1. Introduction
Subunit vaccines rely on the activity of adjuvants to enhance immunogenicity and generate sufficiently robust immune responses against the target pathogen to provide protection from disease. While a number of adjuvants have been approved for human or veterinary use [
1,
2], they may not always be suitable for new vaccines due to their activity profile (i.e., insufficient immunogenicity, induction of humoral vs. cellular responses, etc.). In addition, they may not be readily available due to supply or intellectual property issues. Indeed, the COVID-19 pandemic has reaffirmed the importance of vaccines and the need for a steady supply of effective and diverse adjuvants [
3].
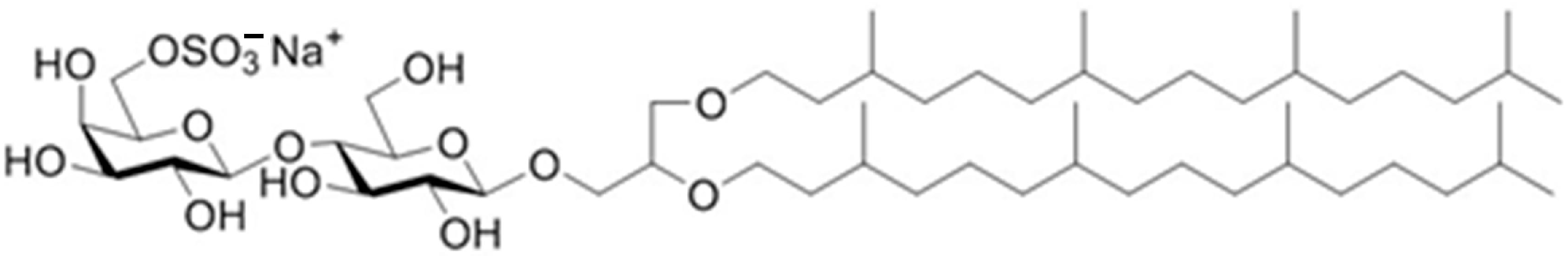
Archaeol-based liposomes (archaeosomes) contain lipids characterized by an archaea-specific structure where lipid tails consist of phytanyl side chains linked via an ether bond to the glycerol backbone [
4]. Archaeosomes have been shown to be strong vaccine adjuvants in a number of preclinical models, greatly enhancing humoral and cellular antigen-specific immune responses [
5,
6]. Archaeosomes of sulfated lactosyl archaeol (SLA), generated semi-synthetically through the attachment of sulfated lactose to a biologically produced archaeol core, have been shown to robustly adjuvant the immunogenicity of a number of disease antigens, including hepatitis B surface antigen (HBsAg), hepatitis C E1/E2 glycoproteins, influenza hemagglutinin (HA),
Schistosoma mansoni cathepsin B and SARS-CoV-2 spike in mice or hamsters [
7,
8,
9,
10,
11,
12]. As the formulation consists of a single lipid, SLA archaeosomes can be easily characterized and have been shown to be equally active with admixed antigen as with antigen encapsulated within the liposome [
8,
9,
13]. They induce local cytokine/chemokine secretion, immune cell recruitment and antigen retention at the injection site [
7,
14,
15]. Furthermore, SLA produced through a fully synthetic chemical process retain their adjuvant activity [
16].
Vaccines are an important tool to maintain the health of companion animals or economically important livestock, with a large number of vaccines on the market targeting a wide range of diseases [
1]. In addition, immunizing animals against zoonotic diseases could be an effective strategy to reduce transmission to humans [
17]. Rabbit hemorrhagic disease virus (RHDV) is a highly contagious and lethal calicivirus that mainly infects
Oryctolagus cuniculus rabbits [
18]. While not a human pathogen, effective vaccines targeting RHDV are required, as it does pose a serious risk to the health of rabbits in the wild, those kept as companion animals or those used in industry. The RHDV genome consists of a positive-strand RNA encoding a number of nonstructural genes, as well as the VP60 capsid protein [
19]. Classical RHDV, RHDV1, was identified in the 1980s and quickly spread across Europe and Asia [
18]. In 2010, a new strain emerged, RHDV2, capable of infecting multiple hare species as well as
Oryctolagus cuniculus. It has emerged as a serious threat, leading to outbreaks in many parts of the world.
Due to the largely conserved nature of the mammalian immune system, the technologies used in vaccines for animals are very similar to those used in humans. Vaccines targeting RHDV are generally of two types: (1) inactivated virus adjuvanted with aluminum salts or mineral oil (e.g., Filavac
® and Eravac) and (2) live viral vectored vaccine (e.g., Nobivac
® Myxo-RHD) where the RHDV capsid protein is expressed by a genetically modified myxoma virus [
20,
21]. While these vaccines have been highly effective against the original RHDV1 and more novel RHDV2 strains in areas with high disease burden, these vaccines have specific drawbacks. As the virus mainly replicates in rabbit liver cells which cannot be maintained in culture in vitro, the virus for the inactivated vaccines is routinely produced in live animals, which requires the use of more costly high containment laboratories to generate the material and then dispose of the contaminated tissues/waste. As there is a general risk of viral transmission/reversion with live viral vectored vaccines [
22], their use for RHDV has been limited in certain regions. With an increased understanding of RHDV biology, the development of effective subunit vaccines that bypass these concerns is possible. Multiple studies have shown that the capsid protein, VP60, from RHDV1 or RHDV2 can self-assemble into genome-free virus-like particles (VLPs) that can induce protection at high antigen doses in a challenge model [
23,
24,
25,
26,
27]. In addition, recent reports have identified a specific domain within RHDV1 VP60 that mediates cellular entry through interaction with nucleolin [
28]. Immunization with a 17mer peptide corresponding to this VP60 domain mediated partial protection in a challenge model. However, to date, no marketed subunit vaccines for RHDV have been developed.
