Psychiatric Manifestation of Anti-LGI1 Encephalitis
Abstract
:1. Background
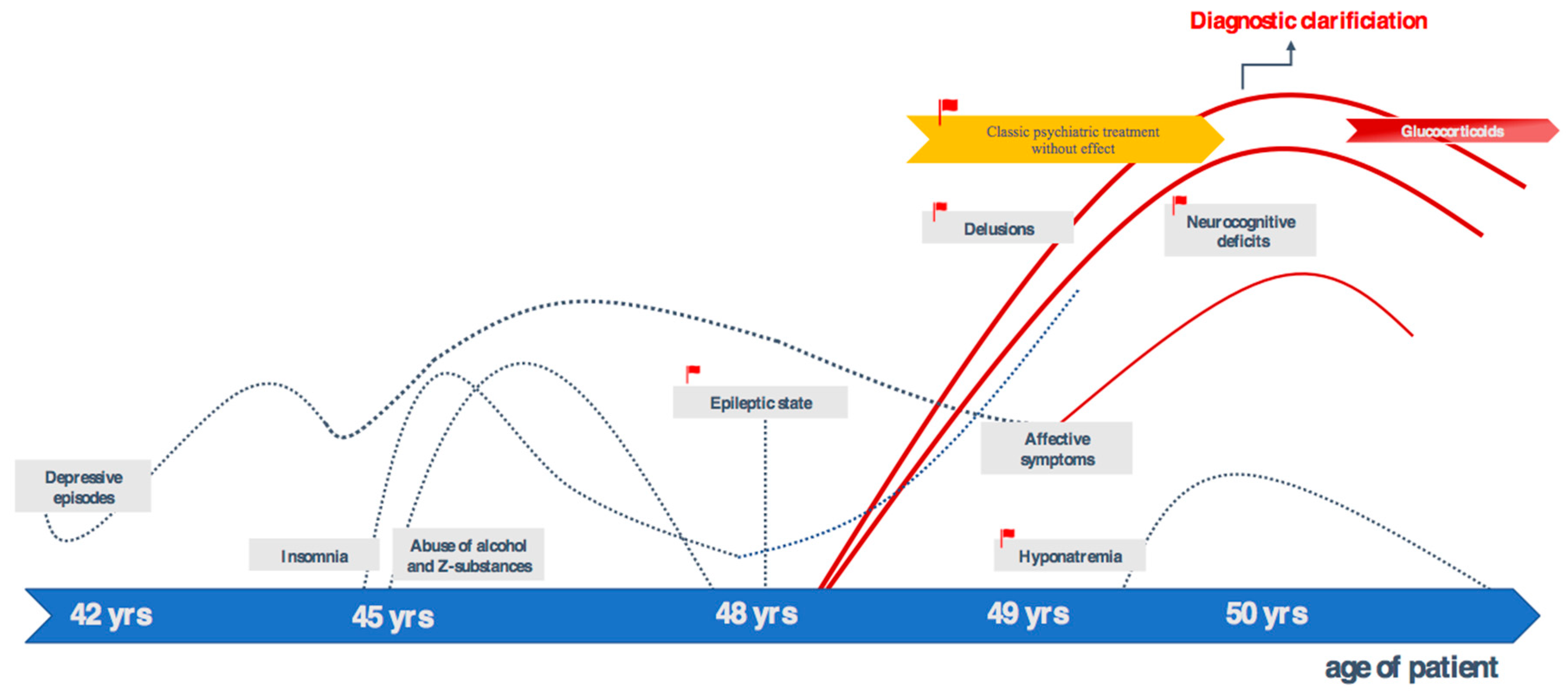
2. Case Presentation
3. Discussion
3.1. Diagnostic Assessment
3.2. Limitations
3.3. Clinical Consequences
3.4. Conceptual Considerations
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Van Sonderen, A.; Thijs, R.D.; Coenders, E.C.; Jiskoot, L.C.; Sanchez, E.; de Bruijn, M.A.; van Coevorden-Hameete, M.H.; Wirtz, P.W.; Schreurs, M.W.; Sillevis Smitt, P.A.; et al. Anti-LGI1 encephalitis: Clinical syndrome and long-term follow-up. Neurology 2016, 87, 1449–1456. [Google Scholar] [CrossRef]
- Van Sonderen, A.; Petit-Pedrol, M.; Dalmau, J.; Titulaer, M.J. The value of LGI1, Caspr2 and voltage-gated potassium channel antibodies in encephalitis. Nat Rev Neurol. 2017, 13, 290–301. [Google Scholar] [CrossRef]
- Dalmau, J.; Graus, F. Antibody-Mediated Encephalitis. N. Engl. J. Med. 2018, 378, 840–851. [Google Scholar] [CrossRef] [Green Version]
- Graus, F.; Titulaer, M.J.; Balu, R.; Benseler, S.; Bien, C.G.; Cellucci, T.; Cortese, I.; Dale, R.C.; Gelfand, J.M.; Geschwind, M.; et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 2016, 15, 391–404. [Google Scholar] [CrossRef] [Green Version]
- Van Sonderen, A.; Schreurs, M.W.; Wirtz, P.W.; Sillevis Smitt, P.A.; Titulaer, M.J. From VGKC to LGI1 and Caspr2 encephalitis: The evolution of a disease entity over time. Autoimmun Rev. 2016, 15, 970–974. [Google Scholar] [CrossRef] [Green Version]
- Prüss, H.; Lennox, B.R. Emerging psychiatric syndromes associated with antivoltage-gated potassium channel complex antibodies. J. Neurol. Neurosurg. Psychiatry 2016, 87, 1242–1247. [Google Scholar] [CrossRef] [PubMed]
- Dalmau, J.; Geis, C.; Graus, F. Autoantibodies to Synaptic Receptors and Neuronal Cell Surface Proteins in Autoimmune Diseases of the Central Nervous System. Physiol Rev. 2017, 97, 839–887. [Google Scholar] [CrossRef] [PubMed]
