Cellular Stress Pathways Are Linked to Acetamiprid-Induced Apoptosis in SH-SY5Y Neural Cells
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Cell Culture Conditions and Treatments
2.3. MTT Assay
2.4. Oxidative Stress Parameters
2.4.1. Flow Cytometry
2.4.2. GSH Content Determination
2.4.3. TAS and TOS Analysis
2.5. Calcium Mobilization
2.6. ER Stress
2.7. Apoptosis/Necrosis
2.7.1. Flow Cytometry
2.7.2. Fluorescent Microscopic Imaging
2.8. mRNA Expression Levels of Apoptosis- and ER Stress-Related Genes
2.9. Western Blot Analysis of NF-κB and MAPK8
2.10. Protein Expression Levels of TNF-α
2.11. Statistical Analysis
3. Results
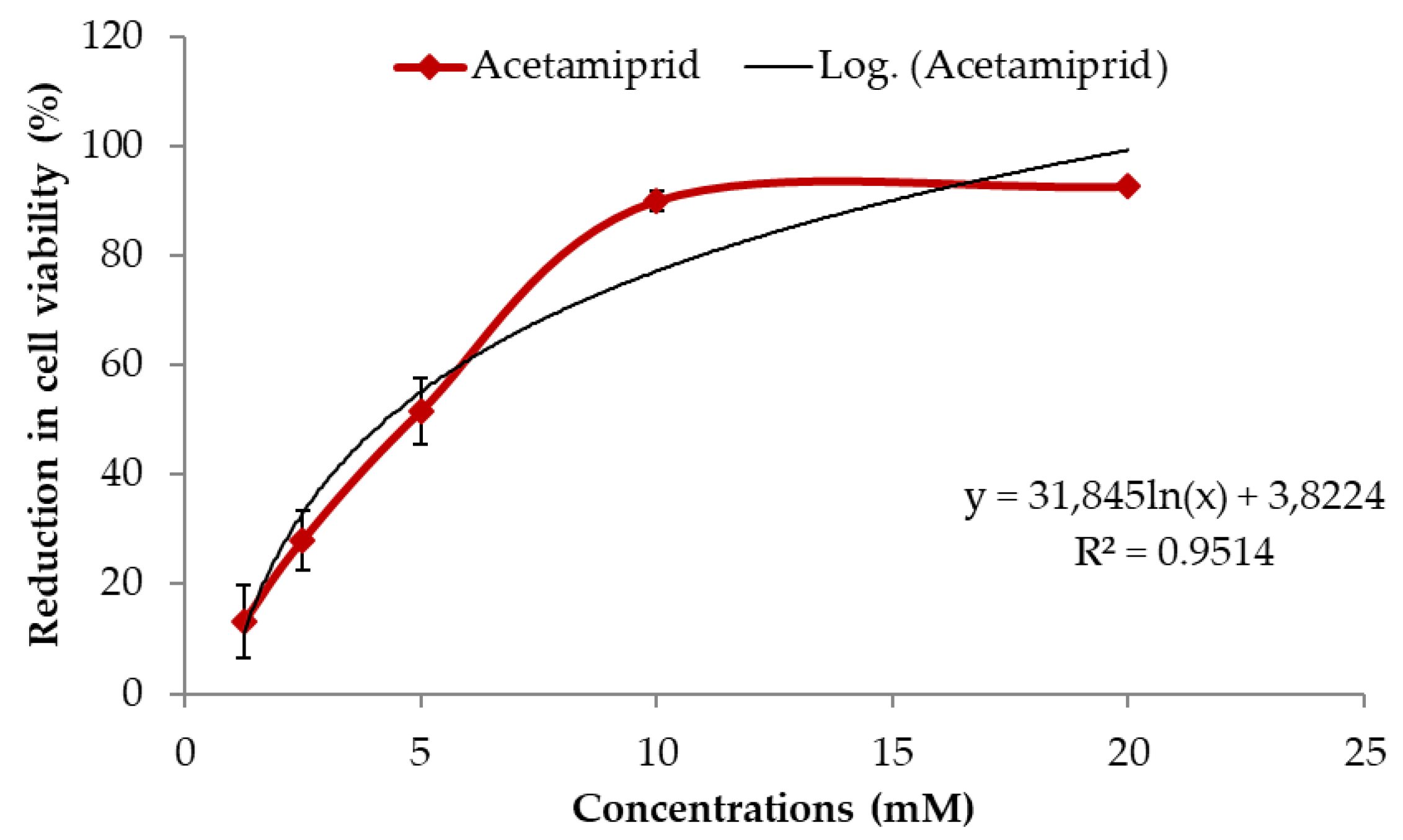
3.1. Effects of ACE on Cell Viability in SH-SY5Y Cells
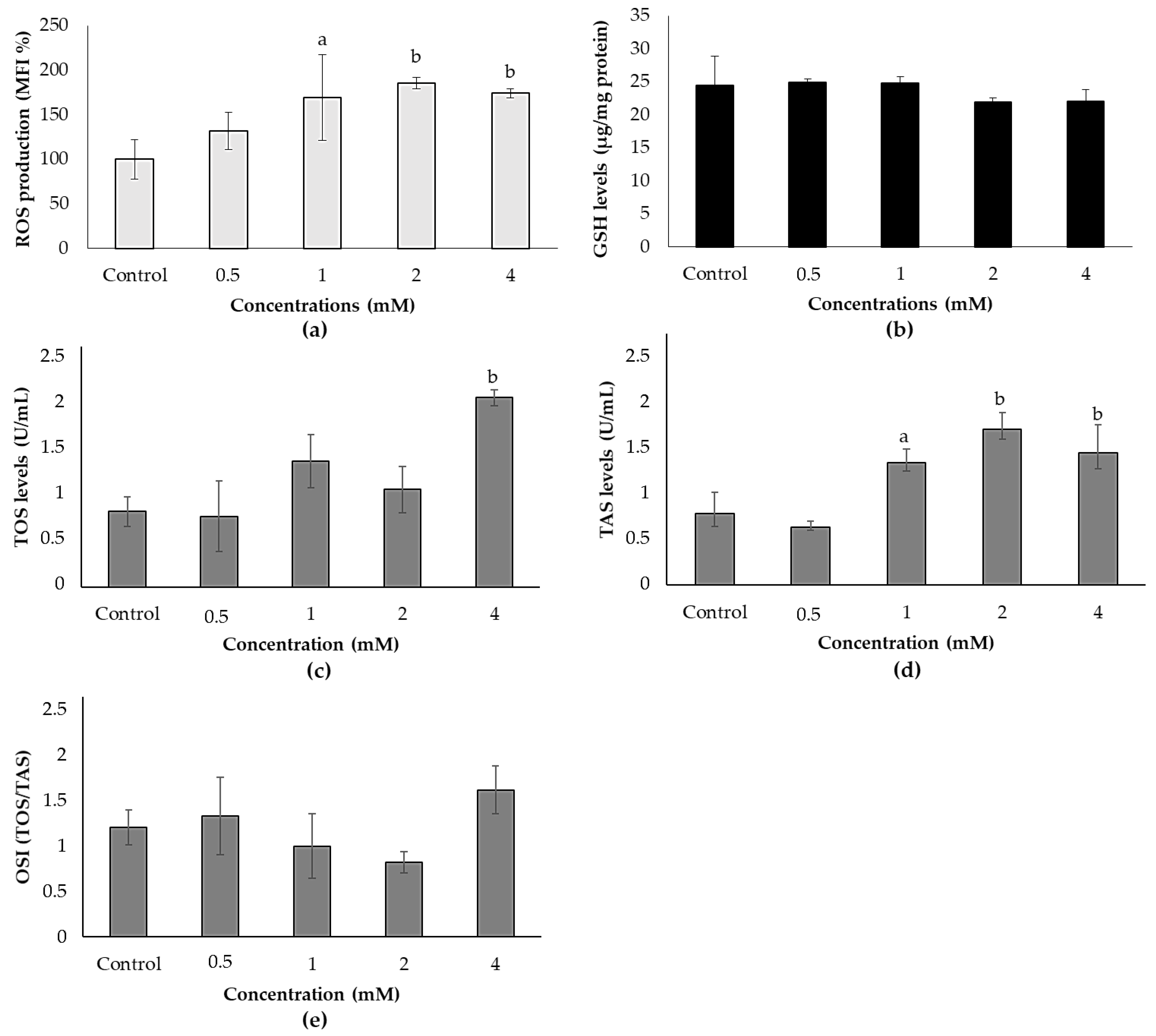
3.2. Effects of ACE on Oxidative Stress in SH-SY5Y Cells
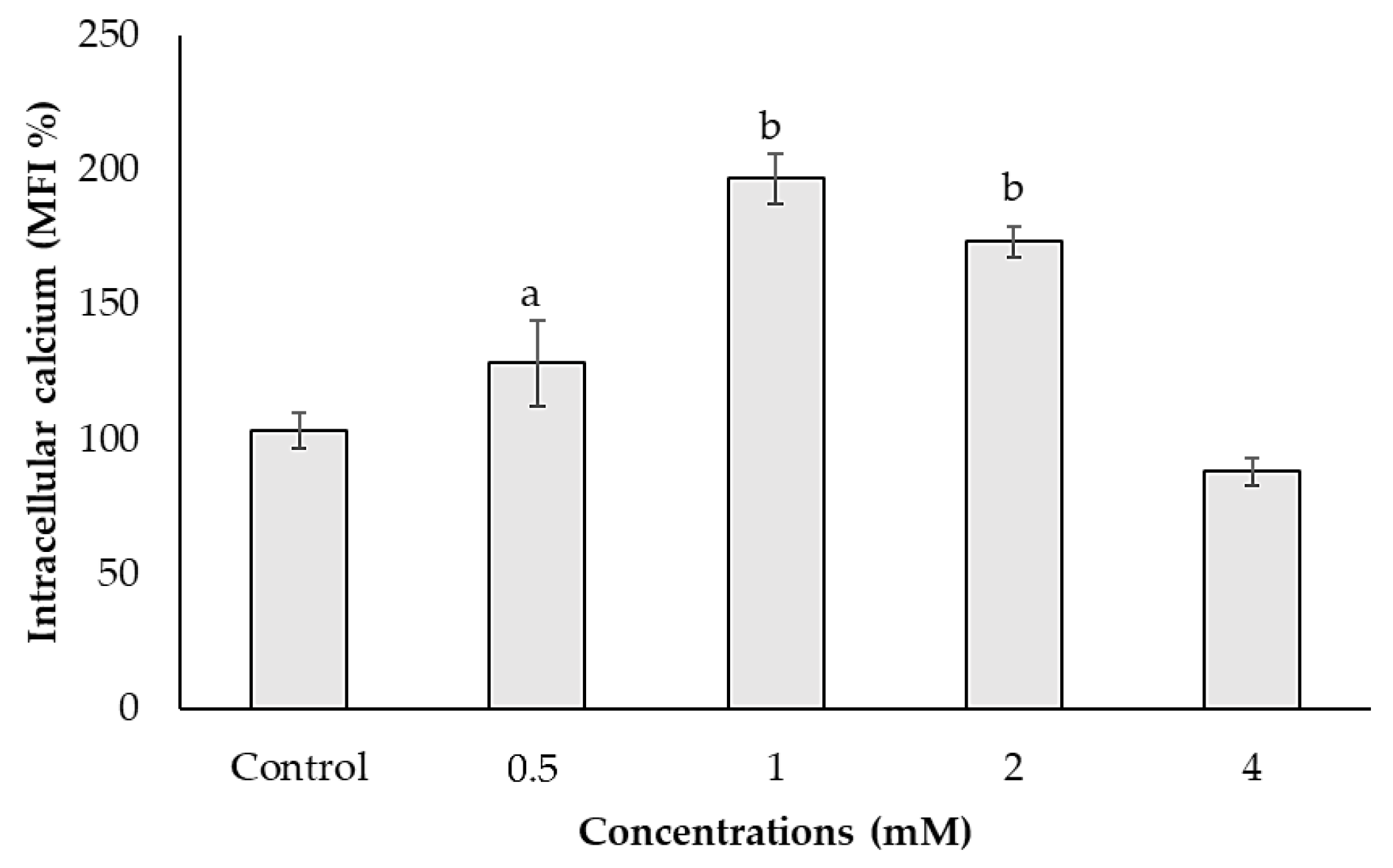
3.3. Effects of ACE on Calcium Mobilization in SH-SY5Y Cells
3.4. Effects of ACE on Endoplasmic Reticulum Stress in SH-SY5Y Cells
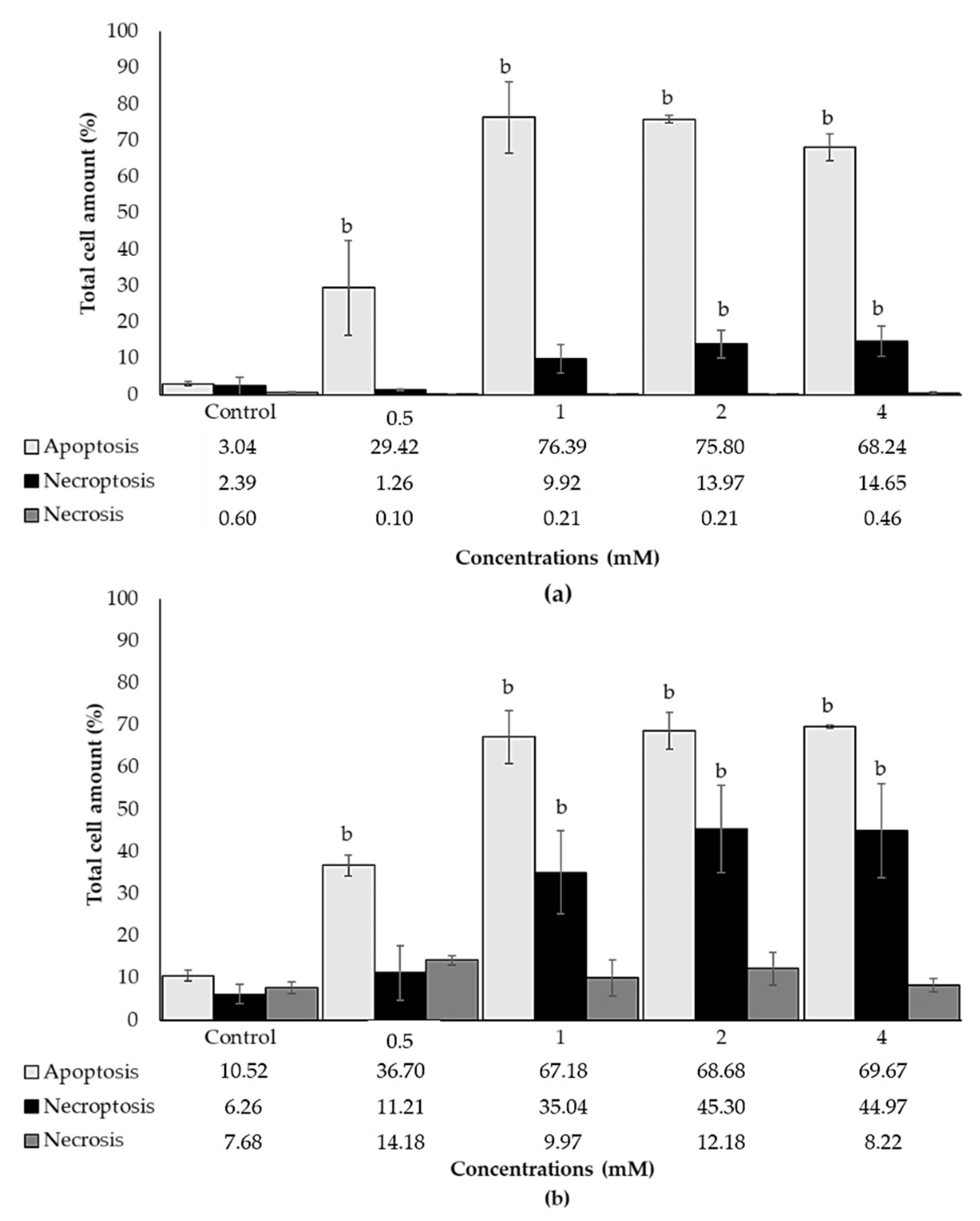
3.5. Effects of ACE on Apoptosis and Necrosis in SH-SY5Y Cells
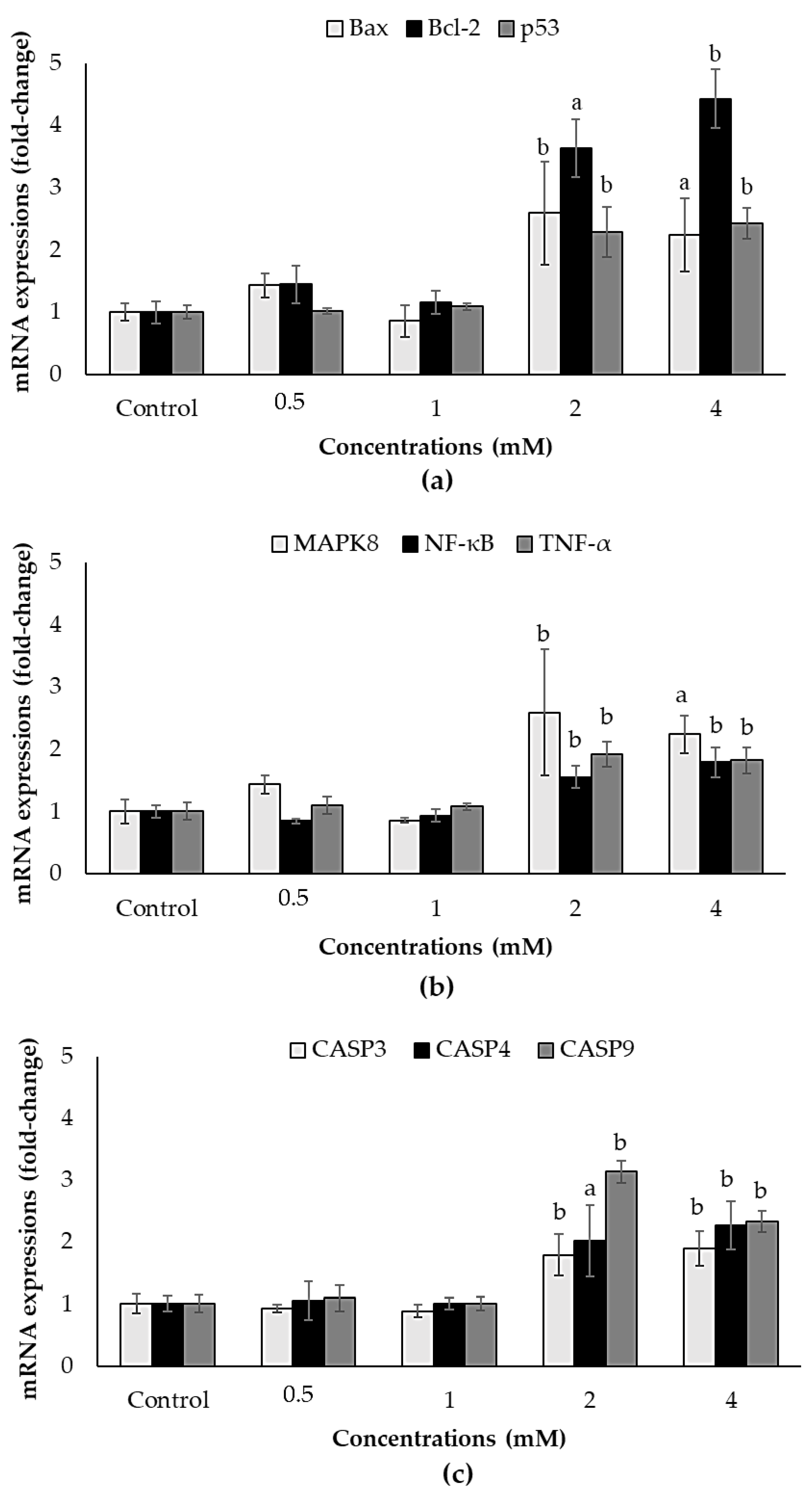
3.6. Effects of ACE on mRNA Expression Levels of Apoptosis and ER Stress Related Genes in SH-SY5Y Cells
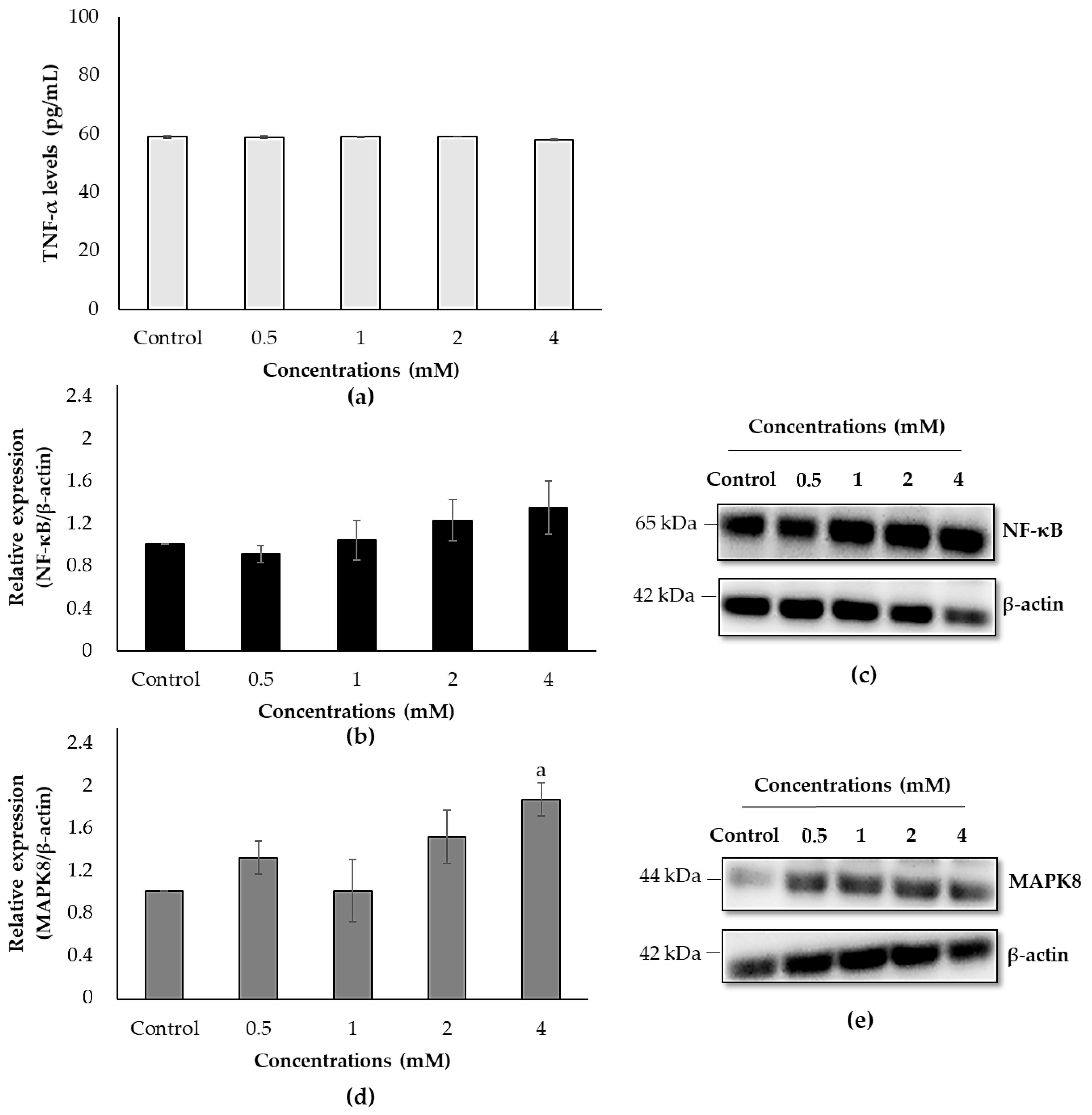
3.7. Effects of ACE on Protein Expression Levels in SH-SY5Y Cells
4. Discussion
5. Conclusions Remarks
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Simon-Delso, N.; Amaral-Rogers, V.; Belzunces, L.P.; Bonmatin, J.M.; Chagnon, M.; Downs, C.; Furlan, L.; Gibbons, D.W.; Giorio, C.; Girolami, V.; et al. Systemic insecticides (neonicotinoids and fipronil): Trends, uses, mode of action and metabolites. Environ. Sci. Pollut. Res. Int. 2015, 22, 5–34. [Google Scholar] [CrossRef] [PubMed]
- Nauen, R.; Denholm, I. Resistance of insect pests to neonicotinoid insecticides: Current status and future prospects. Arch. Insect Biochem. Physiol. 2005, 58, 200–215. [Google Scholar] [CrossRef] [PubMed]
- Tomizawa, M.; Casida, J.E. Neonicotinoid insecticide toxicology: Mechanisms of selective action. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 247–268. [Google Scholar] [CrossRef] [Green Version]
- Bass, C.; Denholm, I.; Williamson, M.S.; Nauen, R. The global status of insect resistance to neonicotinoid insecticides. Pestic. Biochem. Physiol. 2015, 121, 78–87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, P.C.; Lin, H.J.; Liao, Y.Y.; Guo, H.R.; Chen, K.T. Acute poisoning with neonicotinoid insecticides: A case report and literature review. Basic Clin. Pharmacol. Toxicol. 2013, 112, 282–286. [Google Scholar] [CrossRef]
