Biomarker Potential of Vimentin in Oral Cancers
Abstract
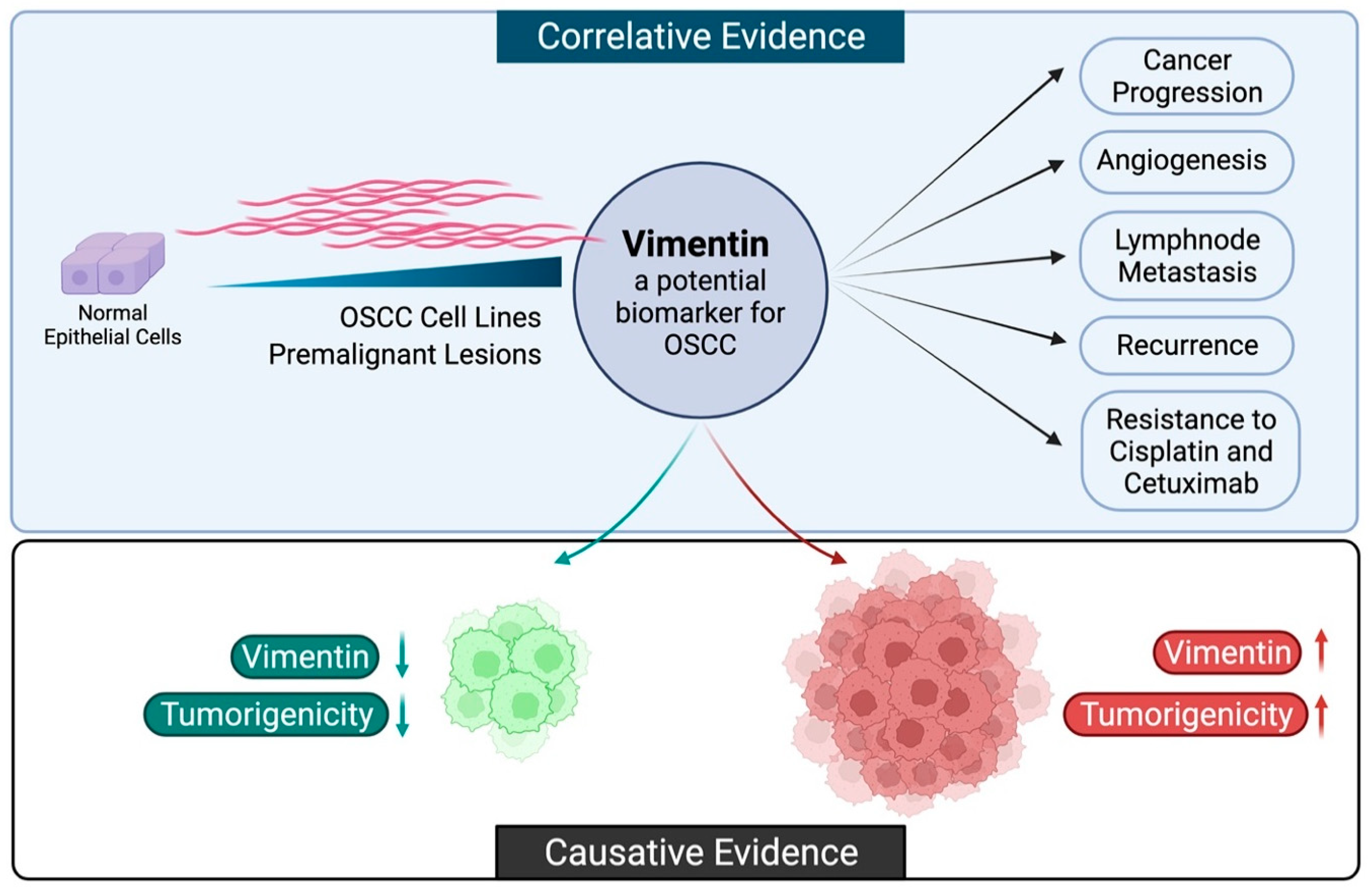
:1. Introduction
2. Regulators of Vimentin
3. Aberrant Expression of Vimentin in Premalignant Oral Lesions
4. Role of Vimentin in Oral Cancer Progression
5. Vimentin in Late Stages of Oral Cancer
6. Vimentin in Lymph Node Metastasis
7. Vimentin in Angiogenesis
8. Vimentin in Recurrence
9. Vimentin in Therapy Resistance
10. Association of Oral Cancer Risk Factors with Vimentin
11. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Green, K.J.; Böhringer, M.; Gocken, T.; Jones, J.C.R. Intermediate Filament Associated Proteins. In Advances in Protein Chemistry; Academic Press: Cambridge, MA, USA, 2005; Volume 70, pp. 143–202. [Google Scholar]
- Kirmse, R.; Portet, S.; Mücke, N.; Aebi, U.; Herrmann, H.; Langowski, J. A Quantitative Kinetic Model for the in Vitro Assembly of Intermediate Filaments from Tetrameric Vimentin*. J. Biol. Chem. 2007, 282, 18563–18572. [Google Scholar] [CrossRef] [Green Version]
- Fuchs, E.; Weber, K. INTERMEDIATE FILAMENTS: Structure, Dynamics, Function and Disease. Ann. Rev. Biochem. 1994, 63, 345–382. [Google Scholar] [CrossRef]
- Herrmann, H.; Hesse, M.; Reichenzeller, M.; Reichenzeller, M.; Aebi, U.; Aebi, U.; Magin, T.M.; Magin, T.M. Functional complexity of intermediate filament cytoskeletons: From structure to assembly to gene ablation. Int. Rev. Cytol. 2003, 223, 83–175. [Google Scholar] [PubMed]
- Evans, R.M. Vimentin: The conundrum of the intermediate filament gene family. Bioassays 1998, 20, 79–86. [Google Scholar] [CrossRef]
- Herrmann, H.; Bär, H.; Kreplak, L.; Strelkov, S.V.; Aebi, U. Intermediate filaments: From cell architecture to nanomechanics. Nat. Rev. Mol. Cell Biol. 2007, 8, 562–573. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, H.; Aebi, U. Intermediate Filaments: Molecular Structure, Assembly Mechanism, and Integration Into Functionally Distinct Intracellular Scaffolds. Ann. Rev. Biochem. 2004, 73, 749–789. [Google Scholar] [CrossRef] [PubMed]