- Urbach, H.; Rauer, S.; Mader, I.; Paus, S.; Wagner, J.; Malter, M.P.; Prüss, H.; Lewerenz, J.; Kassubek, J.; Hegen, H.; et al. Supratentorial white matter blurring associated with voltage-gated potassium channel-complex limbic encephalitis. Neuroradiology 2015, 57, 1203–1209. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, A.; Rauer, S.; Mader, I.; Meyer, P.T. Cerebral FDG-PET and MRI findings in autoimmune limbic encephalitis: Correlation with autoantibody types. J. Neurol. 2013, 260, 2744–2753. [Google Scholar] [CrossRef]
- Wegner, F.; Wilke, F.; Raab, P.; Tayeb, S.B.; Boeck, A.L.; Haense, C.; Trebst, C.; Voss, E.; Schrader, C.; Logemann, F.; et al. Anti-leucine rich glioma inactivated 1 protein and anti-N-methyl-D-aspartate receptor encephalitis show distinct patterns of brain glucose metabolism in 18F-fluoro-2-deoxy-d-glucose positron emission tomography. BMC Neurol. 2014, 14, 136. [Google Scholar] [CrossRef] [Green Version]
- Jang, Y.; Lee, S.T.; Bae, J.Y.; Kim, T.J.; Jun, J.S.; Moon, J.; Jung, K.H.; Park, K.I.; Irani, S.R.; Chu, K.; et al. LGI1 expression and human brain asymmetry: Insights from patients with LGI1-antibody encephalitis. J. Neuroinflamm. 2018, 15, 279. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, M.; Tripathi, M.; Roy, S.G.; Parida, G.K.; Ihtisham, K.; Dash, D.; Damle, N.; Shamim, S.A.; Bal, C. Metabolic topography of autoimmune non-paraneoplastic encephalitis. Neuroradiology 2018, 60, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Blinder, T.; Lewerenz, J. Cerebrospinal Fluid Findings in Patients with Autoimmune Encephalitis-A Systematic Analysis. Front Neurol. 2019, 10, 804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kornau, H.C.; Kreye, J.; Stumpf, A.; Fukata, Y.; Parthier, D.; Sammons, R.P.; Imbrosci, B.; Kurpjuweit, S.; Kowski, A.B.; Fukata, M.; et al. Human Cerebrospinal Fluid Monoclonal LGI1 Autoantibodies Increase Neuronal Excitability. Ann. Neurol. 2020, 87, 405–418. [Google Scholar] [CrossRef] [Green Version]