- Goulson, D. Review: An overview of the environmental risks posed by neonicotinoid insecticides. J. Appl. Ecol. 2013, 50, 977–987. [Google Scholar] [CrossRef]
- Shakthi Devan, R.K.; Prabu, P.C.; Panchapakesan, S. Immunotoxicity assessment of sub-chronic oral administration of acetamiprid in Wistar rats. Drug Chem. Toxicol. 2015, 38, 328–336. [Google Scholar] [CrossRef]
- Yamada, T.; Takahashi, H.; Hatano, R. A novel insecticide, acetamiprid. In Nicotinoid Insecticides and the Nicotinic Acetylcholine Receptor; Yamamoto, I., Casida, J.E., Eds.; Springer: Tokyo, Japan, 1999; pp. 149–176. [Google Scholar]
- US EPA. Acetamiprid Reason for Issuance: Conditional Registration. Pesticide Fact. Sheet; US EPA: Washington, DC, USA, 2002; pp. 1–14.
- Republic of Turkey, Ministry of Agriculture and Forestry. Agriculture and Livestock. General Directorate of Food and Control, Department of Plant Protection Products, Active Ingredient Details: 20% Acetamiprid. Available online: https://bku.tarim.gov.tr/AktifMadde/Details/839 (accessed on 20 May 2021).
- Chakroun, S.; Ezzi, L.; Grissa, I.; Kerkeni, E.; Neffati, F.; Bhouri, R.; Sallem, A.; Najjar, M.F.; Hassine, M.; Mehdi, M.; et al. Hematological, biochemical, and toxicopathic effects of subchronic acetamiprid toxicity in Wistar rats. Environ. Sci. Pollut. Res. Int. 2016, 23, 25191–25199. [Google Scholar] [CrossRef]
- Zhang, J.-J.; Wang, Y.; Xiang, H.-Y.; Li, M.-X.; Li, W.-H.; Ma, K.-G.; Wang, X.-Z.; Zhang, J.-H. Oxidative stress: Role in acetamiprid-induced impairment of the male mice reproductive system. Agric. Sci. China 2011, 10, 786–796. [Google Scholar] [CrossRef]
- Annabi, E.; Ben Salem, I.; Abid-Essefi, S. Acetamiprid, a neonicotinoid insecticide, induced cytotoxicity and genotoxicity in PC12 cells. Toxicol. Mech. Methods 2019, 29, 580–586. [Google Scholar] [CrossRef]
- Marfo, J.T.; Fujioka, K.; Ikenaka, Y.; Nakayama, S.M.; Mizukawa, H.; Aoyama, Y.; Ishizuka, M.; Taira, K. Relationship between urinary N-desmethyl-acetamiprid and typical symptoms including neurological findings: A prevalence case-control study. PLoS ONE 2015, 10, e0142172. [Google Scholar] [CrossRef] [Green Version]
- Cobley, J.N.; Fiorello, M.L.; Bailey, D.M. 13 reasons why the brain is susceptible to oxidative stress. Redox Biol. 2018, 15, 490–503. [Google Scholar] [CrossRef]
- Constantinescu, R.; Constantinescu, A.T.; Reichmann, H.; Janetzky, B. Neuronal differentiation and long-term culture of the human neuroblastoma line SH-SY5Y. J. Neural Transm. Suppl. 2007, 17–28. [Google Scholar] [CrossRef]