RHDV subunit vaccine formulations could be further optimized for clinical use through use of robust adjuvants such as SLA. As SLA has only been evaluated in rodents to date, it was not clear whether it would have similar immunostimulatory effects in rabbits. Herein, we evaluated the immunogenicity and efficacy of SLA-adjuvanted vaccine formulations targeting RHDV in mice and rabbits. Antigens consisted of self-assembled RHDV2 VP60 VLPs or RHDV peptides targeting the nucleolin interaction domain conjugated to cross-reactive material 197 (CRM
197), a mutant nontoxic form of diphtheria toxin. CRM
197 is a well-defined carrier protein, which has been used in multiple conjugated vaccines for a broad range of disease indications [
29].
2. Materials and Methods
2.1. Vaccine Components
To produce the SLA adjuvant, total polar lipids were first extracted from
Halobacterium salinarum (ATCC 33170) biomass, hydrolyzed and archaeol core purified as previously described [
30]. Using the archaeol core, sulfated lactosyl archaeol (SLA; 6′-sulfate-β-d-Galp-(1,4)-β-d-Glcp-(1,1)-archaeol;
Figure 1) was synthesized as reported previously [
16,
31]. Archaeosomes were formed by hydrating the SLA lipid in water as previously described [
32]. SLA was diluted to the desired concentration in phosphate-buffered saline (PBS) and stored at 4 °C until use. SLA archaeosomes have been shown to be stable for up to 6 months when stored at 4 or 37 °C [
33].
For the RHDV–CRM
197 peptide conjugates, terminal N-acetylated peptides corresponding to the domain identified as responsible for nucleolin interaction with the RHDV1 capsid amino acids 468–484 [
28] (Ac-RRTGDVNAAAGSTNGTQK) and the homologous region from RHDV2 (Ac-RRTGDINAEAGSTNGTQK) were synthesized by JPT Peptide Technologies GmbH (Berlin, Germany). The peptides were designed to allow conjugation to the CRM
197 solely at the peptide’s C-terminal end. This was accomplished through addition of an N-terminal acetyl group and a C-terminal lysine residue which contains an amine sidechain. The peptides were dissolved in dimethyl sulfoxide (DMSO) and activated in PBS-based solution by 13 M disuccinimidyl suberate (DSS; ThermoFisher Scientific, Waltham, MA, USA). The reaction was monitored by high-pressure liquid chromatography (HPLC) using a C18 column with gradient 0.1% TFA-water/0.1% TFA-acetonitrile solvent system. After 6 h, the solution was directly injected onto a C18 column. Pure fractions were pooled and lyophilized. CRM
197 (Fina Biosolutions, Rockville, MD, USA) solution was exchanged with conjugation buffer (0.1 M phosphate buffer, pH 6.8, 1.25% sucrose) using a 30K Amicon
® filter (Merck Millipore, Burlington, MA, USA). The activated peptide was dissolved in DMSO and added slowly to the CRM
197 with stirring over 15 min. The reaction was monitored by HPLC (Agilent 1260 Infinity Analytical system, Santa Clara, CA, USA) using size exclusion chromatography (SEC; Superose 12, GE Healthcare, Chicago, IL, USA) and PBS as eluent. After 16 h, the conjugate was washed five times by adding 8 mL of 0.1 M phosphate buffer, pH 6.8, 1.25% sucrose and spinning through a 30K Amicon
® filter. The resulting solution was filter sterilized using a 0.22 µm filter and characterized by size exclusion (SE)-HPLC, matrix-assisted laser desorption/ionization (MALDI) and bicinchoninic acid assay. The conjugate was stored at −80 °C until further use. Solvents were obtained from Sigma-Aldrich (Burlington, MA, USA), Caledon Laboratory Chemicals (Georgetown, ON, Canada) or Fisher Scientific (Pittsburgh, PA, USA) and used without further purification.
The RHDV2 VP60 proteins were generated by GenScript (Piscataway, NJ, USA) using a Baculovirus expression system. The construct encoding the following amino acid sequence included a TEV protease recognition site (underlined) to enable cleavage of the 6x Histidine tag following protein purification: MHHHHHHENLYFQGEGKARAAPQGETAGTATTASVPGTTTDGMDPGVVATTSVVTTENASTSIATAGIGGPPQQVDQQETWRTNFYYNDVFTWSVADAPGNILYTVQHSPQNNPFTAVLSQMYAGWAGGMQFRFIVAGSGVFGGRLVAAVIPPGIEIGPGLEVRQFPHVVIDARSLEPVTITMPDLRPNMYHPTGNPGLVPTLVLSVYNNLINPFGGSTSAIQVTVETRPSEDFEFVMIRAPSSKTVDSISPADLLTTPVLTGVGTDNRWNGEIVGLQPVPGGFSTCNRHWNLNGSTFGWSSPRFAAIDHDGGNASFPGSSSSNVLELWYASAGSATDNPISQIAPDGFPDMSFVPFSGSTIPTAGWVGFGGIWNSNNGAPFVTTMQAYELGFATGAPSNPQPTTTTSGAQIVAKSIYGVANGINQTTAGLFVMASGVISTPNSSAITYTPQPNRIVNAPGTPAAAPIGKNTPIMFASVVRRTGDINAEAGSTNGTQYGAGSQPLPVTVGLSLNNYSSALMPGQFFVWQLNFASGFMELGLSVDGYFYAGTGALATLIDLSELVDIRPVGPRPSTSTLVYNLGGTTNGFSYV. The nucleolin interaction domain is in bold.
Following removal of the tag, the purified protein was provided in a 50 mM Tris-HCl, 500 mM NaCl, 20% Glycerol, 1 mM DTT, pH 8.0 buffer. The aliquoted protein was stored at −80 °C until further use. Protein purity was confirmed to be >90% by SDS-PAGE. The absence of endotoxin contamination in all described antigens/adjuvants was confirmed using Endosafe cartridge-based Limulus amebocyte lysate tests (Charles River Laboratories, Charleston, SC, USA).
