A Quantitative Benefit–Risk Analysis of ChAdOx1 nCoV-19 Vaccine among People under 60 in Italy
Abstract
:1. Introduction
2. Methods
2.1. Parameters of the Simulation
2.2. Updated Values of the Parameters
2.3. Ethics Committee Approval
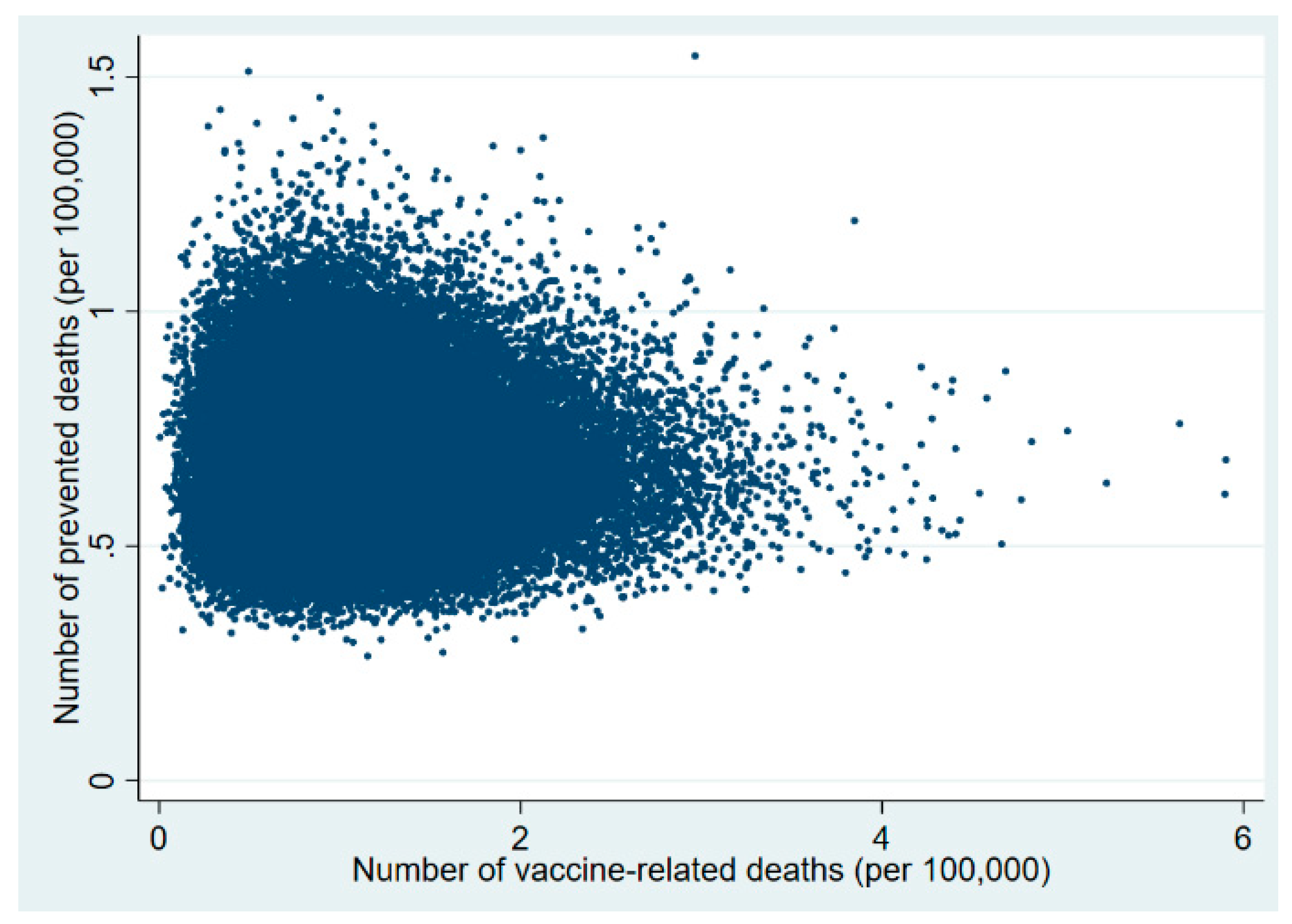
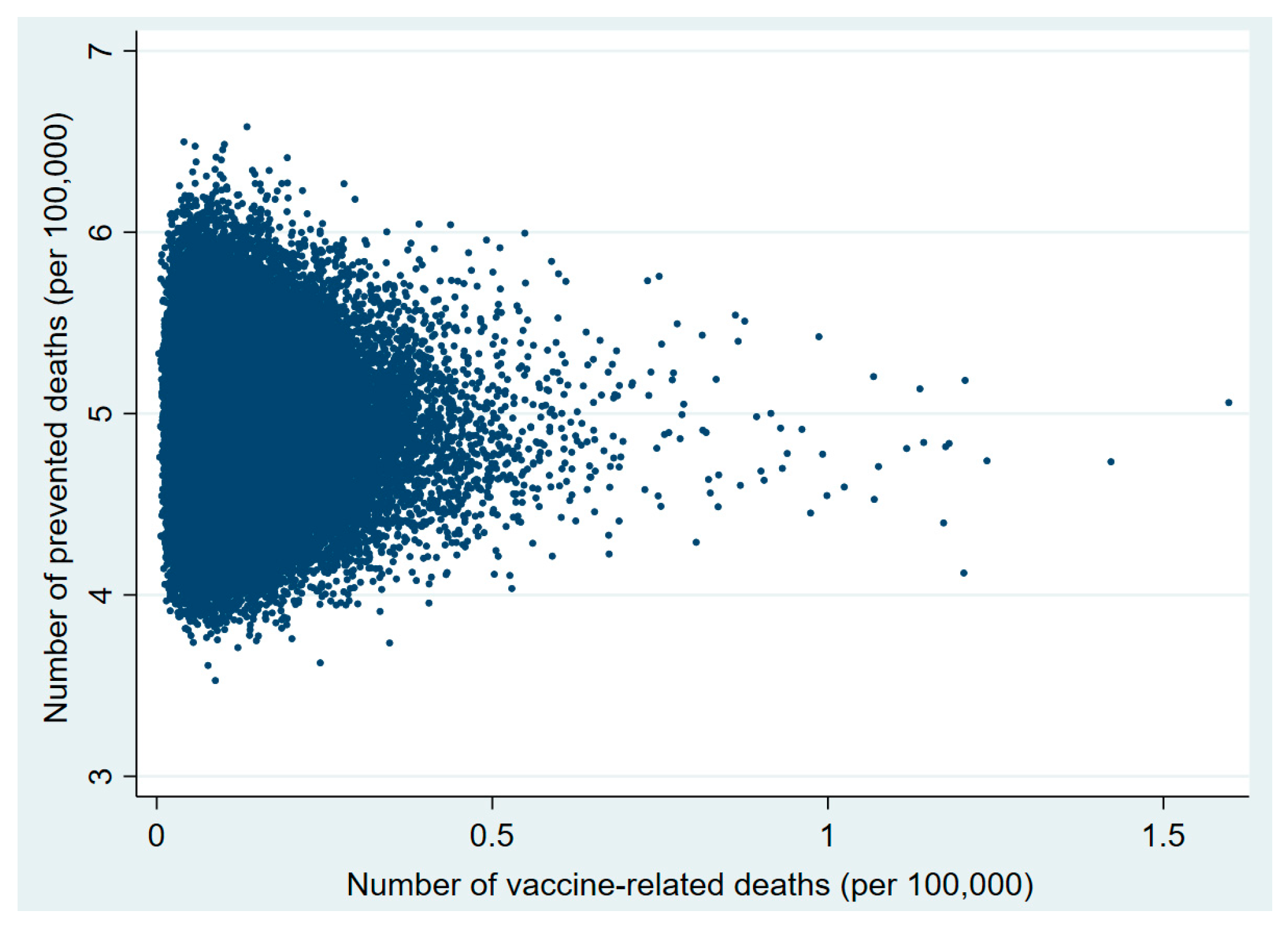
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Greinacher, A.; Thiele, T.; Warkentin, T.E.; Weisser, K.; Kyrle, P.A.; Eichinger, S. Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Emary, K.R.W.; Golubchik, T.; Aley, P.K.; Ariani, C.V.; Angus, B.; Bibi, S.; Blane, B.; Bonsall, D.; Cicconi, P.; Charlton, S.; et al. Efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine against SARS-CoV-2 variant of concern 202012/01 (B.1.1.7): An exploratory analysis of a randomised controlled trial. Lancet 2021, 397, 1351–1362. [Google Scholar] [CrossRef]
- Pharmacovigilance Risk Assessment Committee (PRAC): Signal Assessment Report on Embolic and Thrombotic Events (SMQ) with COVID-19 Vaccine (ChAdOx1-S [Recombinant])—COVID-19 Vaccine AstraZeneca (Other Viral Vaccines); European Medicines Agency: Amsterdam, The Netherlands, 2021.
