1. Introduction
Diabetes mellitus (DM) is a multifactorial-polygenic disease characterized by chronic hyperglycemia and altered metabolism of carbohydrates, lipids, and proteins. There are two major clinical forms: Insulin-dependent DM (type 1: T1DM) and non-insulin-dependent DM (type 2: T2DM), being the latter the most common presentation, accounting for 90% of all diabetics [
1]. DM displays alarming growth rates worldwide, being a serious health concern with an estimated number of affected people of 600 million by the year 2030 [
2,
3].
Microvascular changes are clinically manifested during the natural course of DM. Population studies reported that 20% of diabetics had retinopathy (DR) at the time of diagnosis [
4]. The main pathogenic mechanisms in the diabetic retina include the activation of oxidative stress (OS), advanced glycation end-product generation, inflammation, activation of protein kinase C, polyol, and hexosamine pathways, etc. [
5]. In addition, a series of endogenous/exogenous risk factors have been pointed as contributing highly to the DR initiation and progression [
6].
The incomplete reduction of molecular oxygen (O
2) leads to the formation of reactive oxygen species (ROS) with unpaired electrons in its outermost orbital, making these molecules extremely unstable and reactive. The generation of superoxide anion (O
2−) and hydrogen peroxide (H
2O
2) is prone to start the free radical chain reaction due to hydroxyl radical formation (OH
−), by the Fenton and Haber Weiss chemical reactions [
7,
8]. Furthermore, nitric oxide (NO) is a non-reactive free radical implicated in a variety of biological functions, such as vasodilation, immune response, neurotransmission, and apoptosis [
9,
10].
It is well known that redox status influences the regulation of intracellular signaling with enormous repercussions in health and disease [
11]. In this context, epidemiologic and experimental studies demonstrated that DR is noticeably related to OS [
5,
12,
13,
14,
15].
The concern of nutritional supplements in ocular health and disease has been largely addressed [
16,
17]. Nutritional control has been proposed as a potential intervention for slowing DR initiation/progression, but studies on dietary assessments in diabetics showed contradictory results [
18,
19,
20,
21,
22,
23]. Moreover, nutraceuticals cannot replace eating adequate amounts of a high variety of foods, but more than half of Americans and many Europeans take at least one dietary supplement on a regular basis [
16,
17]. These may be especially beneficial to individuals with poor nutrition caused by a variety of processes, including DM. International science-based regulatory considerations in nutrition and nutraceutical supplements are needed to update its recommendations for the ophthalmological population [
18,
19,
20,
21,
22,
23]. Antioxidant (A) vitamins (vit) are essential for a wide variety of biological functions. Among them, the reduced form of ascorbic acid (AscH2; L-ascorbic acid; vit C), a hydrophilic vitamin, enzymatic cofactor, and ROS scavenger, is involved in the protection against a variety of disorders. Abnormal dietary intake and/or plasma levels of vit C (deficiency defined as a concentration equal of less than 35 µmol/L) have been associated to a higher risk of getting sick, including the development of xerosis and other ophthalmic pathologies [
24]. In this concern, vit C absorption and distribution to tissues and organs is achieved by active transport across the cell membranes. Two specific protein transporters encoded by the sodium-dependent vit C transporter genes, SLC23A1 and SLC23A2 [
25], are involved in this fact. The latter is responsible for the regulation of the intracellular levels of vit C to protect cells from OS, as well as to promote the type I collagen maturation. In fact, genetic variations in the SLC23A2 gene expression modify the vit C availability [
24,
25].
Given that early diagnosis can delay or prevent DR-related blindness, there is considerable interest in exploring the role of its molecular diagnostics [
17,
18]. Up to today, studies on DR biomarkers have dealt with pathogenic processes and risk factors involved in DR development/progression [
2,
3,
5,
26,
27]. The need for more accurate diagnostics that allow advance ophthalmologic and vision care is currently providing a critical window of opportunity for diabetics. The present study deals with improving knowledge about the classical and new potential diagnostic/prognostic candidate biomarkers to verify/validate them by using various endpoints like ophthalmic indicators, classical blood biochemistry, oxidative and antioxidant parameters, as well as the
SLC23A2 gene. The secondary endpoint was to analyze the effect of a course of A and omega 3 fatty acids (A/ω3) in the form of mono-combination on the OS presumptive biomarkers, in order to better managing the diabetic eyes.