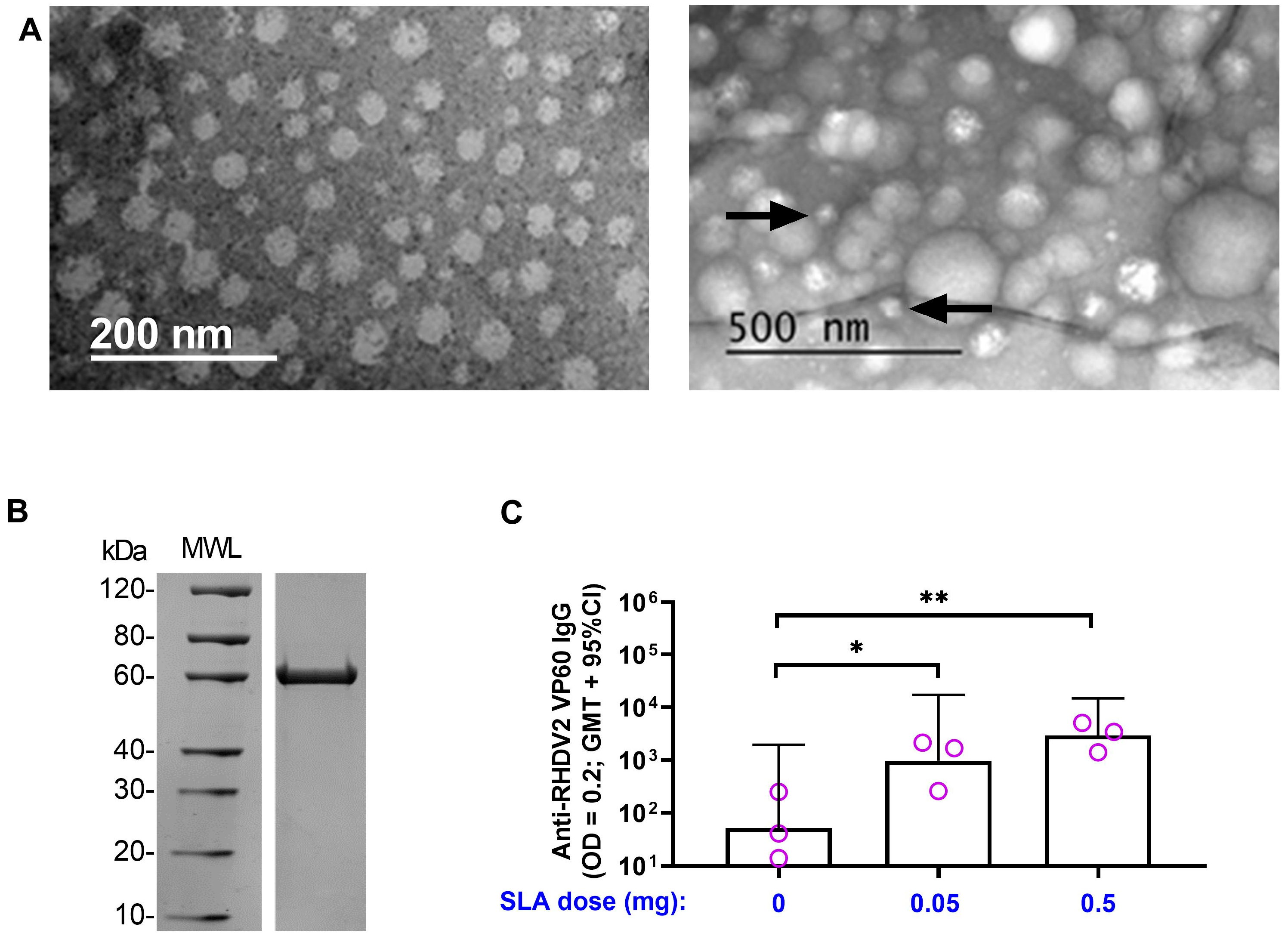
Transmission electron microscopy (TEM) was used to visualize the VLPs. TEM grids (formvar/carbon supported copper, 200 mesh, Sigma-Aldrich) were glow discharged prior to sample deposition. RHDV2 VP60 protein was applied to the surface of the grid and left for 30 s to allow for the sample to adsorb to the grid surface. Any extra sample was removed using filter paper, followed by washing two times with deionized water. Uranyl acetate (1%) was applied to the carbon surface of the grid, with excess solution removed using filter paper. Grids were left to air dry overnight before TEM imaging. Images were acquired on an FEI Titan3 80−300 TEM operated at 300 keV and equipped with a CEOS aberration corrector for the probe-forming lens and a monochromated field-emission gun.
2.2. Immunogenicity Studies in Mice and Rabbits
Female BALB/c mice and female New Zealand white rabbits were obtained from Charles River Laboratories (Saint-Constant, QC, Canada). Animals were housed at the animal facility of the National Research Council Canada (NRC) according to the guidelines of the Canadian Council on Animal Care. Antigen and preformed SLA archaeosomes were admixed and diluted in PBS on the day of immunization. Vaccine formulations were administered in a final volume of 50 or 500 µL per dose in mice or rabbits, respectively. In both species, the scruff of the neck was targeted for subcutaneous (s.c.) injections. Meanwhile, intramuscular injections (i.m.) were administered into the left cranial tibial muscle and left hind limb in mice and rabbits, respectively. Mice were bled via the submandibular vein for the collection of serum in serum separator tubes (Becton Dickinson, East Rutherford, NJ, USA), while blood in rabbits was collected in lithium-heparin-treated tubes (Becton Dickinson) via the central ear artery for the isolation of plasma and peripheral blood mononuclear cells (PBMCs) as described below. All rabbit procedures were conducted under slight sedation with acepromazine (Boehringer Ingelheim, Ingelheim, Germany), while mice were anesthetized with isoflurane (Baxter International, Deerfield, IL, USA) prior to immunizations. For the tolerability assessment, rabbits were monitored daily, with body weight and temperature recorded. For body temperature, transponders (Model IPTT-300, BioMedic Data Systems Inc., Seaford, DE, USA) were implanted subcutaneously prior to study start using a 12-gauge needle and the temperature was read in a noninvasive manner through the skin with a probe. In addition, local reactogenicity (i.e., erythema and edema) and clinical signs (i.e., lethargy, hunched posture, inappetence, body condition and fecal output) were monitored.
