Plausibility of Early Life in a Relatively Wide Temperature Range: Clues from Simulated Metabolic Network Expansion
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Sources
2.2. Thermodynamically Constrained Network Expansion Simulation
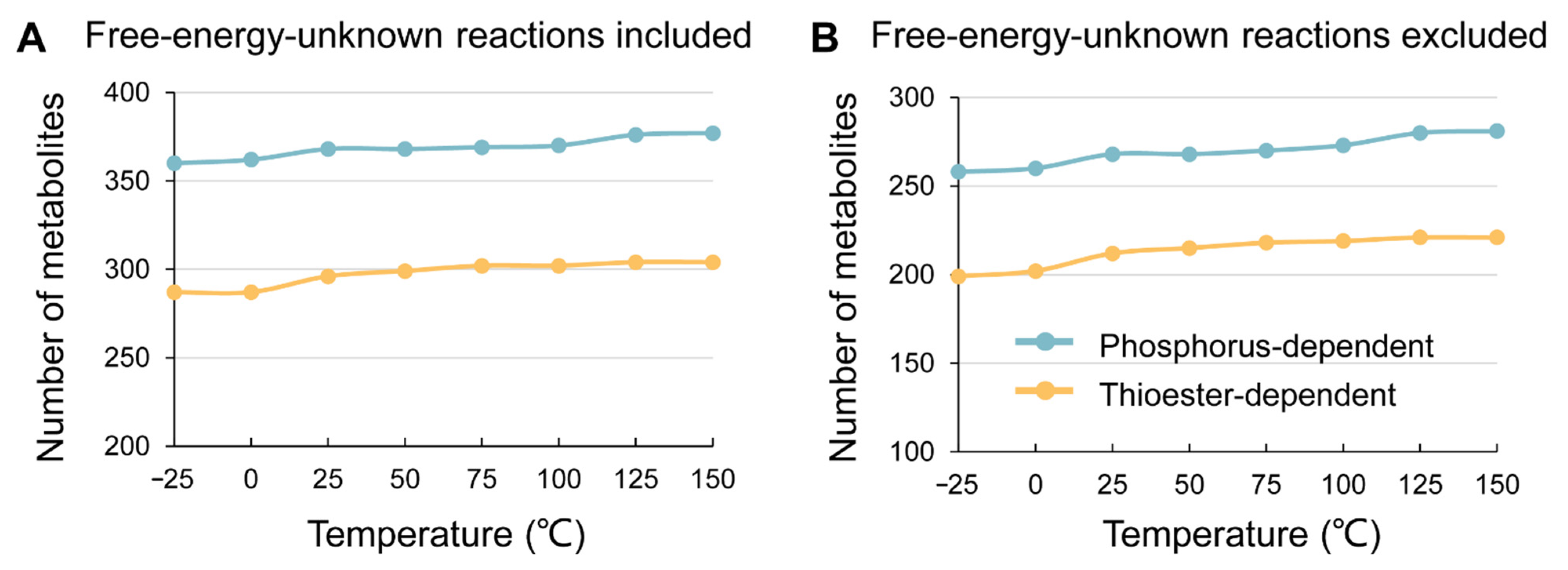
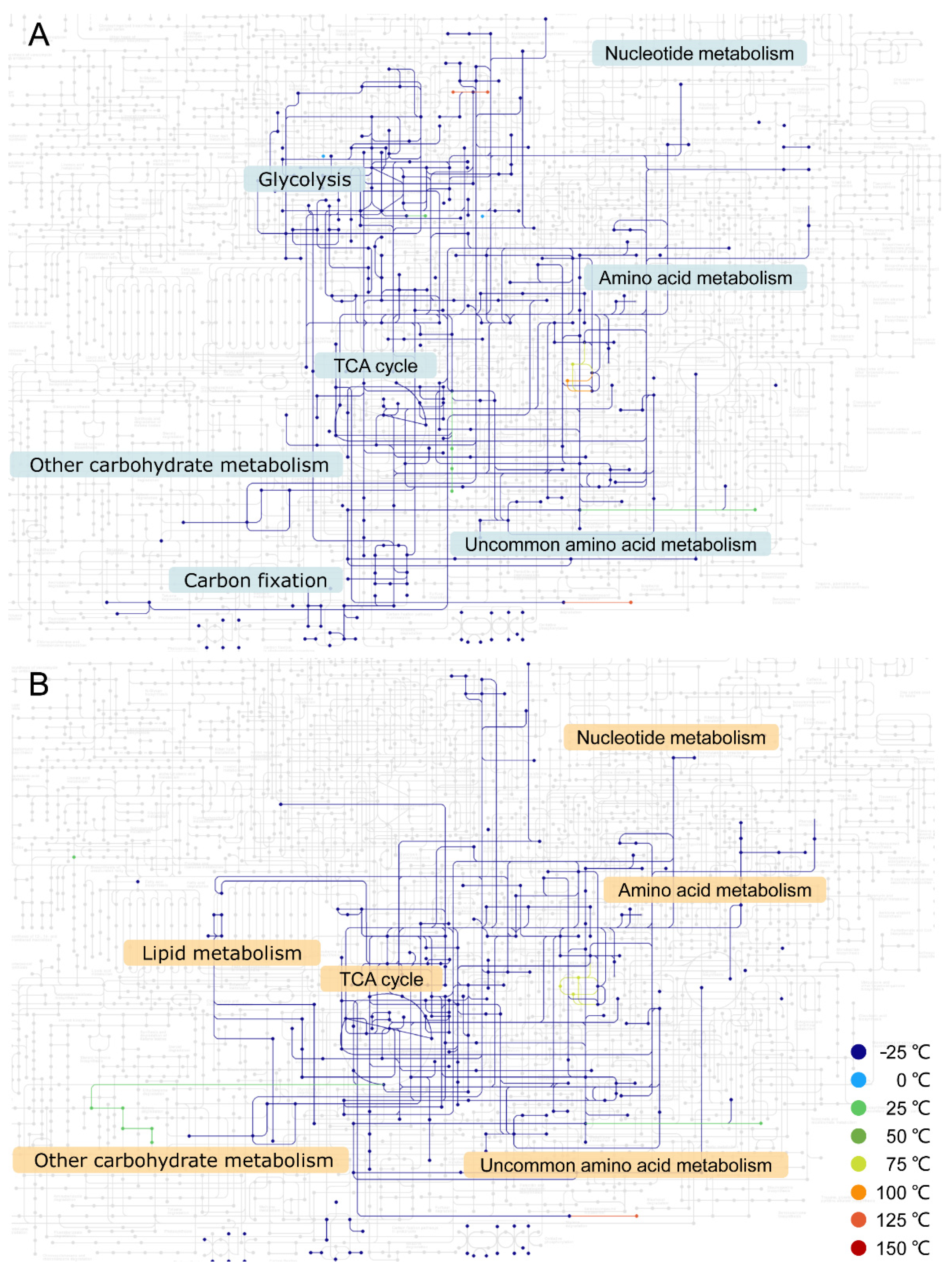
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Woese, C.R. Bacterial evolution. Microbiol. Rev. 1987, 51, 221–271. [Google Scholar] [CrossRef] [PubMed]
- Achenbach-Richter, L.; Gupta, R.; Zillig, W.; Woese, C.R. Rooting the archaebacterial tree: The pivotal role of Thermococcus celer in archaebacterial evolution. Syst. Appl. Microbiol. 1988, 10, 231–240. [Google Scholar] [CrossRef]
- Pace, N.R. Origin of life—Facing up to the physical setting. Cell 1991, 65, 531–533. [Google Scholar] [CrossRef]
- Brochier, C.; Philippe, H. Phylogeny: A non-hyperthermophilic ancestor for bacteria. Nature 2002, 417, 244. [Google Scholar] [CrossRef] [PubMed]
- Galtier, N.; Tourasse, N.; Gouy, M. A nonhyperthermophilic common ancestor to extant life forms. Science 1999, 283, 220–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boussau, B.; Blanquart, S.; Necsulea, A.; Lartillot, N.; Gouy, M. Parallel adaptations to high temperatures in the Archaean eon. Nature 2008, 456, 942–945. [Google Scholar] [CrossRef] [Green Version]
- Groussin, M.; Boussau, B.; Charles, S.; Blanquart, S.; Gouy, M. The molecular signal for the adaptation to cold temperature during early life on Earth. Biol. Lett. 2013, 9, 20130608. [Google Scholar] [CrossRef] [PubMed]
- Eck, R.V.; Dayhoff, M.O. Evolution of the structure of ferredoxin based on living relics of primitive amino acid sequences. Science 1966, 152, 363–366. [Google Scholar] [CrossRef]
- Hartman, H. Speculations on the origin and evolution of metabolism. J. Mol. Evol. 1975, 4, 359–370. [Google Scholar] [CrossRef]
- Morowitz, H.J.; Kostelnik, J.D.; Yang, J.; Cody, G.D. The origin of intermediary metabolism. Proc. Natl. Acad. Sci. USA 2000, 97, 7704–7708. [Google Scholar] [CrossRef] [Green Version]
- Lanier, K.A.; Williams, L.D. The origin of life: Models and data. J. Mol. Evol. 2017, 84, 85–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ebenhöh, O.; Handorf, T.; Heinrich, R. Structural analysis of expanding metabolism networks. Genome Inform. 2004, 15, 35–45. [Google Scholar] [PubMed]
