bhangoo, S.K.; White, F.A. Cytokine and chemokine regulation of sensory neuron function. Handb Exp. Pharmacol. 2009, 417–449. [Google Scholar] [CrossRef] [Green Version]
- Jeon, S.M.; Lee, K.M.; Cho, H.J. Expression of monocyte chemoattractant protein-1 in rat dorsal root ganglia and spinal cord in experimental models of neuropathic pain. Brain Res. 2009, 1251, 103–111. [Google Scholar] [CrossRef]
- Thacker, M.A.; Clark, A.K.; Bishop, T.; Grist, J.; Yip, P.K.; Moon, L.D.; Thompson, S.W.; Marchand, F.; McMahon, S.B. CCL2 is a key mediator of microglia activation in neuropathic pain states. Eur. J. Pain 2009, 13, 263–272. [Google Scholar] [CrossRef]
- Zhang, H.; Boyette-Davis, J.A.; Kosturakis, A.K.; Li, Y.; Yoon, S.Y.; Walters, E.T.; Dougherty, P.M. Induction of monocyte chemoattractant protein-1 (MCP-1) and its receptor CCR2 in primary sensory neurons contributes to paclitaxel-induced peripheral neuropathy. J. Pain 2013, 14, 1031–1044. [Google Scholar] [CrossRef] [Green Version]
- Jeon, S.M.; Lee, K.M.; Park, E.S.; Jeon, Y.H.; Cho, H.J. Monocyte chemoattractant protein-1 immunoreactivity in sensory ganglia and hindpaw after adjuvant injection. Neuroreport 2008, 19, 183–186. [Google Scholar] [CrossRef]
- Sun, J.H.; Yang, B.; Donnelly, D.F.; Ma, C.; LaMotte, R.H. MCP-1 enhances excitability of nociceptive neurons in chronically compressed dorsal root ganglia. J. Neurophysiol. 2006, 96, 2189–2199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arruda, J.L.; Sweitzer, S.; Rutkowski, M.D.; DeLeo, J.A. Intrathecal anti-IL-6 antibody and IgG attenuates peripheral nerve injury-induced mechanical allodynia in the rat: Possible immune modulation in neuropathic pain. Brain Res. 2000, 879, 216–225. [Google Scholar] [CrossRef]
- Zavala, F.; Abad, S.; Ezine, S.; Taupin, V.; Masson, A.; Bach, J.F. G-CSF therapy of ongoing experimental allergic encephalomyelitis via chemokine- and cytokine-based immune deviation. J. Immunol. 2002, 168, 2011–2019. [Google Scholar] [CrossRef] [PubMed]








© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, M.-F.; Hsu, J.-L.; Lu, K.-T.; Chao, P.-K.; Cheng, M.-Y.; Hsu, H.-C.; Lo, A.-L.; Lee, Y.-L.; Hung, Y.-H.; Lyu, R.-K.; et al. Granulocyte Colony Stimulating Factor (GCSF) Can Attenuate Neuropathic Pain by Suppressing Monocyte Chemoattractant Protein-1 (MCP-1) Expression, through Upregulating the Early MicroRNA-122 Expression in the Dorsal Root Ganglia. Cells 2020, 9, 1669. https://doi.org/10.3390/cells9071669
Liao M-F, Hsu J-L, Lu K-T, Chao P-K, Cheng M-Y, Hsu H-C, Lo A-L, Lee Y-L, Hung Y-H, Lyu R-K, et al. Granulocyte Colony Stimulating Factor (GCSF) Can Attenuate Neuropathic Pain by Suppressing Monocyte Chemoattractant Protein-1 (MCP-1) Expression, through Upregulating the Early MicroRNA-122 Expression in the Dorsal Root Ganglia. Cells. 2020; 9(7):1669. https://doi.org/10.3390/cells9071669
Chicago/Turabian StyleLiao, Ming-Feng, Jung-Lung Hsu, Kwok-Tung Lu, Po-Kuan Chao, Mei-Yun Cheng, Hui-Ching Hsu, Ai-Lun Lo, Yun-Lin Lee, Yu-Hui Hung, Rong-Kuo Lyu, and et al. 2020. "Granulocyte Colony Stimulating Factor (GCSF) Can Attenuate Neuropathic Pain by Suppressing Monocyte Chemoattractant Protein-1 (MCP-1) Expression, through Upregulating the Early MicroRNA-122 Expression in the Dorsal Root Ganglia" Cells 9, no. 7: 1669. https://doi.org/10.3390/cells9071669