2.3. Antibody ELISA
Anti-RHDV2 VP60 IgG titers in serum or plasma were quantified by ELISA as previously described [
34]. Briefly, 100 µL of 1 µg/mL RHDV2 protein (His-tagged version of protein described above) diluted in PBS was added per well of a 96-well high-binding ELISA plate (Thermo Fisher Scientific). Plates were coated overnight at room temperature (RT) and then washed five times with PBS/0.05% Tween20 (PBS-T; Sigma-Aldrich, St. Louis, MO, USA). To block the plates, 200 µL of 10% fetal bovine serum (FBS; Thermo Fisher Scientific) in PBS was added to each well and then plates were incubated for 1 h at 37 °C. After another round of washing as above, serially diluted samples in PBS-T with 10% FBS were added in 100 µL volumes and incubated for 1 h at 37 °C. After five washes with PBS-T (Sigma-Aldrich), 100 µL of goat anti-mouse IgG-HRP (Southern Biotech, Birmingham, AL, USA) or goat anti-rabbit IgG-HRP (Southern Biotech) was added at optimal dilution for 1 h at 37 °C. After a final set of washes with PBS-T, 100 µL/well of the substrate o-phenylenediamine dihydrochloride (OPD, Sigma-Aldrich) diluted in 0.05 M citrate buffer (pH 5.0) was added and plates kept in the dark. After 30 min, the reaction was stopped with 50 µL/well of 4N sulfuric acid (H
2SO
4). Plates were read spectrophotometrically at 450 nm. Titers for IgG in serum were defined as the dilution that resulted in an absorbance value (OD 450) of 0.2 and were calculated using XLfit software (ID Business Solutions, Guildford, UK). Samples that did not reach the target OD were assigned the value of the lowest tested dilution (i.e., 10 or 100) for analysis purposes.
For the peptide-specific ELISAs, plates were coated overnight at 4 °C with 10 µg/mL neutravidin (Thermo Fisher Scientific) diluted in PBS. Plates were washed three times with PBS alone, and then incubated for 2 h at room temperature with 1 µg/mL of biotinylated-RHDV1 or RHDV2 peptides diluted in PBS. Plates were then washed five times with PBS-T, blocked with 10% FBS and then treated the same as for the RHDV2 protein ELISA above.
2.4. ELISpot
The levels of CRM
197- or RHDV2-specific T cells were quantified by ELISpot using a rabbit IFN-γ kit (Mabtech Inc., Cincinnati, OH, USA) as previously described [
35]. PBMCs were isolated from 5 mL of rabbit blood by density gradient centrifugation. Blood was passed through a 70 µm cell strainer and overlaid on Histopaque
®-1083 in 15 mL Accuspin™ tubes (Sigma-Aldrich) as per manufacturer’s recommendations. Blood was centrifuged for 30 min at 800×
g at room temperature with the brake off. The buffy coat was collected and washed three times with PBS prior to resuspension in RPMI media (Thermo Fisher Scientific) containing 10% FBS (Thermo Fisher Scientific), 1% penicillin/streptomycin (Thermo Fisher Scientific), 1% glutamine (Thermo Fisher Scientific) and 55 µM 2-Mercaptoethanol (Thermo Fisher Scientific). Once PBMC yields were determined on a Cellometer (Nexcelom, Lawrence, MA, USA), cells were diluted to the appropriate concentration with the supplemented RPMI media. Whole protein (His-tagged RHDV2 or CRM
197 as described above) was used to stimulate 4 × 10
5 cells in duplicate at a final concentration of 5 µg/mL. Final volume per well was 200 µL. Cells were also incubated without any stimulants to measure background responses. Plates were incubated for approximately 20 h at 37 °C with 5% CO
2, at which point the plates were washed and developed according to the manufacturer’s instructions. AEC substrate (Becton Dickinson) was used to visualize the spots. Spots were counted using an automated ELISpot plate reader (BioSys, Miami, FL, USA). For each animal, values obtained with media alone were subtracted from those obtained with the protein-stimulated cells to yield an overall number of antigen-specific IFN-γ
+ spot-forming cells (SFCs)/10
6 PBMCs per animal.
2.5. RHDV Challenge Studies in Rabbits
New Zealand white rabbits greater than 12 weeks of age were obtained and housed at the Elizabeth Macarthur Agricultural Institute. Animals were randomly assigned to 5 treatment groups consisting of 8 rabbits each, with 4 males and 4 females per group. They were confirmed to be free from detectable antibodies against RHDV1 and RHDV2. Animals were immunized s.c. at the scruff of the neck with vaccine formulations prepared as above. As a positive control, one group of animals received a commercial vaccine, Filavac
® VHD K C+V (Filavie, Sevremoine, France). On Days 14, 21 and 28 postimmunization, serum was collected from each animal to evaluate serological responses as determined by RHDV2-blocking ELISA [
36]. Following blood collection on Day 28, rabbits were challenged orally with 0.5 mL of live RHDV2 virus suspension (EMAI strain M215; diluted in PBS, with each dose containing 1000 ID
50 (infectious dose—50% end point)) [
37]. Rabbits were housed individually in cages following challenge and monitored daily for clinical signs (e.g., lethargy, inappetence and seizures). All surviving rabbits were euthanized on Day 42 (14 days post-challenge). Liver samples were collected from each rabbit at time of death for testing for RHDV2 viral load by qRT-PCR. Total nucleic acid was extracted with the MagMAX-96 RNA Isolation Kit (Thermo Fisher Scientific) on a KingFisher™ 96 (Thermo Fisher Scientific) magnetic particle handling system following the manufacturer’s protocol. Nucleic acid was eluted in a 50 μL volume. The RHDV2 qRT-PCR assay utilized the AgPath-IDTM One-Step RT-PCR kit (Thermo Fisher Scientific). A total of 5 μL of purified nucleic acid was added to a 20 μL reaction mix. Primers and the probe [
38] were added and samples run in an ABI 7500 Fast thermal cycler (Applied Biosystems, Waltham, MA, USA) according to the master mix manufacturer’s standard parameters. Results were expressed in terms of the cycle number at which the sample fluorescence plot crossed the 0.05 threshold (the Ct value). Collected sera were tested for antibodies directed towards RHDV2 by an anti-RHDV blocking assay similar to that previously described [
36,
37]. The ELISA plates were first coated with a polyclonal anti-RHDV2 rabbit antiserum as the capture antibody, followed by a semi-purified RHDV2 antigen (both anti-sera and antigen were produced at EMAI). The test serum was then added to the plate and, after an incubation period of 30 min, a peroxidase-conjugated RHDV2-specific monoclonal antibody (4H12, supplied by IZSLER, Brescia, Italy) was added to measure the level of blocking of the monoclonal antibody by the test serum. Results were calculated as a percentage inhibition (PI), with a cut-off point for a positive result set at >40%.
