Biochemical and Morphological Mechanisms Underlying the Performance and Preference of Fall Armyworm (Spodoptera frugiperda) on Wheat and Faba Bean Plants
Abstract
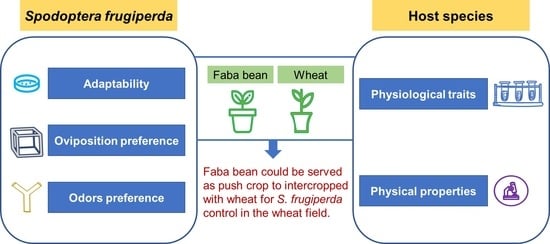
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Plants
2.3. Adaptability Assays
2.4. Plant Leaf Nutrients and Metabolites Analyses
2.5. Oviposition Preference Tests
2.6. Y-Tube Olfactometer Bioassays
2.7. Morphological Characteristics of Plant Leaves
2.8. Statistical Analyses
3. Results
3.1. Developmental Duration of S. frugiperda on Wheat and Faba Bean Leaves
3.2. Survival Rate of S. frugiperda Larvae on Wheat and Faba Bean Leaves
3.3. Pupal Weight of S. frugiperda after Reared on Wheat and Faba Bean Leaves
3.4. Contents of Biochemical Components in Wheat and Faba Bean Leaves
3.5. Oviposition Preference of S. frugiperda Female Moths to Host Species
3.6. Preference of S. frugiperda Female Moths to Volatiles Emitted from Two Host Species
3.7. Observation of Trichome Types and Density on Wheat and Faba Bean Leaves
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sparks, A.N. A review of the biology of the fall armyworm. Fla. Èntomol. 1979, 62, 82–87. [Google Scholar] [CrossRef]
- Feldmann, F.; Rieckmann, U.; Winter, S. The spread of the fall armyworm Spodoptera frugiperda in Africa—What should be done next? J. Plant Dis. Prot. 2019, 126, 97–101. [Google Scholar] [CrossRef]
- Mallapur, C.; Naik, A.; Hagari, S. Status of alien pest fall armyworm, Spodoptera frugiperda (J.E. Smith) on maize in Northern Karnataka. J. Entomol. Zool. Stud. 2018, 6, 432–436. [Google Scholar]
- CABI. Spodoptera frugiperda (Fall Armyworm). Available online: https://www.cabi.org/isc/datasheet/29810#todistribution (accessed on 1 November 2021).
- Guo, J.F.; Zhao, J.Z.; He, K.L.; Zhang, F.; Wang, Z.Y. Potential invasion of the crop-devastating insect pest fall armyworm Spodoptera frugiperda to China. Plant Prot. 2018, 44, 1–10. [Google Scholar]
- Sun, X.-X.; Hu, C.-X.; Jia, H.-R.; Wu, Q.-L.; Shen, X.-J.; Zhao, S.-Y.; Jiang, Y.-Y.; Wu, K.-M. Case study on the first immigration of fall armyworm, Spodoptera frugiperda invading into China. J. Integr. Agric. 2021, 20, 664–672. [Google Scholar] [CrossRef]
- China National Agro-Tech Extension and Service Center. Occurrence Trend of Fall Armyworm, Spodoptera frugiperda in Autumn Maize. Available online: https://www.natesc.org.cn/news/des?id=b4ca3130-c5a2-4f0d-9762-f248e0b94985&Category=%E5%85%A8%E6%96%87%E6%90%9C%E7%B4%A2&CategoryId=07e72766-0a38-4dbd-a6a3-c823ce1172bd (accessed on 3 November 2021).
