Distinguishing of Histopathological Staging Features of H-E Stained Human cSCC by Microscopical Multispectral Imaging
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Equipment
2.2. Experimental Samples
2.3. Data Collection
2.4. Image Stitching
2.5. Statistical Analysis
3. Results
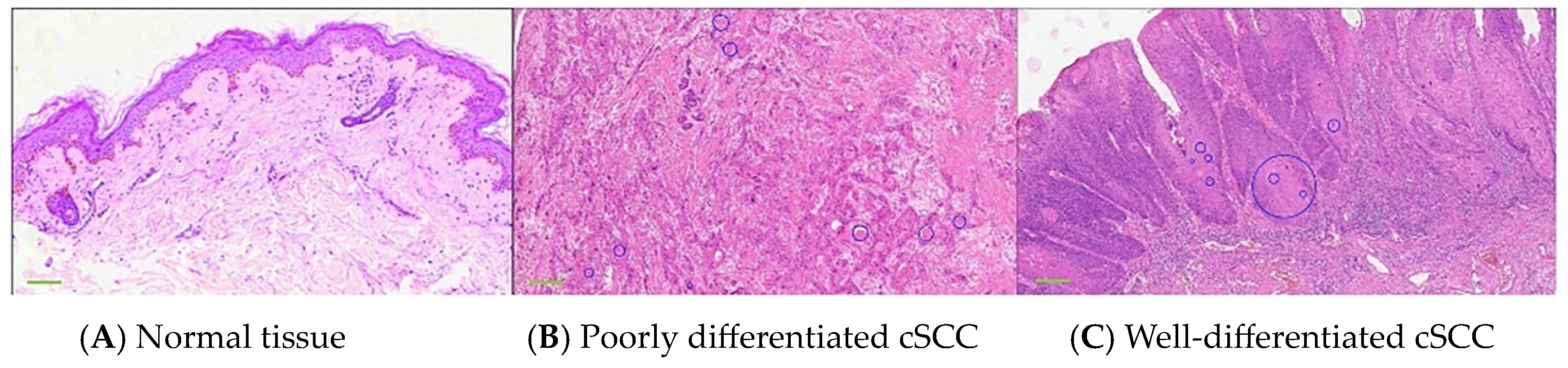
3.1. Human cSCC Manifestation in Pathology
3.2. Features of Human cSCC Were Highlighted by an MSI System
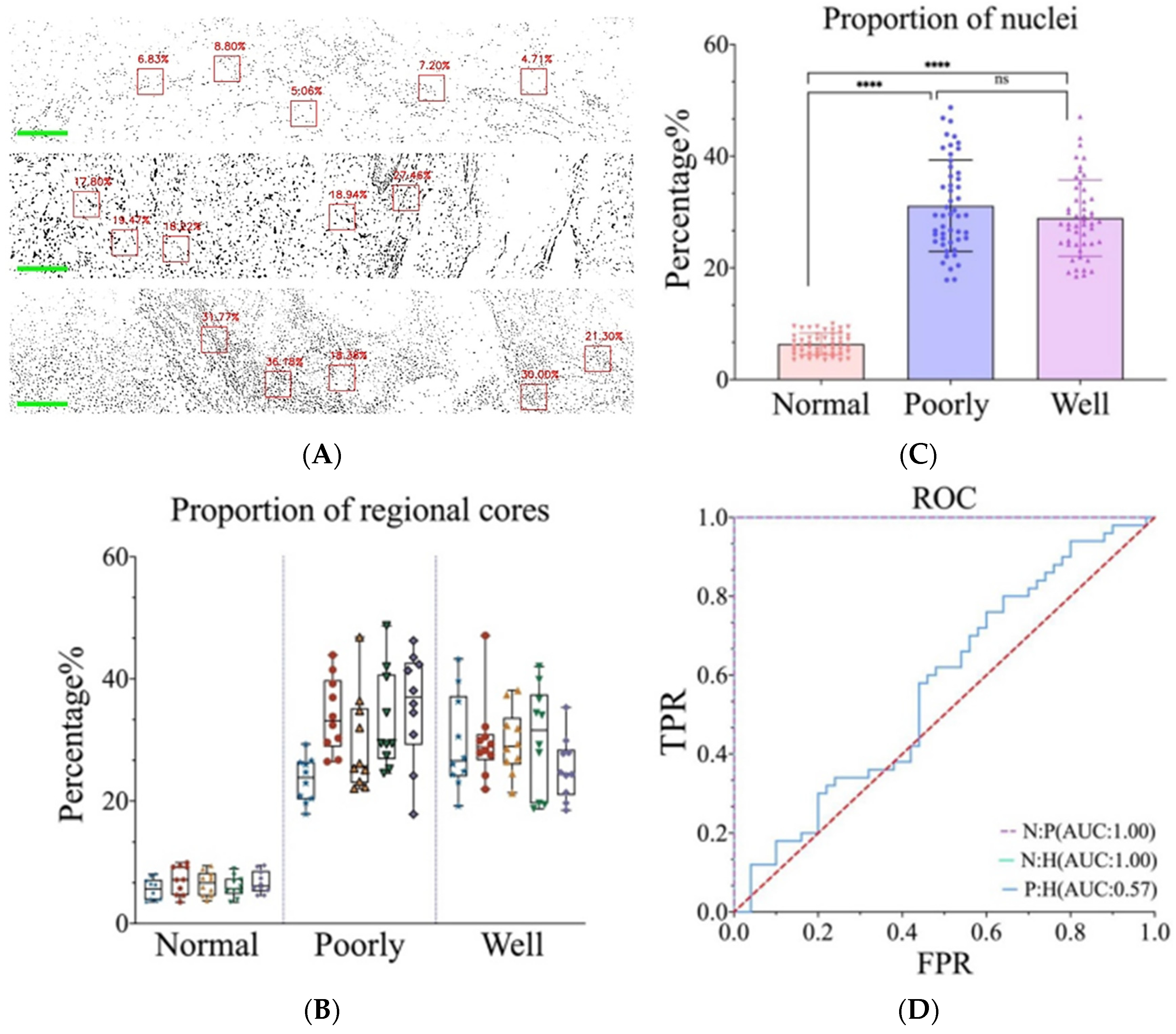
3.3. Differentiation between Normal Skin and cSCC Was Accomplished through Nucleo-Plasmic Ratio
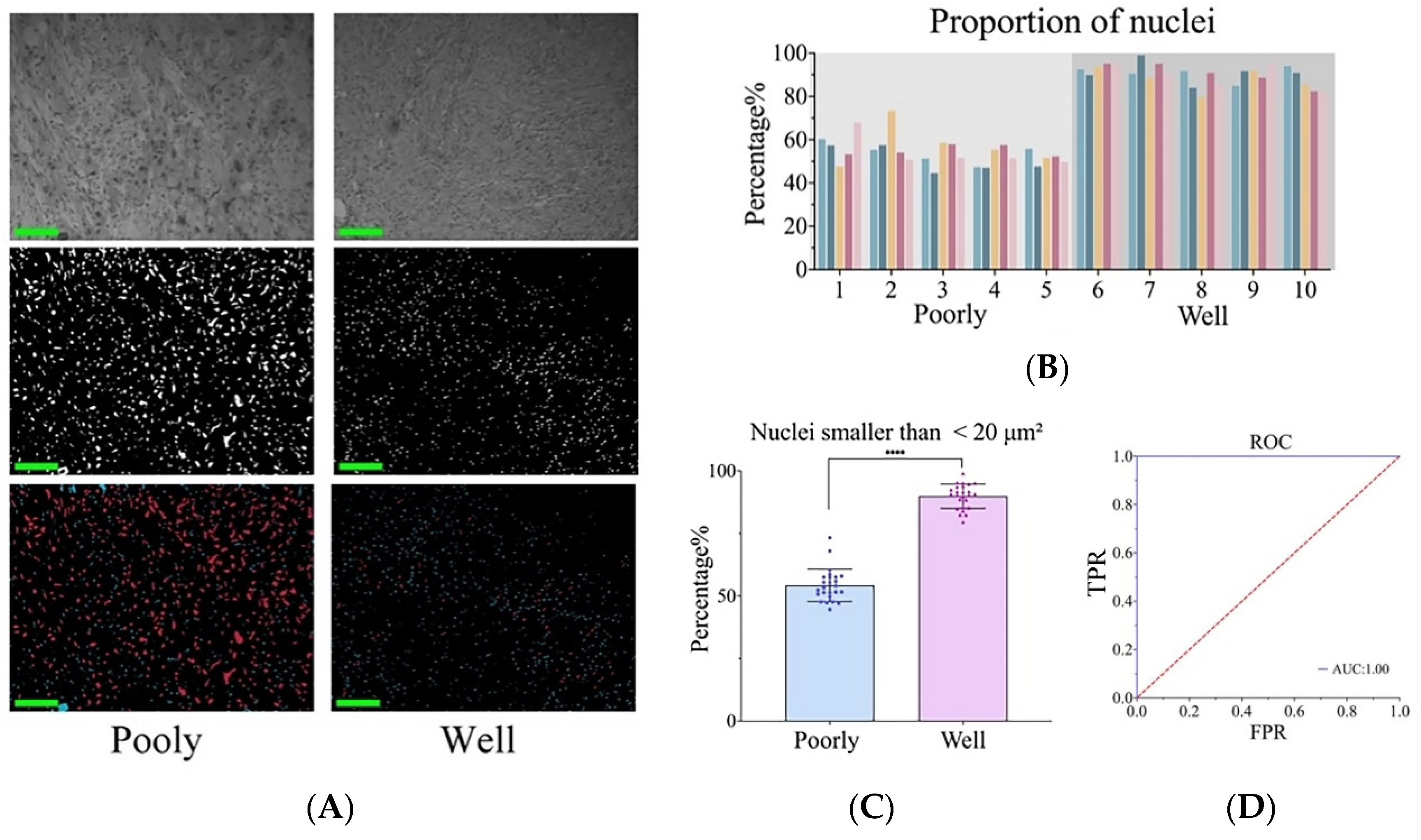
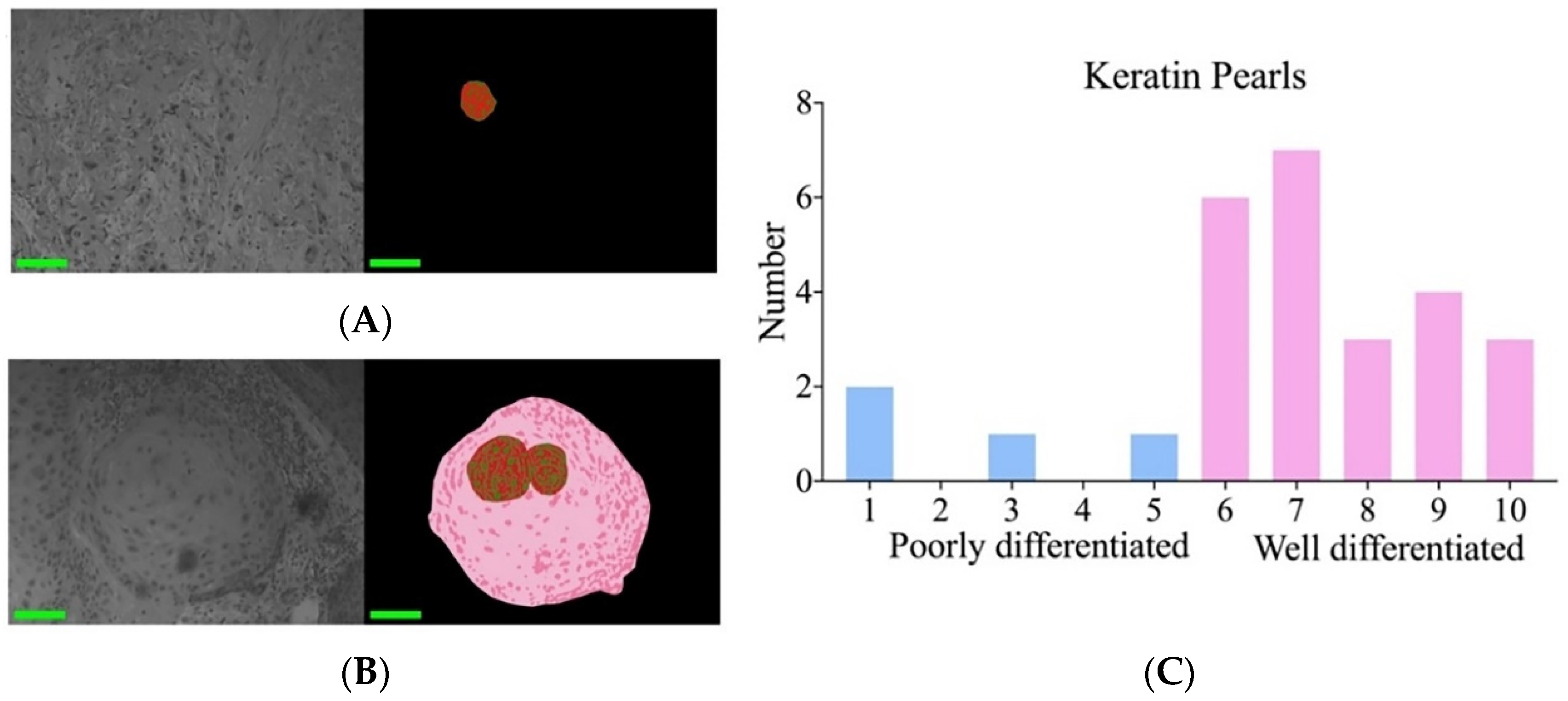
3.4. Poorly and Well-Differentiated cSCC Was Identified via Nuclear Atypia and Keratin Pearls
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Que, S.K.T.; Zwald, F.O.; Schmults, C.D. Cutaneous squamous cell carcinoma: Incidence, risk factors, diagnosis, and staging. J. Am. Acad. Dermatol. 2018, 78, 237–247. [Google Scholar] [CrossRef]
- Rogers, H.; Beveridge, M.; Puente, J.; Wixson, S.; Loy, B.; Happe, L.E. Incidence of nonmelanoma skin cancer in the United States population aged 65 years and older, 2014. J. Am. Acad. Dermatol. 2021, 85, 741–743. [Google Scholar] [CrossRef] [PubMed]
- Schmults, C.D.; Karia, P.S.; Carter, J.B.; Han, J.; Qureshi, A.A. Factors predictive of recurrence and death from cutaneous squamous cell carcinoma: A 10-year, single-institution cohort study. JAMA Dermatol. 2013, 149, 541–547. [Google Scholar] [CrossRef]
- Bennardo, L.; Bennardo, F.; Giudice, A.; Passante, M.; Dastoli, S.; Morrone, P.; Provenzano, E.; Patruno, C.; Nistico, S.P. Local Chemotherapy as an Adjuvant Treatment in Unresectable Squamous Cell Carcinoma: What Do We Know So Far? Curr. Oncol. 2021, 28, 2317–2325. [Google Scholar] [CrossRef]
