1. Introduction
Temperature is one of the major environmental factors for regulating animal health and survival [
1]. It also affects production performance or reduces animal welfare. In critical environmental conditions, the vital physiological processes of homoeothermic animals are usually compromised [
1]. Cold stress often occurs in animal management [
2]. Low temperature has a negative influence on organisms, including leading to metabolic suppression [
3], disrupting hormone release [
4], and affecting the activity of protective enzymes [
5], which can inhibit growth and increase mortality [
2,
6]. Low temperature also increases the immune-suppression level, free radical load, and leads to oxidative stress [
7]. The animal immune system is often vulnerable to low temperatures and through the exacerbation of pathophysiological conditions [
8]. Cold stress can also cause damage to the bursa of poultry, heart, intestinal tissue, and other slow development, thus affecting the growth of poultry and even causing death [
9,
10,
11].
Low temperature is known as a major risk factor for diminishing the growth performance and survival of chicks [
12]. Typical features of chicks include short villi, hypoplasia of organs, and an inadequate thermoregulatory system [
13]. The hatching and brood temperature of chicks is higher than most other animals such as cattle and sheep. Chicks just out of the shell are highly susceptible to low temperatures, and once the ambient temperature is not sufficient for the chick to grow, this often leads to a high mortality rate [
14]. Low temperature also reduces chicken antioxidant capacity [
15,
16], immune function [
17], and damages tissues and organs in chicken [
18]. In addition, low temperature can reduce appetite, feed intake, and feed utilization of chicks, which can affect growth performance and product quality [
14]. Low temperatures tend to limit poultry production, especially causing serious economic losses to chick production, which is a bottleneck that needs to be solved [
15].
Traditional measures to reduce the impact of low-temperature stress on chicks include increasing padding, raising the environmental temperature, adding vitamins and antibiotics, etc., [
19,
20,
21], which are costly, energy consuming, and environmentally unfriendly [
20]. Using medicinal plant additives has been one of the common trends to improve the low-temperature stress on chicks in recent years [
22,
23,
24]. Feeding Chinese herbal medicines (CHM) can promote animal growth, enhance antioxidant levels, and enhance immune ability [
23,
25], in order to reduce the impact of environmental stress on the animal.
Eucommia ulmoides is a precious traditional CHM. Its bark, leaves, and flowers have various active ingredients, which can be used as medicine [
26]. The medicinal value of
Eucommia ulmoides has attracted more and more attention for its chemical composition [
27].
Eucommia ulmoides is rich in chemical components, such as iridoids, flavonoids, phenylpropanoids, lignans, and so on [
19,
28,
29].
Eucommia ulmoides has a variety of biological activities, including growth promotion [
30], metabolic regulation [
28], antioxidant abilities [
31], immune enhancement [
32], anti-inflammatory abilities [
28], and other effects. Chlorogenic acid, flavonoid, and rutin in
Eucommia ulmoides have an obvious effect of promoting growth [
33,
34,
35]. Chlorogenic acid, flavonoid, and quercetin have an antioxidant effect [
36,
37,
38,
39]. Furthermore, flavonoids can improve the immune ability of the body [
40]. Studies have reported that
Eucommia ulmoides and its extracts can be used as feed additives, which have the functions of improving growth performance, antioxidant capacity, and immune response [
41,
42]. However, to the best of our knowledge, very few studies have been conducted using
Eucommia ulmoides or its extracts for animal cold stress protection. In this study,
Eucommia ulmoides leaves (EUL) were used as raw materials to extract the active components and fed to chicks under cold stress, and their growth performance, organ development, serum antioxidant capacity, and immunoglobulin content were determined. The aim of the present study was to explore the effects of EUE on chicks in low-temperature environments, so as to provide a scientific basis for the application of EUE as a feed additive to protect cold-stressed chicks.
2. Materials and Methods
All procedures of animal experiments were performed according to the protocol approved by the Guizhou University Subcommittee of Experimental Animal Ethics (EAE-GZU-2020-P020, Guizhou, China). The chicks used in this study have been treated humanely. Great efforts have been made to minimize pain.
