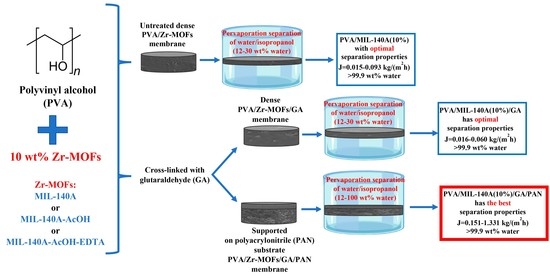
3.1.1. Pervaporation Performance of the Uncross-Linked PVA and PVA/Zr-MOFs Membranes
To optimize the concentration of Zr-MOFs in the polymer matrix, up to 15 wt% of unmodified Zr-MOF (MIL-140A) was introduced into the PVA matrix. The performance of the developed uncross-linked PVA and PVA/MIL-140A membranes were studied in pervaporation separation of water/isopropanol mixtures (12, 20, and 30 wt% water). The dependence of the permeation flux on the water content in the feed for the dehydration of isopropanol is shown in
Figure 1.
The data presented in
Figure 1 demonstrate that the permeation flux for the freestanding PVA and PVA/MIL-140A membranes increased with the rise of water content in the feed. This is related to the higher water concentration in the feed, resulting in increased swelling of the PVA-based membranes in the separated mixture (confirmed by the swelling data presented below). The introduction of MIL-140A (5–15 wt%) into the PVA matrix increased the permeation flux compared to the unmodified membrane that can be related to the formation of interfacial defects or gaps, the change of morphology and hydrophilic–hydrophobic surface balance which facilitated the penetrants diffusion [
68]. The introduction of 5 wt% MIL-140A did not suffice to change significantly the performance of the PVA membrane, due to insignificant structural changes (SEM data presented below), surface roughness (AFM data presented below) and swelling degree (proven by swelling degree data presented below) during the modification. Further, 10 wt% of MIL-140A was shown to be the optimal concentration in the PVA matrix to get enhanced pervaporation performance of the PVA membrane, since PVA+MIL-140A(10%) membrane had the highest values of permeation flux due to morphology changes (SEM data presented below), increased surface roughness (AFM data presented below), and the highest swelling degree in water/isopropanol mixture among PVA and PVA+MIL-140A(5 and 15%) membranes (proven by swelling degree data presented below). The membrane modified with 10 wt% MIL-140A had ca. 1.6 times higher permeation flux than for the unmodified PVA membrane in pervaporation dehydration of isopropanol (30 wt% water). The increase in MIL-140A concentration to 15 wt% in the PVA matrix led to the formation of Zr-MOF agglomerates in the membrane (proven by SEM and AFM data presented below), hindering the penetration of components and decreasing the permeation flux. It should be noted that all membranes showed high selectivity with respect to water (99.9 wt% water in permeate).
The addition of MOF without functional groups is known to possibly cause high permeability and low selectivity, due to the existence of interfacial defects. However, the functionalization of MOFs can decrease or exclude interface defects [
69]. Thus, the effect of the introduction of functionalized MOFs (modified Zr-MOFs (MIL-140A-AcOH and MIL-140A-AcOH-EDTA)) was investigated. Based on the data presented in
Figure 1, showing that 10 wt% MIL-140A is the optimal concentration, the same concentration of modified Zr-MOFs was added to compare MOFs’ properties to non-functionalized MIL-140A. The dependence of the permeation flux on the water content in the feed in pervaporation dehydration of isopropanol (12, 20, and 30 wt% water) is shown in
Figure 2 for the developed uncross-linked PVA/MIL-140A-AcOH(10%) and PVA/MIL-140A-AcOH-EDTA(10%) membranes.
Figure 2 also shows the permeation fluxes of the PVA and PVA/MIL-140A(10%) membranes for comparison.