- WHO Lists Two Additional COVID-19 Vaccines for Emergency Use and COVAX Roll-Out. Available online: https://www.who.int/news/item/15-02-2021-who-lists-two-additional-covid-19-vaccines-for-emergency-use-and-covax-roll-out (accessed on 20 May 2021).
- Recommendation for an Emergency Use of AZD12222 Submitted by AstraZeneca AB and Manufactured by SK Bioscience Co Ltd. Available online: https://extranet.who.int/pqweb/sites/default/files/documents/AZD1222_TAG_REPORT_EUL%20vaccine_FEB2021_v2.pdf (accessed on 20 May 2021).
- Wise, J. COVID-19: European countries suspend use of Oxford-AstraZeneca vaccine after reports of blood clots. BMJ 2021, 372, n699. [Google Scholar] [CrossRef] [PubMed]
- European Medicine Agency: Committee for Medicinal Products for Human Use (CHMP); Assessment report EMA/239822/2021; European Medicines Agency: Amsterdam, The Netherlands, 2021.
- Mishra, H.; Singh, J.; Karmakar, S.; Kumar, R. An integrated approach for modeling uncertainty in human health risk assessment. Environ. Sci. Pollut. Res. Int. 2021. [Google Scholar] [CrossRef] [PubMed]
- Arlegui, H.; Bollaerts, K.; Salvo, F.; Bauchau, V.; Nachbaur, G.; Begaud, B.; Praet, N. Benefit-Risk Assessment of Vaccines. Part I: A Systematic Review to Identify and Describe Studies About Quantitative Benefit-Risk Models Applied to Vaccines. Drug Saf. 2020, 43, 1089–1104. [Google Scholar] [CrossRef] [PubMed]
- COVID-19 Integrated Surveillance Bulletin. Available online: https://www.epicentro.iss.it/coronavirus/bollettino/Bollettino-sorveglianza-integrata-COVID-19_25-novembre-2020.pdf (accessed on 25 November 2020).
- Istituto Superiore di Sanitá: COVID-19 Integrated Surveillance Bulletin (14-4-2020). Available online: https://www.epicentro.iss.it/en/coronavirus/sars-cov-2-integrated-surveillance-data (accessed on 20 May 2021).
- ACCESS Background Incidence Rates of Embolic and Thrombotic Events Following Immunisation with COVID-19 Vaccines. Available online: http://www.encepp.eu/phact_links.shtml (accessed on 20 May 2021).
- European Database of Suspected Adverse Drug Reactions. Available online: http://www.adrreports.eu/en/index.html (accessed on 20 May 2021).
- Doria-Rose, N.; Suthar, M.S.; Makowski, M.; O’Connell, S.; McDermott, A.B.; Flach, B.; Ledgerwood, J.E.; Mascola, J.R.; Graham, B.S.; Lin, B.C.; et al. Antibody Persistence through 6 Months after the Second Dose of mRNA-1273 Vaccine for Covid-19. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Banca dati ISTAT. Available online: http://demo.istat.it/popres/index.php?anno=2020&lingua=ita (accessed on 20 May 2021).
- Data on COVID-19 Vaccination in the EU/EEA. Available online: https://www.ecdc.europa.eu/en/publications-data/data-covid-19-vaccination-eu-eea (accessed on 20 May 2021).