- Hruska, A.J.; Gould, F. Fall armyworm (Lepidoptera: Noctuidae) and Diatraea lineolata (Lepidoptera: Pyralidae): Impact of larval population level and temporal occurrence on maize yield in Nicaragua. J. Econ. Èntomol. 1997, 90, 611–622. [Google Scholar] [CrossRef]
- Montezano, D.G.; Specht, A.; Sosa-Gómez, D.R.; Roque-Specht, V.F.; Sousa-Silva, J.C.; Paula-Moraes, S.V.; Peterson, J.A.; Hunt, T.E. Host plants of Spodoptera frugiperda (Lepidoptera: Noctuidae) in the Americas. Afr. Entomol. 2018, 26, 286–300. [Google Scholar] [CrossRef] [Green Version]
- Guo, J.-F.; Zhang, M.-D.; Gao, Z.-P.; Wang, D.-J.; He, K.-L.; Wang, Z.-Y. Comparison of larval performance and oviposition preference of Spodoptera frugiperda among three host plants: Potential risks to potato and tobacco crops. Insect Sci. 2020, 28, 602–610. [Google Scholar] [CrossRef]
- Chen, W.; Weng, Q.; Nie, R.; Zhang, H.; Jing, X.; Wang, M.; Li, Y.; Mao, J.; Zhang, L. Optimizing photoperiod, exposure time, and host-to-parasitoid ratio for mass-rearing of Telenomus remus, an egg parasitoid of Spodoptera frugiperda, on Spodoptera litura Eggs. Insects 2021, 12, 1050. [Google Scholar] [CrossRef]
- National Bureau of Statistics. Crops Planting Area in China in 2020. Available online: https://data.stats.gov.cn/easyquery.htm?cn=C01&zb=A0D0E&sj=2020 (accessed on 30 November 2021).
- Jiang, Y.Y.; Liu, J.; Wu, Q.L.; Ciren, Z.G.; Zeng, J. Investigation on winter breeding and overwintering areas of Spodoptera frugiperda in China. Plant Prot. 2021, 47, 212–217. [Google Scholar] [CrossRef]
- China National Agro-Tech Extension and Service Center. The National Agro-Tech Extension and Service Center Organized Experts to Investigate the Overwintering Base of Fall Armyworm, Spodoptera frugiperda. Available online: https://www.natesc.org.cn/news/des?id=874a0841-45a6-4225-8d63-a44328986a3a&Category=%E5%85%A8%E6%96%87%E6%90%9C%E7%B4%A2&CategoryId=07e72766-0a38-4dbd-a6a3-c823ce1172bd (accessed on 3 November 2021).
- Wang, W.; He, P.; Zhang, Y.; Liu, T.; Jing, X.; Zhang, S. The population growth of Spodoptera frugiperda on six cash crop species and implications for its occurrence and damage potential in China. Insects 2020, 11, 639. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, Y.; Chen, J.L. Feeding preference and adaptability of Spodoptera frugiperda (Lepidoptera: Noctuidae) on different wheat cultivars in relation to leaf biochemical contents. Acta Entomol. Sin. 2021, 64, 230–239. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, X.G.; Lv, G.Q.; Hao, X.Z.; Li, S.H.; Li, G.P.; Feng, H.Q. Comparison of population fitness of Spodoptera frugiperda (Lepidoptera: Noctuidae) feeding on wheat and different varieties of maize. Plant Prot. 2020, 46, 126–131. [Google Scholar] [CrossRef]
- Hailu, G.; Niassy, S.; Zeyaur, K.R.; Ochatum, N.; Subramanian, S. Maize–legume intercropping and push–pull for management of fall armyworm, stemborers, and striga in Uganda. Agron. J. 2018, 110, 2513–2522. [Google Scholar] [CrossRef] [Green Version]