- Fotakis, G.; Timbrell, J.A. In vitro cytotoxicity assays: Comparison of LDH, neutral red, MTT and protein assay in hepatoma cell lines following exposure to cadmium chloride. Toxicol. Lett. 2006, 160, 171–177. [Google Scholar] [CrossRef]
- Eruslanov, E.; Kusmartsev, S. Identification of ROS using oxidized DCFDA and flow-cytometry. In Advanced Protocols in Oxidative Stress II; Armstrong, D., Ed.; Humana Press: Totowa, NJ, USA, 2010; pp. 57–72. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Wu, R.; Feng, J.; Yang, Y.; Dai, C.; Lu, A.; Li, J.; Liao, Y.; Xiang, M.; Huang, Q.; Wang, D.; et al. Significance of serum total oxidant/antioxidant status in patients with colorectal cancer. PLoS ONE 2017, 12, e0170003. [Google Scholar] [CrossRef] [PubMed]
- Martin, V.V.; Beierlein, M.; Morgan, J.L.; Rothe, A.; Gee, K.R. Novel fluo-4 analogs for fluorescent calcium measurements. Cell Calcium 2004, 36, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.Y.; Kaufman, R.J. The unfolded protein response. J. Cell Sci. 2003, 116, 1861–1862. [Google Scholar] [CrossRef] [Green Version]
- Crowley, L.C.; Marfell, B.J.; Scott, A.P.; Waterhouse, N.J. Quantitation of apoptosis and necrosis by annexin V binding, propidium iodide uptake, and flow cytometry. Cold Spring Harb. Protoc. 2016, 2016. [Google Scholar] [CrossRef]
- Karaman, E.F.; Ozden, S. Alterations in global DNA methylation and metabolism-related genes caused by zearalenone in MCF7 and MCF10F cells. Mycotoxin Res. 2019, 35, 309–320. [Google Scholar] [CrossRef]
- Ali, A.G.; Mohamed, M.F.; Abdelhamid, A.O.; Mohamed, M.S. A novel adamantane thiadiazole derivative induces mitochondria-mediated apoptosis in lung carcinoma cell line. Bioorg. Med. Chem. 2017, 25, 241–253. [Google Scholar] [CrossRef]
- Vasu, S.; McClenaghan, N.H.; McCluskey, J.T.; Flatt, P.R. Cellular responses of novel human pancreatic beta-cell line, 1.1B4 to hyperglycemia. Islets 2013, 5, 170–177. [Google Scholar] [CrossRef] [Green Version]
- Gobel, G.; Szanyi, I.; Revesz, P.; Bauer, M.; Gerlinger, I.; Nemeth, A.; Ember, I.; Gocze, K.; Gombos, K. Expression of NFkB1, GADD45A and JNK1 in salivary gland carcinomas of different histotypes. Cancer Genom. Proteom. 2013, 10, 81–87. [Google Scholar]
- Dong, Y.; Liu, Y.; Kou, X.; Jing, Y.; Sun, K.; Sheng, D.; Yu, G.; Yu, D.; Zhao, Q.; Zhao, X.; et al. The protective or damaging effect of Tumor necrosis factor-alpha in acute liver injury is concentration-dependent. Cell Biosci. 2016, 6, 8. [Google Scholar] [CrossRef] [Green Version]