- Vaccini, il dilemma AstraZeneca. Figliuolo in Pressing Perché Venga Usato Anche sui Cinquantenni. Available online: https://www.repubblica.it/cronaca/2021/05/11/news/il_dilemma_astrazeneca_figliuolo_pressa_perche_venga_suggerito_anche_ai_cinquantenni_gli_esperti_non_decidono-300443140/ (accessed on 20 May 2021).
- Loomba, S.; de Figueiredo, A.; Piatek, S.J.; de Graaf, K.; Larson, H.J. Measuring the impact of COVID-19 vaccine misinformation on vaccination intent in the UK and USA. Nat. Hum. Behav. 2021, 5, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Palladino, R.; Bollon, J.; Ragazzoni, L.; Barone-Adesi, F. Excess Deaths and Hospital Admissions for COVID-19 Due to a Late Implementation of the Lockdown in Italy. Int. J. Environ. Res. Public Health 2020, 17, 5644. [Google Scholar] [CrossRef] [PubMed]
- Caristia, S.; Ferranti, M.; Skrami, E.; Raffetti, E.; Pierannunzio, D.; Palladino, R.; Carle, F.; Saracci, R.; Badaloni, C.; Barone-Adesi, F.; et al. Effect of national and local lockdowns on the control of COVID-19 pandemic: A rapid review. Epidemiol. Prev. 2021, 44, 60–68. [Google Scholar]
- Palladino, R.; Bollon, J.; Ragazzoni, L.; Barone-Adesi, F. Effect of Implementation of the Lockdown on the Number of COVID-19 Deaths in Four European Countries. Dis. Med. Public Heal. Prep. 2020, 2020, 1–3. [Google Scholar] [CrossRef] [PubMed]
- COVID-19 Projections. Available online: https://covid19.healthdata.org/global?view=cumulative-deaths&tab=trend (accessed on 20 May 2021).
- Coronavirus (COVID-19) Vaccinations. Available online: https://ourworldindata.org/coronavirus (accessed on 20 May 2021).
- Vasileiou, E.; Simpson, C.R.; Shi, T.; Kerr, S.; Agrawal, U.; Akbari, A.; Bedston, S.; Beggs, J.; Bradley, D.; Chuter, A.; et al. Interim findings from first-dose mass COVID-19 vaccination roll-out and COVID-19 hospital admissions in Scotland: A national prospective cohort study. Lancet 2021, 397, 1646–1657. [Google Scholar] [CrossRef]




| Original Estimates | Updated Estimates | |||
|---|---|---|---|---|
| Parameter | Estimate (95%CI) | Ref. | Estimate (95%CI) | Ref. |
| Vaccine Efficacy | 72.30% (63.10–79.30%) | [3] | ||
| COVID-19 death rate (20–29) | 1.37 (0.94–1.94) | [11,12] | ||
| COVID-19 death rate (30–49) | 10.20 (9.40–11.10) | [11,12] | ||
| COVID-19 death rate (50–59) | 55.70 (53.20–58.10) | [11,12] | ||
| CVST incidence rate (20–29) | 0.64 (0.29–1.42) | [4] | ||
| CVST incidence rate (30–49) | 1.81 (1.29–2.32) | [4] | ||
| CVST incidence rate (50–59) | 1.00 (0.62–1.61) | [4] | ||
| DIC incidence rate (20–29) | 0.60 (0.34–1.06) | [4] | ||
| DIC incidence rate (30–49) | 1.08 (0.26–1.90) | [4] | ||
| DIC incidence rate (50–59) | 3.07 (2.50–3.77) | [4] | ||
| TEE incidence rate (20–29) | 40.14 (33.75–47.46) | [4] | ||
| TEE incidence rate (30–49) | 85.08 (79.40–91.12) | [4] | ||
| TEE incidence rate (50–59) | 200.73 (189.54–212.56) | [4] | ||