- Midega, C.A.; Pittchar, J.O.; Pickett, J.A.; Hailu, G.W.; Khan, Z.R. A climate-adapted push-pull system effectively controls fall armyworm, Spodoptera frugiperda (J.E. Smith), in maize in East Africa. Crop Prot. 2018, 105, 10–15. [Google Scholar] [CrossRef]
- Scheidegger, L.; Niassy, S.; Midega, C.; Chiriboga, X.; Delabays, N.; Lefort, F.; Zürcher, R.; Hailu, G.; Khan, Z.; Subramanian, S. The role of Desmodium intortum, Brachiaria sp. and Phaseolus vulgaris in the management of fall armyworm Spodoptera frugiperda (J. E. Smith) in maize cropping systems in Africa. Pest Manag. Sci. 2021, 77, 2350–2357. [Google Scholar] [CrossRef] [PubMed]
- Bai, W.L.; Zhang, M.Y.; Ren, J.B.; Tang, L.; Zheng, Y.; Xiao, J.X. Simulation of crop growth curve and analysis of interspecific interaction in wheat and faba bean intercropping system. J. Appl. Ecol. 2018, 29, 4037–4046. [Google Scholar]
- Tang, Y.F.; Xu, Y.B.; Zheng, Y.; Lei, B.K. Effects of wheat and faba bean intercropping on microorganism involved in nitrogen transformation in the rhizosphere soils. J. Agric. Resour. Environ. 2016, 33, 482–490. [Google Scholar] [CrossRef]
- Lopes, T.; Hatt, S.; Xu, Q.; Chen, J.; Liu, Y.; Francis, F. Wheat (Triticum aestivum L.)-based intercropping systems for biological pest control. Pest Manag. Sci. 2016, 72, 2193–2202. [Google Scholar] [CrossRef]
- Yang, J.C.; Liu, J.J.; An, Z.Y.; Zhu, Y.Y.; Li, C.Y.; Chen, X.D.; Lu, Y.E.; Li, H.Y.; Dian, L.F. Analyses on effect of interplanting on diseases and pests control and yield increase of wheat and faba bean. J. Yunnan Agric. Univ. 2009, 24, 340–348. [Google Scholar]
- Wang, X.; Shen, C.; Meng, P.; Tan, G.; Lv, L. Analysis and review of trichomes in plants. BMC Plant Biol. 2021, 21, 70. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, S.R. Impact of Soybean Trichomes on Aphis glycines (Hemiptera: Aphididae) and Their Interaction with Natural Enemy Abundance and Predation. Ph.D. Thesis, Iowa State University, Ames, IA, USA, 2017. [Google Scholar]
- Coapio, G.G.; Cruz-López, L.; Guerenstein, P.; Malo, E.A.; Rojas, J.C. Oviposition preference and larval performance and behavior of Trichoplusia ni (Lepidoptera: Noctuidae) on host and nonhost plants. Arthropod-Plant Interact. 2018, 12, 267–276. [Google Scholar] [CrossRef]
- Ode, P.J. Plant chemistry and natural enemy fitness: Effects on herbivore and natural enemy interactions. Annu. Rev. Èntomol. 2006, 51, 163–185. [Google Scholar] [CrossRef] [Green Version]
- Coppola, M.; Diretto, G.; Digilio, M.C.; Woo, S.L.; Giuliano, G.; Molisso, D.; Pennacchio, F.; Lorito, M.; Rao, R. Transcriptome and metabolome reprogramming in tomato plants by Trichoderma harzianum strain T22 primes and enhances defense responses against aphids. Front. Physiol. 2019, 10, 745. [Google Scholar] [CrossRef] [PubMed]
- Birke, A.; Aluja, M. Do mothers really know best? Complexities in testing the preference-performance hypothesis in polyphagous frugivorous fruit flies. Bull. Èntomol. Res. 2018, 108, 674–684. [Google Scholar] [CrossRef] [PubMed]
- Silva, D.B.; Urbaneja, A.; Pérez-Hedo, M. Response of mirid predators to synthetic herbivore-induced plant volatiles. Entomol. Exp. Appl. 2021, 169, 125–132. [Google Scholar] [CrossRef]
