Effect of Dietary Standardized Ileal Digestible Arginine to Lysine Ratio on Reproductive Performance, Plasma Biochemical Index, and Immunity of Gestating Sows
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Swine and Diet
2.2. Recording and Sampling
2.3. Chemical Analyses
2.4. Statistical Analysis
3. Results
3.1. Reproductive Performance of Sows
3.2. Colostrum Composition
3.3. Plasma IgA, IgG, IgM
3.4. Plasma Biochemical Indexes and Hormones
3.5. Plasma Concentrations of Free Amino Acids
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Kim, S.W.; Mateo, R.D.; Yin, Y.L.; Wu, G. Functional amino acids and fatty acids for enhancing production performance of sows and piglets. Asian-Australas. J. Anim. Sci. 2006, 20, 295–306. [Google Scholar] [CrossRef]
- Kong, X.; Tan, B.; Yin, Y.; Gao, H.; Li, X.; Jaeger, L.A.; Bazer, F.W.; Wu, G. L-Arg stimulates the mTOR signaling pathway and protein synthesis in porcine trophectoderm cells. J. Nutr. Biochem. 2012, 23, 1178–1183. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Bazer, F.W.; Satterfield, M.C.; Li, X.; Wang, X.; Johnson, G.A.; Burghardt, R.C.; Dai, Z.; Wang, J.; Wu, Z. Impacts of Arg nutrition on embryonic and fetal development in mammals. Amino Acids 2013, 45, 241–256. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Jiang, Z.; Lin, Y.; Zheng, C.; Zhou, G.; Chen, F.; Yang, L.; Wu, G. Dietary L-Arg supplementation enhances placental growth and reproductive performance in sows. Amino Acids 2012, 42, 2207–2214. [Google Scholar] [CrossRef]
- Guo, P.; Jiang, Z.Y.; Gao, K.G.; Wang, L.; Yang, X.F.; Hu, Y.J.; Zhang, J.; Ma, X.Y. Low-level Arg supplementation (0.1%) of wheat-based diets in pregnancy increases the total and live-born litter sizes in gilts. Anim. Prod. Sci. 2016, 57, 1091–1096. [Google Scholar] [CrossRef]
- NRC. Nutrient Requirements of Swine, 11th ed.; National Academic Press: Washington, DC, USA, 2012. [Google Scholar]
- Assis, K.; Botelho, D.; Marques, D.; Filardi, C.; Campos, D.; Saraiva, A. Nutrition influence on sow reproductive performance and conceptuses development and survival: A review about L-Arg supplementation. Livest. Sci. 2019, 228, 97–103. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 18th ed.; Association of Official Analytical Chemists International: Rockville, MD, USA, 2000. [Google Scholar]
- Ma, X.; Lin, Y.; Jiang, Z.; Zheng, C.; Zhou, G.; Yu, D.; Cao, T.; Wang, J.; Chen, F. Dietary Arg supplementation enhances antioxidative capacity and improves meat quality of finishing pigs. Amino Acids 2009, 38, 95–102. [Google Scholar] [CrossRef]
- Krogh, U.; Oksbjerg, N.; Purup, S.; Ramaekers, P.; Theil, P.K. Colostrum and milk production in multiparous sows fed supplementary Arg during gestation and lactation. J. Anim. Sci. 2016, 94 (Suppl. S3), 22–25. [Google Scholar] [CrossRef]
- Robbins, K.R.; Saxton, A.M.; Southern, L.L. Estimation of nutrient requirements using broken-line regression analysis. J. Anim. Sci. 2006, 84 (Suppl. S13), E155. [Google Scholar] [CrossRef]