- Vigano, E.; Diamond, C.E.; Spreafico, R.; Balachander, A.; Sobota, R.M.; Mortellaro, A. Human caspase-4 and caspase-5 regulate the one-step non-canonical inflammasome activation in monocytes. Nat. Commun. 2015, 6, 8761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Souza Prestes, A.; Dos Santos, M.M.; Ecker, A.; de Macedo, G.T.; Fachinetto, R.; Bressan, G.N.; da Rocha, J.B.T.; Barbosa, N.V. Methylglyoxal disturbs the expression of antioxidant, apoptotic and glycation responsive genes and triggers programmed cell death in human leukocytes. Toxicol. Vitro 2019, 55, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Rosa, S.C.; Goncalves, J.; Judas, F.; Mobasheri, A.; Lopes, C.; Mendes, A.F. Impaired glucose transporter-1 degradation and increased glucose transport and oxidative stress in response to high glucose in chondrocytes from osteoarthritic versus normal human cartilage. Arthritis Res. Ther. 2009, 11, R80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeschke, P.; Nauen, R.; Schindler, M.; Elbert, A. Overview of the status and global strategy for neonicotinoids. J. Agric. Food Chem. 2011, 59, 2897–2908. [Google Scholar] [CrossRef]
- Matsuda, K.; Buckingham, S.D.; Kleier, D.; Rauh, J.J.; Grauso, M.; Sattelle, D.B. Neonicotinoids: Insecticides acting on insect nicotinic acetylcholine receptors. Trends Pharmacol. Sci. 2001, 22, 573–580. [Google Scholar] [CrossRef]
- Chen, M.; Tao, L.; McLean, J.; Lu, C. Quantitative analysis of neonicotinoid insecticide residues in foods: Implication for dietary exposures. J. Agric. Food Chem. 2014, 62, 6082–6090. [Google Scholar] [CrossRef] [PubMed]
- Morrissey, C.A.; Mineau, P.; Devries, J.H.; Sanchez-Bayo, F.; Liess, M.; Cavallaro, M.C.; Liber, K. Neonicotinoid contamination of global surface waters and associated risk to aquatic invertebrates: A review. Environ. Int. 2015, 74, 291–303. [Google Scholar] [CrossRef]
- EFSA Panel on Plant Protection Products and their Residues (PPR). Scientific Opinion on the developmental neurotoxicity potential of acetamiprid and imidacloprid. EFSA J. 2013, 11, 3471. [Google Scholar] [CrossRef]
- Devan, R.K.S.; Mishra, A.; Prabu, P.C.; Mandal, T.K.; Panchapakesan, S. Sub-chronic oral toxicity of acetamiprid in Wistar rats. Toxicol. Environ. Chem. 2015, 97, 1236–1252. [Google Scholar] [CrossRef]
- Kimura-Kuroda, J.; Komuta, Y.; Kuroda, Y.; Hayashi, M.; Kawano, H. Nicotine-like effects of the neonicotinoid insecticides acetamiprid and imidacloprid on cerebellar neurons from neonatal rats. PLoS ONE 2012, 7, e32432. [Google Scholar] [CrossRef]