3. Results
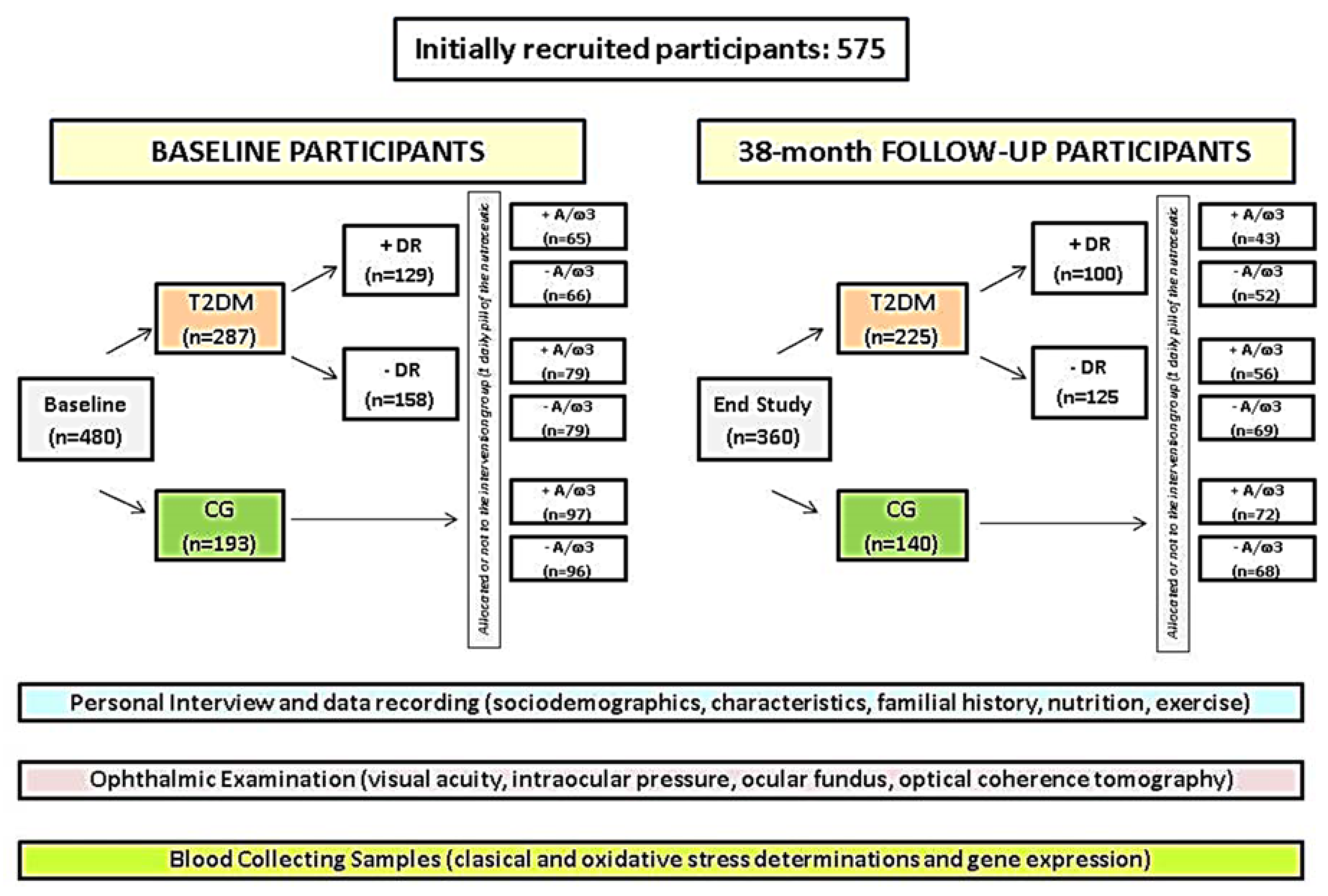
Out of the suitable initial 480 participants, 365 (rate of response from baseline: 76%) completed their 38-month study course (
Figure 1). The overall dropout rate was 24%. Deterioration of health/lack of motivation (7%), confusing ocular fundus or retinographies, and/or progression to PDR or DME (10%), and blood samples failing to show any data (7%) constituted common causes of withdrawn.