- Chernyatina, A.A.; Nicolet, S.; Aebi, U.; Herrmann, H.; Strelkov, S.V. Atomic structure of the vimentin central α-helical domain and its implications for intermediate filament assembly. Proc. Natl. Acad. Sci. USA 2012, 109, 13620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aziz, A.; Hess, J.F.; Budamagunta, M.S.; Voss, J.C.; Kuzin, A.P.; Huang, Y.J.; Xiao, R.; Montelione, G.T.; FitzGerald, P.G.; Hunt, J.F. The Structure of Vimentin Linker 1 and Rod 1B Domains Characterized by Site-directed Spin-labeling Electron Paramagnetic Resonance (SDSL-EPR) and X-ray Crystallography*. J. Biol. Chem. 2012, 287, 28349–28361. [Google Scholar] [CrossRef] [Green Version]
- Goldman, R.D.; Khuon, S.; Chou, Y.H.; Opal, P.; Steinert, P.M. The function of intermediate filaments in cell shape and cytoskeletal integrity. J. Cell Biol. 1996, 134, 971–983. [Google Scholar] [CrossRef]
- Cheng, F.; Shen, Y.; Mohanasundaram, P.; Lindström, M.; Ivaska, J.; Ny, T.; Eriksson, J.E. Vimentin coordinates fibroblast proliferation and keratinocyte differentiation in wound healing via TGF-β–Slug signaling. Proc. Natl. Acad. Sci. USA 2016, 113, E4320. [Google Scholar] [CrossRef] [Green Version]
- Nieminen, M.; Henttinen, T.; Merinen, M.; Marttila–Ichihara, F.; Eriksson, J.E.; Jalkanen, S. Vimentin function in lymphocyte adhesion and transcellular migration. Nat. Cell Biol. 2006, 8, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Eckes, B.; Dogic, D.; Colucci-Guyon, E.; Wang, N.; Maniotis, A.; Ingber, D.; Merckling, A.; Langa, F.; Aumailley, M.; Delouvée, A.; et al. Impaired mechanical stability, migration and contractile capacity in vimentin-deficient fibroblasts. J. Cell Sci. 1998, 111, 1897–1907. [Google Scholar] [CrossRef]
- Richardson, A.M.; Havel, L.S.; Koyen, A.E.; Konen, J.M.; Shupe, J.; Wiles, W.G.; Martin, W.D.; Grossniklaus, H.E.; Sica, G.; Gilbert-Ross, M.; et al. Vimentin Is Required for Lung Adenocarcinoma Metastasis via Heterotypic Tumor Cell–Cancer-Associated Fibroblast Interactions during Collective Invasion. Clin. Cancer Res. 2018, 24, 420. [Google Scholar] [CrossRef] [Green Version]
- Peuhu, E.; Virtakoivu, R.; Mai, A.; Wärri, A.; Ivaska, J. Epithelial vimentin plays a functional role in mammary gland development. Development 2017, 144, 4103–4113. [Google Scholar] [CrossRef] [Green Version]
- The Human Protein Atlas. Available online: https://www.proteinatlas.org/ENSG00000026025-VIM/tissue (accessed on 10 December 2021).