- Kong, D.; Zhang, J.; Hou, X.; Zhang, S.; Tan, J.; Chen, Y.; Yang, W.; Zeng, J.; Han, Y.; Liu, X.; et al. Acetamiprid inhibits testosterone synthesis by affecting the mitochondrial function and cytoplasmic adenosine triphosphate production in rat Leydig cells. Biol. Reprod. 2017, 96, 254–265. [Google Scholar] [CrossRef] [Green Version]
- Mandal, P.; Mondal, S.; Karnam, S.; Purohit, K. A behavioral study on learning a memory in adult Sprague Dawley rat in induced acetamiprid toxicity. Explor. Anim. Med. Res. 2015, 5, 27–32. [Google Scholar]
- Zhang, Q.; Li, Z.; Chang, C.H.; Lou, J.L.; Zhao, M.R.; Lu, C. Potential human exposures to neonicotinoid insecticides: A review. Environ. Pollut. 2018, 236, 71–81. [Google Scholar] [CrossRef]
- Şenyildiz, M.; Kilinc, A.; Ozden, S. Investigation of the genotoxic and cytotoxic effects of widely used neonicotinoid insecticides in HepG2 and SH-SY5Y cells. Toxicol. Ind. Health 2018, 34, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Lu, Y.; Zhao, Z.; Hoogenboom, R.; Zhang, Q.; Liu, X.; Song, W.; Guan, S.; Song, W.; Rao, Q. Assessing the combined toxicity effects of three neonicotinoid pesticide mixtures on human neuroblastoma SK-N-SH and lepidopteran Sf-9 cells. Food Chem. Toxicol. 2020, 145, 111632. [Google Scholar] [CrossRef] [PubMed]
- Cavas, T.; Cinkilic, N.; Vatan, O.; Yilmaz, D. Effects of fullerenol nanoparticles on acetamiprid induced cytoxicity and genotoxicity in cultured human lung fibroblasts. Pestic. Biochem. Physiol. 2014, 114, 1–7. [Google Scholar] [CrossRef]
- Gomez, S.D.; Bustos, P.S.; Sanchez, V.G.; Ortega, M.G.; Guinazu, N. Trophoblast toxicity of the neonicotinoid insecticide acetamiprid and an acetamiprid-based formulation. Toxicology 2020, 431, 152363. [Google Scholar] [CrossRef]
- Yao, X.H.; Min, H.; Lv, Z.M. Response of superoxide dismutase, catalase, and ATPase activity in bacteria exposed to acetamiprid. Biomed. Environ. Sci. 2006, 19, 309–314. [Google Scholar]
- Gasmi, S.; Rouabhi, R.; Kebieche, M.; Salmi, A.; Boussekine, S.; Toualbia, N.; Taib, C.; Henine, S.; Bouteraa, Z.; Djabri, B. Neurotoxicity of acetamiprid in male albino rats and the opposite effect of quercetin. Biotechnol. Ind. J. 2016, 12, 113. [Google Scholar]
- Gasmi, S.; Kebieche, M.; Rouabhi, R.; Touahria, C.; Lahouel, A.; Lakroun, Z.; Henine, S.; Soulimani, R. Alteration of membrane integrity and respiratory function of brain mitochondria in the rats chronically exposed to a low dose of acetamiprid. Environ. Sci. Pollut. Res. Int. 2017, 24, 22258–22264. [Google Scholar] [CrossRef] [PubMed]