The introduction of modified Zr-MOFs (10 wt%) into the PVA matrix increased the permeation flux, maintaining a high content of water in the permeate (99.9 wt%), compared to the pristine PVA membrane. The increased permeation flux of Zr-MOFs modified membranes was related to the change of membrane morphology, an increase in the swelling degree in water/isopropanol mixtures (12 and 30 wt% water), and surface roughness during the modification by the modified Zr-MOFs. The lowest values of permeation flux for all membranes were observed when separating an azeotropic water/isopropanol mixture, containing 12 wt% water, since the lowest values of the swelling degree were noted in the azeotropic mixture (proven by swelling degree data presented below). The membrane modified with MIL-140A (10 wt%) had the highest values of permeation flux compared to the pristine PVA and other modified PVA/Zr-MOFs(10%) membranes. The enhancement of the permeation flux for the PVA+MIL-140A(10%) membrane may be explained by the highest swelling in the separated mixture (confirmed by swelling data presented below) and surface roughness (confirmed by AFM data presented below) compared to the uncross-linked PVA and PVA/Zr-MOFs (PVA+MIL-140A-AcOH(10%) and PVA+MIL-140A-AcOH-EDTA(10%)) membranes. The introduction of AcOH and EDTA functional groups into the MOF structure increased pore size and specific surface area on the one hand (
Figure S3 in
Supplementary Materials) and altered particle shape and size on the other hand (
Figure S4 in
Supplementary Materials). Particle shape and size changes reduced membrane surface roughness (proven by AFM data presented below). Also, the modification with additional functional groups increased pore size in the particles, which could lead to the flow of the polymer into the pores of the MOF [
70] and eliminate interfacial defects causing the decrease of the permeation flux. An increase in the specific surface area usually decreases the permeation flux [
71,
72,
73,
74]. Thus, the PVA+MIL-140A(10%) membrane was selected as optimal among the studied uncross-linked membranes.
3.1.3. Structure and Physicochemical Properties of the Freestanding PVA and PVA/Zr-MOFs Membranes
Fourier-transform infrared spectroscopy (FTIR) was used to study structural changes of the uncross-linked and cross-linked PVA and PVA/Zr-MOFs(10%) membranes (
Figure 4).
The FTIR spectrum shown in
Figure 4a for the PVA membrane shows a broad band at 3284 cm
−1 and a peak with a maximum at 2934 cm
−1, which correspond to vibrations of O-H and C-H bonds, respectively [
75]. The absorption bands with maxima at 1327 and 1086 cm
−1 correspond to vibrations associated with -C-O-H- group [
75]. After the introduction of Zr-MOFs into the PVA matrix, FTIR spectra changed slightly. A shift of the peak at 3284 cm
−1 for the PVA membrane to 3293 cm
−1, 3301 cm
−1, and 3290 cm
−1 for the PVA+MIL-140A(10%), PVA+MIL-140A-AcOH(10%), and PVA+MIL-140A-AcOH-EDTA(10%) membranes, respectively, was noted. This shift may be attributed to the formation of hydrogen bonds between the -OH groups of PVA and the -OH and/or -COOH groups of Zr-MOFs [
45].
The cross-linking of the PVA membrane with GA slightly changed the spectrum for the PVA/GA membrane (
Figure 4b). There is a shift of the peak from 1418 cm
−1 to 1431 cm
−1, related to the deformation vibrations of the –OH groups; the decrease of band intensity in the region 3200–3350 cm
−1 and the appearance of an intensive peak in the region 1650–1720 cm
−1, referring to carbonyl groups [
76]. The cross-linking of PVA chains with GA occurs with the formation of acetyl groups, according to the mechanism described in the study [
76]. For the cross-linked PVA/Zr-MOFs/GA, the following changes in the FTIR spectra were noted: an increase in the intensity of the peak at 1690 cm
−1 for the PVA/MIL-140A membrane, 1711 cm
−1 for the PVA/MIL-140A-AcOH membrane and 1691 cm
−1 for the PVA/MIL-140A-AcOH-EDTA membrane. These absorption bands may correspond to the stretching vibrations of C=O groups.