2.6. Statistical Analysis
GraphPad Prism® version 9 (GraphPad Software, San Diego, CA, USA) was used to analyze the data. Statistical significance of the difference between groups was calculated by: (1) for 2 groups, unpaired two-tailed Student’s t-test or (2) for 3 or more groups, one-way analysis of variance (ANOVA) followed by post hoc analysis using Tukey’s (comparison across all groups) multiple comparison test. Antibody and ELISpot data were log transformed prior to statistical analysis. Survival curve analyses were carried out using the Gehan–Breslow–Wilcoxon method. For all analyses, differences were considered to be not significant with p > 0.05. Significance was indicated in the graphs as follows: * p < 0.05, ** p < 0.01, *** p < 0.001 and **** p < 0.0001.
4. Discussion
SLA’s adjuvant effects were first established in rodent species such as mice. In this study, we have demonstrated the activity and utility of the SLA adjuvant system in a larger mammalian species, rabbits. SLA was able to augment antigen-specific antibodies and T cells in rabbits to two different types of antigens: a carrier-linked peptide vaccine and a recombinant protein VLP. In the case of the RHDV2 VLPs, SLA was able to induce functional responses that were efficacious in protecting rabbits from disease in a challenge model.
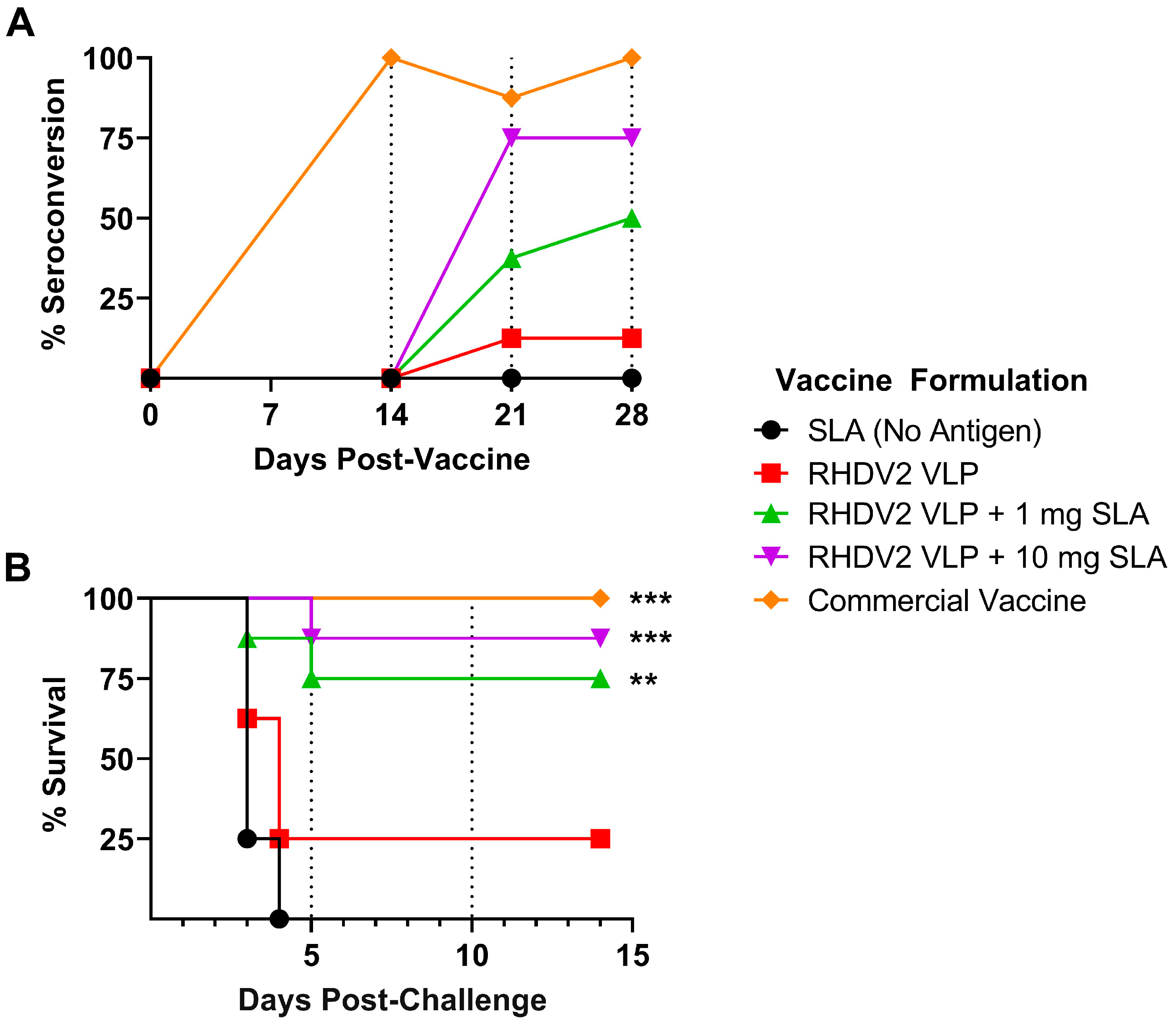
The use of recombinant VP60-based vaccines targeting RHDV1 or RHDV2 have been evaluated previously. In studies measuring responses after a single vaccine dose, the amount of purified antigen included in the formulations varied greatly, ranging from 500 to 5000 µg per dose [
23,
27,
40]. While most of these did not incorporate an adjuvant, one did include a water-in-oil-in-water adjuvant Montanide™ ISA 201 VG [
40]. One of the first studies to attempt this recombinant VP60-based vaccine approach formulated 30 µg of antigen with water-in-oil adjuvant Montanide™ ISA 50 but utilized a multiple-vaccination strategy (two vaccine doses, 14 days apart) to demonstrate survival from viral challenge with RHDV1 [
24]. In our study, we clearly demonstrated that inclusion of SLA in the vaccine formulation allowed for up to 88% survival following a single immunization with a vaccine containing only 30 µg of antigen. While we did not achieve 100% efficacy in our stringent challenge model, it is conceivable that this could be achieved with the use of slightly higher antigen doses or following a booster dose. This would be of interest to evaluate in future studies. The amount of antigen dose sparing discussed above would have important implications on the cost of the formulation. The cost of including the adjuvant would also need to be taken into account when designing the vaccine formulation. The SLA generated for this study was made through a semi-synthetic process where the sulfated lactose domain is chemically conjugated to biologically derived archaeol, which has been purified from the biomass of the aerobically cultured archaeal species
H. salinarum. There is also the option of manufacturing a purely synthetic SLA, generated chemically without any archaeal precursors, as it was shown to be equally active in vivo [
16]. We are currently optimizing and adapting the manufacturing process to generate SLA cost-effectively at a larger scale suitable for clinical applications. This work will better inform us on the ultimate cost of generating SLA for commercial applications.
