High Expression of Solute Carrier Family 2 Member 1 (SLC2A1) in Cancer Cells Is an Independent Unfavorable Prognostic Factor in Pediatric Malignant Peripheral Nerve Sheath Tumor
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Immunohistochemistry
2.3. Markers of Systemic Inflammation
2.4. Statistical Analysis
3. Results
3.1. Expression of Hypoxic Markers by MPNST
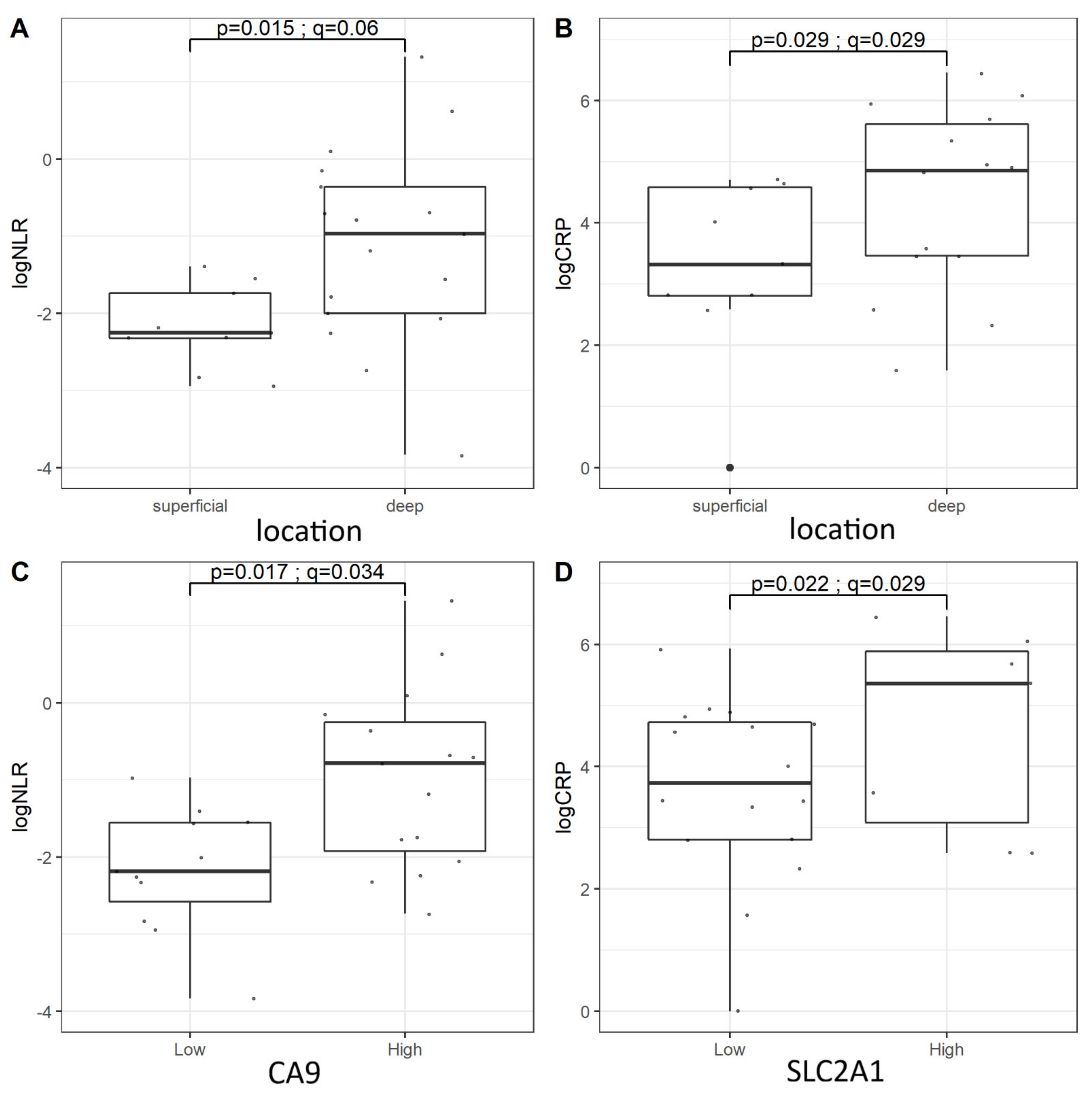
3.2. Complete Blood Count, LDH, and CRP
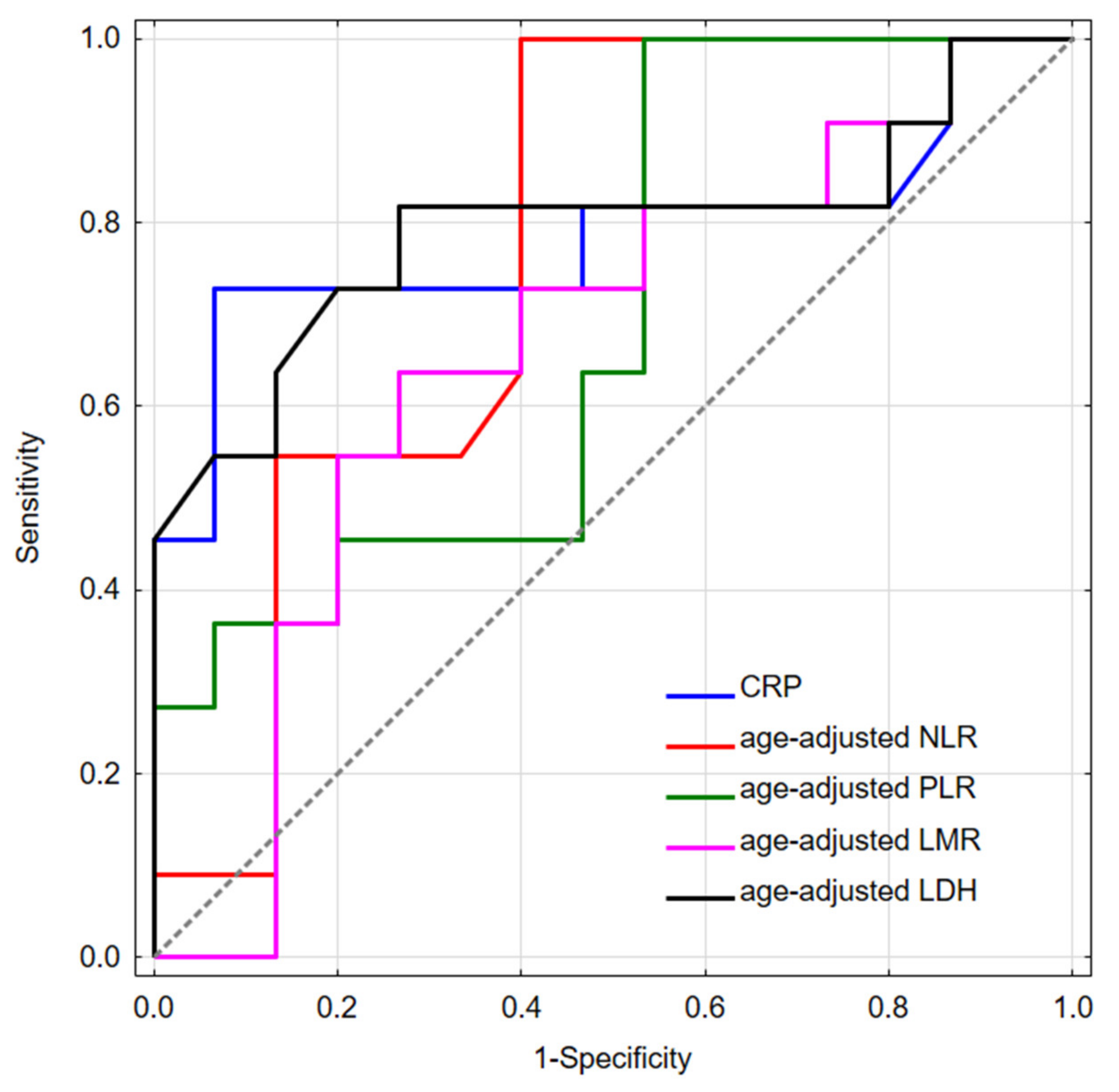
3.3. Survival Analysis—Markers of Hypoxia
3.4. Survival Analysis—NLR and CRP
3.5. Multivariable Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Noesel, M.M.; Orbach, D.; Brennan, B.; Kelsey, A.; Zanetti, I.; Salvo, G.L.; Gaze, M.N.; Craigie, R.J.; McHugh, K.; Francotte, N.; et al. Outcome and prognostic factors in pediatric malignant peripheral nerve sheath tumors: An analysis of the European Pediatric Soft Tissue Sarcoma Group (EpSSG) NRSTS-2005 prospective study. Pediatr. Blood Cancer 2019, 66, e27833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martin, E.; Coert, J.H.; Flucke, U.E.; Slooff, W.B.M.; van de Sande, M.A.J.; van Noesel, M.M.; Grünhagen, D.J.; Wijnen, M.H.W.A.; Verhoef, C. Neurofibromatosis-associated malignant peripheral nerve sheath tumors in children have a worse prognosis: A nationwide cohort study. Pediatr. Blood Cancer 2020, 67, e28138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meister, M.T.; Scheer, M.; Hallmen, E.; Stegmaier, S.; Vokuhl, C.; Kalle, T.; Fuchs, J.; Münter, M.; Niggli, F.; Ladenstein, R.; et al. Malignant peripheral nerve sheath tumors in children, adolescents, and young adults: Treatment results of five Cooperative Weichteilsarkom Studiengruppe (CWS) trials and one registry. J. Surg. Oncol. 2020, 122, 1337–1347. [Google Scholar] [CrossRef] [PubMed]