- Che, L.; Yang, P.; Fang, Z.; Lin, Y.; Wu, D. Effects of dietary Arg supplementation on reproductive performance and immunity of sows. Czech J. Anim. Sci. 2013, 58, 167–175. [Google Scholar] [CrossRef]
- Nuntapaitoon, M.; Muns, R.; Theil, P.K.; Tummaruk, P. l-Arg supplementation in sow diet during late gestation decrease stillborn piglet, increase piglet birth weight and increase immunoglobulin G concentration in colostrum. Theriogenology 2018, 121, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Wu, G. Board-invited review: Intrauterine growth retardation: Implications for the animal sciences. J. Anim. Sci. 2006, 84, 2316–2337. [Google Scholar] [CrossRef]
- Mateo, R.D.; Wu, G.; Bazer, F.W.; Tummaruk, P. Dietary L-Arg supplementation enhances the reproductive performance of gilts. J. Nutr. 2007, 137, 652–656. [Google Scholar] [CrossRef] [PubMed]
- Quesnel, H.; Quiniou, N.; Roy, H.; Lottin, A.; Boulot, S.; Gondret, F. Supplying dextrose before insemination and L-Arg during the last third of pregnancy in sow diets. Effects on within-litter variation of piglet birth weight. J. Anim. Sci. 2014, 92, 1445–1450. [Google Scholar] [CrossRef] [PubMed]
- Oliviero, C.; Kothe, S.; Heinonen, M.; Valros, A.; Peltoniemi, O. Prolonged duration of farrowing is associated with subsequent decreased fertility in sows. Theriogenology 2013, 79, 1095–1099. [Google Scholar] [CrossRef] [PubMed]
- Manu, H.; Pangeni, D.; Wilcock, P.; Baidoo, S.K. The Effect of Superdosing Phytase from 109 Days of Gestation through Lactation on Farrowing Duration, Piglet and Sow Performance. J. Anim. Sci. 2018, 96 (Suppl. S2), 148. [Google Scholar] [CrossRef]
- Herpin, P.; Le Dividich, J.; Hulin, J.C.; Fillaut, M.; De Marco, F.; Bertin, R. Effects of the level of asphyxia during delivery on viability at birth and early postnatal vitality of newborn pigs. J. Anim. Sci. 1996, 74, 2067–2075. [Google Scholar] [CrossRef]
- Yun, J.; Björkman, S.; Oliviero, C.; Soede, N.; Peltoniemi, O. The effect of farrowing duration and parity on preovulatory follicular size and oxytocin release of sows at subsequent oestrus. Reprod. Domest. Anim. 2018, 53, 776–783. [Google Scholar] [CrossRef]
- Bass, B.E.; Bradley, C.L.; Johnson, Z.B.; Zier-Rush, C.E.; Boyd, R.D.; Usry, J.L.; Maxwell, C.V.; Frank, J.W. Influence of dietary L-Arg supplementation of sows during late pregnancy on piglet birth weight and sow and litter performance during lactation. J. Anim. Sci. 2017, 95, 248–256. [Google Scholar] [CrossRef]
- Hines, E.A.; Romoser, M.R.; Kiefer, Z.E.; Lottin, A.; Boulot, S.; Gondret, F. The impact of dietary supplementation of Arg during gestation in a commercial swine herd: I. Gilt reproductive performance. J. Anim. Sci. 2019, 97, 3617–3625. [Google Scholar] [CrossRef]