- Cives, M.; Mannavola, F.; Lospalluti, L.; Sergi, M.C.; Cazzato, G.; Filoni, E.; Cavallo, F.; Giudice, G.; Stucci, L.S.; Porta, C.; et al. Non-Melanoma Skin Cancers: Biological and Clinical Features. Int. J. Mol. Sci. 2020, 21, 5394. [Google Scholar] [CrossRef]
- Karia, P.S.; Han, J.; Schmults, C.D. Cutaneous squamous cell carcinoma: Estimated incidence of disease, nodal metastasis, and deaths from disease in the United States, 2012. J. Am. Acad. Dermatol. 2013, 68, 957–966. [Google Scholar] [CrossRef]
- Hoque, M.Z.; Keskinarkaus, A.; Nyberg, P.; Xu, H.; Seppanen, T. Invasion depth estimation of carcinoma cells using adaptive stain normalization to improve epidermis segmentation accuracy. Comput. Med. Imaging Graph. 2023, 108, 102276. [Google Scholar] [CrossRef]
- Scheau, C.; Caruntu, C.; Caruntu, A. Current Approach and Future Directions in the Diagnosis and Prognosis of Keratinocyte Carcinomas. J. Clin. Med. 2023, 12, 3974. [Google Scholar] [CrossRef]
- Farasat, S.; Yu, S.S.; Neel, V.A.; Nehal, K.S.; Lardaro, T.; Mihm, M.C.; Byrd, D.R.; Balch, C.M.; Califano, J.A.; Chuang, A.Y.; et al. A new American Joint Committee on Cancer staging system for cutaneous squamous cell carcinoma: Creation and rationale for inclusion of tumor (T) characteristics. J. Am. Acad. Dermatol. 2011, 64, 1051–1059. [Google Scholar] [CrossRef]
- Bautista, P.A.; Yagi, Y. Multispectral enhancement method to increase the visual differences of tissue structures in stained histopathology images. Anal. Cell. Pathol. 2012, 35, 407–420. [Google Scholar] [CrossRef]