- Light, D.M.; Flath, R.A.; Buttery, R.G.; Zalom, F.G.; Rice, R.E.; Dickens, J.C.; Jang, E.B. Host-plant green-leaf volatiles synergize the synthetic sex pheromones of the corn earworm and codling moth (Lepidoptera). Chemoecology 1993, 4, 145–152. [Google Scholar] [CrossRef]
- McCormick, A.C.; Reinecke, A.; Gershenzon, J.; Unsicker, S.B. Feeding experience affects the behavioral response of polyphagous gypsy moth caterpillars to herbivore-induced poplar volatiles. J. Chem. Ecol. 2016, 42, 382–393. [Google Scholar] [CrossRef] [Green Version]
- Züst, T.; Agrawal, A.A. Trade-offs between plant growth and defense against insect herbivory: An emerging mechanistic synthesis. Annu. Rev. Plant Biol. 2017, 68, 513–534. [Google Scholar] [CrossRef] [Green Version]
- Agrawal, A.A.; Fishbein, M.; Jetter, R.; Salminen, J.; Goldstein, J.B.; Freitag, A.E.; Sparks, J.P. Phylogenetic ecology of leaf surface traits in the milkweeds (Asclepias spp.): Chemistry, ecophysiology, and insect behavior. New Phytol. 2009, 183, 848–867. [Google Scholar] [CrossRef]
- Rashmi, M.A.; Verghese, A.; Shivashankar, S.; Chakravarthy, A.; Sumathi, M.; Kandakoor, S. Does change in tannin content in mango (Mangifera indica) fruits influence the extent of fruit fly (Bactrocera dorsalis Hendel) herbivory? J. Entomol. Zool. Stud. 2017, 381, 381–385. [Google Scholar]
- Sotelo-Cardona, P.; Chuang, W.-P.; Lin, M.-Y.; Chiang, M.-Y.; Ramasamy, S. Oviposition preference not necessarily predicts offspring performance in the fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae) on vegetable crops. Sci. Rep. 2021, 11, 15885. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Guo, J.F.; Wang, Q.Y.; He, K.L.; Wang, Z.Y. Resistance evaluation of corn hybrid Jingke 968 silks to Ostrinia furnacalis and related resistance mechanism. J. Environ. Entomol. 2019, 41, 25–32. [Google Scholar] [CrossRef]
- Lattanzio, V.; Terzano, R.; Cicco, N.; Cardinali, A.; Di Venere, D.; Linsalata, V. Seed coat tannins and bruchid resistance in stored cowpea seeds. J. Sci. Food Agric. 2005, 85, 839–846. [Google Scholar] [CrossRef]
- Carrasco, D.; Larsson, M.C.; Anderson, P. Insect host plant selection in complex environments. Curr. Opin. Insect Sci. 2015, 8, 1–7. [Google Scholar] [CrossRef]
- Bruce, T.J.; Wadhams, L.J.; Woodcock, C.M. Insect host location: A volatile situation. Trends Plant Sci. 2005, 10, 269–274. [Google Scholar] [CrossRef]
- Thöming, G.; Norli, H.R. Olfactory cues from different plant species in host selection by female pea moths. J. Agric. Food Chem. 2015, 63, 2127–2136. [Google Scholar] [CrossRef]
- Pan, J.W.; Volkan, P.C. Mechanisms of development and evolution of the insect olfactory system. Cell Dev. Biol. 2013, 2, 130. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Cui, Z.; Si, P.; Liu, Y.; Zhou, Q.; Wang, G. Characterization of a specific odorant receptor for linalool in the Chinese citrus fly Bactrocera minax (Diptera: Tephritidae). Insect Biochem. Mol. Biol. 2020, 122, 103389. [Google Scholar] [CrossRef]