- Goldford, J.E.; Hartman, H.; Smith, T.F.; Segrè, D. Remnants of an ancient metabolism without phosphate. Cell 2017, 168, 1126–1134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goldford, J.E.; Hartman, H.; Marsland, R.; Segrè, D. Environmental boundary conditions for the origin of life converge to an organo-sulfur metabolism. Nat. Ecol. Evol. 2019, 3, 1715–1724. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Chu, X.-Y.; Yang, Y.; Zhang, X.; Liu, Y.-M.; Gao, J.; Ma, B.-G.; Zhang, H.-Y. Phosphates as energy sources to expand metabolism networks. Life 2019, 9, 43. [Google Scholar] [CrossRef] [Green Version]
- Schwartz, A.W. Phosphorus in prebiotic chemistry. Philos. Trans. R. Soc. B 2006, 361, 1743–1749. [Google Scholar] [CrossRef] [Green Version]
- De Duve, C. A research proposal on the origin of life. Orig. Life Evol. Biosph. 2003, 33, 559–574. [Google Scholar] [CrossRef]
- Piast, R.W.; Wieczorek, R.M. Origin of life and the phosphate transfer catalyst. Astrobiology 2017, 17, 277–285. [Google Scholar] [CrossRef]
- Glindemann, D.; De Graaf, R.M.; Schwartz, A.W. Chemical reduction of phosphate on the primitive earth. Orig. Life Evol. Biosph. 1999, 29, 555–561. [Google Scholar] [CrossRef]
- Pasek, M.A. Schreibersite on the early Earth: Scenarios for prebiotic phosphorylation. Geosci. Front. 2017, 8, 329–335. [Google Scholar] [CrossRef] [Green Version]
- Yamagata, Y.; Watanabe, H.; Saitoh, M.; Namba, T. Volcanic production of polyphosphates and its relevance to prebiotic evolution. Nature 1991, 352, 516–519. [Google Scholar] [CrossRef]
- Benner, S.A.; Kim, H.J.; Carrigan, M.A. Asphalt, water, and the prebiotic synthesis of ribose, ribonucleosides, and RNA. Acc. Chem. Res. 2012, 45, 2025–2034. [Google Scholar] [CrossRef] [PubMed]
- Ogata, H.; Goto, S.; Sato, K.; Fujibuchi, W.; Bono, H.; Kanehisa, M. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 1999, 27, 29–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flamholz, A.; Noor, E.; Bar-Even, A.; Milo, R. eQuilibrator—The biochemical thermodynamics calculator. Nucleic Acids Res. 2012, 40, D770–D775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noor, E.; Haraldsdóttir, H.S.; Milo, R.; Fleming, R.M.T. Consistent estimation of Gibbs energy using component contributions. PLoS Comput. Biol. 2013, 9, e1003098. [Google Scholar] [CrossRef] [Green Version]