- Krawczyk, M.A.; Karpinsky, G.; Izycka-Swieszewska, E.; Gabrych, A.; Kunc, M.; Fatyga, A.; Garstka, M.; Styczewska, M.; Sokolewicz, E.M.; Szlagatys-Sidorkiewicz, A.; et al. Immunohistochemical assessment of cyclin D1 and p53 is associated with survival in childhood malignant peripheral nerve sheath tumor. Cancer Biomark. 2019, 24, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Eltzschig, H.K.; Carmeliet, P. Hypoxia and inflammation. N. Engl. J. Med. 2011, 364, 656–665. [Google Scholar] [CrossRef] [Green Version]
- Liao, D.; Johnson, R.S. Hypoxia: A key regulator of angiogenesis in cancer. Cancer Metastasis Rev. 2007, 26, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene 2010, 29, 625–634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masson, N.; Ratcliffe, P.J. Hypoxia signaling pathways in cancer metabolism: The importance of co-selecting interconnected physiological pathways. Cancer Metab. 2014, 2, 3. [Google Scholar] [CrossRef] [Green Version]
- Zambrano, A.; Molt, M.; Uribe, E.; Salas, M. Glut 1 in cancer cells and the inhibitory action of resveratrol as a potential therapeutic strategy. Int. J. Mol. Sci. 2019, 20, 3374. [Google Scholar] [CrossRef] [Green Version]
- Pastorek, J.; Pastorekova, S. Hypoxia-induced carbonic anhydrase IX as a target for cancer therapy: From biology to clinical use. Semin. Cancer Biol. 2015, 31, 52–64. [Google Scholar] [CrossRef]
- Semenza, G.L. HIF-1: Upstream and downstream of cancer metabolism. Curr. Opin. Genet. Dev. 2010, 20, 51–56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lucarini, L.; Magnelli, L.; Schiavone, N.; Crisci, A.; Innocenti, A.; Puccetti, L.; Cianchi, F.; Peri, S.; Supuran, C.T.; Papucci, L.; et al. Plasmatic carbonic anhydrase IX as a diagnostic marker for clear cell renal cell carcinoma. J. Enzyme Inhib. Med. Chem. 2018, 33, 234–240. [Google Scholar] [CrossRef] [Green Version]