The inner structures of the uncross-linked and cross-linked PVA and PVA/Zr-MOFs membranes were studied by scanning electron microscopy (SEM). The cross-sectional SEM micrographs for the uncross-linked and cross-linked PVA and PVA/Zr-MOFs membranes are presented in
Figure 5;
Figure 6, respectively.
The presented SEM micrographs demonstrate that the uncross-linked PVA membrane had a rather smooth and uniform cross-sectional structure (
Figure 5a). The introduction of 5 wt% MIL-140A (
Figure 5b) slightly increased the roughness of the cross-section. The introduction of 10 and 15 wt% MIL-140A (
Figure 5c,d) into the PVA matrix visualize MOF particles on the membrane cross-sections, the number of which increases with the rise of MIL-140A content. The introduction of 10 wt% MIL-140A-AcOH (
Figure 5e) and MIL-140A-AcOH-EDTA (
Figure 5f) into the PVA matrix also altered the cross-sectional structure of the membranes. These included: the appearance of plastic deformations, which are cross-sectional irregularities caused by immersion membrane in liquid nitrogen with subsequent cleavage and enhanced by embedded particles, and the visibility of particles Zr-MOFs, in particular MIL-140A-AcOH. The cross-sections of the modified membranes also differed depending on the introduced Zr-MOFs, due to the different shapes and structures (
Figure S4 in
Supplementary Materials) [
69].
The cross-linking with GA created roughness on the cross-sectional structure. For cross-linked modified PVA/Zr-MOFs/GA membranes, Zr-MOFs particles are also visible on the membrane’s cross-sections. The changes in the inner morphology during the GA cross-linking and Zr-MOFs modification of the PVA were reflected significantly in the permeation fluxes of the developed membranes.
The surface roughness of the uncross-linked and cross-linked PVA and PVA/Zr-MOFs membranes was studied by atomic force microscopy (AFM). AFM images with a scan size of 10 × 10 μm are presented in
Figure 7;
Figure 8 for the uncross-linked and cross-linked PVA and PVA/Zr-MOFs membranes, respectively.
The surface roughness characteristics (average, Ra, and root-mean-squared roughness, Rq) of the uncross-linked PVA and PVA/Zr-MOFs membranes were calculated based on the AFM images (
Figure 7) and are presented in
Table 1.
The data presented in
Table 1 demonstrate that the introduction of Zr-MOFs into the PVA matrix increased surface average and root-mean-squared roughness. The increase of MIL-140A content in the PVA matrix from 5 to 15 wt% led to the rise of surface roughness characteristics. The PVA+MIL-140A(15%) membrane had the highest values of surface roughness due to the formation of particles agglomerates (confirmed by SEM data,
Figure 5d). In comparison to the pristine PVA, PVA+MIL-140A-AcOH(10%) and PVA+MIL-140A-AcOH-EDTA(10%) membranes, the PVA+MIL-140A(10%) membrane had the highest surface roughness (Ra of 33.76 nm, Rq of 36.61 nm), which affects the facilitated sorption of feed components on the membrane surface (swelling degree data presented below), resulting to the highest permeation flux (
Figure 2) among the uncross-linked PVA and PVA/Zr-MOFs(10%) membranes. The highest roughness values of this membrane among uncross-linked PVA, PVA+MIL-140A-AcOH(10%), and PVA+MIL-140A-AcOH-EDTA(10%) membranes can be associated with the MIL-140A shape, which is a narrow cylinder (
Figure S4 in
Supplementary Materials), and the smallest pore size [
69].
Surface average (Ra) and root-mean-squared (Rq) roughness of the cross-linked PVA and PVA/Zr-MOFs(10%) membranes are presented in
Table 2.