- Danielsson, F.; Peterson, M.K.; Caldeira Araújo, H.; Lautenschläger, F.; Gad, A.K. Vimentin Diversity in Health and Disease. Cells 2018, 7, 147. [Google Scholar] [CrossRef] [Green Version]
- Battaglia, R.A.; Delic, S.; Herrmann, H.; Snider, N.T. Vimentin on the move: New developments in cell migration. F1000 Res. 2018, 7, 1796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colucci-Guyon, E.; Giménez, Y.R.M.; Maurice, T.; Babinet, C.; Privat, A. Cerebellar defect and impaired motor coordination in mice lacking vimentin. Glia 1999, 25, 33–43. [Google Scholar] [CrossRef]
- Antfolk, D.; Sjöqvist, M.; Cheng, F.; Isoniemi, K.; Duran, C.L.; Rivero-Muller, A.; Antila, C.; Niemi, R.; Landor, S.; Bouten, C.V.C.; et al. Selective regulation of Notch ligands during angiogenesis is mediated by vimentin. Proc. Natl. Acad. Sci. USA 2017, 114, E4574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Triolo, D.; Dina, G.; Taveggia, C.; Vaccari, I.; Porrello, E.; Rivellini, C.; Domi, T.; La Marca, R.; Cerri, F.; Bolino, A.; et al. Vimentin regulates peripheral nerve myelination. Development 2012, 139, 1359–1367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Franke, W.W.; Grund, C.; Kuhn, C.; Jackson, B.W.; Illmensee, K. Formation of Cytoskeletal Elements During Mouse Embryogenesis: III. Primary Mesenchymal Cells and the First Appearance of Vimentin Filaments. Differentiation 1982, 23, 43–59. [Google Scholar] [CrossRef]
- Petrie, R.J.; Koo, H.; Yamada, K.M. Generation of compartmentalized pressure by a nuclear piston governs cell motility in a 3D matrix. Science 2014, 345, 1062–1065. [Google Scholar] [CrossRef] [Green Version]
- Blanchoin, L.; Boujemaa-Paterski, R.; Sykes, C.; Plastino, J. Actin Dynamics, Architecture, and Mechanics in Cell Motility. Physiol. Rev. 2014, 94, 235–263. [Google Scholar] [CrossRef] [Green Version]
- Mor-Vaknin, N.; Punturieri, A.; Sitwala, K.; Markovitz, D.M. Vimentin is secreted by activated macrophages. Nat. Cell Biol. 2003, 5, 59–63. [Google Scholar] [CrossRef]
- Xu, B.; deWaal Robert, M.; Mor-Vaknin, N.; Hibbard, C.; Markovitz David, M.; Kahn Mark, L. The Endothelial Cell-Specific Antibody PAL-E Identifies a Secreted Form of Vimentin in the Blood Vasculature. Mol. Cell. Biol. 2004, 24, 9198–9206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Byun, Y.; Chen, F.; Chang, R.; Trivedi, M.; Green, K.J.; Cryns, V.L. Caspase cleavage of vimentin disrupts intermediate filaments and promotes apoptosis. Cell Death Differ. 2001, 8, 443–450. [Google Scholar] [CrossRef]
- Hol, E.M.; Capetanaki, Y. Type III Intermediate Filaments Desmin, Glial Fibrillary Acidic Protein (GFAP), Vimentin, and Peripherin. Cold Spring Harb. Perspect. Biol. 2017, 9, a021642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanaka, H.; Goto, H.; Inoko, A.; Makihara, H.; Enomoto, A.; Horimoto, K.; Matsuyama, M.; Kurita, K.; Izawa, I.; Inagaki, M. Cytokinetic Failure-induced Tetraploidy Develops into Aneuploidy, Triggering Skin Aging in Phosphovimentin-deficient Mice. J. Biol. Chem. 2015, 290, 12984–12998. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dmello, C.; Sawant, S.; Alam, H.; Gangadaran, P.; Mogre, S.; Tiwari, R.; D’Souza, Z.; Narkar, M.; Thorat, R.; Patil, K. Vimentin regulates differentiation switch via modulation of keratin 14 levels and their expression together correlates with poor prognosis in oral cancer patients. PLoS ONE 2017, 12, e0172559. [Google Scholar] [CrossRef] [PubMed]
- Dmello, C.; Srivastava, S.S.; Tiwari, R.; Chaudhari, P.R.; Sawant, S.; Vaidya, M.M. Multifaceted role of keratins in epithelial cell differentiation and transformation. J. Biosci. 2019, 44, 33. [Google Scholar] [CrossRef]