- Triner, D.; Shah, Y.M. Hypoxic regulation of neutrophils in cancer. Int. J. Mol. Sci. 2019, 20, 4189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imtiyaz, H.Z.; Simon, M.C. Hypoxia-inducible factors as essential regulators of inflammation. Curr. Top. Microbiol. Immunol. 2010, 345, 105–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Proctor, M.J.; Talwar, D.; Balmar, S.M.; O’Reilly, D.S.J.; Foulis, A.K.; Horgan, P.G.; Morrison, D.S.; McMillan, D.C. The relationship between the presence and site of cancer, an inflammation-based prognostic score and biochemical parameters. Initial results of the Glasgow Inflammation Outcome Study. Br. J. Cancer 2010, 103, 870–876. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Dong, X.; Qu, Z.; Peng, K.; Sun, X.; Chen, R. Correlation between peripheral blood neutrophil-lymphocyte ratio and CD34 expression in prostate cancer. BMC Cancer 2020, 20, 900. [Google Scholar] [CrossRef]
- Nayak, A.; McDowell, D.T.; Kellie, S.J.; Karpelowsky, J. Elevated preoperative neutrophil–lymphocyte ratio is predictive of a poorer prognosis for pediatric patients with solid tumors. Ann. Surg. Oncol. 2017, 24, 3456–3462. [Google Scholar] [CrossRef]
- Seng, D.; Fang, Q.; Li, P.; Liu, F.; Liu, S.; Vasquez, L.; León, E.; Beltran, B.; Maza, I.; Oscanoa, M.; et al. Prognostic value of the pretreatment neutrophil-to-lymphocyte ratio in pediatric parotid cancer. J. Pediatr. Hematol. Oncol. 2017, 7, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Vasquez, L.; León, E.; Beltran, B.; Maza, I.; Oscanoa, M.; Geronimo, J. Pretreatment neutrophil-to-lymphocyte ratio and lymphocyte recovery: Independent prognostic factors for survival in pediatric sarcomas. J. Pediatr. Hematol. Oncol. 2017, 39, 538–546. [Google Scholar] [CrossRef]