Table 2 shows the main socio-demographic data, medical history, and laboratory parameters of the study participants. 26% of the baseline population had DR. Lead time between the DM diagnosis and the last study visit was 22 (5) years in the +DR and 15 (3) years in the −DR (
p = 0.002).
Eating habits were addressed during the study course to check whether the participants had nutrient adequacy and adherence to the MedDiet. Higher nutritional control was observed in the T2DM − DR (41%) than in the T2DM + DR (30%) (p = 0.001). Moreover, a higher percentage of diabetics +DR (32%) displayed a lower MedDiet questionnaire score as compared to the T2DM − DR patients (19%) (p = 0.0001). Significantly higher BMI was seen in diabetics vs. the CG (p = 0.001).
No significant differences were observed between the T2DM patients and the CG, regarding age, gender, and physical exercise routine, but statistically significant differences (
p = 0.000001) in glucose and HbA1c determinations were detected between the main study groups (
Table 2).
Ocular fundus was explored and photographed in the CG participants, showing a normal appearance without any DR signs (
Figure 3A), in contrast to the diabetics with mild-to-moderate retinal complications, as reflected in the ocular fundus of
Figure 3B.
Furthermore, the series of ocular fundus examinations of the T2DM group revealed a mild increment in NPDR development and progression with respect to baseline. Among the participants who completed the study altogether, those with retinopathy showed impairment of retinal affectation grading (54%: Mild DR; 35%: Moderate DR; 11%: Severe DR) from baseline (76% mild; 21% moderate; 3% severe).
Morphologic and morphometric evaluation of the OCT images was performed on the T2DM patients and the CG individuals (
Figure 3C,D). Representative OCT images, as shown in
Figure 3C,D, did not show noticeable differences in central retinal thickness from each eye when the diabetic patients and the control individuals were compared (
Figure 3E). We have to emphasize that this tool improved our capacity to document diabetic retinal lesions in the posterior eye pole.
Table 3 shows the main parameters of the ophthalmologic examination in the study participants. These data revealed noticeable differences in structural/functional values between groups, distributed into participants assigned to the A/ω3 regimen versus the no supplemented ones.
A relevant point of this follow-up study was the evaluation of DR progression. In fact, the ETDRS grading scale [
28] (that is more popular among the researchers that other similar DR classification systems) was used in the present work to define DR progression. The presence of hemorrhages, hard exudates, and cotton-wool spots was equivalent to the ETDRS level 35 of a mild NPDR [
28]. Moderate and severe NPDR forms have also been conveniently staged, as reflected in
Figure 3.
From the 129 T2DM patients with DR (NPDR forms) at baseline, 22% showed a cumulative incidence of DR impairment at the end of follow-up, the majority of them (13%) corresponding to mild NPDR stages, 5% to moderate stage, and 4% corresponding to the severe NPDR form. Among the diabetics that showed clinical signs and symptoms of retinopathy progression, 65% pertained to the −A/ω3 subgroup. From the 158 initial T2DM patients without NPDR, (27%) developed DR at the end of the study, 12% mild; 9% moderate; 5% severe. Among the diabetics that initially did not suffer DR, 58% pertained to the −A/ω3 subgroup as shown in the
Table 4.