- Satelli, A.; Li, S. Vimentin in cancer and its potential as a molecular target for cancer therapy. Cell. Mol. Life Sci. 2011, 68, 3033–3046. [Google Scholar] [CrossRef] [Green Version]
- Sommers, C.L.; Skerker, J.M.; Chrysogelos, S.A.; Bosseler, M.; Gelmann, E.P. Regulation of vimentin gene transcription in human breast cancer cell lines. Cell Growth Differ. 1994, 5, 839–846. [Google Scholar] [PubMed]
- Rittling, S.R.; Baserga, R. Functional analysis and growth factor regulation of the human vimentin promoter. Mol. Cell. Biol. 1987, 7, 3908–3915. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhang, X.; Salmon, M.; Lin, X.; Zehner, Z.E. TGFβ1 regulation of vimentin gene expression during differentiation of the C2C12 skeletal myogenic cell line requires Smads, AP-1 and Sp1 family members. Biochim. Biophys. Acta Mol. Cell Res. 2007, 1773, 427–439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rittling, S.R.; Coutinho, L.; Amram, T.; Kolbe, M. AP-1/jun binding sites mediate serum inducibility of the human vimentin promoter. Nucleic Acids Res. 1989, 17, 1619–1633. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.H.; Vercamer, C.; Li, Z.; Paulin, D.; Vandenbunder, B.; Stehelin, D. PEA3 transactivates vimentin promoter in mammary epithelial and tumor cells. Oncogene 1996, 13, 1667–1675. [Google Scholar]
- Zhang, X.; Diab, I.H.; Zehner, Z.E. ZBP-89 represses vimentin gene transcription by interacting with the transcriptional activator, Sp1. Nucleic Acids Res. 2003, 31, 2900–2914. [Google Scholar] [CrossRef] [Green Version]
- Sergio, S.; Coluccia, A.M.L.; Lemma, E.D.; Spagnolo, B.; Vergara, D.; Maffia, M.; De Vittorio, M.; Pisanello, F. 3D-microenvironments initiate TCF4 expression rescuing nuclear β-catenin activity in MCF-7 breast cancer cells. Acta Biomater. 2020, 103, 153–164. [Google Scholar] [CrossRef]
- Gilles, C.; Polette, M.; Mestdagt, M.; Nawrocki-Raby, B.; Ruggeri, P.; Birembaut, P.; Foidart, J.-M. Transactivation of Vimentin by β-Catenin in Human Breast Cancer Cells. Cancer Res. 2003, 63, 2658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez-Tilló, E.; de Barrios, O.; Siles, L.; Cuatrecasas, M.; Castells, A.; Postigo, A. β-catenin/TCF4 complex induces the epithelial-to-mesenchymal transition (EMT)-activator ZEB1 to regulate tumor invasiveness. Proc. Natl. Acad. Sci. USA 2011, 108, 19204. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Diab, I.; Zhang, X.; Izmailova, E.S.; Zehner, Z.E. Stat3 enhances vimentin gene expression by binding to the antisilencer element and interacting with the repressor protein, ZBP-89. Oncogene 2004, 23, 168–178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chernyatina, A.A.; Strelkov, S.V. Stabilization of vimentin coil2 fragment via an engineered disulfide. J. Struct. Biol. 2012, 177, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Snider, N.T.; Omary, M.B. Post-translational modifications of intermediate filament proteins: Mechanisms and functions. Nat. Rev. Mol. Cell Biol. 2014, 15, 163–177. [Google Scholar] [CrossRef] [Green Version]
- Kraxner, J.; Lorenz, C.; Menzel, J.; Parfentev, I.; Silbern, I.; Denz, M.; Urlaub, H.; Schwappach, B.; Köster, S. Post-translational modifications soften vimentin intermediate filaments. Nanoscale 2021, 13, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Hyder, C.L.; Pallari, H.-M.; Kochin, V.; Eriksson, J.E. Providing cellular signposts—Post-translational modifications of intermediate filaments. FEBS Lett. 2008, 582, 2140–2148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Subramonian, D.; Raghunayakula, S.; Olsen, J.V.; Beningo, K.A.; Paschen, W.; Zhang, X.-D. Analysis of Changes in SUMO-2/3 Modification during Breast Cancer Progression and Metastasis. J. Proteome Res. 2014, 13, 3905–3918. [Google Scholar] [CrossRef]