The cross-linking of PVA-based membranes with GA results in a reduction in values of surface average and root-mean-squared roughness compared to the uncross-linked membranes (
Table 2). The dependence trend of the surface roughness of the cross-linked membranes is noted as in the case of the uncross-linked membranes (
Table 1), which is in agreement with the obtained permeation fluxes (
Figure 3). The cross-linked membrane modified with MIL-140A (PVA+MIL-140A(10%)/GA membrane) had the highest surface roughness values due to the particle pore size, as well as its shape, resulting in the maximum values of permeation flux among the cross-linked membranes.
The thermal stability of the uncross-linked and cross-linked PVA and PVA/Zr-MOFs(10%) membranes was investigated by thermogravimetric analysis (TGA). The obtained thermograms are presented in
Figure 9.
Figure 9a shows three stages of weight loss for the uncross-linked PVA and PVA/Zr-MOFs(10%) membranes at the following temperature ranges: (1) 30–170 °C; (2) 170–398 °C; (3) >398 °C for the PVA membrane; (1) 30–178 °C; (2) 178–360 °C; (3) >360 °C for the PVA+MIL-140A(10%) membrane; (1) 30–159 °C; (2) 159–359 °C; (3) >359 °C for the PVA+MIL-140A-AcOH(10%) membrane; and (1) 30–168 °C; (2) 168–383 °C; (3) >383 °C for the PVA+MIL-140A-AcOH-EDTA(10%) membrane. The first stage is associated with the evaporation of water, which is present in the membranes, due to the absorption of atmospheric moisture. The weight loss for this step is approximately the same for all samples at 2.2–2.5%. The second step of weight loss is different for all samples: 78.5% at 398 °C for the PVA membrane, 61.5% at 360 °C for the PVA+MIL-140A(10%) membrane, 63.5% at 359 °C for the PVA+MIL-140A-AcOH(10%) membrane, 54.4% at 383 °C for the PVA+MIL-140A-AcOH-EDTA(10%) membrane. This step is attributed to the removal of hydroxyl groups attached to the polymer backbone. The last step of weight loss of samples refers to the decomposition of the membrane material. Wherein, the introduction of Zr-MOFs in the PVA matrix increased the thermal stability of membranes, the PVA+MIL-140A(10%) membrane has the highest thermal stability. The weight loss for the PVA membrane was 94.3%, while for the PVA+MIL140A(10%) membrane—80.5% at 550 °C.
There were four stages of weight loss for the cross-linked PVA/GA and PVA/Zr-MOFs(10%)/GA membranes at the following temperature ranges (
Figure 9b): (1) 30–175°C; (2) 175–314 °C; (3) 314–513 °C; (4) >513 °C for the PVA/GA membrane; (1) 30–167 °C; (2) 167–279 °C; (3) 279–380°C; (4) >380 °C for the PVA+MIL-140A(10%)/GA membrane; (1) 30–170 °C; (2) 170–296 °C; (3) 296–379 °C; (4) >379 °C for the PVA+MIL-140A-AcOH(10%)/GA membrane; and (1) 30–164 °C; (2) 164–290 °C; (3) 290–398 °C; (4) >398 °C for the PVA+MIL-140A-AcOH-EDTA(10%)/GA membrane. The first area, as in the case of the uncross-linked membranes, was associated with the evaporation of water. The weight loss for this step was approximately the same for all samples being 2.5–4.4%. The following three steps differed significantly for the cross-linked PVA/GA membrane and cross-linked PVA/Zr-MOFs(10%)/GA membranes. The second step of weight loss was 26.6% at 314 °C for the unmodified PVA/GA membrane, 20.3–22.7% at 279–296 °C for PVA/Zr-MOFs(10%)/GA membranes and was attributed to the degradation of functional groups (for example, hydroxyl) attached to the polymer backbone. For the unmodified PVA/GA membrane, the weight loss was 94.1% at 513 °C, for PVA/Zr-MOFs(10%)/GA membranes the weight loss was 49.5–50.4% at the third step. The third step may correspond to the thermal decomposition of cross-linked PVA chains [
77]. The final weight loss is related to the decomposition of the polymer’s main backbones and the decomposition of the PVA [
78]. The PVA+MIL-140A(10%)/GA membrane had the highest thermal stability among the cross-linked membranes. The weight loss for the PVA/GA membrane was 94.3%, while for the PVA+MIL140A(10%)/GA membrane was 80.0% at 550 °C.