- Quintana, M.M.; Rivero Osimani, V.; Magnarelli, G.; Rovedatti, M.G.; Guinazu, N. The insecticides chlorpyrifos and acetamiprid induce redox imbalance in umbilical cord blood erythrocytes in vitro. Pestic. Biochem. Physiol. 2018, 148, 87–92. [Google Scholar] [CrossRef]
- Chen, H.; Bi, W.; Cao, B.; Yang, Z.; Chen, S.; Shang, H.; Yu, P.; Yang, J. A novel podophyllotoxin derivative (YB-1EPN) induces apoptosis and down-regulates express of P-glycoprotein in multidrug resistance cell line KBV200. Eur. J. Pharmacol. 2010, 627, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.Y.; Han, M.H.; Jin, C.Y.; Kim, G.Y.; Choi, B.T.; Nam, T.J.; Kim, S.K.; Choi, Y.H. Apoptosis induction of human leukemia cells by Streptomyces sp. SY-103 metabolites through activation of caspase-3 and inactivation of Akt. Int. J. Mol. Med. 2010, 25, 31–40. [Google Scholar] [PubMed]
- Chien, C.C.; Wu, M.S.; Shen, S.C.; Ko, C.H.; Chen, C.H.; Yang, L.L.; Chen, Y.C. Activation of JNK contributes to evodiamine-induced apoptosis and G2/M arrest in human colorectal carcinoma cells: A structure-activity study of evodiamine. PLoS ONE 2014, 9, e99729. [Google Scholar] [CrossRef]
- Bose, D.; Banerjee, S.; Das, S.; Chatterjee, N.; Saha, K.D. Heat killed attenuated Leishmania induces apoptosis of HepG2 cells through ROS mediated p53 dependent mitochondrial pathway. Cell. Physiol. Biochem. 2016, 38, 1303–1318. [Google Scholar] [CrossRef]
- Kitamura, M. The unfolded protein response triggered by environmental factors. Semin. Immunopathol. 2013, 35, 259–275. [Google Scholar] [CrossRef]
- Ren, Z.; Chen, S.; Qing, T.; Xuan, J.; Couch, L.; Yu, D.; Ning, B.; Shi, L.; Guo, L. Endoplasmic reticulum stress and MAPK signaling pathway activation underlie leflunomide-induced toxicity in HepG2 Cells. Toxicology 2017, 392, 11–21. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Zhang, X.; Niu, Z.; Qi, Y.; Huang, D.; Zhang, Y. 2,4,6-Trichlorophenol cytotoxicity involves oxidative stress, endoplasmic reticulum stress, and apoptosis. Int. J. Toxicol. 2014, 33, 532–541. [Google Scholar] [CrossRef]
- Tirasophon, W.; Welihinda, A.A.; Kaufman, R.J. A stress response pathway from the endoplasmic reticulum to the nucleus requires a novel bifunctional protein kinase/endoribonuclease (Ire1p) in mammalian cells. Genes Dev. 1998, 12, 1812–1824. [Google Scholar] [CrossRef] [Green Version]