The OS markers were assayed in plasma samples from the groups and subgroups of participants. At baseline, comparison of the CG and the T2DM patients reflected significantly higher plasma levels of MDA/TBARS [2.9 (0.1) µM vs. 2.1 (0.2) µM (
p = 0.001)] and significantly lower plasma levels of TAC [2.3 (0.1) mM vs. 3.1 (0.3) mM (
p = 0.001)], GSH [2 (0.2) µM vs. 2.8 (0.2) µM (
p = 0.002)] and vit C [40 (19) µM/mL vs. 56 (20) µM/mL (
p = 0.001)]. Therefore, diabetics displayed significantly higher circulating pro-oxidant markers and lower antioxidant levels than the non-diabetic study population. Furthermore, significantly higher plasma levels of MDA/TBARS, lower TAC, as well as decreased vit C were observed in the T2DM + DR with respect to the T2DM − DR patients, both at baseline and at the end of follow-up.
Table 5 illustrates the effects of the A/ω3 administration in mono-combination on biochemical blood traits, as the patients were categorized in relation to their response to the nutraceutical regimen. Therefore, participants randomly assigned to the oral +A/ω3 improved their OS background with statistical differences between the T2DM + DR vs. T2DM − DR patients throughout the 38-month follow-up (
Table 5). A regular A/ω3 supplementation course reduced the OS by conferring a potential neuroprotective effect to the diabetic retina, which is a priority and challenging topic for our current research and the foreseeable future.
In our study population, we observed that aging, higher DM length, bad metabolic control (as reflected by the elevated levels of glycemia and HbA1c) in the presence of significant oxidative load were frequent findings that helped to identify diabetics at risk of DR and DR progression. We also found that overweight patients had poorer overall glycemic control also suffered a more advanced stage of NPDR than the normal-weight diabetics throughout the 38-month follow-up.
Regarding the gene expression assays, we detected a differential expression profile of the SLC23A2 gene (responsible for the regulation of the intracellular levels of vit C) between the main groups and subgroups of the study participants at baseline (
Figure 4).
When corrected, these data for multiple testing, significantly lowered SLC23A2 gene expression (fold change) in the T2DM vs. the CG (p < 0.05) and in the T2DM +DR vs. −DR (p < 0.05) was detected. Furthermore, we found that the SLC23A2 gene was down-regulated in T2DM patients, and it was associated with significantly lower vit C plasma levels (p = 0.001). The above association remained statistically significant (p < 0.05) even after a multivariate adjustment for major potential confounders (sex, age, eating habits, and BMI) was carried out in the data processing.
In addition, we analyzed the correlation between the main OS markers and the SLC23A2 gene expression in blood samples of the study participants at baseline. Spearman Rho correlation between the plasma MDA/TBARS values and SLC23A2 blood gene expression was statistically significant [0.290 (p = 0.026) for the T2DM and 0.188 (p = 0.048) for the CG, and 0.651 (p = 0.030) for the T2DM + DR vs. 0.711 (p = 0.0002) for the T2DM-DR]. Spearman Rho correlation between the plasma TAC and SLC23A2 blood gene expression was statistically significant for the comparison of diabetics and controls [0.294 (p = 0.027) in the T2DM vs. 0.038 (p = 0.012) in the CG], as well as for the T2DM + DR patients [0.0427 (p = 0.001)], but no significant correlation was detected for the T2DM-DR patients [0.084 (p = 0.144)].
The final rate of compliance with the A/ω3 regimen was 89% for the T2DM and 80% for the CG. The highest response was for the T2DM + DR group than those from the T2DM − DR patients. There were no observed/reported side effects of the oral supplementation, except a few cases of mild gastrointestinal complaints.
4. Discussion
The main results from this population study from baseline to the 38-month follow-up were: (1) DR progression was more likely with higher DM duration, BMI, and Hb1Ac in the context of outstanding OS, and (2) Noticeable amelioration over six consecutive screening intervals, was detected in major hematological parameters and OS markers in the T2DM + DR patients randomly assigned to the oral A/ω3 regimen, as compared to the non-supplemented participants.