- Zhu, Q.S.; Rosenblatt, K.; Huang, K.L.; Lahat, G.; Brobey, R.; Bolshakov, S.; Nguyen, T.; Ding, Z.; Belousov, R.; Bill, K.; et al. Vimentin is a novel AKT1 target mediating motility and invasion. Oncogene 2011, 30, 457–470. [Google Scholar] [CrossRef] [Green Version]
- Lim, Y.-P.; Wong, C.Y.; Ooi, L.L.; Druker, B.J.; Epstein, R.J. Selective Tyrosine Hyperphosphorylation of Cytoskeletal and Stress Proteins in Primary Human Breast Cancers. Clin. Cancer Res. 2004, 10, 3980. [Google Scholar] [CrossRef] [Green Version]
- van der Velden, L.A.; Manni, J.J.; Ramaekers, F.C.; Kuijpers, W. Expression of intermediate filament proteins in benign lesions of the oral mucosa. Eur. Arch. Otorhinolaryngol. 1999, 256, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Sawant, S.S.; Vaidya, M.M.; Chaukar, D.A.; Alam, H.; Dmello, C.; Gangadaran, P.; Kannan, S.; Kane, S.; Dange, P.P.; Dey, N.; et al. Clinical significance of aberrant vimentin expression in oral premalignant lesions and carcinomas. Oral Dis. 2014, 20, 453–465. [Google Scholar] [CrossRef]
- Akhtar, K.; Ara, A.; Siddiqui, S.A.; Sherwani, R.K. Transition of Immunohistochemical Expression of E-Cadherin and Vimentin from Premalignant to Malignant Lesions of Oral Cavity and Oropharynx. Oman Med. J. 2016, 31, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Tao, D.; Xu, Q.; Gao, Z.; Tang, D. Expression of E-cadherin and vimentin in oral squamous cell carcinoma. Int. J. Clin. Exp. Pathol. 2015, 8, 3150–3154. [Google Scholar]
- Dmello, C.; Sawant, S.; Chaudhari, P.R.; Dongre, H.; Ahire, C.; D’Souza, Z.C.; Charles, S.E.; Rane, P.; Costea, D.E.; Chaukar, D.; et al. Aberrant expression of vimentin predisposes oral premalignant lesion derived cells towards transformation. Exp. Mol. Pathol. 2018, 105, 243–251. [Google Scholar] [CrossRef]
- Dmello, C.; Sawant, S.; Alam, H.; Gangadaran, P.; Tiwari, R.; Dongre, H.; Rana, N.; Barve, S.; Costea, D.E.; Chaukar, D.; et al. Vimentin-mediated regulation of cell motility through modulation of beta4 integrin protein levels in oral tumor derived cells. Int. J. Biochem. Cell Biol. 2016, 70, 161–172. [Google Scholar] [CrossRef]
- De Araujo, V.C.; Pinto, D.S.; De Sousa, S.O.M.; Nunes, F.D.; de Araujo, N.S. Vimentin in oral squamous cell carcinoma. Eur. Arch. Oto-Rhino-Laryngol. 1993, 250, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.K.; Agarwal, S.; Singh, S.N.; Sehra, R.; Jat, P.S.; Singhal, P. Role of Vimentin and E-cadherin Expression in Premalignant and Malignant Lesions of Oral Cavity. Indian J. Otolaryngol. Head Neck Surg. 2021, 1–6. [Google Scholar] [CrossRef]
- Anjum, A.; Akhtar, K.; Siddiqui, S.; Sherwani, R. Diagnostic and Prognostic Significance of E-Cadherin and Vimentin in Oral Cancer Metastasis. Ann. Pathol. Lab. Med. 2019, 3, 8–13. [Google Scholar]
- Costa, L.C.; Leite, C.F.; Cardoso, S.V.; Loyola, A.M.; Faria, P.R.; Souza, P.E.; Horta, M.C. Expression of epithelial-mesenchymal transition markers at the invasive front of oral squamous cell carcinoma. J. Appl. Oral Sci. 2015, 23, 169–178. [Google Scholar] [CrossRef]
- Lazarevic, M.; Milosevic, M.; Jelovac, D.; Milenkovic, S.; Tepavcevic, Z.; Baldan, F.; Suboticki, T.; Toljic, B.; Trisic, D.; Dragovic, M.; et al. Marked epithelial to mesenchymal transition in surgical margins of oral cancer-an in vitro study. Oncol. Lett. 2020, 19, 3743–3750. [Google Scholar] [CrossRef] [Green Version]