- Soltani, S.; Ojaghi, A.; Qiao, H.; Kaza, N.; Li, X.; Dai, Q.; Osunkoya, A.O.; Robles, F.E. Prostate cancer histopathology using label-free multispectral deep-UV microscopy quantifies phenotypes of tumor aggressiveness and enables multiple diagnostic virtual stains. Sci. Rep. 2022, 12, 9329. [Google Scholar] [CrossRef] [PubMed]
- Habibalahi, A.; Allende, A.; Michael, J.; Anwer, A.G.; Campbell, J.; Mahbub, S.B.; Bala, C.; Coroneo, M.T.; Goldys, E.M. Pterygium and Ocular Surface Squamous Neoplasia: Optical Biopsy Using a Novel Autofluorescence Multispectral Imaging Technique. Cancers 2022, 14, 1591. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Chen, Q.; Gao, T.; Guo, S.; Xiang, H.; Zheng, G.; Zhang, D.; Wang, X. Segmentation and Recognition of the Pathological Features of Squamous Cell Carcinoma of the Skin Based on Multispectral Imaging. J. Clin. Med. 2022, 11, 3815. [Google Scholar] [CrossRef]
- Wu, X.; Thigpen, J.; Shah, S.K. Multispectral microscopy and cell segmentation for analysis of thyroid fine needle aspiration cytology smears. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2009, 2009, 5645–5648. [Google Scholar] [PubMed]
- Veuthey, T.V.; Herrera, M.G.; Dodero, V.I. Dyes and stains: From molecular structure to histological application. Front. Biosci. 2014, 19, 91–112. [Google Scholar] [CrossRef] [PubMed]
- Bautista, P.A.; Hashimoto, N.; Yagi, Y. Color standardization in whole slide imaging using a color calibration slide. J. Pathol. Inform. 2014, 5, 4. [Google Scholar] [CrossRef] [PubMed]
- Spreinat, A.; Selvaggio, G.; Erpenbeck, L.; Kruss, S. Multispectral near infrared absorption imaging for histology of skin cancer. J. Biophotonics 2020, 13, e201960080. [Google Scholar] [CrossRef]