- Yalon, M.; Toren, A.; Jabarin, D.; Fadida, E.; Constantini, S.; Mehrian-Shai, R. Elevated NLR may be a feature of pediatric brain cancer patients. Front. Oncol. 2019, 9, 327. [Google Scholar] [CrossRef]
- Bien, E.; Rapala, M.; Krawczyk, M.; Balcerska, A. The serum levels of soluble interleukin-2 receptor α and lactate dehydrogenase but not of B2-microglobulin correlate with selected clinico-pathological prognostic factors and response to therapy in childhood soft tissue sarcomas. J. Cancer Res. Clin. Oncol. 2010, 136, 293–305. [Google Scholar] [CrossRef]
- Krawczyk, M.A.; Styczewska, M.; Sokolewicz, E.M.; Kunc, M.; Gabrych, A.; Fatyga, A.; Izycka-Swieszewska, E.; Kazanowska, B.; Adamkiewicz-Drozynska, E.; Bien, E. Tumour expressions of hypoxic markers predict the response to neo-adjuvant chemotherapy in children with inoperable srcoma. Biomarkers 2019, 24, 538–548. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24275-0. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Fukushima, S.; Endo, M.; Matsumoto, Y.; Fukushi, J.; Matsunobu, T.; Kawaguchi, K.; Setsu, N.; IIda, K.; Yokoyama, N.; Nakagawa, M.; et al. Hypoxia-inducible factor 1 alpha is a poor prognostic factor and potential therapeutic target in malignant peripheral nerve sheath tumor. PLoS ONE 2017, 12, e0178064. [Google Scholar] [CrossRef]
- Rad, E.; Dodd, K.; Thomas, L.; Upadhyaya, M.; Tee, A. STAT3 and HIF1α signaling drives oncogenic cellular phenotypes in malignant peripheral nerve sheath tumors. Mol. Cancer Res. 2015, 13, 1149–1160. [Google Scholar] [CrossRef] [Green Version]
- Zhang, B.; Xie, Z.; Li, B. The clinicopathologic impacts and prognostic significance of GLUT1 expression in patients with lung cancer: A meta-analysis. Gene 2019, 689, 76–83. [Google Scholar] [CrossRef]