In the challenge study, the efficacy of our SLA-adjuvanted RHDV2 VLP formulations was compared to a commercially available RHDV vaccine. The rates of survival induced by these formulations were statistically similar, but subtle differences in the kinetics and degree of seroconversion were observed. The commercial vaccine’s antigen component consisted of homogenized liver extract from RHDV-infected rabbits which had been treated to inactivate the virus. To enhance its immunogenicity, the liver extract was paired with an aluminum salt-based adjuvant. It should also be noted that the commercial vaccine contained antigens from both RHDV1 and RHDV2 genotypes. The amount of virus included in each dose is unclear. As the viral antigen is generated in rabbits, it is possible that it contains certain features (e.g., glycosylation pattern) that would more closely mimic the capsid protein found on the viral pathogen than the insect cell-derived antigen used in the SLA-adjuvanted formulations. Therefore, differences in antigen type, format and quantity, as well as the adjuvant used, could explain the minor differences seen between the commercial and experimental formulations tested above. In future studies, it would be informative to compare the physical characteristics and immunogenicity (when combined with SLA) of a mammalian vs. insect cell-produced RHDV VP60 antigen. In addition, it would be interesting to confirm the efficacy of SLA-adjuvanted bivalent formulations including a combination of RHDV1- and RHDV2-based VLPs against both viruses.
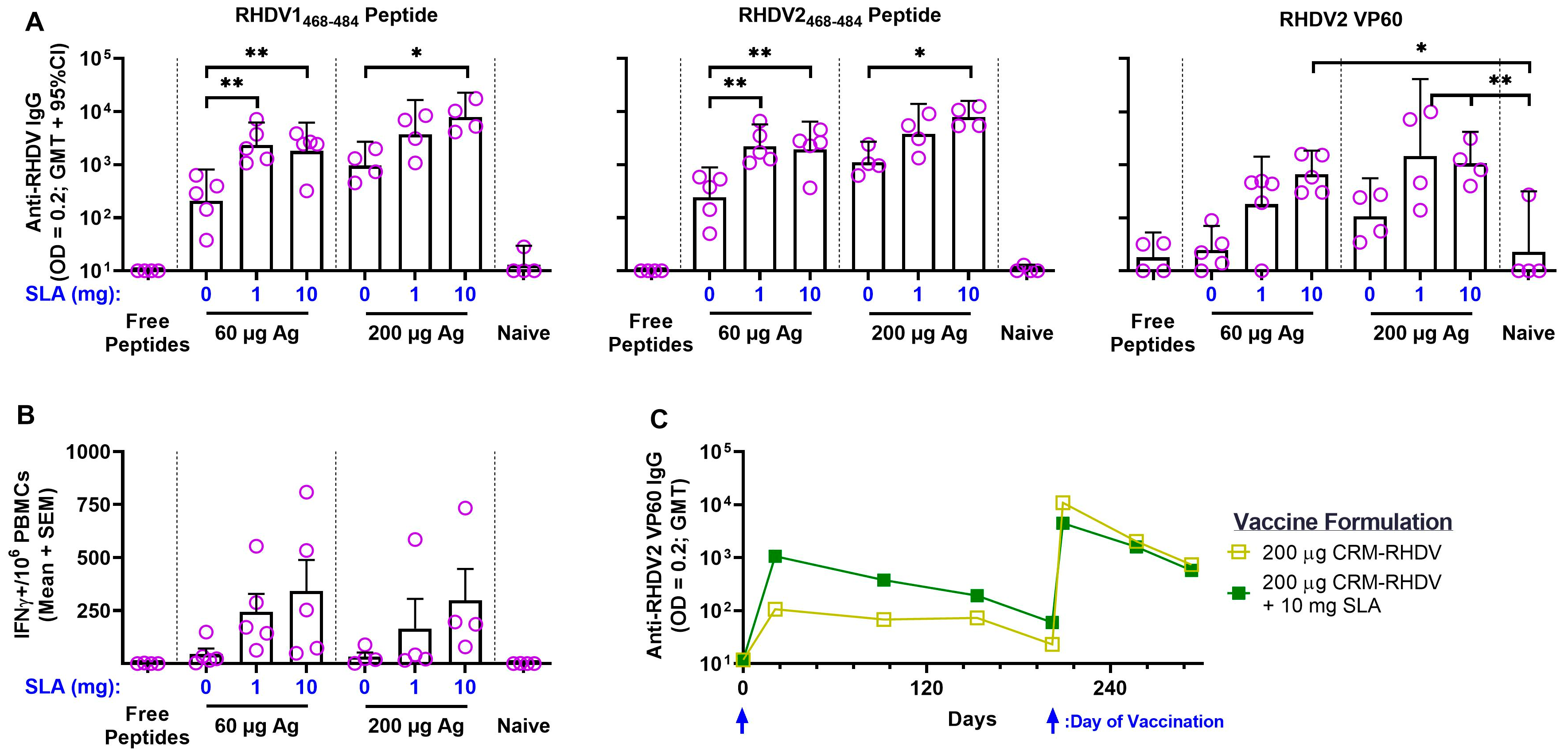
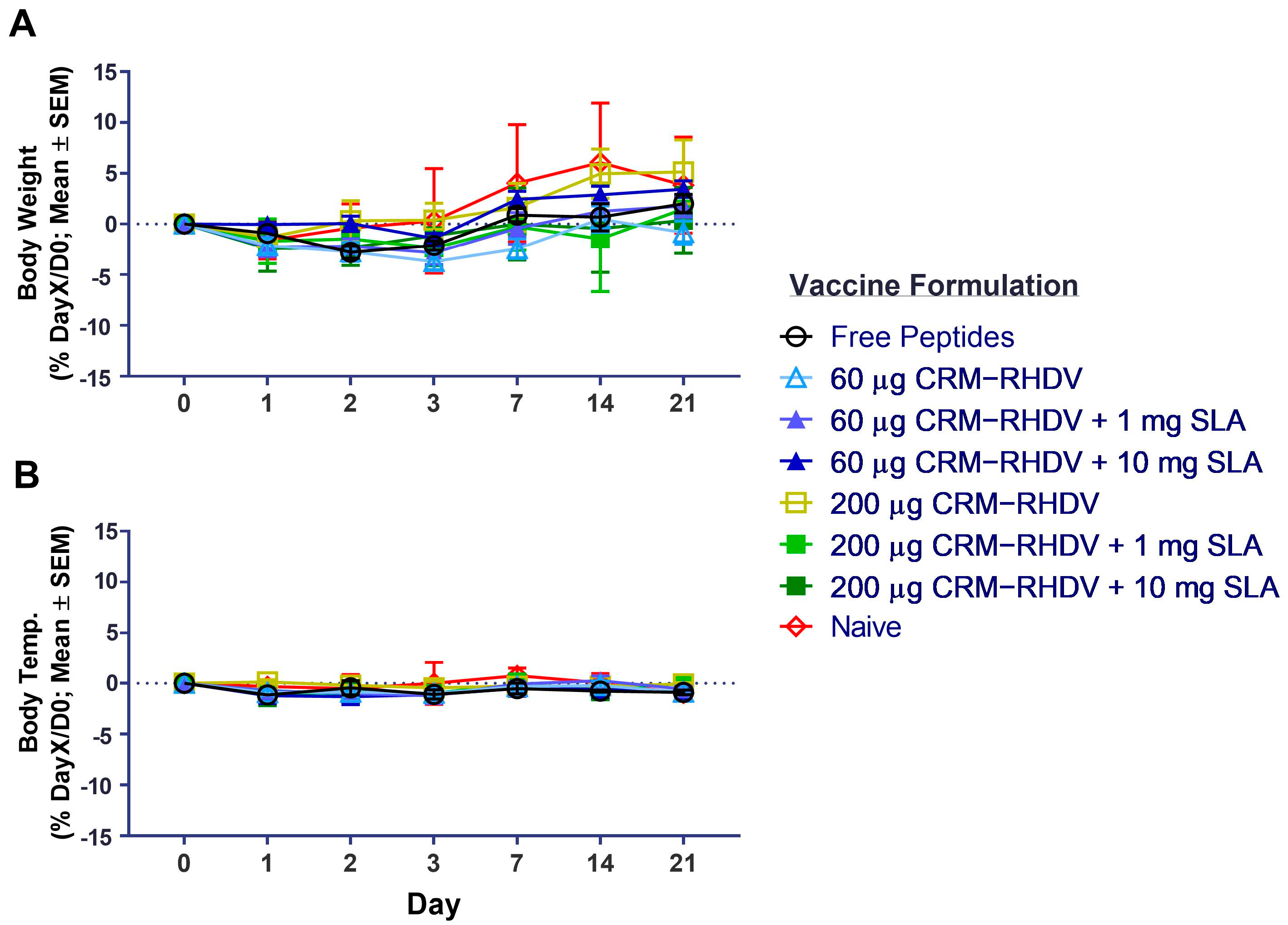
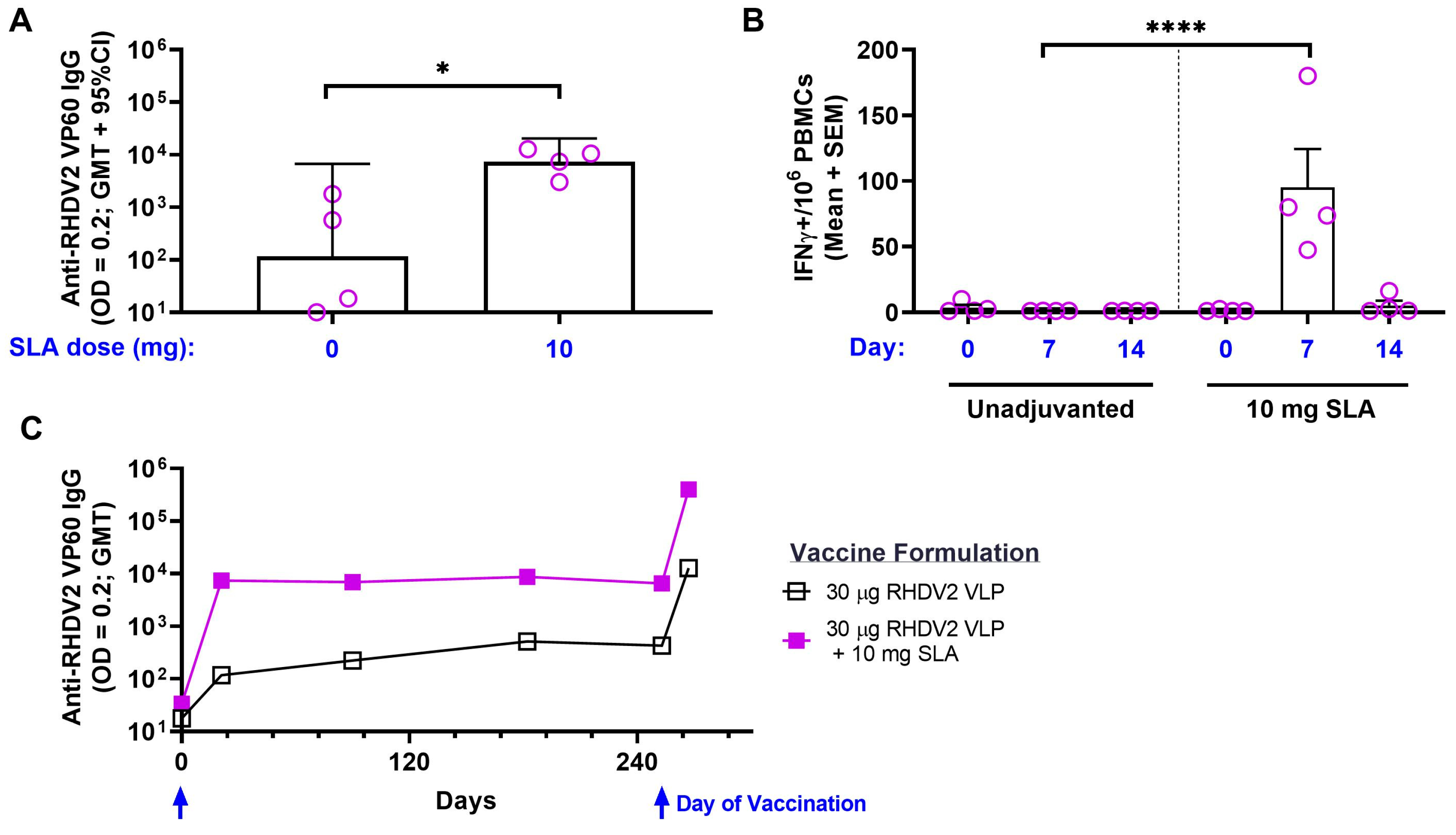
In this study, we were also able to confirm the high tolerability profile of SLA in another mammalian species. Previous studies in mice had shown that it was safe and well tolerated at doses of up to 10 mg [