- Wangmo, C.; Charoen, N.; Jantharapattana, K.; Dechaphunkul, A.; Thongsuksai, P.A.-O. Epithelial-Mesenchymal Transition Predicts Survival in Oral Squamous Cell Carcinoma. Pathol. Oncol. Res. 2020, 26, 1511–1518. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.-F.; Kang, B.-H.; Wu, Y.-M.; Sun, J.-H.; Yen, L.-M.; Fu, T.-Y.; Lin, Y.-C.; Liou, H.-H.; Lin, Y.-S.; Sie, H.-C.; et al. Vimentin is a potential prognostic factor for tongue squamous cell carcinoma among five epithelial–mesenchymal transition-related proteins. PLoS ONE 2017, 12, e0178581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohamed, H.; Haglund, C.; Jouhi, L.; Atula, T.; Hagström, J.; Mäkitie, A. Expression and Role of E-Cadherin, β-Catenin, and Vimentin in Human Papillomavirus–Positive and Human Papillomavirus–Negative Oropharyngeal Squamous Cell Carcinoma. J. Histochem. Cytochem. 2020, 68, 595–606. [Google Scholar] [CrossRef]
- Bu, J.Q.; Chen, F. TGF-β1 promotes cells invasion and migration by inducing epithelial mesenchymal transformation in oral squamous cell carcinoma. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 2137–2144. [Google Scholar] [PubMed]
- Liu, S.; Liu, L.; Ye, W.; Ye, D.; Wang, T.; Guo, W.; Liao, Y.; Xu, D.; Song, H.; Zhang, L.; et al. High Vimentin Expression Associated with Lymph Node Metastasis and Predicated a Poor Prognosis in Oral Squamous Cell Carcinoma. Sci. Rep. 2016, 6, 38834. [Google Scholar] [CrossRef]
- Bhardwaj, M.; Sen, S.A.-O.; Chosdol, K.; Bakhshi, S.; Pushker, N.A.-O.; Sharma, A.; Kashyap, S.; Bajaj, M.S.; Singh, V.K. Vimentin overexpression as a novel poor prognostic biomarker in eyelid sebaceous gland carcinoma. Br. J. Ophthalmol. 2020, 104, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Morohashi, S.; Sato, F.; Kudo, Y.; Akasaka, H.; Tsutsumi, S.; Ogasawara, H.; Miyamoto, K.; Wajima, N.; Kawasaki, H.; et al. Vimentin expression of esophageal squamous cell carcinoma and its aggressive potential for lymph node metastasis. Biomed. Res. 2010, 31, 105–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, H.; Hakamada, K. Vimentin expression in lymph node metastatic lesions of squamous cell carcinoma of the esophagus. J. Clin. Oncol. 2016, 34, e15547. [Google Scholar] [CrossRef]
- Balasundaram, P.; Singh, M.K.; Dinda, A.K.; Thakar, A.; Yadav, R. Study of β-catenin, E-cadherin and vimentin in oral squamous cell carcinoma with and without lymph node metastases. Diagn. Pathol. 2014, 9, 145. [Google Scholar] [CrossRef] [Green Version]
- Steinmetz, N.F.; Maurer, J.; Sheng, H.; Bensussan, A.; Maricic, I.; Kumar, V.; Braciak, T.A. Two Domains of Vimentin Are Expressed on the Surface of Lymph Node, Bone and Brain Metastatic Prostate Cancer Lines along with the Putative Stem Cell Marker Proteins CD44 and CD133. Cancers 2011, 3, 2870–2885. [Google Scholar] [CrossRef] [Green Version]
- Pantel, K.; Brakenhoff, R.H.; Brandt, B. Detection, clinical relevance and specific biological properties of disseminating tumour cells. Nat. Rev. Cancer 2008, 8, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, M.L.; Møller, C.K.; Rasmussen, L.; Boisen, L.; Pedersen, H.; Kristensen, P. An anti vimentin antibody promotes tube formation. Sci. Rep. 2017, 7, 3576. [Google Scholar] [CrossRef] [Green Version]
- Gonzales, M.; Weksler, B.; Tsuruta, D.; Goldman, R.D.; Yoon, K.J.; Hopkinson, S.B.; Flitney, F.W.; Jones, J.C. Structure and function of a vimentin-associated matrix adhesion in endothelial cells. Mol. Biol. Cell 2001, 12, 85–100. [Google Scholar] [CrossRef] [Green Version]
- Irani, S.; Dehghan, A. The Expression and Functional Significance of Vascular Endothelial-Cadherin, CD44, and Vimentin in Oral Squamous Cell Carcinoma. J. Int. Soc. Prev. Community Dent. 2018, 8, 110–117. [Google Scholar] [CrossRef]