- Deng, Y.; Zou, J.; Deng, T.; Liu, J. Clinicopathological and prognostic significance of GLUT1 in breast cancer A meta-analysis. Medicine 2018, 97, e12961. [Google Scholar] [CrossRef]
- Yang, J.; Wen, J.; Tian, T.; Lu, Z.; Wang, Y.; Wang, Z.; Wang, X.; Yang, Y. GLUT-1 overexpression as an unfavorable prognostic biomarker in patients with colorectal cancer. Oncotarget 2017, 8, 11788–11796. [Google Scholar] [CrossRef] [Green Version]
- Achalandabaso Boira, M.; Di Martino, M.; Gordillo, C.; Adrados, M.; Martín-Pérez, E. GLUT-1 as a predictor of worse prognosis in pancreatic adenocarcinoma: Immunohistochemistry study showing the correlation between expression and survival. BMC Cancer 2020, 20, 909. [Google Scholar] [CrossRef]
- Hernández, F.; Navarro, M.; Encinas, J.L.; López Gutiérrez, J.C.; López Santamaría, M.; Leal, N.; Martínez, L.; Patrón, M.; Tovar, J.A. The role of GLUT1 immunostaining in the diagnosis and classification of liver vascular tumors in children. J. Pediatr. Surg. 2005, 40, 801–804. [Google Scholar] [CrossRef]
- Pinheiro, C.; Granja, S.; Longatto-Filho, A.; Faria, A.M.; Fragoso, M.C.B.V.; Lovisolo, S.M.; Bonatelli, M.; Costa, R.F.A.; Lerário, A.M.; Almeida, M.Q.; et al. GLUT1 expression in pediatric adrenocortical tumors: A promising candidate to predict clinical behavior. Oncotarget 2017, 8, 63835–63845. [Google Scholar] [CrossRef] [Green Version]
- Rakheja, D.; Khokhar, S.; Mitui, M.; Cost, N.G. Immunohistochemical expression of GLUT1 and its correlation with unfavorable histology and TP53 codon 72 polymorphism in wilms tumors. Pediatr. Dev. Pathol. 2012, 15, 286–292. [Google Scholar] [CrossRef]
- Ramani, P.; Headford, A.; May, M.T. GLUT1 protein expression correlates with unfavourable histologic category and high risk in patients with neuroblastic tumours. Virchows Arch. 2013, 462, 203–209. [Google Scholar] [CrossRef]