- Xie, Y.; Huang, Q.; Rao, Y.; Hong, L.; Zhang, D. Efficacy of Origanum vulgare essential oil and carvacrol against the housefly, Musca domestica L. (Diptera: Muscidae). Environ. Sci. Pollut. Res. 2019, 26, 23824–23831. [Google Scholar] [CrossRef]
- Müller, G.C.; Junnila, A.; Butler, J.; Kravchenko, V.D.; Revay, E.E.; Weiss, R.W.; Schlein, Y. Efficacy of the botanical repellents geraniol, linalool, and citronella against mosquitoes. J. Vector Ecol. 2009, 34, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Zhu, Q.Z.; Tan, Y.T.; Ma, Q.L.; Wang, R.F.; Zhang, M.F.; Xu, H.H.; Zhang, Z.X. Artificial diets and rearing technique of Spodoptera frugiperda (J.E. Smith) in laboratory. J. Environ. Entomol. 2019, 41, 742–747. [Google Scholar] [CrossRef]
- Akinci, S.; Losel, D.M. The soluble sugars determination in cucurbitaceae species under water stress and recovery periods. Adv. Environ. Biol. 2009, 3, 175–183. [Google Scholar]
- He, L.-M.; Wang, T.-L.; Chen, Y.-C.; Ge, S.-S.; Wyckhuys, K.A.; Wu, K.-M. Larval diet affects development and reproduction of East Asian strain of the fall armyworm, Spodoptera frugiperda. J. Integr. Agric. 2021, 20, 736–744. [Google Scholar] [CrossRef]
- Batyrshina, Z.S.; Yaakov, B.; Shavit, R.; Singh, A.; Tzin, V. Comparative transcriptomic and metabolic analysis of wild and domesticated wheat genotypes reveals differences in chemical and physical defense responses against aphids. BMC Plant Biol. 2020, 20, 19. [Google Scholar] [CrossRef] [Green Version]
- Xu, Q.; Hatt, S.; Lopes, T.; Zhang, Y.; Bodson, B.; Chen, J.; Francis, F. A push–pull strategy to control aphids combines intercropping with semiochemical releases. J. Pest Sci. 2018, 91, 93–103. [Google Scholar] [CrossRef]
- Xie, H.-C.; Chen, J.-L.; Cheng, D.-F.; Zhou, H.-B.; Sun, J.-R.; Liu, Y.; Francis, F. Impact of wheat-mung bean intercropping on English grain aphid (Hemiptera: Aphididae) populations and its natural enemy. J. Econ. Èntomol. 2012, 105, 854–859. [Google Scholar] [CrossRef]
- He, L.-M.; Wu, Q.-L.; Gao, X.-W.; Wu, K.-M. Population life tables for the invasive fall armyworm, Spodoptera frugiperda fed on major oil crops planted in China. J. Integr. Agric. 2021, 20, 745–754. [Google Scholar] [CrossRef]
- Lin, X.Q.; Huang, J.S.; Cai, S.P.; Ke, Y.Z.; Zeng, L.Q.; He, X.Y.; Chen, S.L.; Wu, H.Z.; Li, X.M. Correlation of inclusions contents of different Casuarina spp. strains and resistibility to Anoplophora chinensis. J. Cent. South Univ. For. Technol. 2014, 34, 37–41. [Google Scholar] [CrossRef]
- Capinera, J.L.; Renaud, A.R.; Roehrig, N.E. Chemical basis for host selection by Hemileuca oliviae: Role of tannins in preference of C4 grasses. J. Chem. Ecol. 1983, 9, 1425–1437. [Google Scholar] [CrossRef] [PubMed]
- Hare, J.D. Ecological role of volatiles produced by plants in response to damage by herbivorous insects. Annu. Rev. Èntomol. 2011, 56, 161–180. [Google Scholar] [CrossRef]
