3.1.2. Mineralogical Characterization and Microstructure
The mineralogical analyses of the slags are summarized in
Table 3 and
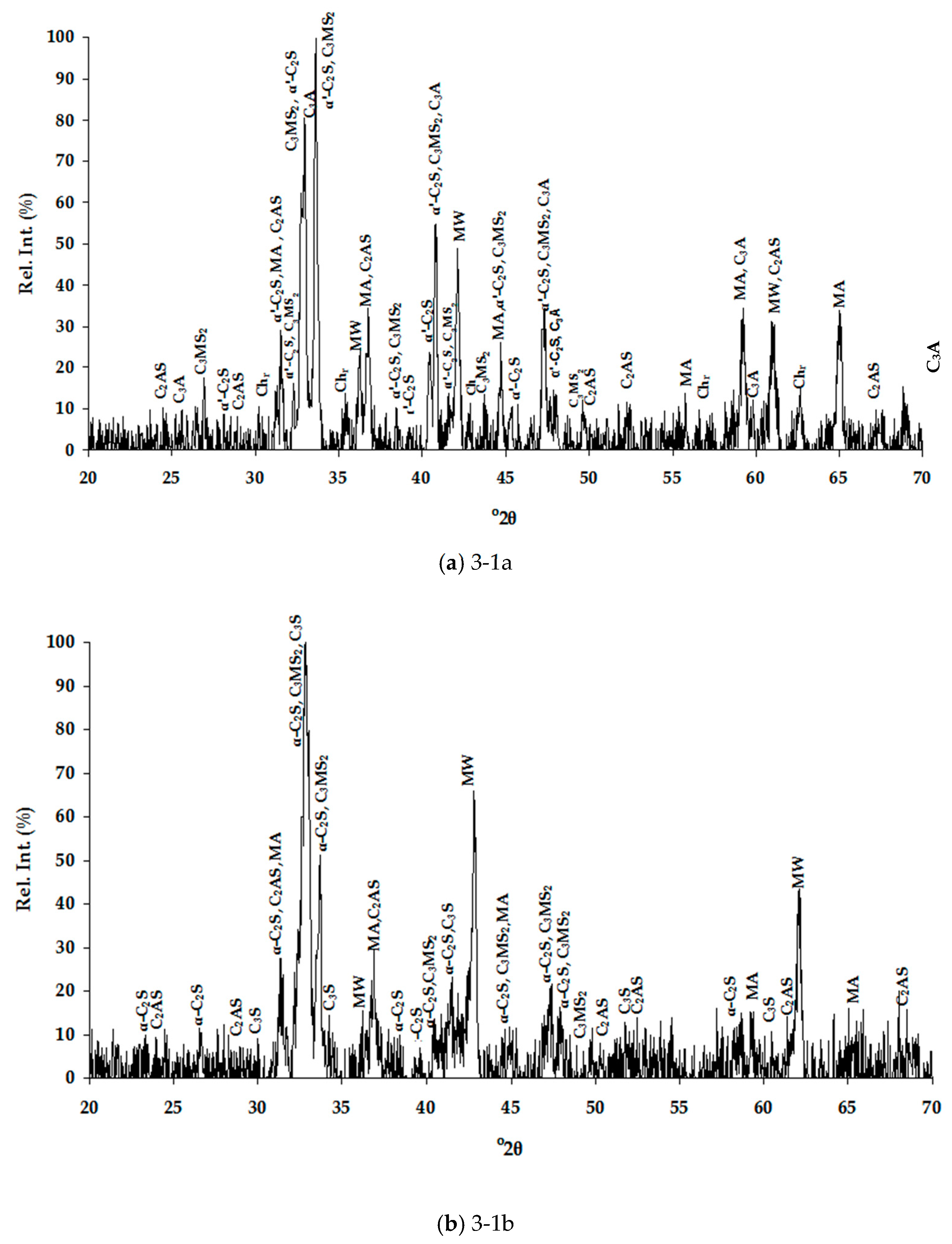
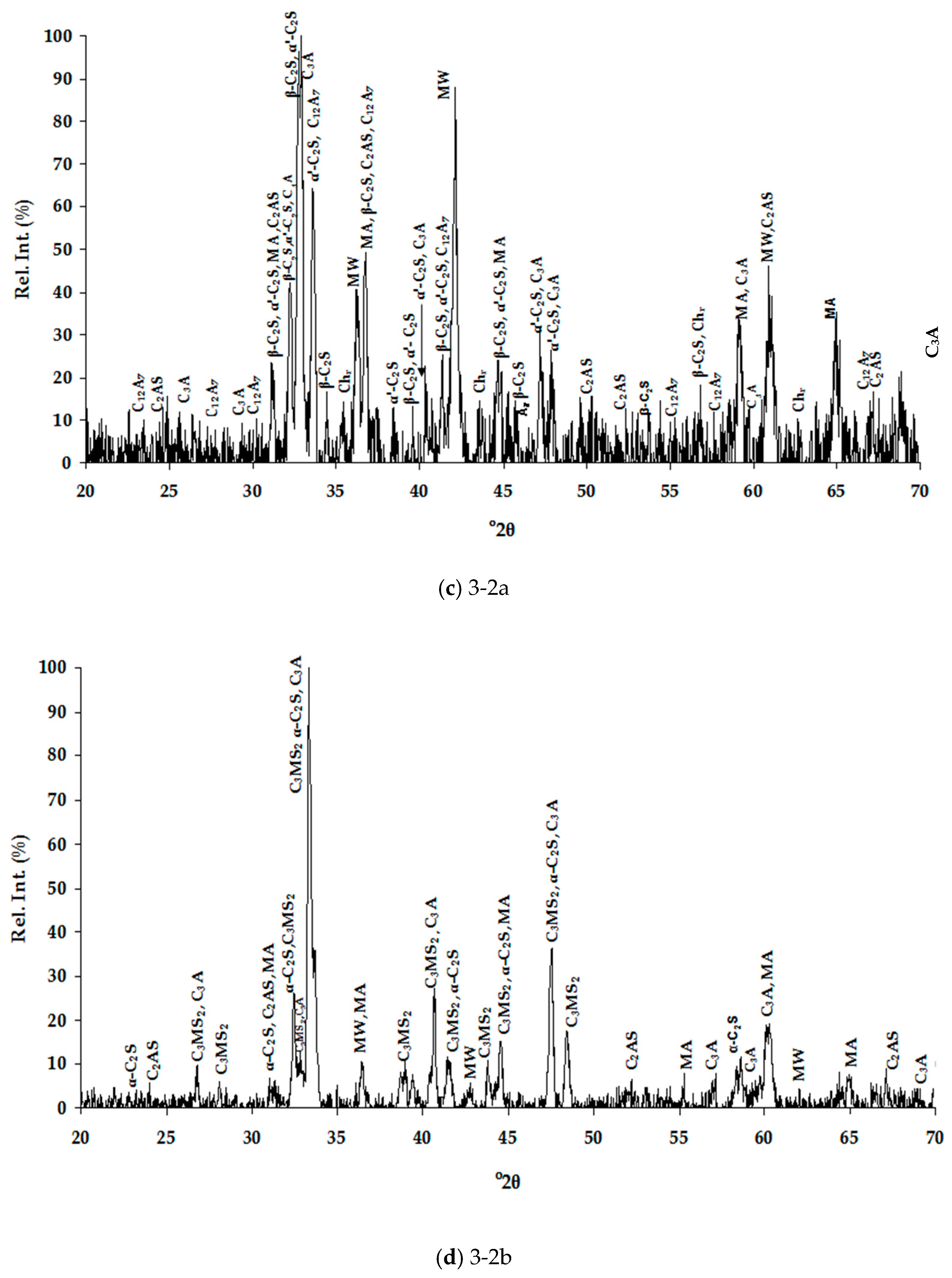
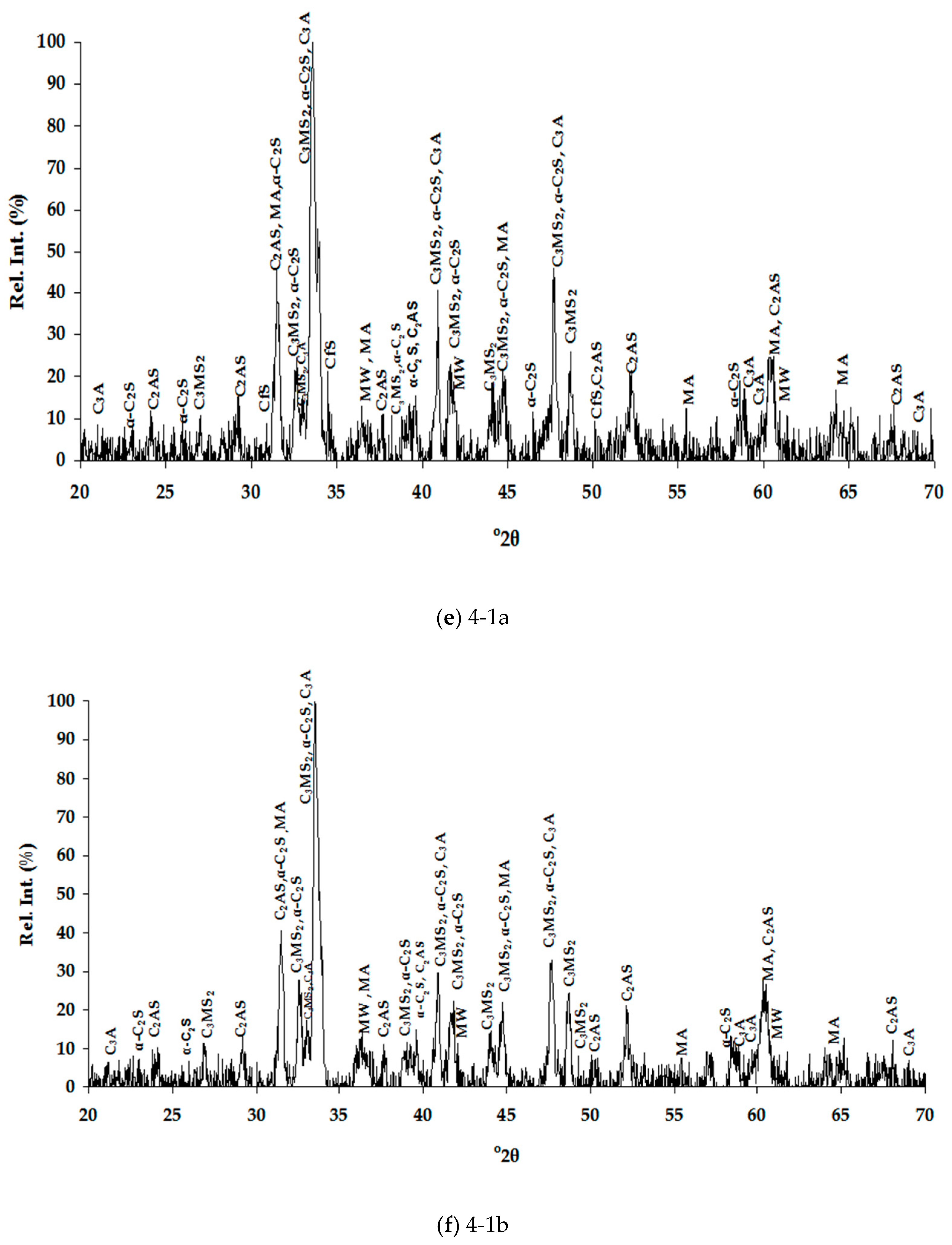