- Coggins, A.J.; Powner, M.W. Prebiotic synthesis of phosphoenol pyruvate by phosphorylation controlled triose glycolysis. Nat. Chem. 2017, 9, 310–317. [Google Scholar] [CrossRef]
- Keller, M.A.; Turchyn, A.V.; Ralser, M. Non-enzymatic glycolysis and pentose phosphate pathway-like reactions in a plausible Archean ocean. Mol. Syst. Biol. 2014, 10, 725. [Google Scholar] [CrossRef]
- Bar-Even, A.; Flamholz, A.; Noor, E.; Milo, R. Thermodynamic constraints shape the structure of carbon fixation pathways. BBA Bioenergetics 2012, 1817, 1646–1659. [Google Scholar] [CrossRef] [Green Version]
- Mykytczuk, N.C.S.; Foote, S.J.; Omelon, C.R.; Southam, G.; Greer, C.W.; Whyte, L.G. Bacterial growth at −15 °C; molecular insights from the permafrost bacterium Planococcus halocryophilus Or1. ISME J. 2013, 7, 1211–1226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takai, K.; Nakamura, K.; Toki, T.; Tsunogai, U.; Miyazaki, M.; Miyazaki, J.; Hirayama, H.; Nakagawa, S.; Nunoura, T.; Horikoshi, K. Cell proliferation at 122 °C and isotopically heavy CH4 production by a hyperthermophilic methanogen under high-pressure cultivation. Proc. Natl. Acad. Sci. USA 2008, 105, 10949–10954. [Google Scholar] [CrossRef] [Green Version]
- Chu, X.Y.; Zhang, H.Y. Cofactors as molecular fossils to trace the origin and evolution of proteins. ChemBioChem 2020, 21, 3161–3168. [Google Scholar] [CrossRef] [PubMed]
- Whicher, A.; Camprubi, E.; Pinna, S.; Herschy, B.; Lane, N. Acetyl phosphate as a primordial energy currency at the origin of life. Orig. Life Evol. Biosph. 2018, 48, 159–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamagata, Y. Prebiotic formation of ADP and ATP from AMP, calcium phosphates and cyanate in aqueous solution. Orig. Life Evol. Biosph. 1999, 29, 511–520. [Google Scholar] [CrossRef]
- Akouche, M.; Jaber, M.; Maurel, M.C.; Lambert, J.F.; Georgelin, T. Phosphoribosyl pyrophosphate: A molecular vestige of the origin of life on minerals. Angew. Chem. Int. Ed. Engl. 2017, 56, 7920–7923. [Google Scholar] [CrossRef] [PubMed]
- Yadav, M.; Kumar, R.; Krishnamurthy, R. Chemistry of abiotic nucleotide synthesis. Chem. Rev. 2020, 120, 4766–4805. [Google Scholar] [CrossRef] [PubMed]
- Peretó, J.; Bada, J.L.; Lazcano, A. Charles Darwin and the origin of life. Orig. Life Evol. Biosph. 2009, 39, 395–406. [Google Scholar] [CrossRef] [Green Version]