- Kim, S.W.; Weaver, A.C.; Shen, Y.B.; Zhao, Y. Improving efficiency of sow productivity: Nutrition and health. J. Anim. Sci. Biotechnol. 2013, 4, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xu, T.; Cai, A.; Wu, Y.; Wei, H.; Jiang, S.; Peng, J. Excessive backfat of sows at 109 d of gestation induces lipotoxic placental environment and is associated with declining reproductive performance. J. Anim. Sci. 2018, 96, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Tummaruk, P.; Lundeheim, N.; Einarsson, S.; Dalin, A.M. Effect of birth litter size, birth parity number, growth rate, backfat thickness and age at first mating of gilts on their reproductive performance as sows. Anim. Reprod. Sci. 2001, 66, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Filha, W.S.A.; Bernardi, M.L.; Wentz, I.; Bortolozzo, F.P. Reproductive performance of gilts according to growth rate and backfat thickness at mating. Anim. Reprod. Sci. 2010, 121, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Houde, A.A.; Méthot, S.; Murphy, B.D.; Bordignon, V.; Palin, M.F. Relationships between backfat thickness and reproductive efficiency of sows: A two-year trial involving two commercial herds fixing backfat thickness at breeding. Can. Vet. J. Rev. Vet. Can. 2010, 90, 429–436. [Google Scholar] [CrossRef]
- Thongkhuy, S.; Chuaychu, S.B.; Burarnrak, P.; Ruangjoy, P.; Juthamanee, P.; Nuntapaitoon, M.; Tummaruk, P. Effect of backfat thickness during late gestation on farrowing duration, piglet birth weight, colostrum yield, milk yield and reproductive performance of sows. Livest. Sci. 2020, 234, 103983. [Google Scholar] [CrossRef]
- Devillers, N.; Farmer, C.; Le Dividich, J.; Prunier, A. Variability of colostrum yield and colostrum intake in pigs. Animal 2007, 1, 1033–1041. [Google Scholar] [CrossRef]
- Quesnel, H.; Farmer, C.; Devillers, N. Colostrum intake: Influence on piglet performance and factors of variation. Livest. Sci. 2012, 146, 105–114. [Google Scholar] [CrossRef]
- Elliott, R.F.; Noot, G.W.V.; Gilbreath, R.L.; Fisher, H. Effect of dietary protein level on composition changes in sow colostrum and milk. J. Anim. Sci. 1971, 32, 1128–1137. [Google Scholar] [CrossRef]
- Loisel, F.; Farmer, C.; Ramaekers, P.; Quesnel, H. Effects of high fiber intake during late pregnancy on sow physiology, colostrum production, and piglet performance. J. Anim. Sci. 2013, 91, 5269–5279. [Google Scholar] [CrossRef]
- Dividich, J.L.; Herpin, P.; Paul, E.; Strullu, F. Effect of fat content of colostrum on voluntary colostrum intake and fat utilization in newborn pig. J. Anim. Sci. 1997, 75, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Szyndler-Nędza, M.; Mucha, A.; Tyra, M. The effect of colostrum lactose content on growth performance of piglets from Polish Large White and Polish Landrace sows. Livest. Sci. 2020, 234, 103997. [Google Scholar] [CrossRef]
- Hurley, W.L. Composition of colostrum and milk. In The Gestating and Lactating Sow; Farmer, C., Ed.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2015; pp. 193–229. [Google Scholar]