- Endres, D.; Maier, S.; Feige, B.; Mokhtar, N.B.; Nickel, K.; Goll, P.; Meyer, S.A.; Matthies, S.; Ebert, D.; Philipsen, A.; et al. Increased rates of intermittent rhythmic delta and theta activity in the electroencephalographies of adult patients with attention-deficit hyperactivity disorder. Epilepsy Behav. 2017, 75, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Salmon, E.; Garraux, G.; Delbeuck, X.; Collette, F.; Kalbe, E.; Zuendorf, G.; Perani, D.; Fazio, F.; Herholz, K. Predominant ventromedial frontopolar metabolic impairment in frontotemporal dementia. Neuroimage 2003, 20, 435–440. [Google Scholar] [CrossRef]
- Minoshima, S.; Frey, K.A.; Koeppe, R.A.; Foster, N.L.; Kuhl, D.E. A diagnostic approach in Alzheimer’s disease using three-dimensional stereotactic surface projections of fluorine-18-FDG PET. J. Nucl. Med. 1995, 36, 1238–1248. [Google Scholar]
- Heine, J.; Prüss, H.; Bartsch, T.; Ploner, C.J.; Paul, F.; Finke, C. Imaging of autoimmune encephalitis—Relevance for clinical practice and hippocampal function. Neuroscience 2015, 309, 68–83. [Google Scholar] [CrossRef]
- Dodich, A.; Cerami, C.; Iannaccone, S.; Marcone, A.; Alongi, P.; Crespi, C.; Canessa, N.; Andreetta, F.; Falini, A.; Cappa, S.F.; et al. Neuropsychological and FDG-PET profiles in VGKC autoimmune limbic encephalitis. Brain Cogn. 2016, 108, 81–87. [Google Scholar] [CrossRef]
- Chen, C.; Wang, X.; Zhang, C.; Cui, T.; Shi, W.X.; Guan, H.Z.; Ren, H.T.; Shao, X.Q. Seizure semiology in leucine-rich glioma-inactivated protein 1 antibody-associated limbic encephalitis. Epilepsy Behav. 2017, 77, 90–95. [Google Scholar] [CrossRef]
- Shan, W.; Liu, X.; Wang, Q. Teaching NeuroImages: 18F-FDG-PET/SPM analysis in 3 different stages from a patient with LGI-1 autoimmune encephalitis. Neurology 2019, 93, e1917–e1918. [Google Scholar] [CrossRef] [PubMed]