- Hare, J.D. How insect herbivores drive the evolution of plants. Science 2012, 338, 50–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Züst, T.; Heichinger, C.; Grossniklaus, U.; Harrington, R.; Kliebenstein, D.J.; Turnbull, L.A. Natural enemies drive geographic variation in plant defenses. Science 2012, 338, 116–119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Webster, B.; Gezan, S.; Bruce, T.; Hardie, J.; Pickett, J. Between plant and diurnal variation in quantities and ratios of volatile compounds emitted by Vicia faba plants. Phytochemistry 2010, 71, 81–89. [Google Scholar] [CrossRef]
- Piesik, D.; Rochat, D.; Delaney, K.J.; Marion-Poll, F. Orientation of European corn borer first instar larvae to synthetic green leaf volatiles. J. Appl. Èntomol. 2012, 137, 234–240. [Google Scholar] [CrossRef] [Green Version]
- Piesik, D.; Rochat, D.; Delaney, K.J.; Marion-Poll, F. Pulsed odors from maize or spinach elicit orientation in European corn borer neonate larvae. J. Chem. Ecol. 2009, 35, 1032–1042. [Google Scholar] [CrossRef]
- Guhling, O.; Kinzler, C.; Dreyer, M.; Bringmann, G.; Jetter, R. Surface composition of myrmecophilic plants: Cuticular wax and glandular trichomes on leaves of Macaranga tanarius. J. Chem. Ecol. 2005, 31, 2323–2341. [Google Scholar] [CrossRef]
- Ramaswamy, S.B. Host finding by moths: Sensory modalities and behaviours. J. Insect Physiol. 1988, 34, 235–249. [Google Scholar] [CrossRef]
- Lambert, L.; Beach, R.M.; Kilen, T.C.; Todd, J.W. Soybean pubescence and its influence on larval development and oviposition preference of lepidopterous insects. Crop Sci. 1992, 32, 463–466. [Google Scholar] [CrossRef]







| Stage | Wheat | Faba Bean | Significant | ||
|---|---|---|---|---|---|
| n | Days | n | Days | ||
| Egg | 100 | 2.00 ± 0.00 | 100 | 2.00 ± 0.00 | ns |
| Larva | |||||
| 1st instar | 100 | 2.23 ± 0.045 | 100 | 5.09 ± 0.16 | *** |
| 2nd instar | 100 | 2.32 ± 0.053 | 69 | 4.32 ± 0.21 | *** |
| 3rd instar | 100 | 2.60 ± 0.065 | 39 | 5.31 ± 0.29 | *** |
| 4th instar | 99 | 2.54 ± 0.056 | 34 | 4.03 ± 0.21 | *** |
| 5th instar | 99 | 2.56 ± 0.071 | 30 | 4.03 ± 0.23 | *** |
| 6th instar | 96 | 2.01 ± 0.060 | 27 | 3.22 ± 0.26 | *** |
| Pupa | 86 | 7.91 ± 0.13 | 25 | 7.40 ± 0.40 | ns |
| Adult | 80 | 5.83 ± 0.10 | 20 | 5.90 ± 0.25 | ns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, H.; Cheng, Y.; Wang, X.; Francis, F.; Wang, Q.; Liu, X.; Zhang, Y.; Chen, J. Biochemical and Morphological Mechanisms Underlying the Performance and Preference of Fall Armyworm (Spodoptera frugiperda) on Wheat and Faba Bean Plants. Insects 2022, 13, 317. https://doi.org/10.3390/insects13040317
Liu H, Cheng Y, Wang X, Francis F, Wang Q, Liu X, Zhang Y, Chen J. Biochemical and Morphological Mechanisms Underlying the Performance and Preference of Fall Armyworm (Spodoptera frugiperda) on Wheat and Faba Bean Plants. Insects. 2022; 13(4):317. https://doi.org/10.3390/insects13040317
Chicago/Turabian StyleLiu, Huan, Yumeng Cheng, Xiaoqing Wang, Frédéric Francis, Qian Wang, Xiaobei Liu, Yong Zhang, and Julian Chen. 2022. "Biochemical and Morphological Mechanisms Underlying the Performance and Preference of Fall Armyworm (Spodoptera frugiperda) on Wheat and Faba Bean Plants" Insects 13, no. 4: 317. https://doi.org/10.3390/insects13040317