2.1. Preparation of EUE
EUL was picked from the South Campus of Guizhou University. The Eucommia ulmoides were dried at 80 °C and crushed by an ultrafine grinder. After being passed through an −80 mesh sieve, the powders were stored at −20 °C. The powders were supplemented with 10 volumes of distilled water and soaked at room temperature for 2 h accompanied by stirring several times. Then, the mixtures were extracted in a water bath at 80–90 °C for 1 h and centrifuged at 10,000 rpm for 5 min at 25 °C. The supernatants were dried in an oven at 60 °C to obtain EUE. Fresh EUL were dried and crushed into EUL powder.
2.2. Composition Determination of EUL and EUE by LC-MS
Sample preparations. First, 0.2 g EUE and EUL powders were respectively placed in 10 mL centrifugal tubes. The samples were ultrasonically extracted with 50 mL of 8% methanol for 40 min at 45 °C and left to rest for 5 min. The supernatants were removed, placed in new centrifuge tubes and centrifuged at 13,000 rpm for 10 min, and filtered with a 0.22 µm microporous membrane to obtain the extractions, which were stored in a refrigerator at 4 °C.
Chromatographic conditions. Extractions were injected into a C18 column (100 mm × 2.1 mm, 1.8 μm) at a 0.3 mL/min flow rate. The column temperature was set at 35 °C and the sample volume was 5 μL. Gradient elution was performed with mobile phases A (0.1% formic acid aqueous solution) and B (acetonitrile) by HPLC (LC-30A, Shimadzu, Kyoto, Japan). The elution procedure was shown in
Table 1.
Mass Spectrometry Conditions. Electrospray ionization was used in the positive ion mode with the following details: Temperature, 600 °C; spray voltage, 5500 V; declustering potential (DP), 100 V; collision energy (CE), 35 eV; collision energy spread (CES), 15 eV; nebulizing gas, nitrogen; curtain gas, 35 psi; auxiliary gas1, 60 psi; auxiliary gas 2, 50 psi; mass scan, 50 to 1000 m/z. Electrospray ionization was used in the negative ion mode: Spray voltage, −4500 V; collision energy (CE), −35 eV. The rest of the experimental conditions remained in positive ion mode.
2.3. Animals and Experimental Design
The animal experiment design was referenced from previous literature [
43]. A total of 90 2-week-old healthy chicks (iron-footed hemp variety) with similar BWs were randomly divided into two equal groups (45 chicks each group), one of which was randomly selected as the control group and the other as the treatment group. Then, chicks were randomly assigned to 3 stages of the experiment, which were 0 d, 7 d, and 15 d, respectively. Each pen had 5 chicks and 3 replicate pens. Chicks in each pen were reared in the same breeding house equipped with a feeder, an automatic drinker, and straw pellets as bedding to ensure the same feeding environment. The control group was fed with a basal diet (CS) using the Broiler chicken compound diet 510 (Guiyang Special Drive Hope Agricultural Science and Technology Co., LTD, Guiyang, China). In addition, the treatment group was fed a basal diet supplemented with 0.8% EUE according to the relevant literature [
37,
44]. The basal diet ingredients are presented in
Table 2. In order to investigate the repeated response of the chicks to the low-temperature environment and avoid long-term low-temperature stress leading to the death of the chicks, the environmental temperature control design was carried out according to the relevant literature [
45]. Chicks of the above two groups were exposed to 10 ± 1 °C from 22:00 to 6:00 to simulate cold stress, and returned to a normal temperature at 25 ± 1 °C from 6:00 to 22:00; this lasted for 15 d. The relative humidity was 65 ± 5% and the light:dark cycle was 10 h:14 h per day. Water and feed were provided ad libitum to the chicks.
2.4. Growth Performance
The BW and feed intake (FI) of each replicate were recorded at 0 d, 7 d, and 15 d. The ADG, average feed intake (AFI), and F/G were calculated for 0–7 d, 8–15 d, and 0–15 d, respectively. The mortality rate was recorded daily and was used to calculate the mortality-corrected FCR.