- John, K. Histological and Histochemical Methods; Scion Publishing Ltd.: Oxford, UK, 2015. [Google Scholar]
- Horobin, R.; John, K. (Eds.) Conn’s Biological Stains: A Handbook of Dyes, Stains and Fluorochromes for Use in Biology and Medicine; Taylor & Francis: Abingdon, UK, 2020. [Google Scholar]
- Kim, J.Y.S.; Kozlow, J.H.; Mittal, B.; Moyer, J.; Olenecki, T.; Rodgers, P. Guidelines of care for the management of cutaneous squamous cell carcinoma. J. Am. Acad. Dermatol. 2018, 78, 560–578. [Google Scholar] [CrossRef]
- Schmults, C.D.; Blitzblau, R.; Aasi, S.Z.; Alam, M.; Andersen, J.S.; Baumann, B.C.; Bordeaux, J.; Chen, P.L.; Chin, R.; Contreras, C.M.; et al. NCCN Guidelines® Insights: Squamous Cell Skin Cancer, Version 1.2022. J. Natl. Compr. Cancer Netw. 2021, 19, 1382–1394. [Google Scholar] [CrossRef]
- Dessinioti, C.; Stratigos, A.J. Recent Advances in the Diagnosis and Management of High-Risk Cutaneous Squamous Cell Carcinoma. Cancers 2022, 14, 3556. [Google Scholar] [CrossRef]
- Mullen, J.T.; Feng, L.; Xing, Y.; Mansfield, P.F.; Gershenwald, J.E.; Lee, J.E.; Ross, M.I.; Cormier, J.N. Invasive squamous cell carcinoma of the skin: Defining a high-risk group. Ann. Surg. Oncol. 2006, 13, 902–909. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.S.; Schmults, C.D. Sentinel lymph node biopsy in cutaneous squamous cell carcinoma: A systematic review of the English literature. Dermatol. Surg. 2006, 32, 1309–1321. [Google Scholar] [CrossRef] [PubMed]