- Newey, C.R.; Sarwal, A. Hyponatremia and Voltage Gated Potassium Channel Antibody Associated Limbic Encephalitis. J. Neurol Neurophysiol. 2014, 5, 195. [Google Scholar] [CrossRef] [Green Version]
- Black, N.; Hamada, H. Possible anti-VGKC autoimmune limbic encephalitis associated with SIADH. BMJ Case Rep. 2018, bcr–2017. [Google Scholar] [CrossRef] [PubMed]
- Head, K.; Gong, S.; Joseph, S.; Wang, C.; Burkhardt, T.; Rossi, M.R.; LaDuca, J.; Matsui, S.; Vaughan, M.; Hicks, D.G.; et al. Defining the expression pattern of the LGI1 gene in BAC transgenic mice. Mamm. Genome. 2007, 18, 328–337. [Google Scholar] [CrossRef]
- Sechi, G.; Serra, A. Wernicke’s encephalopathy: New clinical settings and recent advances in diagnosis and management. Lancet Neurol. 2007, 6, 442–455. [Google Scholar] [CrossRef]
- Prüß, H.; Köhler, S.; Müller, S. Autoimmune encephalitis-Diagnostic and therapeutic decision tree from a psychiatric, neurological and ethico-legal point of view: Approach in cases of lack of ability to give consent and permissibility of compulsory treatment. Der Nervenarzt 2020, 91, 122–130. [Google Scholar] [CrossRef] [Green Version]
- Ho, C.S.H.; Ho, R.C.M.; Quek, A.M.L. Chronic Manganese Toxicity Associated with Voltage-Gated Potassium Channel Complex Antibodies in a Relapsing Neuropsychiatric Disorder. Int. J. Environ. Res. Public Health 2018, 15, 783. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.; Hao, Q.; He, L.; Wang, Q. LGI1 antibody encephalitis and psychosis. Australas. Psychiatry 2018, 26, 612–614. [Google Scholar] [CrossRef]
- Ellul, P.; Groc, L.; Tamouza, R.; Leboyer, M. The Clinical Challenge of Autoimmune Psychosis: Learning from Anti-NMDA Receptor Autoantibodies. Front Psychiatry 2017, 8, 54. [Google Scholar] [CrossRef] [Green Version]