The swelling of freestanding membranes was studied in water/isopropanol mixtures (12/88, 30/70 wt%) and water. For the uncross-linked PVA and PVA/Zr-MOFs membranes, the swelling degree is presented only for water/isopropanol (12/88, 30/70 wt%) mixtures, since these membranes instantly dissolve in pure water. The swelling data is shown in
Table 3.
The data presented in
Table 3 demonstrate that the addition of Zr-MOFs increased the swelling degree in the water/isopropanol (12/88 and 30/70 wt%) mixtures compared to the PVA and PVA/GA membranes. A slight increase in the swelling degree was caused by an increased number of sorption centers on the surface of the freestanding membranes. The introduction of MIL-140A (5 and 10 wt%) into the PVA matrix increased the swelling degree with the rise of its content in the membrane. However, the introduction of 15 wt% MIL-140A in the PVA membrane causes a slightly decreased swelling degree, compared to the PVA+MIL-140A(10%) membrane. It may be associated with the increased size of agglomerates and change of the membrane morphology, related to high modifier content (confirmed by SEM,
Figure 5d). The introduction of 10 wt% modified Zr-MOFs (MIL-140A-AcOH and MIL-140A-AcOH-EDTA) increased the swelling compared to the PVA membrane but the swelling was slightly decreased compared to the MIL-140 modifier. The stability of PVA-based membranes in water was achieved by cross-linking with GA, as the membranes were found to be stable in water media for at least 14 days during the swelling study. Also, cross-linking of PVA chains resulted in a slight decrease in swelling degree in the water/isopropanol (12/88 and 30/70 wt%) mixtures compared to the uncross-linked membranes. The membranes modified with 10 wt% MIL-140A (PVA+MIL-140A(10%) and PVA+MIL-140A(10%)/GA) have the highest values of the swelling degree among the freestanding membranes based on PVA, PVA/MIL-140A-AcOH(10%), and PVA/MIL-140A-AcOH-EDTA(10%), which is in agreement with the highest surface roughness (
Table 1 and
Table 2) and permeation flux (
Figure 2 and
Figure 3) among them.
To study changes in the surface properties of the PVA membrane during modification with Zr-MOFs, the contact angle of water for the cross-linked membranes was measured (
Table 4). For the uncross-linked PVA and PVA/Zr-MOFs(10%) membranes, the contact angles were not possible to test since these membranes instantly dissolve in water.
The data presented in
Table 4 demonstrate that modification of the membranes based on PVA with Zr-MOFs led to almost identical contact angle data. This may be explained by a number of factors that hampers a qualitative estimation of the changes in the hydrophilic-hydrophobic balance of the surface. These also include: changes in membrane roughness upon the introduction of a modifier with different functional groups which can also modulate membrane surface hydrophilicity.
To study the changes in the free volume of the PVA membrane during cross-linking and modification with 10 wt% Zr-MOFs, the density of the freestanding membranes were measured. The data are presented in
Table 5.
The data presented in
Table 5 demonstrate that the density of the PVA membrane was 1.26 g/cm
3, which is comparable with the data presented in the literature [
79]. The density of the PVA membranes modified with Zr-MOFs particles increased compared to the pristine PVA membranes, which was associated with the addition of MOF modifiers with a high density (density of Zr-MOFs ~0.5 g/cm
3) [
80]. Additionally, it can be associated with a denser polymer structure around the MOF particles [
80] that caused the decreased free volume in the modified membranes.