- Miller, S.L. A production of amino acids under possible primitive earth conditions. Science 1953, 117, 528–529. [Google Scholar] [CrossRef] [Green Version]
- Deamer, D.; Damer, B.; Kompanichenko, V. Hydrothermal chemistry and the origin of cellular life. Astrobiology 2019, 19, 1523–1537. [Google Scholar] [CrossRef]
- Damer, B.; Deamer, D. The hot spring hypothesis for an origin of life. Astrobiology 2020, 20, 429–452. [Google Scholar] [CrossRef] [Green Version]
- Omran, A.; Pasek, M. A constructive way to think about different hydrothermal environments for the origins of life. Life 2020, 10, 36. [Google Scholar] [CrossRef] [Green Version]
- Ross, D.; Deamer, D. Dry/wet cycling and the thermodynamics and kinetics of prebiotic polymer synthesis. Life 2016, 6, 28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, C.; Fan, C.; Wan, R.; Tong, C.; Miao, Z.; Zhao, Y. Phosphorylation of adenosine with trimetaphosphate under simulated prebiotic conditions. Orig. Life. Evol. Biosph. 2002, 32, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.; Harada, K.; Kendrick, J. Production of spherules from proteinoids and hot water. Science 1959, 129, 1221–1223. [Google Scholar] [CrossRef]
- Yu, S.-S.; Solano, M.; Blanchard, M.K.; Soper-Hopper, M.; Krishnamurthy, R.; Fernandez, F.M.; Hud, N.V.; Schork, F.J.; Grover, M.A. Elongation of model prebiotic proto-peptides by continuous monomer feeding. Macromolecules 2017, 50, 9286–9294. [Google Scholar] [CrossRef]
- De Guzman, V.; Vercoutere, W.; Shenasa, H.; Deamer, D.W. Generation of oligonucleotides under hydrothermal conditions by non-enzymatic polymerization. J. Mol. Evol. 2014, 78, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Bains, W.; Xiao, Y.; Yu, C. Prediction of the maximum temperature for life based on the stability of metabolites to decomposition in water. Life 2015, 5, 1054–1100. [Google Scholar] [CrossRef] [Green Version]
- Miller, S.L.; Bada, J.L. Submarine hot springs and the origin of life. Nature 1988, 334, 609–611. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.L.; Lazcano, A. The origin of life—Did it occur at high temperatures? J. Mol. Evol. 1995, 41, 689–692. [Google Scholar] [CrossRef] [PubMed]
- Matthews, C.N.; Minard, R.D. Hydrogen cyanide polymers, comets and the origin of life. Faraday Discuss. 2006, 133, 393–401. [Google Scholar] [CrossRef]