| RR CVST in vaccine users (20–29) | 21.80 (0.28–121.30) | [4] | 44.92 (18.05–92.55) | [4,13,14] |
| RR CVST in vaccine users (30–49) | 3.67 (1.90–6.40) | [4] | 7.98 (5.56–11.11) | [4,13,14] |
| RR CVST in vaccine users (50–59) | 1.40 (0.20–5.10) | [4] | 8.33 (4.31–14.56) | [4,13,14] |
| RR DIC in vaccine users (20–29) | 23.30 (0.30–129.40) | [4] | 6.84 (0.17–38.1) | [4,13,14] |
| RR DIC in vaccine users (30–49) | 2.02 (0.54–5.16) | [4] | 4.14 (2.07–7.42) | [4,13,14] |
| RR DIC in vaccine users (50–59) | 0.23 (0.01–1.27) | [4] | 0.68 (0.14–1.98) | [4,13,14] |
| RR TEE in vaccine users (20–29) | 3.82 (1.91–6.84) | [4] | ||
| RR TEE in vaccine users (30–49) | 1.30 (1.03–1.62) | [4] | ||
| RR TEE in vaccine users (50–59) | 0.41 (0.29–0.56) | [4] | ||
| DIC fatality rate | 0.57 (0.18–0.90) | [4] | 0.53 (0.26–0.79) | [4,13,14] |
| CVST fatality rate | 0.33 (0.13–0.59) | [4] | 0.28 (0.16–0.42) | [4,13,14] |
| TEE fatality rate | 0.22 (0.17–0.29) | [4] | ||
| COVID-19 Deaths Prevented per Excess Death for Embolic and Thrombotic Events | COVID-19 Deaths Prevented per Excess Death for Disseminated Intravascular Coagulation and Cerebral Venous Sinus Thrombosis | ||||
|---|---|---|---|---|---|
| Based on Original Estimates | Based on Original Estimates | Based on Updated Estimates | |||
| Age | B-R Ratio (95% UI) | Age | B-R Ratio (95% UI) | Age | B-R Ratio (95% UI) |
| 20–29 | 0.70 (0.27–2.11) | 20–29 | 0.93 (0.06–14.3) | 18–24 | 1.48 (0.33–5.54) |
| 30–49 | 22.9 (10.1–186.4) | 30–49 | 52.2 (16.6–179.1) | 25–49 | 22.7 (10.9–44.7) |
| 50–59 | 1577.1 (1176.9–2121.5) | 50–59 | 3506.7 (166.0–54,427.9) | 50–59 | 305 (109.7–851.9) |
| 30% under-Reporting | 50% under-Reporting | 80% under-Reporting | |
|---|---|---|---|
| Age | B-R Ratio (95% UI) | B-R Ratio (95% UI) | B-R Ratio (95% UI) |
| 18–24 | 0.49 (0.19–1.46) | 0.34 (0.13–1.04) | 0.14 (0.05–0.42) |
| 25–49 | 16.1 (7.10–131.6) | 11.4 (5.03–98.1) | 4.59 (2.03–36.5) |
| 50–59 | 1104.4 (823.2–1483.4) | 788.9 (588.7–1058.7) | 315.6 (235.7–422.6) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palladino, R.; Ceriotti, D.; De Ambrosi, D.; De Vito, M.; Farsoni, M.; Seminara, G.; Barone-Adesi, F. A Quantitative Benefit–Risk Analysis of ChAdOx1 nCoV-19 Vaccine among People under 60 in Italy. Vaccines 2021, 9, 618. https://doi.org/10.3390/vaccines9060618
Palladino R, Ceriotti D, De Ambrosi D, De Vito M, Farsoni M, Seminara G, Barone-Adesi F. A Quantitative Benefit–Risk Analysis of ChAdOx1 nCoV-19 Vaccine among People under 60 in Italy. Vaccines. 2021; 9(6):618. https://doi.org/10.3390/vaccines9060618
Chicago/Turabian StylePalladino, Raffaele, Daniele Ceriotti, Damiano De Ambrosi, Marta De Vito, Marco Farsoni, Giuseppina Seminara, and Francesco Barone-Adesi. 2021. "A Quantitative Benefit–Risk Analysis of ChAdOx1 nCoV-19 Vaccine among People under 60 in Italy" Vaccines 9, no. 6: 618. https://doi.org/10.3390/vaccines9060618