2.5. Antioxidant Index and Malondialdehyde Content
At 0 d, 7 d, and 15 d, 5 chicks in each repeated group were slaughtered and sampled (15 chicks in each group, with a total of 90 chicks). Blood was collected from the jugular vein and placed in the procoagulant tube. After standing for 1–2 h at 4 °C, the samples were centrifuged for 10 min at 4 °C (3000 rpm) to separate the serum. Five chicks’ serum from each replicate group was mixed (in equal quantity) (
n = 15) [
46,
47]. The levels of total antioxidant capability (T-AOC), superoxide dismutase (SOD), glutathione-peroxidase (GSH-Px), and malondialdehyde (MDA) in serum were individually determined using the protein or MDA enzyme-linked immunosorbent assay (ELISA)kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). The operation was carried out according to the manufacturer’s instructions and repeated three times.
2.6. Immune Index
The levels of IgA, IgG, and IgM contents in chick serum were measured at 0 d, 7 d, and 15 d. The collection procedure of the serum sample was the same as above (n = 15). Total IgA, IgG, and IgM in serum were respectively determined by immunoglobulin ELISA kits (Shanghai Jining Biological Technology Co., Ltd., Shanghai, China). The operation was carried out according to the manufacturer’s instructions and repeated three times.
2.7. Determination of Organ Index
One chick from each replicate representing the mean weight of the pen was selected, slaughtered, and eviscerated (
n = 3). The liver, heart, spleen, thymus, bursa of Fabricius (BF), duodenum, and musculature were separated and sucked with filter paper. After the blood had dried, their weights were valued, and the organ indexes were then calculated as follows [
48]: Organ index = organ weight (g)/body weight (g) × 100%.
2.8. Histopathological Examination
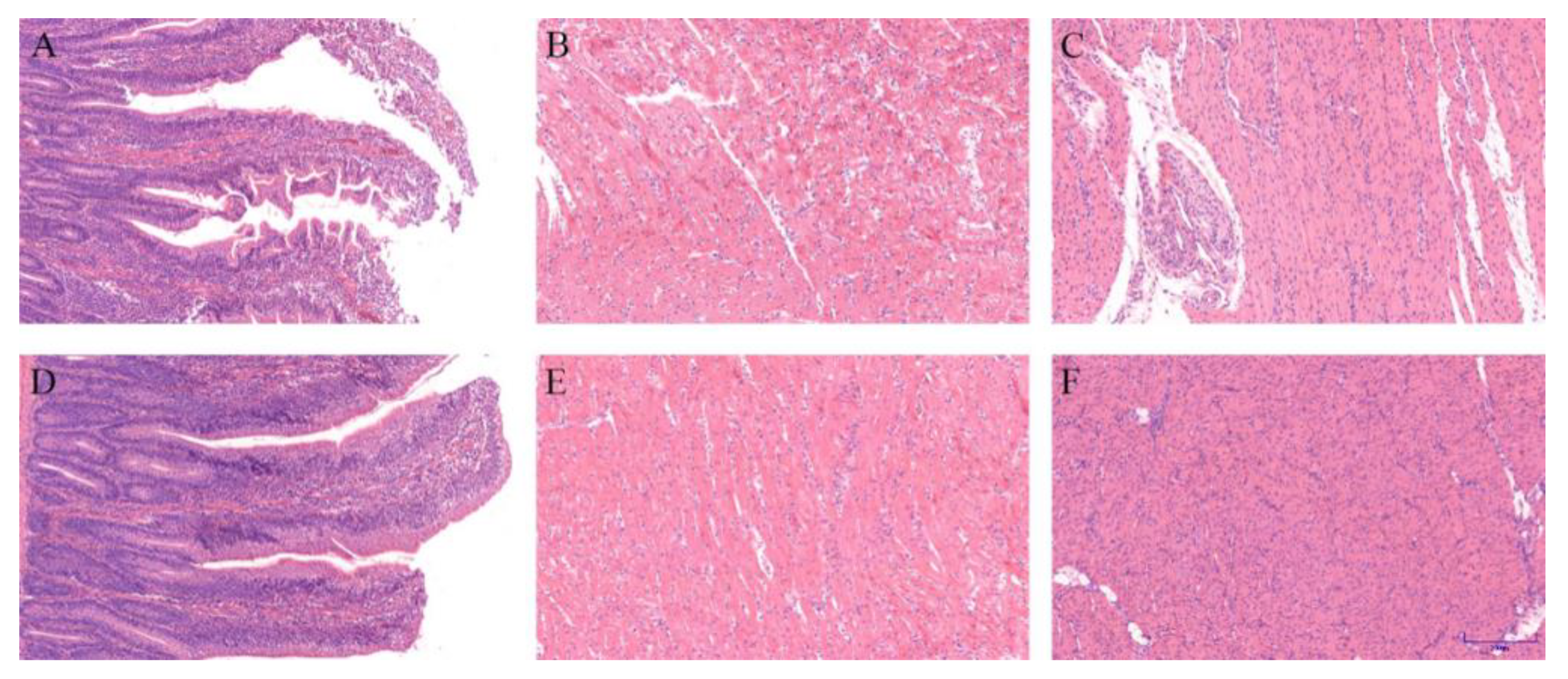
When slaughtered at 15 d, heart, liver, and duodenum samples were collected from 1 chick in each replicate for histological analysis (n = 3). The tissues were fixed in tissue fixative (Wuhan Saiweier Biological Technology Co., Ltd., Wuhan, China) for 24 h and later dehydrated by consecutive washes with ethyl alcohol (70–100%). Subsequently, xylol was used to diaphanize the samples, then embedded in paraffin wax, and serial paraffin sections (4 μm) were obtained. The sections were cut by a microtome and fixed on slides. Sections were stained with hematoxylin and eosin (H&E) (Beyotime, Shanghai, China) and then observed under a microscope.
2.9. Statistical Analysis