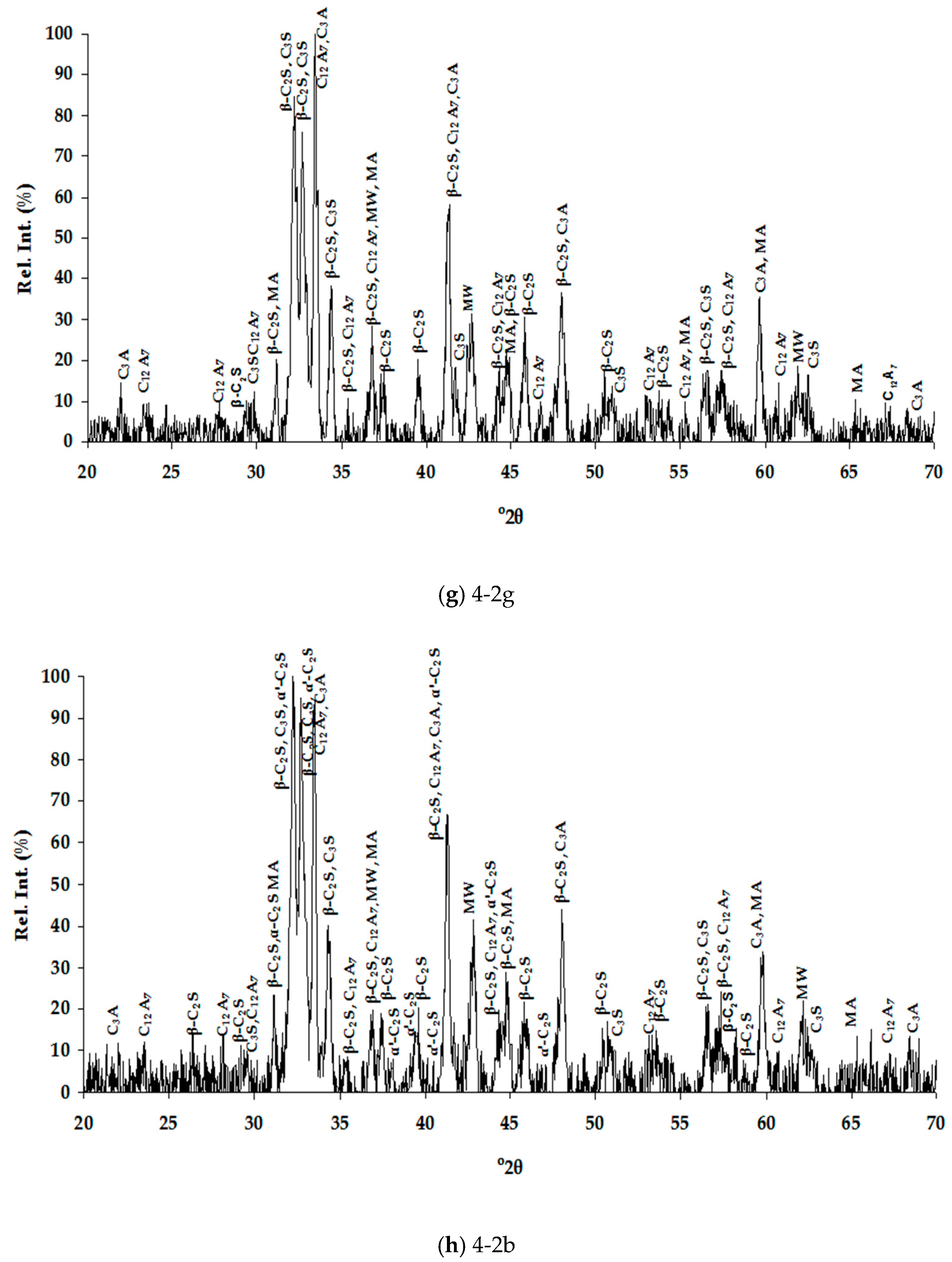
Table 4 for the air and water cooled samples, respectively. The high number of phases detected in each XRD pattern (
Figure 1), therefore high overlapping of peaks, made their distinguishing quite difficult and SEM/EDS analysis assisted substantially towards this direction (
Table 5). Special care was taken to present selected microanalyses that are either stoichiometric or closely approximate the stoichiometry, since the fractured analyzed surfaces of samples prevented accurate quantitative analyses. Moreover, characteristic microphotographs of their structure obtained by optical microscopy and SEM are presented in
Figure 2,
Figure 3,
Figure 4 and
Figure 5.