- Tan, B.; Li, X.G.; Kong, X.; Huang, R.L.; Ruan, Z.; Yao, K.; Deng, Z.Y.; Xie, M.Y.; Shinzato, I.; Yin, Y.L.; et al. Dietary l-Arg supplementation enhances the immune status in early-weaned piglets. Amino Acids 2009, 37, 323–331. [Google Scholar] [CrossRef]
- Li, P.; Yin, Y.L.; Li, D.; Kim, S.W.; Wu, G. Amino acids and immune function. Br. J. Nutr. 2007, 98, 237–252. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Bazer, F.W.; Hu, J.; Johnson, G.A.; Spencer, T.E. Polyamine synthesis from proline in the developing porcine placenta. Biol. Reprod. 2005, 72, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, L.P.; Redmer, D.A. Angiogenesis in the placenta. Biol. Reprod. 2001, 64, 1033–1040. [Google Scholar] [CrossRef]
- Kwon, H.; Wu, G.; Meininger, C.J.; Bazer, F.W.; Spencer, T.E. Developmental changes in nitric oxide synthesis in the ovine placenta. Biol. Reprod. 2004, 70, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Yin, Y.L.; Liu, Y.Q.; Liu, X.D.; Liu, Z.Q.; Li, T.J.; Deng, Z.Y. Effect of dietary Arg and N-carbamoylglutamate supplementation on reproduction and gene expression of eNOS, VEGFA and PlGF1 in placenta in late pregnancy of sows. Anim. Reprod. Sci. 2012, 132, 187–192. [Google Scholar] [CrossRef]
- Morris, S.M., Jr. Arg: Beyond protein. Am. J. Clin. Nutr. 2006, 83, 508S–512S. [Google Scholar] [CrossRef]
- Schneider, F.; Kanitz, E.; Gerrard, D.E.; Kuhn, G.; Brüssow, K.P.; Nürnberg, K.; Rehfeldt, C. Administration of recombinant porcine somatotropin (rpST) changes hormone and metabolic status during early pregnancy. Domest. Anim. Endocrinol. 2002, 23, 455–474. [Google Scholar] [CrossRef]
- Cochard, A.; Guilhermet, R.; Bonneau, M. Plasma growth hormone (GH), insulin and amino acid responses to Arg with or without aspartic acid in pigs. Effect of the dose. Reprod. Nutr. Dev. 1998, 38, 331–343. [Google Scholar] [CrossRef] [PubMed]
- Newsholme, P.; Keane, K.N.; Elahy, M.; Cruzat, V.F. L-Arg, Pancreatic Beta Cell Function, and Diabetes: Mechanisms of Stimulated Insulin Release and Pathways of Metabolism. In L-Arg in Clinical Nutrition; Humana Press: Cham, Switzerland, 2017; pp. 85–94. [Google Scholar] [CrossRef]
- Wu, G. Amino acids: Metabolism, functions, and nutrition. Amino Acids 2009, 37, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Maul, H.; Longo, M.; Saade, G.R.; Garfield, R.E. Nitric oxide and its role during pregnancy: From ovulation to delivery. Curr. Pharm. Des. 2003, 9, 359–380. [Google Scholar] [CrossRef]
- Southern, L.L.; Baker, D.H. Performance and concentration of amino acids in plasma and urine of young pigs fed diets with excesses of either Arg or lysine. J. Anim. Sci. 1982, 55, 857–866. [Google Scholar] [CrossRef] [PubMed]

| Item | SID Arg: Lys Ratio | ||||
|---|---|---|---|---|---|
| 0.91 | 1.02 | 1.14 | 1.25 | 1.38 | |
| Ingredient, g/kg | |||||
| Corn | 565.8 | 566.6 | 567.4 | 568.3 | 569.1 |
| Wheat | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Soybean meal, 44% | 134.0 | 134.0 | 134.0 | 134.0 | 134.0 |