- Li, C.; Li, Q.; Cai, Y.; He, Y.; Lan, X.; Wang, W.; Liu, J.; Wang, S.; Zhu, G.; Fan, J.; et al. Overexpression of angiopoietin 2 promotes the formation of oral squamous cell carcinoma by increasing epithelial–mesenchymal transition-induced angiogenesis. Cancer Gene Ther. 2016, 23, 295–302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Otsuki, S.; Inokuchi, M.; Enjoji, M.; Ishikawa, T.; Takagi, Y.; Kato, K.; Yamada, H.; Kojima, K.; Sugihara, K. Vimentin expression is associated with decreased survival in gastric cancer. Oncol. Rep. 2011, 25, 1235–1242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Maghrabi, J. Vimentin immunoexpression is associated with higher tumor grade, metastasis, and shorter survival in colorectal cancer. Int. J. Clin. Exp. Pathol. 2020, 13, 493–500. [Google Scholar]
- Ling, Z.; Cheng, B.; Tao, X.A.-O. Epithelial-to-mesenchymal transition in oral squamous cell carcinoma: Challenges and opportunities. Int. J. Cancer 2021, 148, 1548–1561. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.K.; Jiang, X.Y.; Zhou, X.-X.; Wang, D.-M.; Song, X.-L.; Jiang, H.-B. Upregulation of vimentin and aberrant expression of E-cadherin/beta-catenin complex in oral squamous cell carcinomas: Correlation with the clinicopathological features and patient outcome. Mod. Pathol. 2010, 23, 213–224. [Google Scholar] [CrossRef] [Green Version]
- Aires, F.T.; Lin, C.S.; Matos, L.L.; Kulcsar, M.A.V.; Cernea, C.R. Risk Factors for Distant Metastasis in Patients with Oral Cavity Squamous Cell Carcinoma Undergoing Surgical Treatment. ORL 2017, 79, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.D.; Ding, X.; Du, H.M.; Wu, Y.N.; Li, H.Q.; Wu, H.M.; Zhang, X.M. FOXM1 is a novel predictor of recurrence in patients with oral squamous cell carcinoma associated with an increase in epithelial-mesenchymal transition. Mol. Med. Rep. 2019, 19, 4101–4108. [Google Scholar] [CrossRef]
- Ngan, C.Y.; Yamamoto, H.; Seshimo, I.; Tsujino, T.; Man-i, M.; Ikeda, J.I.; Konishi, K.; Takemasa, I.; Ikeda, M.; Sekimoto, M.; et al. Quantitative evaluation of vimentin expression in tumour stroma of colorectal cancer. Br. J. Cancer 2007, 96, 986–992. [Google Scholar] [CrossRef]
- Ye, Z.; Zhang, X.; Luo, Y.; Li, S.; Huang, L.; Li, Z.; Li, P.; Chen, G. Prognostic Values of Vimentin Expression and Its Clinicopathological Significance in Non-Small Cell Lung Cancer: A Meta-Analysis of Observational Studies with 4118 Cases. PLoS ONE 2016, 11, e0163162. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Helfand, B.T.; Jang, T.L.; Zhu, L.J.; Chen, L.; Yang, X.J.; Kozlowski, J.; Smith, N.; Kundu, S.D.; Yang, G.; et al. Nuclear Factor-κB-Mediated Transforming Growth Factor-β-Induced Expression of Vimentin Is an Independent Predictor of Biochemical Recurrence after Radical Prostatectomy. Clin. Cancer Res. 2009, 15, 3557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, L.; Li, J.; Lei, L.; He, H.; Chen, E.; Dong, J.; Yang, J. High Vimentin Expression Predicts a Poor Prognosis and Progression in Colorectal Cancer: A Study with Meta-Analysis and TCGA Database. BioMed Res. Int. 2018, 2018, 6387810. [Google Scholar] [CrossRef]
- Liu, S.-C.; Wu, Y.-C.; Huang, C.-M.; Hsieh, M.-S.; Huang, T.-Y.; Huang, C.-S.; Hsu, T.-N.; Huang, M.-S.; Lee, W.-H.; Yeh, C.-T.; et al. Inhibition of Bruton’s tyrosine kinase as a therapeutic strategy for chemoresistant oral squamous cell carcinoma and potential suppression of cancer stemness. Oncogenesis 2021, 10, 20. [Google Scholar] [CrossRef]
- Khoo, X.-H.; Paterson, I.C.; Goh, B.-H.; Lee, W.-L. Cisplatin-Resistance in Oral Squamous Cell Carcinoma: Regulation by Tumor Cell-Derived Extracellular Vesicles. Cancers 2019, 11, 1166. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.-F.; Nieh, S.; Jao, S.-W.; Liu, C.-L.; Wu, C.-H.; Chang, Y.-C.; Yang, C.-Y.; Lin, Y.-S. Quercetin Suppresses Drug-Resistant Spheres via the p38 MAPK–Hsp27 Apoptotic Pathway in Oral Cancer Cells. PLoS ONE 2012, 7, e49275. [Google Scholar] [CrossRef] [Green Version]