According to the obtained results, well-formed crystals were also detected in the slags cooled in water as shown in
Figure 1. The constrained amorphous (glass) content was also verified by the optical observation under polarized light of the water-cooled 35–45 μm slags fraction as indicatively shown in
Figure 2a. In order to obtain vitreous granulated slag, as in GGBS, high pressure water jets are required [
19]. The main differences in the mineralogical phases identified between air and water-cooled slags of the same chemical composition were the polymorphs of dicalcium silicate attributed to the different rates of cooling. The environmental conditions during the smelting trials and thus slags tapping (temperature <0 °C, rainfall) and formation of microcrystalline material prevented the formation of γ-C
2S (γ-2CaO·SiO
2) and the disintegration of the cooled slag phase due to β→γ-C
2S transformation even in the air-cooled slags. The later takes place in cases of slow cooling rate and its presence is not desired in cementitious materials, since it exhibits slight hydraulic properties not contributing to strength development. The co-existence of α and/or α′ and β-C
2S phases in the slag samples indicates that the theoretical thermodynamic transformation was not completed due to rapid temperature drop mainly in the initial phase of the cooling process [
30]. The incorporation of Μg in C
2S could lead to the formation of bredigite, Ca
14Mg
2(SiO
4)
8—once the solubility limits in α and α′-C
2S are exceeded. Usually, its composition varies between Ca
1.75Mg
0.25 and Ca
1.7Mg
0.3 at ~1300 °C per SiO
4 [
31] and its presence has been reported in cases of EAF steelmaking slags [
32]. However, α′-C
2S and bredigite have different structures, it has been confirmed [
31,
33] that the similarity in their XRD patterns makes distinguishing them very difficult (
Figure 1). In this work, the nomenclature of α′-C
2S has been currently attributed to a probable combination of both phases. The semiquantitative analysis obtained by SEM-EDS verified solubility of Mg in C
2S, as well as the existence of purer C
2S micro-areas, while Al, Mn and Fe incorporated in the crystal lattice was also identified (
Table 5,
Figure 2). The incorporation of foreign ions in the structure of dicalcium silicates, as well as deviation from the stoichiometric composition, favor the stabilization of the α′ and β phases [
34].
In all examined samples, magnesiowüstite phase was identified (
Table 5,
Figure 3a). Magnesiowüstite comprises a solid solution between MgO and FeO and it is commonly found in the mineralogical composition of steelmaking slags [
19,
21,
35,
36]. It is reflected in the XRD patterns by displaced peaks located between pure wüstite (41.890 2θ strongest line) and periclase ones (42.917 2θ strongest line), depending on the Mg/Fe
2+ ratio and it has been assigned as Mg
1−xFe
xO (MW). Investigations have shown that the composition of these types of solid solutions in the slag depends on slag chemistry and on the cooling rate. Slower cooling rate will promote the enrichment of FeO into the structure, while MgO predominates by increasing the cooling rate [
21,
36]. This may explain the difference observed when comparing the microanalysis of MW phase in slags, 3-1a versus 3-1b (
Table 5). Nevertheless, it may be noticed that the strongly reduced %FeO in the chemical analysis of 4-2 slag led to rich Mg-MW even in the air-cooled slag sample.