- Stratigos, A.J.; Garbe, C.; Dessinioti, C.; Lebbe, C.; Bataille, V.; Bastholt, L.; Dreno, B.; Fargnoli, M.C.; Forsea, A.M.; Frenard, C.; et al. European interdisciplinary guideline on invasive squamous cell carcinoma of the skin: Part 1. epidemiology, diagnostics and prevention. Eur. J. Cancer 2020, 128, 60–82. [Google Scholar] [CrossRef] [PubMed]
- Gregor, S.; Simoncic, U.; Milanic, M. Imaging perfusion changes in oncological clinical applications by hyperspectral imaging: A literature review. Radiol. Oncol. 2022, 56, 420–429. [Google Scholar]
- Jansen-Winkeln, B.; Barberio, M.; Chalopin, C.; Schierle, K.; Diana, M.; Köhler, H.; Gockel, I.; Maktabi, M. Feedforward artificial neural network-based colorectal cancer detection using hyperspectral imaging: A step towards automatic optical biopsy. Cancers 2021, 13, 967. [Google Scholar] [CrossRef] [PubMed]
- Jansen-Winkeln, B.; Germann, I.; Köhler, H.; Mehdorn, M.; Maktabi, M.; Sucher, R.; Barberio, M.; Chalopin, C.; Diana, M.; Moulla, Y.; et al. Comparison of hyperspectral imaging and fluorescence angiography for the determination of the transection margin in colorectal resections—A comparative study. Int. J. Color. Dis. 2021, 36, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Studier-Fischer, A.; Bressan, M.; Qasim, A.; Özdemir, B.; Sellner, J.; Seidlitz, S.; Haney, C.M.; Egen, L.; Michel, M.; Dietrich, M. Spectral characterization of intraoperative renal perfusion using hyperspectral imaging and artificial intelligence. Sci. Rep. 2024, 14, 17262. [Google Scholar] [CrossRef]
- Berry, S.; Giraldo, N.A.; Green, B.F.; Cottrell, T.R.; Stein, J.E.; Engle, E.L.; Xu, H.; Ogurtsova, A.; Roberts, C.; Wang, D.; et al. Analysis of multispectral imaging with the AstroPath platform informs efficacy of PD-1 blockade. Science 2021, 372, eaba2609. [Google Scholar] [CrossRef]