- Carmeliet, P. VEGF as a key mediator of angiogenesis in cancer. Oncology 2005, 69, 4–10. [Google Scholar] [CrossRef]
- Widemann, B.C.; Lu, Y.; Reinke, D.; Okuno, S.H.; Meyer, C.F.; Cote, G.M.; Chugh, R.; Milhem, M.M.; Hirbe, A.C.; Kim, A.; et al. Targeting sporadic and neurofibromatosis Type 1 (NF1) related refractory malignant peripheral nerve sheath tumors (MPNST) in a Phase II study of everolimus in combination with bevacizumab (SARC016). Sarcoma 2019, 2019, 7656747. [Google Scholar] [CrossRef] [Green Version]
- Geller, J.I.; Fox, E.; Turpin, B.K.; Goldstein, S.L.; Liu, X.; Minard, C.G.; Kudgus, R.A.; Reid, J.M.; Berg, S.L.; Weigel, B.J. A study of axitinib, a VEGF receptor tyrosine kinase inhibitor, in children and adolescents with recurrent or refractory solid tumors: A Children’s Oncology Group phase 1 and pilot consortium trial (ADVL1315). Cancer 2018, 124, 4548–4555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tuomisto, A.E.; Mäkinen, M.J.; Väyrynen, J.P. Systemic inflammation in colorectal cancer: Underlying factors, effects, and prognostic significance. World J. Gastroenterol. 2019, 25, 4383–4404. [Google Scholar] [CrossRef] [PubMed]
- Bousquet, P.A.; Meltzer, S.; Sønstevold, L.; Esbensen, Y.; Dueland, S.; Flatmark, K.; Sitter, B.; Bathen, T.F.; Seierstad, T.; Redalen, K.R.; et al. Markers of mitochondrial metabolism in tumor hypoxia, systemic inflammation, and adverse outcome of rectal cancer. Transl. Oncol. 2019, 12, 76–83. [Google Scholar] [CrossRef] [PubMed]

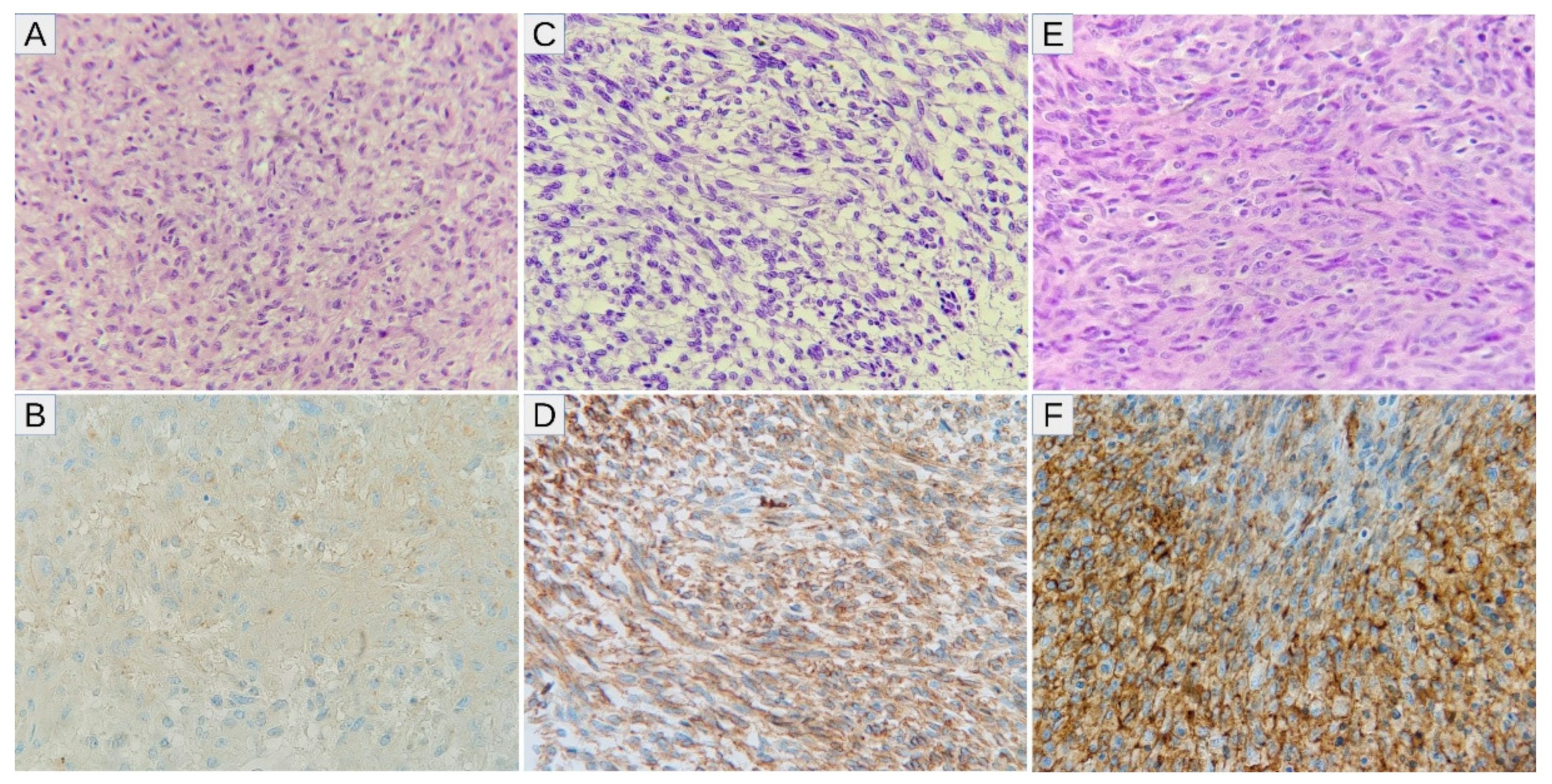

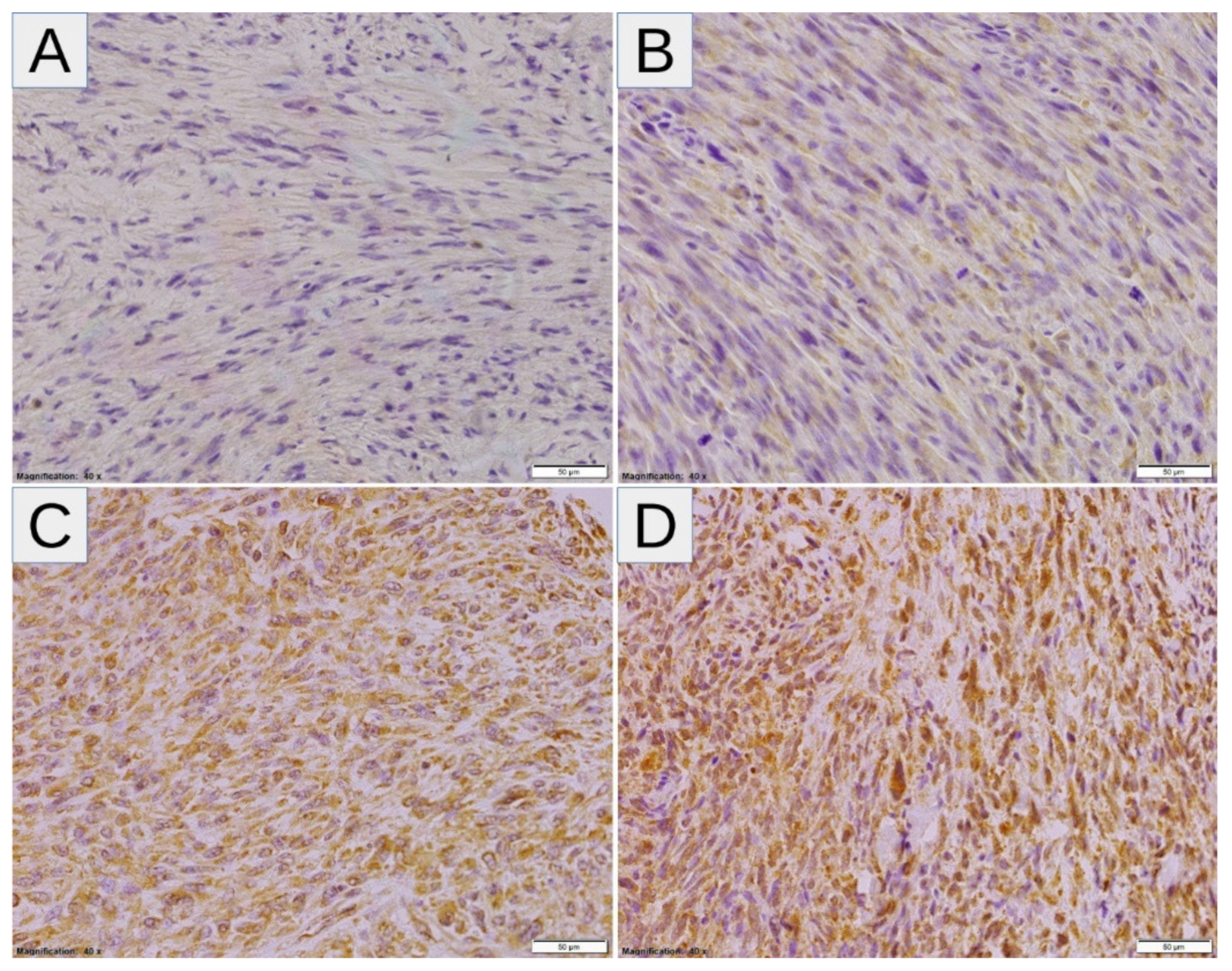





| Percentage of Immunopositive Cells | |||||
|---|---|---|---|---|---|
| Intensity of the IHC staining | >50% | 26–50% | 6–25% | 0–5% | |
| High +++ | 12 | 9 | 6 | 3 | |
| Intermediate ++ | 11 | 8 | 5 | 2 | |
| Low + | 10 | 7 | 4 | 1 | |
| No staining | 0 | ||||