14]. Studies conducted under Good Laboratory Practice (GLP) standards in rats have also shown SLA to have a favorable safety profile when administered alone at doses of up to 10 mg (McCluskie et al., unpublished data). As safety and tolerability of a vaccine formulation may also depend on the specific antigen, confirmatory studies will need to be repeated once the vaccine doses/formulations are established for any clinical formulation. Interestingly, we were able to utilize similar SLA dose levels of 1 to 10 mg in rabbits with average body weights >4 kg compared to mice weighing >100 times less. While responses tended to be slightly better with 10 vs. 1 mg in the rabbit challenge model, we found generally similar activity with both doses of adjuvant when coupled with the CRM
197–RHDV conjugates (
Figure 3). While further study is required, these results indicate that much higher doses of SLA may not be required when moving to even larger animal species or humans. This has been seen with other adjuvant types, such as emulsion or aluminum salt-based adjuvants, where adjuvant concentration may be more relevant than a dose based on the size/weight of the immunized subject [
41,
42]. We were also able to demonstrate that SLA retains its adjuvant activity when administered s.c. Almost all studies conducted to date with SLA have been administered i.m. to mimic the favored immunization route for parenteral vaccines in humans, but data above (
Figure 5,
Figure 6 and
Figure 7) suggest that s.c. administration could be a viable option with SLA-adjuvanted vaccines in the future. This could have important implications for multiple veterinary applications where s.c. is the preferred vaccination route.
SLA was also able to effectively enhance antibody responses to the RHDV peptides encompassing the nucleolin interaction domain. Zhu et al. [