- Kirave, P.; Gondaliya, P.; Kulkarni, B.; Rawal, R.; Garg, R.; Jain, A.; Kalia, K. Exosome mediated miR-155 delivery confers cisplatin chemoresistance in oral cancer cells via epithelial-mesenchymal transition. Oncotarget 2020, 11, 1157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, A.; Wu, Y.; Chu, W.; Yang, X.; Zhu, Z.; Yan, E.; Zhang, W.; Zhou, J.; Ding, X.; Liu, J.; et al. Involvement of miR-619-5p in resistance to cisplatin by regulating ATXN3 in oral squamous cell carcinoma. Int. J. Biol. Sci. 2021, 17, 430–447. [Google Scholar] [CrossRef]
- Kimura, I.; Kitahara, H.; Ooi, K.; Kato, K.; Noguchi, N.; Yoshizawa, K.; Nakamura, H.; Kawashiri, S. Loss of epidermal growth factor receptor expression in oral squamous cell carcinoma is associated with invasiveness and epithelial-mesenchymal transition. Oncol. Lett. 2016, 11, 201–207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.-M.; Gaur, A.B.; Lengyel, E.; Peter, M.E. The miR-200 family determines the epithelial phenotype of cancer cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes Dev. 2008, 22, 894–907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arunkumar, G.; Rao, A.K.D.M.; Manikandan, M.; Rao, H.P.S.; Subbiah, S.; Ilangovan, R.; Murugan, A.K.; Munirajan, A.K. Dysregulation of miR-200 family microRNAs and epithelial-mesenchymal transition markers in oral squamous cell carcinoma. Oncol. Lett. 2018, 15, 649–657. [Google Scholar] [CrossRef]
- Ghosh, R.D.; Ghuwalewala, S.; Das, P.; Mandloi, S.; Alam, S.K.; Chakraborty, J.; Sarkar, S.; Chakrabarti, S.; Panda, C.K.; Roychoudhury, S. MicroRNA profiling of cisplatin-resistant oral squamous cell carcinoma cell lines enriched with cancer-stem-cell-like and epithelial-mesenchymal transition-type features. Sci. Rep. 2016, 6, 23932. [Google Scholar] [CrossRef] [Green Version]
- Biddle, A.; Liang, X.; Gammon, L.; Fazil, B.; Harper, L.J.; Emich, H.; Costea, D.E.; Mackenzie, I.C. Cancer Stem Cells in Squamous Cell Carcinoma Switch between Two Distinct Phenotypes That Are Preferentially Migratory or Proliferative. Cancer Res. 2011, 71, 5317. [Google Scholar] [CrossRef] [Green Version]
- Schmitz, S.; Bindea, G.; Albu, R.I.; Mlecnik, B.; Machiels, J.-P. Cetuximab promotes epithelial to mesenchymal transition and cancer associated fibroblasts in patients with head and neck cancer. Oncotarget 2015, 6, 34288–34299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ram, H.; Sarkar, J.; Kumar, H.; Konwar, R.; Bhatt, M.L.B.; Mohammad, S. Oral Cancer: Risk Factors and Molecular Pathogenesis. J. Maxillofac. Oral Surg. 2011, 10, 132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bagnardi, V.; Rota, M.; Botteri, E.; Tramacere, I.; Islami, F.; Fedirko, V.; Scotti, L.; Jenab, M.; Turati, F.; Pasquali, E.; et al. Light alcohol drinking and cancer: A meta-analysis. Ann. Oncol. 2013, 24, 301–308. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mogre, S.; Makani, V.; Pradhan, S.; Devre, P.; More, S.; Vaidya, M.; Dmello, C. Biomarker Potential of Vimentin in Oral Cancers. Life 2022, 12, 150. https://doi.org/10.3390/life12020150
Mogre S, Makani V, Pradhan S, Devre P, More S, Vaidya M, Dmello C. Biomarker Potential of Vimentin in Oral Cancers. Life. 2022; 12(2):150. https://doi.org/10.3390/life12020150
Chicago/Turabian StyleMogre, Saie, Vidhi Makani, Swapnita Pradhan, Pallavi Devre, Shyam More, Milind Vaidya, and Crismita Dmello. 2022. "Biomarker Potential of Vimentin in Oral Cancers" Life 12, no. 2: 150. https://doi.org/10.3390/life12020150