Data analysis, single-factor ANOVA, and Duncan’s multiple comparisons were performed using Microsoft Excel Tools and SAS 9.4 software (SAS Institute Inc., Cary, NC, USA) for comparing the differences in growth performance, antioxidant, and immune ability between the EUE group and control group. Results were presented as mean values ± standard deviation (SD). Probability values of p < 0.05 were considered statistically significant.
4. Discussion
Eucommia ulmoides is one of the traditional CHMs, which contains a variety of active ingredients in its root, bark, leaf, and flower, with multiple economic and pharmacological values [
49,
50,
51]. The identification of
Eucommia ulmoides and their extracts have been a hot spot in the research and utilization of
Eucommia ulmoides. Our study showed that the water extract of EUL contained lignans, iridoids, phenylpropanoids, flavonoids, and so on, which is consistent with that extracted by organic solvent [
52,
53]. However, the water extract has the advantages of low cost, simple operation, and less pollution. It has been reported that iridoids, flavonoids, phenylpropanoids, and other substances in
Eucommia ulmoides have antioxidant, anti-inflammatory, and immunity effects [
28,
54,
55]. In addition, chlorogenic acid [
36,
37], quercetin [
38], and rutin [
56] increase immune and antioxidant capacity and promote animal growth and development. It was shown that EUE contained abundant chlorogenic acid, galangal, woodruff nucleoside acid, peach leaf coral glycosides, rutin, and quercetin, which may improve the immune function and growth performance of chicks, indicating EUE can be a potential feed additive.
Cold stress can reduce the growth performance of animals, such as BW and feed efficiency [
57,
58,
59]. In the present study, the chicks supplemented with EUE showed better growth performance of higher BW (
p < 0.01) and ADG (
p < 0.05) and lower F/G (
p < 0.001) throughout the study period, and higher AFI at 0–7 d (
p < 0.001). Our study showed EUE had a better protective effect on cold-stressed poultry. Studies have shown that feeding
Eucommia ulmoides can improve animal growth performance [
60]. Previous investigations found that after feeding
Eucommia ulmoides (rich in chlorogenic acid), the weight of grass carp increased by 5.22%, indicating that
Eucommia ulmoides can promote the growth of grass carp [
34]. It was reported earlier that adding 1 g/kg rutin can improve the growth performance of broilers [
61]. Additionally, the addition of EUE containing flavone improved the growth performance of piglets stimulated by dipterous [
35]. The reason EUE can protect chicks under cold stress may be that EUE contains high levels of chlorogenic acid, rutin, flavonoids, etc., which promotes the BW, ADG, and AFI of chicks, decreases the F/G, and alleviates the impact of cold stress.