Several authors reported that metabolic and risk factors control ameliorated DR prognosis [
1,
2,
3,
4,
5,
6,
19,
28]. In the current study, we planned to analyze classic risk factors (T2DM duration, sedentary lifestyle, eating habits, and BMI), as well as blood parameters (glucose/HbA1c) and findings from the circulating candidate DR predictors (MDA/TBARS, TAC, GSH, vit C, and the SLC23A2 gene). Recent reports established that DR prevalence in Spain in the T2DM patients depends on the study population, ranging from 15% [
27] to 26% [
28,
36]. From our initial 480 participants, 26% had DR. In addition, the mean T2DM lastingness at 38-months follow-up was 15 (3) years in −DR patients vs. 22 (5) years for the +DR patients. Nevertheless, additional factors also play critical roles in managing DR at any stage [
1,
2,
3,
4,
5,
6,
19,
26,
27,
28], with a special interest in diabetics without clinically detectable retinopathy and those suffering from the early DR stage [
37,
38].
Nutrition is a pivotal component for diabetic patients care. Regarding DR, researchers have described the role of micronutrients (including A/ω3), macronutrients, several food groups, and dietary aspects and lifestyle, such as the MedDiet, in the initiation and progression of DR [
4,
5,
6,
18,
19,
20,
21,
22,
23,
24]. Despite this, findings remain inconclusive, and current evidence does not inform about the specific dietary components that are likely to reduce (or increase) the risk of DR. Moreover, new pharmacologic therapies for DR are urgently needed [
4,
19,
26,
27,
28].
A growing paradigm in managing DR is the relevance of endogenous pathogenic mechanisms, such as OS and its downstream effectors [
4,
12,
13,
14,
15,
19]. A variety of plasmatic OS biomarkers (MDA/TBARS, TAC, vit C, vit E, and ω3) have been reported in diabetics [
7,
12,
13,
14,
19,
26,
27,
39,
40]. Furthermore, Pan et al. interestingly reported a combinatorial molecular signature that may potentially be used as a marker of PDR [
41]. Regarding the latter report, we similarly have described a differential profile of circulating OS markers in the T2DM patient’s with respect to the non-diabetic individuals, but our diabetic population was only constituted by NPDR patients.
There is growing interest in identifying genes that could trigger OS in diabetics. Our experiments allowed us to demonstrate a statistically significant differential expression profile of the SLC23A2 gene between the T2DM patients and the non-diabetic participants, as well as between the T2DM + DR vs. −DR patients. Being this gene the regulator of vit C intracellular levels (for protecting the cells from OS) and the promoter of type I collagen maturation [
24,
25], we also found that the SLC23A2 gene was down-regulated in T2DM patients and highly associated with significantly lower vit C plasma levels. Multivariate adjustment for major potential confounders (sex, age, eating habits, and BMI) was carried out in the data processing, and the association of lower SLC23A2 expression levels with lower vit C plasma concentration remained statistically significant in our study population (
p < 0.05). Shaghaghi et al. [
42] emphasized the necessity to consider the genetic variation of vitamin C transporters in clinical and epidemiologic studies regarding complex diseases. Encouraged by our results, we strongly suggest the connecting role of the SLC23A2 gene down-regulation and the inappropriate vit C bioavailability, with redox imbalance in the context of chronic hyperglycemia and DR. In this concern, Murgia and Adanski [
43] also reported that genetic variation alters nutritional requirements, and that nutritional genomics provides an outstanding challenge for biomedical research, as in DR.
In addition, a significant plasmatic decrease in MDA/TBARS, as well as an increase in TAC, was found in the T2DM +A/ω3 and the CG +A/ω3 during the study period. Inclusion of multiple components in the tested formula [vit (E, C, B1, B2, B3, B6, B9, and B12), lutein, zeaxanthin, GSH, hydroxytyrosol, trace elements (Se, Mn, Zn, and Cu) and DHA] theoretically targeting multiple and overlapping pathogenic mechanisms implicated in DR, strongly support a wider counter-response [
7,
12,
13,
14,
19,
44,
45,
46]. In fact, the European Union ROS consortium (EU-ROS) stated in 2017 that many diseases had been found to be associated with OS, and subsequently, the theory that OS-related pathologies can be corrected by antioxidant therapy was launched [
47]. However, while experimental studies support this theory, clinical studies still generate controversial results [
48,
49]. In our opinion, the possibility remains that biotherapies based on the redox changes occurring in diabetics may help to protect the retina. In the latter concern, an interesting spectrum of presumptive factors for DR protection has been ultimately proposed, including outstanding antioxidants [
50,
51,
52,
53].