| Variable | SLC2A1 | HIF1A | VEGFA | CA9 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Low | High | p-Value/ q-Value | Low | High | p-Value/ q-Value | Low | High | p-Value/ q-Value | Low | High | p-Value/ q-Value | ||
| Baseline distant and/or nodal metastases | No | 13 | 5 | 0.189/0.378 | 11 | 7 | 0.035/0.420 | 5 | 13 | 0.667/0.889 | 8 | 10 | 1.000/1.000 |
| Yes | 3 | 5 | 1 | 7 | 3 | 5 | 3 | 5 | |||||
| Location | Superficial | 8 | 1 | 0.087/0.552 | 5 | 4 | 0.484/0.830 | 3 | 6 | 1.000/1.000 | 6 | 3 | 0.103/0.412 |
| Deep | 8 | 9 | 7 | 10 | 5 | 12 | 5 | 12 | |||||
| NF1 | No | 12 | 4 | 0.108/0.324 | 8 | 8 | 0.619/0.928 | 5 | 11 | 1.000/1.000 | 9 | 7 | 0.109/0.262 |
| Yes | 4 | 6 | 4 | 6 | 3 | 7 | 2 | 8 | |||||
| SLC2A1 | HIF1A | VEGFA | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Low | High | p-Value/ q-Value | Low | High | p-Value/ q-Value | Low | High | p-Value/ q-Value | ||
| HIF1A | Low | 9 | 3 | 0.191/0.573 | N/A | N/A | ||||
| High | 7 | 7 | ||||||||
| VEGFA | Low | 6 | 2 | 0.419/0.628 | 5 | 3 | 0.265/0.530 | N/A | ||
| High | 10 | 8 | 7 | 11 | ||||||
| CA9 | Low | 10 | 1 | 0.014/0.084 | 5 | 6 | 0.951/1.000 | 3 | 8 | 1.000/1.000 |
| High | 6 | 9 | 7 | 8 | 5 | 10 | ||||
| Variable | AUC | ±95% CI | Cut-Off | p-Value | q-Value |
|---|---|---|---|---|---|
| NLR | 0.76 | 0.57–0.95 | 0.24 | 0.007 | 0.017 |
| PLR | 0.69 | 0.49–0.90 | 0.45 | 0.062 | 0.103 |
| LMR | 0.66 | 0.44–0.88 | 3.88 | 0.149 | 0.149 |
| CRP | 0.78 | 0.57–0.99 | 31 mg/L | 0.007 | 0.035 |
| LDH | 0.66 | 0.44–0.88 | 0.99 | 0.144 | 0.180 |
| Variable | 2-Year RFS [%] | 10-Year RFS [%] | Log-Rank p-Value | Log-Rank q-Value | Multivariate Cox Regression | ||||
|---|---|---|---|---|---|---|---|---|---|
| Low | High | Low | High | HR | 95% CI | p-Value | |||
| SLC2A1 | 67 | 20 | 59 | 0 | 0.007 | 0.021 | 3.31 | 1.08–10.09 | 0.036 |