Incorporation of Manganese, Calcium and Chromium (most possibly divalent) in magnesiowüstite solid solutions was also detected in the course of SEM-EDS analysis in the slag samples. Chromium was also identified in XRD analysis in chromite phase in two of the air-cooled slag samples (3-1,2a). Increase of cooling rate prevents the thermodynamic chromite spinel formation from the MW solid solution with temperature drop, preserving Chromium in the MW phase. In high basicity EAF slags, primary crystallization of Chromium occurs in magnesiowüstite [
21,
36]. This is illustrated in the microanalyses of the mageniowüstite solid solution phases in the higher basicity air-cooled slag samples 4 and 5, (
Table 5). Increase of FeO amount in these solid solutions has also been reported to decrease its leachability from the slag, which is an advantageous property for its further envisaged use [
21,
36].
Overall, iron in the examined slags was mainly in the divalent state bound in the MW phase as described above. In slag 5, where limited reduction of iron oxides was targeted through the applied experimental conditions, brownmillerite, Ca
4Al
2Fe
2O
10 (C
4AF) and calcium ferrite (CaO.2Fe
2O
3) were additionally identified, also indicating presence of trivalent iron in the slag. Brownmillerite is commonly met in the clinker of Portland cement, while calcium ferrite phase in contrast to MW has been reported as phase of hydraulic nature, too [
18]. Micro-areas rich in C
4AF crystals were detected by SEM/EDS examinations in slag 5 sample (
Table 5,
Figure 3b).
Tricalcium silicate 3CaO.SiO
2 (C
3S), being the most important phase in cement for early strength development among the calcium silicates, was mainly detected in the slag products, 4-2a, b and 5, where the basicity was higher, while some traces were also found in slag 3-1b. Typical micrograph and microanalyses of identified C
3S hexagonal crystal in the slags microstructure is shown in
Figure 3c and