- Hitomi, J.; Katayama, T.; Eguchi, Y.; Kudo, T.; Taniguchi, M.; Koyama, Y.; Manabe, T.; Yamagishi, S.; Bando, Y.; Imaizumi, K.; et al. Involvement of caspase-4 in endoplasmic reticulum stress-induced apoptosis and Abeta-induced cell death. J. Cell Biol. 2004, 165, 347–356. [Google Scholar] [CrossRef]
- Zhang, K.; Kaufman, R.J. From endoplasmic-reticulum stress to the inflammatory response. Nature 2008, 454, 455–462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sui, X.; Kong, N.; Ye, L.; Han, W.; Zhou, J.; Zhang, Q.; He, C.; Pan, H. p38 and JNK MAPK pathways control the balance of apoptosis and autophagy in response to chemotherapeutic agents. Cancer Lett. 2014, 344, 174–179. [Google Scholar] [CrossRef]
- Tam, A.B.; Mercado, E.L.; Hoffmann, A.; Niwa, M. ER stress activates NF-κB by integrating functions of basal IKK activity, IRE1 and PERK. PLoS ONE 2012, 7, e45078. [Google Scholar] [CrossRef] [Green Version]
- Bhandary, B.; Marahatta, A.; Kim, H.R.; Chae, H.J. An involvement of oxidative stress in endoplasmic reticulum stress and its associated diseases. Int. J. Mol. Sci. 2012, 14, 434–456. [Google Scholar] [CrossRef]
- Deniaud, A.; Sharaf el dein, O.; Maillier, E.; Poncet, D.; Kroemer, G.; Lemaire, C.; Brenner, C. Endoplasmic reticulum stress induces calcium-dependent permeability transition, mitochondrial outer membrane permeabilization and apoptosis. Oncogene 2008, 27, 285–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patergnani, S.; Suski, J.M.; Agnoletto, C.; Bononi, A.; Bonora, M.; De Marchi, E.; Giorgi, C.; Marchi, S.; Missiroli, S.; Poletti, F.; et al. Calcium signaling around Mitochondria Associated Membranes (MAMs). Cell Commun. Signal. 2011, 9, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hetz, C.; Bernasconi, P.; Fisher, J.; Lee, A.H.; Bassik, M.C.; Antonsson, B.; Brandt, G.S.; Iwakoshi, N.N.; Schinzel, A.; Glimcher, L.H.; et al. Proapoptotic BAX and BAK modulate the unfolded protein response by a direct interaction with IRE1alpha. Science 2006, 312, 572–576. [Google Scholar] [CrossRef] [Green Version]
- Namba, T.; Chu, K.; Kodama, R.; Byun, S.; Yoon, K.W.; Hiraki, M.; Mandinova, A.; Lee, S.W. Loss of p53 enhances the function of the endoplasmic reticulum through activation of the IRE1α/XBP1 pathway. Oncotarget 2015, 6, 19990. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Häcki, J.; Egger, L.; Monney, L.; Conus, S.; Rossé, T.; Fellay, I.; Borner, C. Apoptotic crosstalk between the endoplasmic reticulum and mitochondria controlled by Bcl-2. Oncogene 2000, 19, 2286–2295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Union. Commission Regulation (EU) 2019/88. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32019R0088&from=EN (accessed on 20 May 2021).