From a clinical viewpoint, retinal microvascular changes found in the NPDR eyes are related to basement membranes thickening, pericyte loss, capillary occlusion, and microaneurysms, that can progress towards ischemia, macular edema, and neovascularization [
4,
5,
19,
36,
37,
38]. In this context, image analysis of retinal lesions can provide qualitative and quantitative data to accurately diagnosing and grading DR. Our data suggest relatively lower rates of progression of DR in our cohort during the 38-month follow-up. Regarding the participants that completed the study, only 11% of the T2DM patients +DR progressed to severe NPDR forms. Among the participants assigned to the A/ω3 supplementation, the impairment was noticeably less than in the non-supplemented participants. From the 129 T2DM patients with DR (NPDR forms) at baseline, 22% showed a cumulative incidence of DR impairment at the end of follow-up, the majority (18%) corresponded to the mild-to-moderate NPDR stages. Among the diabetics that showed clinical signs and symptoms of retinopathy progression, 65% pertained to the −A/ω3 subgroup. Moreover, from the 158 initial T2DM patients without NPDR, 27% developed DR at the end of the study. In addition, the majority of these pertain to mild-to-moderate stages of the disease (21%), but 5% progressed to the severe NDPR. Among the T2DM that initially did not suffer DR, 58% were concerned with the −A/ω3 subgroup.
Data also showed that a reduction of hyperglycemia, HbA1c, and BMI may have a beneficial effect on the progression to more severe DR forms. We also confirmed that a regular A/ω3 supplementation course reduces the oxidative load, confers beneficial effects as opposed to detrimental ones, and may potentially act as a dietary prophylaxis/adjunctive intervention for high-risk diabetics. The OCT imaging technique provides retinal cross-section images within the micrometer resolution, allowing the evaluation of structural characteristics of the healthy and pathologic retina [
54]. It is widely known that the OCT examination qualitatively and quantitatively improves the preclinical diagnosis of retinal changes occurring in diabetics [
13,
19,
55,
56,
57,
58,
59,
60]. It was reported in T2DM -DR patients that the foveal and temporal retinal thickness appeared significantly reduced and that a higher HbA1c level was associated with a thinner retina in the temporal perifoveal area in these patients [
58]. Data comparison at the end of follow-up in our study cohort showed a significant trend to stabilization of the ophthalmologic parameters, mainly regarding the BCVA and OCT-derived measurements in the T2DM + DR + A/ω3 vs. the T2DM + DR − A/ω3 groups. This is an interesting point to consider because interventions directed to counteract several of the pathogenic mechanisms involved in the deleterious effects of chronic hyperglycemia on the retinal microvasculature are needed.
Moscos et al. [
61] reported an increase in the foveal thickness of T2DM patients at two years of oral supplementation with carotenoids, suggesting that the above type of intervention may benefit the visual function in diabetics. It has also been said that early oral supplementation with nutraceuticals provides a significant health benefit to prevent DR progression because of its antioxidant and anti-inflammatory properties, reducing both the neural and vascular damage caused in the retina by the hyperglycemia [
7,
12,
19,
62,
63]. The combined A/ω3 formula used herein strongly supported the descriptions of the above authors and our previous works [
19,
35,
39,
40,
49] on the processes of retinal neuroprotection and vasculoprotection. Moreover, Amato et al. [
64] emphasized the possible side effects of systemic neuroprotectants as well as doubts about the appropriate retinal bioavailability of these substances in contrast with the intravitreal injections. We hereby confirm that with the formulation used herein, no adverse effects were reported but scarcely mild gastrointestinal discomfort, while the intravitreal injections may potentially produce endophthalmitis, hemorrhages, rhegmatogenous retinal detachment, lens damage, and/or other complications [
65]. These authors interestingly described the feasibility of delivering magnetic nanoparticles functionalized with a neuroprotectant substance by means of intravitreal injections in mice, suggesting an interesting role of these new approaches to protect the retina in diabetic patients. In this concern, Dolz-Marco et al. [
66] also reported that intravitreal DHA administration (at the specific assayed doses) in rabbits is safe and potentially useful for treating retinal diseases.