- Mansfield, J.R.; Gossage, K.W.; Hoyt, C.C.; Levenson, R.M. Autofluorescence removal, multiplexing, and automated analysis methods for in-vivo fluorescence imaging. J. Biomed. Opt. 2005, 10, 41207. [Google Scholar] [CrossRef]
- Ramos-Vara, J.A.; Miller, M.A. When tissue antigens and antibodies get along: Revisiting the technical aspects of immunohistochemistry--the red, brown, and blue technique. Vet. Pathol. 2014, 51, 42–87. [Google Scholar] [CrossRef]
- Stack, E.C.; Wang, C.; Roman, K.A.; Hoyt, C.C. Multiplexed immunohistochemistry, imaging, and quantitation: A review, with an assessment of Tyramide signal amplification, multispectral imaging and multiplex analysis. Methods 2014, 70, 46–58. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, R.; Yang, J.; Chen, Q.; Yang, C.; Ge, Q.; Rui, D.; Xiang, H.; Zhang, D.; Wang, C.; Zhao, X. Distinguishing of Histopathological Staging Features of H-E Stained Human cSCC by Microscopical Multispectral Imaging. Biosensors 2024, 14, 467. https://doi.org/10.3390/bios14100467
Wu R, Yang J, Chen Q, Yang C, Ge Q, Rui D, Xiang H, Zhang D, Wang C, Zhao X. Distinguishing of Histopathological Staging Features of H-E Stained Human cSCC by Microscopical Multispectral Imaging. Biosensors. 2024; 14(10):467. https://doi.org/10.3390/bios14100467
Chicago/Turabian StyleWu, Rujuan, Jiayi Yang, Qi Chen, Changxing Yang, Qianqian Ge, Danni Rui, Huazhong Xiang, Dawei Zhang, Cheng Wang, and Xiaoqing Zhao. 2024. "Distinguishing of Histopathological Staging Features of H-E Stained Human cSCC by Microscopical Multispectral Imaging" Biosensors 14, no. 10: 467. https://doi.org/10.3390/bios14100467