- Sofi, F.; Macchi, C.; Abbate, R.; Gensini, G.F.; Casini, A. Mediterranean diet and health. Biofactors 2013, 39, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization/World Health Organization. Pesticide Residues in Food—2011: Toxicological Evaluations. In Proceedings of the Joint Meeting of the FAO Panel of Experts on Pesticide Residues in Food and the Environment and the WHO Core Assessment Group on Pesticide Residues, Geneva, Switzerland, 20–29 September 2011; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- European Union. Agenda Item 12: Establishment of Codex Priority Lists of Pesticides. Available online: https://ec.europa.eu/food/safety/international_affairs/standard_setting_bodies/codex/ccpr/archives_en (accessed on 20 May 2021).

| Gene | Primer Sequences (5′–3′) | Ta | Reference |
|---|---|---|---|
| Bax | F: ACCAAgAAgCTgAgCgAgTATC R: ACAAAgATggTCACggTCTgCC | 60 | [25] |
| Bcl-2 | F: TgTggCCCAgATAggCACCCAg R: ACTTCgCCgAgATgTCCAgCCAg | 65 | [25] |
| p53 | F: AgAgTCTATAggCCCACCCC R: GCTCgACgCTAggATCTgAC | 61 | [25] |
| MAPK8 | F: TTggAACACCATgTCCTgAA R: ATgTACgggTgTTggAgAgC | 57 | [26] |
| NF-κB | F: CACTgCTCAggTCCACTgTC R: CTgTCACTATCCCggAgTTCA | 61 | [27] |
| TNF-α | F: TTCTgTCTACTgAACTTgggggTgATCggTCC R: gTATgAgATAgCAAATCggCTgACggTgTggg | 60 | [28] |
| CASP3 | F: gCTATTgTAggCggTTgT R: TgTTTCCCTgAggTTTgC | 53 | [25] |
| CASP4 | F: AAgAgAAgCAACgTATggCAggAC R: ggACAAAgCTTgAgggCATCTgTA | 62 | [29] |
| CASP9 | F: ACCAgAgATTCgCAAACCAg R: TCACCAAATCCTCCAgAACC | 57 | [30] |
| β-actin | F: AACTACCTTCAACTCCAT R: TgATCTTgATCTTCATTgTg | 48 | [31] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Öztaş, E.; Kara, M.; Boran, T.; Bişirir, E.; Karaman, E.F.; Kaptan, E.; Özhan, G. Cellular Stress Pathways Are Linked to Acetamiprid-Induced Apoptosis in SH-SY5Y Neural Cells. Biology 2021, 10, 820. https://doi.org/10.3390/biology10090820
Öztaş E, Kara M, Boran T, Bişirir E, Karaman EF, Kaptan E, Özhan G. Cellular Stress Pathways Are Linked to Acetamiprid-Induced Apoptosis in SH-SY5Y Neural Cells. Biology. 2021; 10(9):820. https://doi.org/10.3390/biology10090820
Chicago/Turabian StyleÖztaş, Ezgi, Mehtap Kara, Tuğçe Boran, Enes Bişirir, Ecem Fatma Karaman, Engin Kaptan, and Gül Özhan. 2021. "Cellular Stress Pathways Are Linked to Acetamiprid-Induced Apoptosis in SH-SY5Y Neural Cells" Biology 10, no. 9: 820. https://doi.org/10.3390/biology10090820