- Herken, J.; Prüss, H. Red Flags: Clinical Signs for Identifying Autoimmune Encephalitis in Psychiatric Patients. Front Psychiatry 2017, 8, 25. [Google Scholar] [CrossRef] [Green Version]
- Najjar, S.; Steiner, J.; Najjar, A.; Bechter, K. A clinical approach to new-onset psychosis associated with immune dysregulation: The concept of autoimmune psychosis. J. Neuroinflamm. 2018, 15, 40. [Google Scholar] [CrossRef]
- Endres, D.; Bechter, K.; Prüss, H.; Hasan, A.; Steiner, J.; Leypoldt, F.; Tebartz van Elst, L. Autoantibody-associated schizophreniform psychoses: Clinical symptomatology. Der Nervenarzt 2019, 90, 547–563, German. [Google Scholar] [CrossRef]
- Tebartz van Elst, L.; Bechter, K.; Prüss, H.; Hasan, A.; Steiner, J.; Leypoldt, F.; Endres, D. Autoantibody-associated schizophreniform psychoses: Pathophysiology, diagnostics, and treatment. Der Nervenarzt 2019, 90, 745–761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pollak, T.A.; Lennox, B.R.; Müller, S.; Benros, M.E.; Prüss, H.; Tebartz van Elst, L.; Klein, H.; Steiner, J.; Frodl, T.; Bogerts, B.; et al. Autoimmune psychosis: An international consensus on an approach to the diagnosis and management of psychosis of suspected autoimmune origin. Lancet Psychiatry 2020, 7, 93–108. [Google Scholar] [CrossRef]
- Titulaer, M.J.; McCracken, L.; Gabilondo, I.; Armangué, T.; Glaser, C.; Iizuka, T.; Honig, L.S.; Benseler, S.M.; Kawachi, I.; Martinez-Hernandez, E.; et al. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: An observational cohort study. Lancet Neurol. 2013, 12, 157–165. [Google Scholar] [CrossRef] [Green Version]
- McKeon, A.; Marnane, M.; O’connell, M.; Stack, J.P.; Kelly, P.J.; Lynch, T. Potassium channel antibody associated encephalopathy presenting with a frontotemporal dementia like syndrome. Arch. Neurol. 