No data from the interim time points were presented in this work because the main results from the 18-months follow-up were previously published [
19]. Regarding the above data, the BCVA (each eye separately) was significantly higher in the CG as compared to the T2DM group [(CG at baseline: RE: 0.95 ± 0.10 and LE: 0.96 ± 0.08;
p < 0.001/CG; at 18 months: RE: 0.95 ± 0.11 and LE: 0.95 ± 0.10;
p < 0.001) (T2DMG at baseline: RE: 0.81 ± 0.30 and LE: 0.83 ± 0.22;
p < 0.001/T2DMG; at 18 months: RE: 0.78 ± 0.22 and LE: 0.76 ± 0.22;
p < 0.001)]. Regarding the redox status of the study population at 18 months, on the one hand, plasmatic pro-oxidants (MDA/TBARS levels) significantly decreased in the T2DM + A/ω3 subgroup with respect to the T2DM − A/ω3 patients, and on the other hand, the TAC significantly increased in the T2DM + A/ω3 subgroup, while the non-supplemented participants did not show any change. These data are similar to those obtained from the 38-month follow-up during the present work.
The authors are confident that the results obtained during this follow-up study can be useful in designing an integrative document with the clinical (ocular fundus multimodal imaging) and molecular-genetic biomarkers (MDA/TBARS, TAC, vit C, and
SLC23A2 gene) in the form of practical guidelines that can be helpful for primary care physicians as well as to ophthalmologists and ophthalmic researchers. In this concern, classical unmodifiable risk factors, such as the older age and/or longer DM duration have been widely involved in the development and progression of DR, altogether with the modifiable biochemical parameters of higher glycemia and HbA1c as well as the increased BMI and the lack of physical exercise [
1,
2,
3,
4,
5,
6,
19]. All these, and the oxidative stress parameters reported herein (MDA/TBARS, TAC, vit C), can help outline the importance of integrating patient characteristics, as well as clinical and biochemical biomarkers better to identify diabetics at risk of retinopathy and visual disability. This follow-up study strongly emphasizes the benefits of the effects of the A/ω3 administration in mono-combination on both the DR and biochemical blood traits in T2DM patients. Up to today, our group continues to pursue research, consistently looking for new solutions to avoid diabetic blindness.
A study limitation is the relatively small final sample of participants distributed in each group and subgroup reaching the 38-month follow-up. Patients who developed PDR or DME were excluded from data processing to avoid the bias of statistical procedures, mainly because these patients needed to be immediately treated with specific medical, laser, or surgical interventions. Statistical processing of the GSH data showed surprising results. No significant differences were found at the end of the study regarding the A/ω3 supplementation (containing GSH). We may hypothesize that sample handling may induce artificial oxidation of GSH, producing GSSG that interfered with our results. It is needed to proceed with a rapid –SH alkylation with N-ethylmaleimide to improve these results for further research. The study has provided a large amount of data for easy analyses. Because of this, we have focused on the main objectives of our work, and some of the data have been omitted for the present study. In fact, the expression of the
SLC23A2 gene was only assayed at baseline in the main groups and subgroups of participants. No comparison was made of the
SLC23A2 gene expression between the baseline and the end of follow-up and nor in the participants assigned or not to the oral supplement regimen due to unsolvable problems at the end of follow-up. Nevertheless, the differential expression profile of the SLC23A2 gene in diabetics, as compared to the controls, as well as in diabetics with and without NPDR, provided interesting data at the beginning of the study (as reflected in
Figure 4), showing outstanding correlation with the plasma vit C levels. In fact, Hierro et al. [
67] demonstrated a SLC23A2 down-regulation in the kidney and brain from a model of streptozotocin Zucker rats.