| Alfalfa meal | 128.0 | 128.0 | 128.0 | 128.0 | 128.0 |
| Soybean oil | 15.0 | 15.0 | 15.0 | 15.0 | 15.0 |
| Dicalcium phosphate | 9.3 | 9.3 | 9.3 | 9.3 | 9.3 |
| Limestone | 5.3 | 5.3 | 5.3 | 5.3 | 5.3 |
| Salt | 4.0 | 4.0 | 4.0 | 4.0 | 4.0 |
| L-Lysine-HCl | 2.3 | 2.3 | 2.3 | 2.3 | 2.3 |
| L-Arg | 0.0 | 0.8 | 1.6 | 2.4 | 3.2 |
| L-Alanine | 6.5 | 4.9 | 3.3 | 1.6 | 0.0 |
| DL-Methionine | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| L-Threonine | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Premix a | 30.0 | 30.0 | 30.0 | 30.0 | 3.0 |
| Total | 1000.0 | 1000.0 | 1000.0 | 1000.0 | 1000.0 |
| Analyzed nutrient level | |||||
| Crude protein, % | 14.24 | 14.25 | 14.26 | 14.28 | 14.26 |
| Arg | 0.64 | 0.71 | 0.79 | 0.89 | 0.97 |
| Lysine | 0.79 | 0.79 | 0.80 | 0.80 | 0.78 |
| Calculated composition b | |||||
| Digestible energy, Mcal/kg | 3.14 | 3.14 | 3.14 | 3.14 | 3.14 |
| Net energy, Mcal/kg | 2.30 | 2.30 | 2.30 | 2.30 | 2.30 |
| Crude fiber, % | 6.02 | 6.02 | 6.02 | 6.02 | 6.02 |
| Calcium, % | 0.82 | 0.82 | 0.82 | 0.82 | 0.82 |
| Phosphorus, % | 0.48 | 0.48 | 0.48 | 0.48 | 0.48 |
| STTD Phosphoru, % | 0.35 | 0.35 | 0.35 | 0.35 | 0.35 |
| SID Arg, % | 0.58 | 0.65 | 0.73 | 0.80 | 0.88 |
| SID Lysine, % | 0.64 | 0.64 | 0.64 | 0.64 | 0.64 |
| SID Arg: Lys, % | 0.91 | 1.02 | 1.14 | 1.25 | 1.38 |
| SID Methionine + Cysteine, % | 0.41 | 0.41 | 0.41 | 0.41 | 0.41 |
| SID Threonine, % | 0.42 | 0.42 | 0.42 | 0.42 | 0.42 |
| SID Tryptophan, % | 0.14 | 0.14 | 0.14 | 0.14 | 0.14 |
| Item | SID Arg: Lys Ratio | SEM | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.91 | 1.02 | 1.14 | 1.25 | 1.38 | Treatment | Linear | Quadratic | ||
| Sow number, n | 35 | 34 | 35 | 36 | 33 | ||||
| Litter size, n | |||||||||
| Total born | 12.23 | 12.94 | 12.94 | 13.03 | 12.61 | 0.25 | 0.843 | 0.648 | 0.496 |
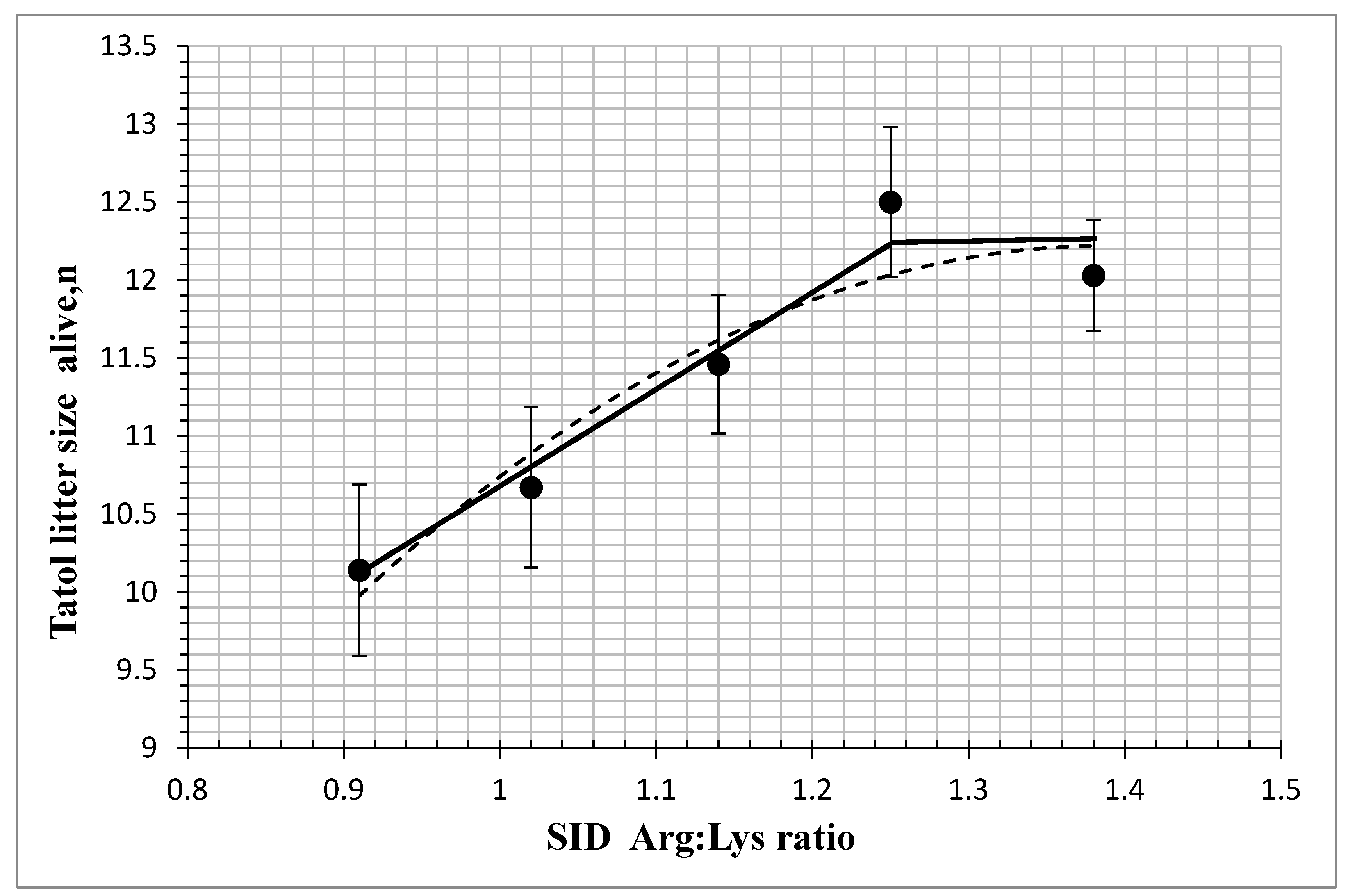
| Born alive | 10.14 c | 10.67 bc | 11.46 b | 12.50 a | 12.03 ab | 0.23 | 0.005 | 0.001 | 0.001 |
| Stillborn | 1.57 a | 1.48 a | 0.89 ab | 0.14 c | 0.27 bc | 0.11 | 0.000 | 0.000 | 0.000 |
| Mummified | 0.09 | 0.30 | 0.23 | 0.14 | 0.03 | 0.03 | 0.061 | 0.236 | 0.033 |
| Average live birth weight, kg | 1.41 | 1.40 | 1.41 | 1.41 | 1.41 | 0.01 | 0.992 | 0.846 | 0.980 |
| Litter weight of born alive, kg | 14.30 b | 14.49 b | 16.15 ab | 17.62 a | 16.96 a | 0.33 | 0.003 | 0.001 | 0.001 |
| CV of total born birth weitght | 0.21 a | 0.20 ab | 0.18 ab | 0.17 b | 0.16 b | 0.03 | 0.020 | 0.002 | 0.008 |
| Farrowing duration, min | 283 a | 220 b | 234 b | 259 ab | 223 b | 7.31 | 0.030 | 0.115 | 0.192 |
| Birth intervals, min | 25.86 a | 18.22 b | 18.61 b | 19.87 b | 18.51 b | 0.82 | 0.014 | 0.025 | 0.013 |
| Backfat thickness, mm | |||||||||
| Day 30 of gestation | 17.17 | 16.76 | 16.83 | 16.94 | 16.76 | 0.25 | 0.984 | 0.718 | 0.985 |
| Day 110 of gestation | 20.14 | 19.45 | 19.80 | 20.03 | 19.67 | 0.26 | 0.925 | 0.833 | 0.959 |