2007, 64, 1528–1530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suppiah, R.; Mukhtyar, C.; Flossmann, O.; Alberici, F.; Baslund, B.; Batra, R.; Brown, D.; Holle, J.; Hruskova, Z.; Jayne, D.R.; et al. A cross-sectional study of the Birmingham Vasculitis Activity Score version 3 in systemic vasculitis. Rheumatology (Oxf.) 2011, 50, 899–905. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Endres, D.; Perlov, E.; Stich, O.; Rauer, S.; Maier, S.; Waldkircher, Z.; Lange, T.; Mader, I.; Meyer, P.T.; van Elst, L.T. Hypoglutamatergic state is associated with reduced cerebral glucose metabolism in anti-NMDA receptor encephalitis: A case report. BMC Psychiatry 2015, 15, 186. [Google Scholar] [CrossRef] [Green Version]
| Neuropsychiatric examination |
|
| Blood analyses |
|
| Cerebrospinal fluid analyses (was performed three times after in-patient admission) |
|
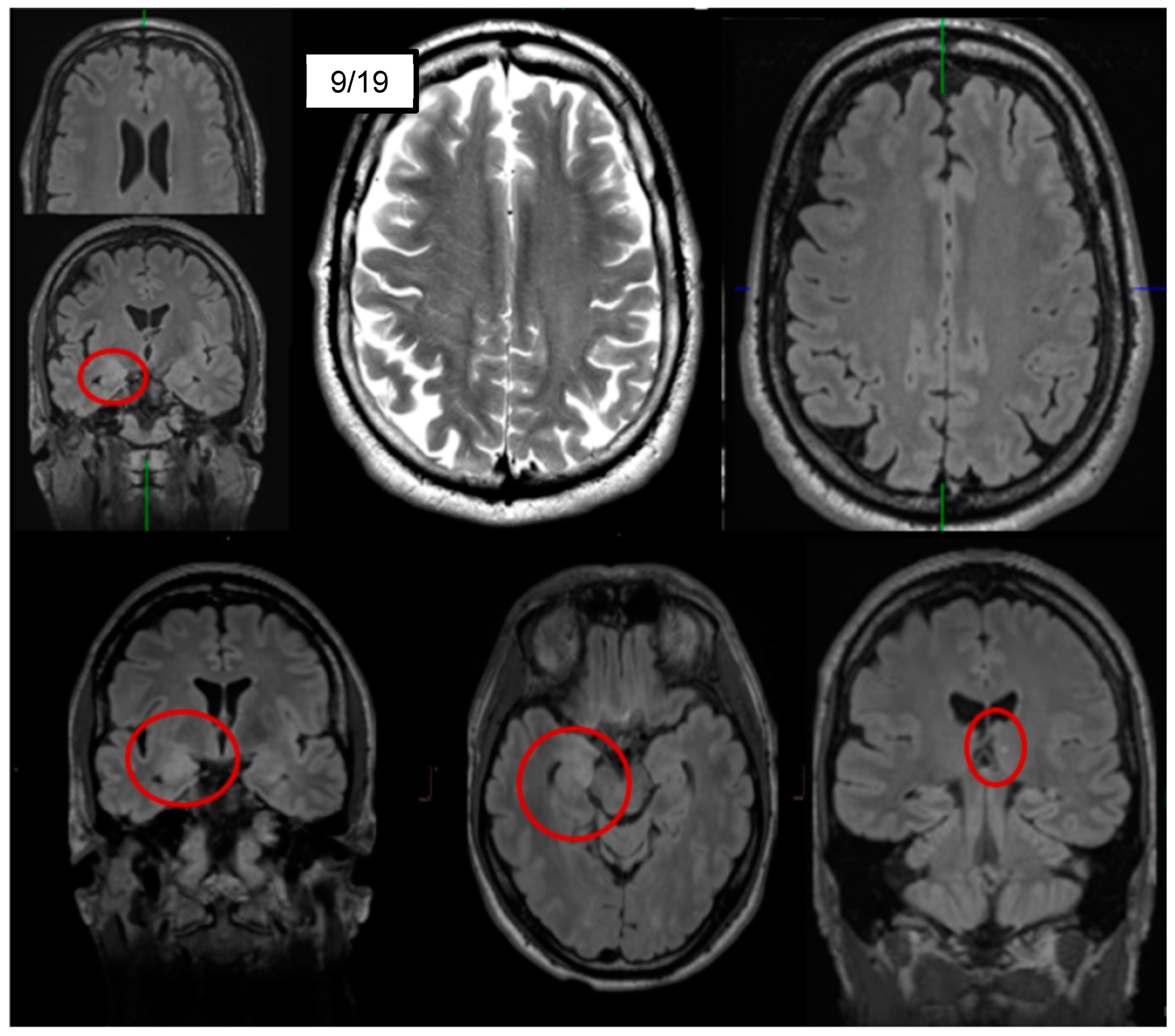
| Cerebral magnetic resonance imaging |
|
| Electroencephalo-graphy |
|
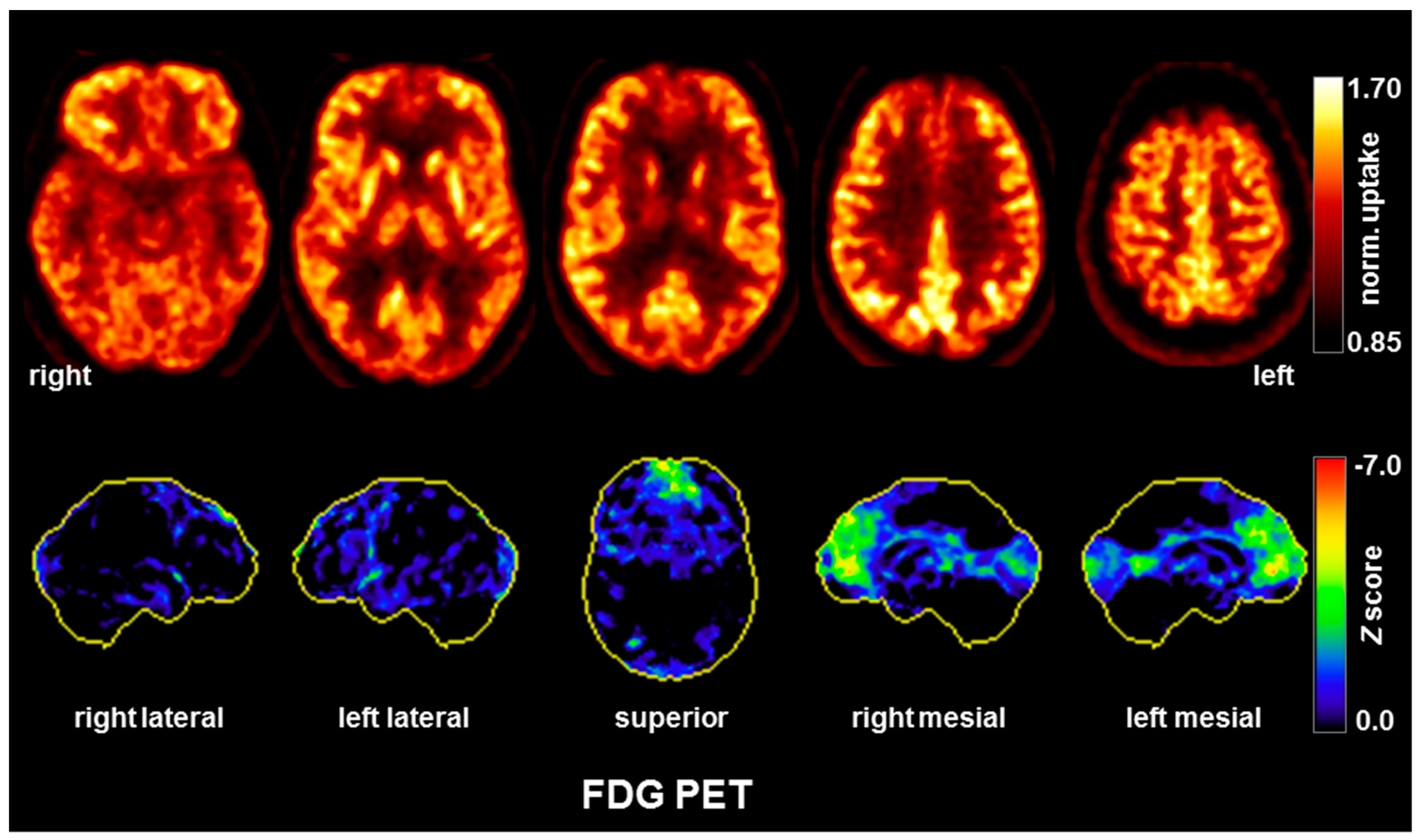
| FDG-PET |
|
| Heart examination |
|
| Computer tomography and x-ray thorax |
|
| Bronchoscopy |
|
| Sonography of the abdomen |
|
| Electromyography |
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Endres, D.; Prüss, H.; Dressing, A.; Schneider, J.; Feige, B.; Schweizer, T.; Venhoff, N.; Nickel, K.; Meixensberger, S.; Matysik, M.; et al. Psychiatric Manifestation of Anti-LGI1 Encephalitis. Brain Sci. 2020, 10, 375. https://doi.org/10.3390/brainsci10060375
Endres D, Prüss H, Dressing A, Schneider J, Feige B, Schweizer T, Venhoff N, Nickel K, Meixensberger S, Matysik M, et al. Psychiatric Manifestation of Anti-LGI1 Encephalitis. Brain Sciences. 2020; 10(6):375. https://doi.org/10.3390/brainsci10060375
Chicago/Turabian StyleEndres, Dominique, Harald Prüss, Andrea Dressing, Johanna Schneider, Bernd Feige, Tina Schweizer, Nils Venhoff, Kathrin Nickel, Sophie Meixensberger, Miriam Matysik, and et al. 2020. "Psychiatric Manifestation of Anti-LGI1 Encephalitis" Brain Sciences 10, no. 6: 375. https://doi.org/10.3390/brainsci10060375