The body’s antioxidant level reflects the ability to resist stress [
62]. T-AOC, SOD, and GSH-Px in serum are all essential components of the antioxidant defense system, and they have very significant impacts on homeostasis between oxidation and antioxidants [
63]; MDA is one of the end-products of lipid peroxidation, and its level reflects the degree of oxidative injury to the organism [
64]. As reported, cold stress could cause an increase in T-AOC, SOD, and GSH-Px levels [
65] and the reduction of MDA levels [
66]. Previous studies reported that EUE improved the antioxidant capacity of weaned piglets [
31,
67]. Additionally, the dietary chlorogenic acid-enriched extract (CGAE) from
Eucommia ulmoides supplementation improved oxidative status in pigs [
60]. Bai et al. (2019) demonstrated that the addition of
Eucommia ulmoides to diet could significantly increase GSH-Px activity and decrease MDA content, thus improving the antioxidant capacity of hemp ducks [
41]. Chlorogenic acid and flavonoids are both natural antioxidants [
68,
69]. The chlorogenic acid-enriched extract (CGAE) from
Eucommia ulmoides leaves significantly elevated T-AOC and SOD activity in the liver and serum of lambs under stress, reduced MDA content, improved antioxidant status, and alleviated oxidative damage of lambs [
42]. The literature also shows that EUE (flavonoids) alleviates the oxidative stress induced by diquat in piglets by reducing the growth performance impairment, pro-inflammatory cytokines secretion, and intestinal barrier dysfunction [
35]. In this study, the results showed that chicks fed with EUE could significantly increase the levels of SOD at 15 d (
p < 0.05) and GSH-Px at 7 d (
p < 0.01), and significantly reduce the level of MDA at 15 d (
p < 0.01). Thus, EUE can reduce the damage of cold stress to the chicks by improving the antioxidant ability, which may be related to chlorogenic acid and flavonoids in the antioxidant substances in EUE.
The variations of ambient temperature affect the immune response of chicks [
70,
71]. The immune system is often subject to low temperatures, which reduces the activity of immunoglobulins such as IgA, IgG, and IgM, resulting in immunosuppression in animals [
72]. A previous study has shown that dietary EUL extracts significantly increase the blood IgG and IgM contents of weaned piglets [
73] and broilers [
74]. Additionally, Flavonoids extracted from Chinese herbs could improve immune function and alleviate immune stress in broilers challenged with lipopolysaccharide [
75]. An animal’s systemic immune status may be reflected by the concentration of IgA, IgG, and serum IgM [
76]. In the present study, the level of IgA (7 d,
p < 0.05), IgG (7 d,
p < 0.01; 15 d,
p < 0.001), and IgM (15 d,
p < 0. 001) in the EUE group were significantly higher than that in the CS group at the same stage under low temperatures. Our study suggests that EUE may enhance chicks’ immune function by increasing the serum immunoglobulin contents (IgA, IgG, and IgM), thus having a protective effect against cold stress on chicks.
The organ index often reflects the relative growth and functional state of organs [
77]. The liver, spleen, and BF are important immune organs, and their organ indexes could reflect the immune function [
78,
79,
80,
81,
82]. EUL extract could increase the liver index and contribute to the alleviation of the weaning stress response in piglets [
64,
83]. In addition, EUL extract can increase the index of spleen and BF and improve the immune function of hemp ducks [
41]. The work presented here showed that EUE had no significant effects on the indexes of heart, liver, spleen, BF, stomach, duodenum, and pancreas of chicks under cold-stress conditions. Interestingly, we observed that, compared with the control group, the EUE group had a certain degree of improvement in all organ indexes except the spleen at 7 d. To a certain extent, feeding EUE could reduce the effects of cold stress on the organ development of chicks. Unfortunately, the differences were not significant. The results may be related to the fact that only 0.8% EUE was added in this experiment.
The heart, duodenum, and stomach are essential organs for metabolism and nutrient absorption, and their health degree reflects the adaptability of an organism to environmental stress [
84,
85]. It has been reported that stresses such as cold exposure can induce heart, duodenum, and stomach injury [
9,
86]. In the present study, cold stress caused damage to the tissue of the heart, duodenum, and stomach by H&E staining, while the damage in the EUE group was milder than the CS group. EUL extract can reduce weaned pigs’ intestinal damage caused by stress [
87]. Consistent with the aforementioned reports, we identified that EUE could reduce the organ damage of chicks under cold stress, which may be related to the EUE elevating metabolism and nutrient absorption and enhancing organ health.