| Backfat gain (d 30 to 110) | 2.97 | 2.70 | 2.97 | 3.08 | 2.91 | 0.06 | 0.268 | 0.515 | 0.808 |
| Item | SID Arg: Lys Ratio | SEM | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.91 | 1.02 | 1.14 | 1.25 | 1.38 | Treatment | Linear | Quadratic | ||
| Milk fat, % | 2.62 | 3.61 | 3.78 | 3.99 | 3.45 | 0.17 | 0.109 | 0.109 | 0.023 |
| Lactose, % | 4.24 a | 3.62 b | 4.02 ab | 3.64 b | 4.01 ab | 0.08 | 0.041 | 0.492 | 0.153 |
| Non-fat solid, % | 17.94 c | 20.20 b | 18.38 c | 22.34 a | 20.13 b | 0.38 | <0.001 | 0.017 | 0.029 |
| Total-solid, % | 20.63 c | 24.12 b | 22.44 bc | 26.83 a | 23.96 b | 0.48 | <0.001 | 0.006 | 0.003 |
| Milk protein, % | 12.01 d | 14.93 b | 12.68 cd | 16.83 a | 14.36 bc | 0.40 | <0.001 | 0.023 | 0.025 |
| Energy, kJ/100 g | 367 c | 419 b | 384 c | 478 a | 423 b | 18.99 | <0.001 | 0.015 | 0.017 |
| Item | SID Arg: Lys Ratio | SEM | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.91 | 1.02 | 1.14 | 1.25 | 1.38 | Treatment | Linear | Quadratic | ||
| IgA μg/mL | |||||||||
| 90 d | 8.77 | 14.34 | 13.06 | 15.34 | 11.59 | 0.48 | 0.060 | 0.148 | 0.028 |
| 110 d | 10.85 | 12.52 | 11.65 | 12.08 | 11.22 | 0.72 | 0.187 | 0.450 | 0.581 |
| IgG μg/mL | |||||||||
| 90 d | 81.52 | 105.27 | 98.42 | 89.84 | 88.62 | 1.56 | 0.235 | 0.145 | 0.169 |
| 110 d | 79.42 | 89.85 | 86.43 | 110.32 | 95.98 | 1.65 | 0.090 | 0.011 | 0.014 |
| IgM μg/mL | |||||||||
| 90 d | 41.19 | 43.68 | 38.90 | 46.08 | 52.41 | 1.72 | 0.109 | 0.037 | 0.042 |
| 110 d | 43.18 | 44.72 | 49.53 | 46.24 | 47.26 | 0.91 | 0.178 | 0.119 | 0.132 |
| Item | SID Arg: Lys Ratio | SEM | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.91 | 1.02 | 1.14 | 1.25 | 1.38 | Treatment | Linear | Quadratic | ||
| Urea nitrogen, mmol/L | |||||||||
| Day 90 | 6.28 | 6.58 | 5.56 | 5.75 | 6.14 | 0.23 | 0.645 | 0.572 | 0.600 |
| Day 110 | 6.33 | 6.05 | 5.75 | 6.12 | 5.58 | 0.16 | 0.622 | 0.204 | 0.450 |
| NO, μmol/L | |||||||||
| Day 90 | 8.89 | 8.89 | 7.86 | 8.37 | 8.93 | 0.25 | 0.701 | 0.496 | 0.531 |
| Day 110 | 7.56 b | 8.56 a | 8.51 a | 8.78 a | 9.33 a | 0.17 | 0.014 | 0.001 | 0.004 |
| IGF-1, ug/L | |||||||||
| Day 90 | 29.98 | 31.27 | 30.01 | 30.61 | 32.14 | 0.49 | 0.613 | 0.280 | 0.483 |
| Day 110 | 30.49 | 30.76 | 30.68 | 30.1 | 30.28 | 0.20 | 0.850 | 0.468 | 0.725 |
| Progesterone, ng/ml | |||||||||
| Day 90 | 343.77 | 388.84 | 341.80 | 369.47 | 414.80 | 13.63 | 0.409 | 0.196 | 0.358 |
| Day 110 | 372.04 | 360.00 | 382.81 | 345.45 | 354.60 | 8.44 | 0.688 | 0.420 | 0.714 |
| GH, ng/ml | |||||||||
| Day 90 | 0.49 | 0.55 | 0.49 | 0.53 | 0.53 | 0.02 | 0.287 | 0.084 | 0.141 |
| Day 110 | 0.54 | 0.53 | 0.56 | 0.51 | 0.55 | 0.01 | 0.843 | 0.706 | 0.930 |
| Insulin, mIU/L | |||||||||
| Day 90 | 12.71 b | 12.79 b | 12.31 b | 18.19 a | 17.08 a | 0.91 | 0.028 | 0.007 | 0.025 |
| Day 110 | 15.37 | 17.02 | 15.99 | 18.86 | 15.59 | 0.84 | 0.702 | 0.730 | 0.640 |
| Estrogen, pmol/L | |||||||||
| Day 90 | 7.43 | 10.02 | 7.44 | 8.55 | 10.99 | 0.74 | 0.472 | 0.270 | 0.478 |