28] have shown that immunization with unconjugated peptide did induce RHDV-specific antibody responses and some protection from RHDV1 challenge. We found that free peptides were weakly immunogenic with limited measurable induction of antigen-specific antibodies. This may be due to slight differences in antigen sequence or structure (presence of N- and C-terminal acetyl group and lysine, respectively) or the purity of the peptides. In our study, the conjugate vaccines did generate significantly higher levels of peptide-specific and RHDV2 VLP-reactive antibodies than the free peptides. In future studies, it would be interesting to evaluate if they would be protective against viral challenge, especially for RHDV2, which has not yet been shown to rely on nucleolin for cellular entry. However, the waning of responses over time in our rabbit study could indicate that some optimization of the RHDV conjugates may be required. It would be interesting to determine if altered peptide loading per CRM
197 molecule or the masking of terminal neo-epitopes on the linear peptides would enhance longevity of responses [
43]. An altered peptide loading profile on the carrier could potentially reduce the induction of anti-carrier antibodies, which have been previously shown to reduce the levels of responses following subsequent vaccinations with conjugate antigens using the same carrier [
44]. While CRM
197 IgG titers were not measured in our study, potential induction of higher levels of anti-CRM
197 antibodies with the SLA-adjuvanted formulation could have impacted the vaccine immunogenicity following the second immunization, leading to a similar level of responses with the unadjuvanted vs. SLA-adjuvanted RHDV–CRM conjugates following boost.
In summary, our data demonstrate the strong adjuvant activity of SLA in larger mammalian species such as rabbits and its potential utility in a recombinant protein-based vaccine targeting RHDV. Our formulation could serve as the basis for a novel safe RHDV vaccine that circumvents some of the safety and manufacturing concerns associated with formulations currently on the market. As such, we believe that SLA in general and these RHDV vaccine formulations in particular warrant further development as novel vaccination technologies.