Table 5, respectively. Presence of merwinite, 3CaO·MgO·2SiO
2 (C
3MS
2), common constituent of EAF steelmaking slags was also identified in the XRD and SEM/EDS analysis of the slags. Characteristic micro-area of solidified merwinite crystals is shown in
Figure 3d and representative C
3MS
2 microanalyses are provided in
Table 5. According to [
15] low grain size merwinite in EAFs contributes to slags cementitious properties exhibiting a hydration behavior similar to that of di- and tri-calcium silicates.
Calcium aluminate phases, such as 3CaO·Al
2O
3 (C
3A) and 12CaO·7Al
2O
3 (C
12A
7), which also contribute to strength development upon hydration, participated in the mineralogical composition of the slags, being more abundant in slags 4-2(
Table 5,
Figure 4), which had the highest content of Al
2O
3. In addition, gehlenite C
2AS (2CaOAl
2O
3SiO
2) was also often detected (
Figure 1,
Table 5). The retention of some of the water-cooled slags in water during their cooling resulted in the fast hydration of part of the calcium aluminate phases. The presence of sulfur in these slags led to the formation of monosulfate, C
4ASH
12 (4CaO·Al
2O
3·SO
3·12H
2O), found as thin hexagonal plates, (
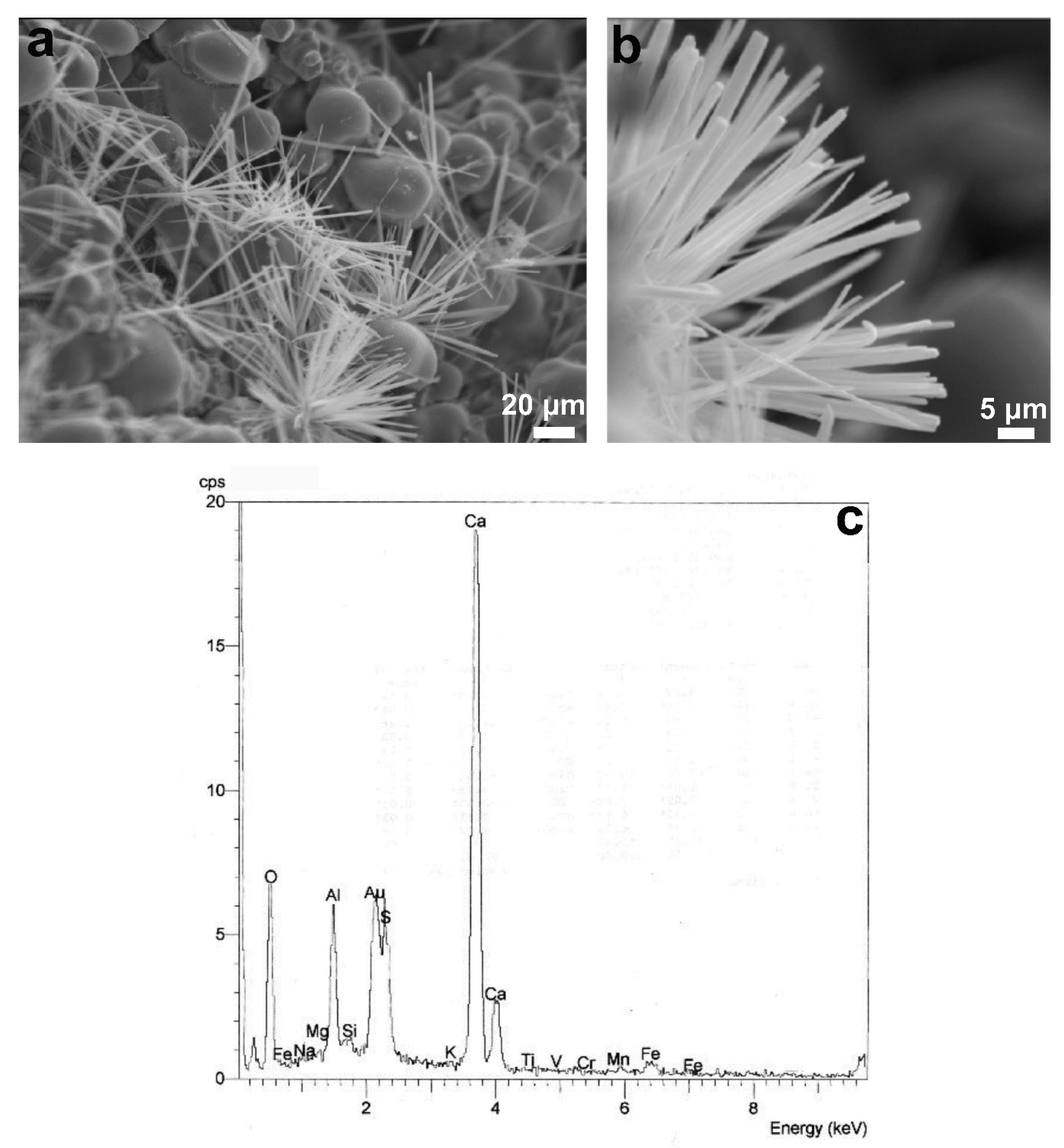
Figure 4c) or ettringiteC
6AS
3H
32 (6CaO·Al
2O
3·3SO
3·32H
2O) detected in the form of needles (
Figure 5). These hydrated phases were only detected locally in SEM backscattered images. It has also been reported that in most cases it is not possible to detect ettringite by XRD [
37]. These phases are usually encountered during the hydration of Portland cements due to the reaction of gypsum with calcium aluminates according to the following reactions:
During cement hydration process, if added gypsum is consumed before the complete hydration of C3A, the remaining C3A reacts with ettringite producing monosulfate. The higher calcium aluminate content, due to higher Al2O3 percentage in the case of slag 4-2b has possibly allowed monosulfate formation by a similar mechanism.