| Day 110 | 7.18 | 8.73 | 9.37 | 8.42 | 9.72 | 0.63 | 0.771 | 0.287 | 0.534 |
| Item | SID Arg: Lys Ratio | SEM | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.91 | 1.02 | 1.14 | 1.25 | 1.38 | Treatment | Linear | Quadratic | ||
| Total protein, g/L | 71.98 | 73.56 | 76.57 | 74.09 | 80.30 | 3.76 | 0.233 | 0.042 | 0.123 |
| Albumin, g/L | 39.08 | 36.27 | 36.65 | 38.12 | 41.25 | 2.54 | 0.336 | 0.275 | 0.094 |
| Creatinine, μmol/L | 174 | 184 | 188 | 197 | 183 | 12.00 | 0.430 | 0.249 | 0.186 |
| Uric acid, μmol/L | 36.17 | 39.55 | 43.28 | 45.36 | 51.31 | 10.08 | 0.628 | 0.100 | 0.264 |
| Glucose, mmol/L | 4.92 | 5.38 | 5.61 | 5.36 | 4.87 | 0.56 | 0.62 | 0.894 | 0.259 |
| Triacylglycerol, mmol/L | 2.94 | 3.68 | 4.28 | 3.20 | 3.04 | 0.74 | 0.050 | 0.379 | 0.215 |
| Cholesterol, mmol/L | 1.33 | 1.13 | 1.37 | 1.31 | 1.40 | 0.14 | 0.346 | 0.304 | 0.463 |
| HDL-Cholesterol, mmol/L | 4.83 | 4.62 | 5.50 | 5.47 | 5.49 | 0.58 | 0.390 | 0.096 | 0.238 |
| LDL-Cholesterol, mmol/L | 0.51 | 0.44 | 0.54 | 0.51 | 0.55 | 0.48 | 0.236 | 0.186 | 0.360 |
| Calcium, mmol/L | 2.89 | 3.03 | 3.13 | 3.16 | 3.34 | 0.256 | 0.521 | 0.069 | 0.198 |
| Phosphorus, mmol/L | 1.69 | 1.76 | 1.57 | 1.67 | 1.80 | 0.091 | 0.126 | 0.524 | 0.196 |
| Item/μmol/L | SID Arg: Lys Ratio | SEM | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.91 | 1.02 | 1.14 | 1.25 | 1.38 | Treatment | Linear | Quadratic | ||
| Taurine | 69 | 63 | 69 | 62 | 64 | 1.97 | 0.697 | 0.474 | 0.744 |
| Aspartate | 14 | 14 | 12 | 13 | 11 | 0.70 | 0.593 | 0.118 | 0.277 |
| Threonine | 204 b | 193 b | 205 b | 163 b | 262 a | 9.13 | 0.005 | 0.156 | 0.023 |
| Serine | 173 | 162 | 159 | 160 | 172 | 4.19 | 0.718 | 0.902 | 0.339 |
| Asparagine | 74 | 70 | 72 | 75 | 83 | 4.03 | 0.907 | 0.465 | 0.592 |
| Glutamate | 171 a | 130 ab | 151 a | 145 ab | 103 b | 7.63 | 0.053 | 0.021 | 0.064 |
| Glutamine | 386 | 374 | 348 | 379 | 330 | 11.45 | 0.517 | 0.177 | 0.402 |
| Glycine | 964 | 863 | 884 | 917 | 1006 | 39.04 | 0.795 | 0.607 | 0.448 |
| Alanine | 990 a | 820 ab | 716 b | 726 b | 666 b | 33.29 | 0.008 | 0.001 | 0.001 |
| Citrulline | 100 | 90 | 101 | 122 | 123 | 6.29 | 0.399 | 0.082 | 0.199 |
| Valine | 299 | 281 | 302 | 282 | 337 | 9.38 | 0.330 | 0.220 | 0.194 |
| Cystine | 15 | 15 | 14 | 12 | 15 | 0.84 | 0.801 | 0.748 | 0.744 |
| Methionine | 75 ab | 87 a | 83 a | 63 b | 90 a | 2.63 | 0.023 | 0.727 | 0.744 |
| Isoleucine | 139 | 140 | 139 | 134 | 157 | 4.71 | 0.605 | 0.375 | 0.379 |
| Leucine | 279 | 263 | 250 | 257 | 289 | 8.33 | 0.584 | 0.786 | 0.237 |
| Tyrosine | 145 b | 131 b | 136 b | 145 b | 169 a | 4.19 | 0.031 | 0.031 | 0.004 |
| Phenylalanine | 106 | 102 | 113 | 107 | 121 | 2.73 | 0.193 | 0.061 | 0.126 |
| Ornithine | 113 b | 135 b | 131 b | 174 a | 175 a | 6.12 | <0.001 | <0.001 | <0.001 |
| Lysine | 187 | 203 | 202 | 178 | 210 | 6.12 | 0.488 | 0.620 | 0.864 |
| Histidine | 121 | 109 | 117 | 120 | 117 | 2.80 | 0.738 | 0.839 | 0.887 |