- Miyakawa, S.; Cleaves, H.J.; Miller, S.L. The cold origin of life: A. implications based on pyrimidines and purines produced from frozen ammonium cyanide solutions. Orig. Life Evol. Biosph. 2002, 32, 195–208. [Google Scholar] [CrossRef]
- Miyakawa, S.; Cleaves, H.J.; Miller, S.L. The cold origin of life: B. implications based on pyrimidines and purines produced from frozen ammonium cyanide solutions. Orig. Life Evol. Biosph. 2002, 32, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.; Miller, S.L.; Oró, J. Production of guanine from NH4CN polymerizations. J. Mol. Evol. 1999, 49, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Doolittle, W.F. Phylogenetic classification and the universal tree. Science 1999, 284, 2124–2129. [Google Scholar] [CrossRef]
- Lingam, M.; Loeb, A. Physical constraints on the likelihood of life on exoplanets. Int. J. Astrobiol. 2018, 17, 116–126. [Google Scholar] [CrossRef] [Green Version]
- Ooka, H.; McGlynn, S.E.; Nakamura, R. Electrochemistry at deep-sea hydrothermal vents: Utilization of the thermodynamic driving force towards the autotrophic origin of life. Chem. Electro. Chem. 2019, 6, 1316–1323. [Google Scholar] [CrossRef] [Green Version]
- Boyd, E.S.; Amenabar, M.J.; Poudel, S.; Templeton, A.S. Bioenergetic constraints on the origin of autotrophic metabolism. Philos. Trans. A Math. Phys. Eng. Sci. 2020, 378, 20190151. [Google Scholar] [CrossRef] [Green Version]
- Brack, A.; Spach, G. Multiconformational synthetic polypeptides. J. Amer. Chem. Soc. 1981, 103, 6319–6323. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chu, X.-Y.; Chen, S.-M.; Zhao, K.-W.; Tian, T.; Gao, J.; Zhang, H.-Y. Plausibility of Early Life in a Relatively Wide Temperature Range: Clues from Simulated Metabolic Network Expansion. Life 2021, 11, 738. https://doi.org/10.3390/life11080738
Chu X-Y, Chen S-M, Zhao K-W, Tian T, Gao J, Zhang H-Y. Plausibility of Early Life in a Relatively Wide Temperature Range: Clues from Simulated Metabolic Network Expansion. Life. 2021; 11(8):738. https://doi.org/10.3390/life11080738
Chicago/Turabian StyleChu, Xin-Yi, Si-Ming Chen, Ke-Wei Zhao, Tian Tian, Jun Gao, and Hong-Yu Zhang. 2021. "Plausibility of Early Life in a Relatively Wide Temperature Range: Clues from Simulated Metabolic Network Expansion" Life 11, no. 8: 738. https://doi.org/10.3390/life11080738