| HIF1A | 67 | 31 | 58 | 11 | 0.176 | 0.264 | * | ||
| CA9 | 55 | 43 | 55 | 22 | 0.275 | 0.275 | * | ||
| VEGFA | 75 | 36 | 75 | 16 | 0.005 | 0.030 | 4.40 | 0.95–20.34 | 0.058 |
| NLR | 75 | 35 | 75 | 17 | 0.009 | 0.018 | * | ||
| CRP | 50 | 44 | 50 | 11 | 0.206 | 0.247 | * | ||
| 2-year OS [%] | 10-year OS [%] | ||||||||
| SLC2A1 | 94 | 40 | 82 | 10 | <0.001 | 0.036 | 12.20 | 2.55–58.33 | 0.002 |
| HIF1A | 92 | 57 | 79 | 23 | 0.016 | 0.024 | * | ||
| CA9 | 82 | 67 | 82 | 37 | 0.161 | 0.193 | * | ||
| VEGFA | 88 | 66 | 73 | 38 | 0.194 | 0.194 | * | ||
| NLR | 100 | 60 | 100 | 31 | 0.007 | 0.014 | * | ||
| CRP | 82 | 56 | 82 | 11 | 0.003 | 0.009 | * | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krawczyk, M.A.; Kunc, M.; Styczewska, M.; Gabrych, A.; Karpinsky, G.; Izycka-Swieszewska, E.; Bien, E. High Expression of Solute Carrier Family 2 Member 1 (SLC2A1) in Cancer Cells Is an Independent Unfavorable Prognostic Factor in Pediatric Malignant Peripheral Nerve Sheath Tumor. Diagnostics 2021, 11, 598. https://doi.org/10.3390/diagnostics11040598
Krawczyk MA, Kunc M, Styczewska M, Gabrych A, Karpinsky G, Izycka-Swieszewska E, Bien E. High Expression of Solute Carrier Family 2 Member 1 (SLC2A1) in Cancer Cells Is an Independent Unfavorable Prognostic Factor in Pediatric Malignant Peripheral Nerve Sheath Tumor. Diagnostics. 2021; 11(4):598. https://doi.org/10.3390/diagnostics11040598
Chicago/Turabian StyleKrawczyk, Malgorzata A., Michal Kunc, Malgorzata Styczewska, Anna Gabrych, Gabrielle Karpinsky, Ewa Izycka-Swieszewska, and Ewa Bien. 2021. "High Expression of Solute Carrier Family 2 Member 1 (SLC2A1) in Cancer Cells Is an Independent Unfavorable Prognostic Factor in Pediatric Malignant Peripheral Nerve Sheath Tumor" Diagnostics 11, no. 4: 598. https://doi.org/10.3390/diagnostics11040598