| Arg | 180 b | 234 b | 237 b | 323 a | 327 a | 14.63 | 0.001 | <0.001 | <0.001 |
| Proline | 309 | 309 | 297 | 322 | 333 | 11.73 | 0.911 | 0.467 | 0.665 |
| Item/μmol/L | SID Arg: Lys Ratio | SEM | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.91 | 1.02 | 1.14 | 1.25 | 1.38 | Treatment | Linear | Quadratic | ||
| Taurine | 48 | 55 | 54 | 58 | 63 | 0.83 | 0.115 | 0.008 | 0.033 |
| Aspartate | 11 | 13 | 12 | 12 | 11 | 0.70 | 0.138 | 0.298 | 0.132 |
| Threonine | 204 | 202 | 201 | 232 | 206 | 6.73 | 0.576 | 0.496 | 0.733 |
| Serine | 142 | 168 | 164 | 179 | 167 | 5.70 | 0.361 | 0.145 | 0.163 |
| Asparagine | 71 | 105 | 115 | 108 | 93 | 6.39 | 0.215 | 0.315 | 0.055 |
| Glutamate | 138 | 146 | 158 | 144 | 139 | 4.53 | 0.649 | 0.965 | 0.400 |
| Glutamine | 254 b | 335 a | 335 a | 385 a | 328 ab | 13.84 | 0.041 | 0.047 | 0.014 |
| Glycine | 534 | 701 | 563 | 647 | 590 | 25.37 | 0.234 | 0.787 | 0.580 |
| Alanine | 1016 a | 999 a | 881 ab | 804 b | 740 b | 29.54 | 0.003 | <0.001 | <0.001 |
| Citrulline | 70 | 70 | 66 | 83 | 61 | 2.69 | 0.121 | 0.733 | 0.467 |
| Valine | 246 | 282 | 288 | 318 | 290 | 10.75 | 0.345 | 0.110 | 0.133 |
| Cystine | 12 | 13 | 9 | 15 | 13 | 0.92 | 0.375 | 0.653 | 0.842 |
| Methionine | 79 b | 93 ab | 110 a | 118 a | 111 a | 4.79 | 0.050 | 0.006 | 0.008 |
| Isoleucine | 115 | 147 | 150 | 159 | 158 | 5.45 | 0.051 | 0.009 | 0.011 |
| Leucine | 234 | 309 | 291 | 317 | 289 | 10.29 | 0.082 | 0.117 | 0.043 |
| Tyrosine | 133 | 139 | 145 | 152 | 143 | 5.49 | 0.862 | 0.389 | 0.562 |
| Phenylalanine | 100 | 110 | 111 | 124 | 111 | 3.95 | 0.459 | 0.205 | 0.250 |
| Ornithine | 159 | 162 | 176 | 186 | 179 | 5.30 | 0.215 | 0.006 | 0.298 |
| Lysine | 140 c | 183 bc | 207 abc | 242 a | 227 ab | 9.73 | 0.003 | <0.001 | <0.001 |
| Histidine | 116 | 118 | 121 | 124 | 109 | 3.02 | 0.645 | 0.649 | 0.395 |
| Arg | 202 b | 206 b | 232 ab | 283 a | 299 a | 12.51 | 0.023 | 0.001 | 0.004 |
| Proline | 356 | 391 | 391 | 379 | 364 | 18.86 | 0.097 | 0.993 | 0.797 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wen, X.; Jiang, Z.; Yang, X.; Xiao, H.; Gao, K.; Wang, L. Effect of Dietary Standardized Ileal Digestible Arginine to Lysine Ratio on Reproductive Performance, Plasma Biochemical Index, and Immunity of Gestating Sows. Animals 2024, 14, 2688. https://doi.org/10.3390/ani14182688
Wen X, Jiang Z, Yang X, Xiao H, Gao K, Wang L. Effect of Dietary Standardized Ileal Digestible Arginine to Lysine Ratio on Reproductive Performance, Plasma Biochemical Index, and Immunity of Gestating Sows. Animals. 2024; 14(18):2688. https://doi.org/10.3390/ani14182688
Chicago/Turabian StyleWen, Xiaolu, Zongyong Jiang, Xuefen Yang, Hao Xiao, Kaiguo Gao, and Li Wang. 2024. "Effect of Dietary Standardized Ileal Digestible Arginine to Lysine Ratio on Reproductive Performance, Plasma Biochemical Index, and Immunity of Gestating Sows" Animals 14, no. 18: 2688. https://doi.org/10.3390/ani14182688







