APR-246—The Mutant TP53 Reactivator—Increases the Effectiveness of Berberine and Modified Berberines to Inhibit the Proliferation of Pancreatic Cancer Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Lines
2.2. Sources of APR-246, BBR and NAX Compounds
2.3. Transduction of MIA-PaCa-2 and PANC-28 Cells with a Vector Encoding WT-TP53 or an Empty Vector Control
2.4. Measurements of Cellular Proliferation
2.5. Clonogenicity Assays
2.6. Statistical Analysis
3. Results
3.1. Effects of the Mutant TP53 Reactivator on Two PDAC Cell Lines
3.2. Interactions between Berberine (BBR), Chemically Modified BBRs (NAX Compounds) and APR-246 in MIA-PaCa-2 + pLXSN and MIA-PaCa-2 + WT-TP53 Cells
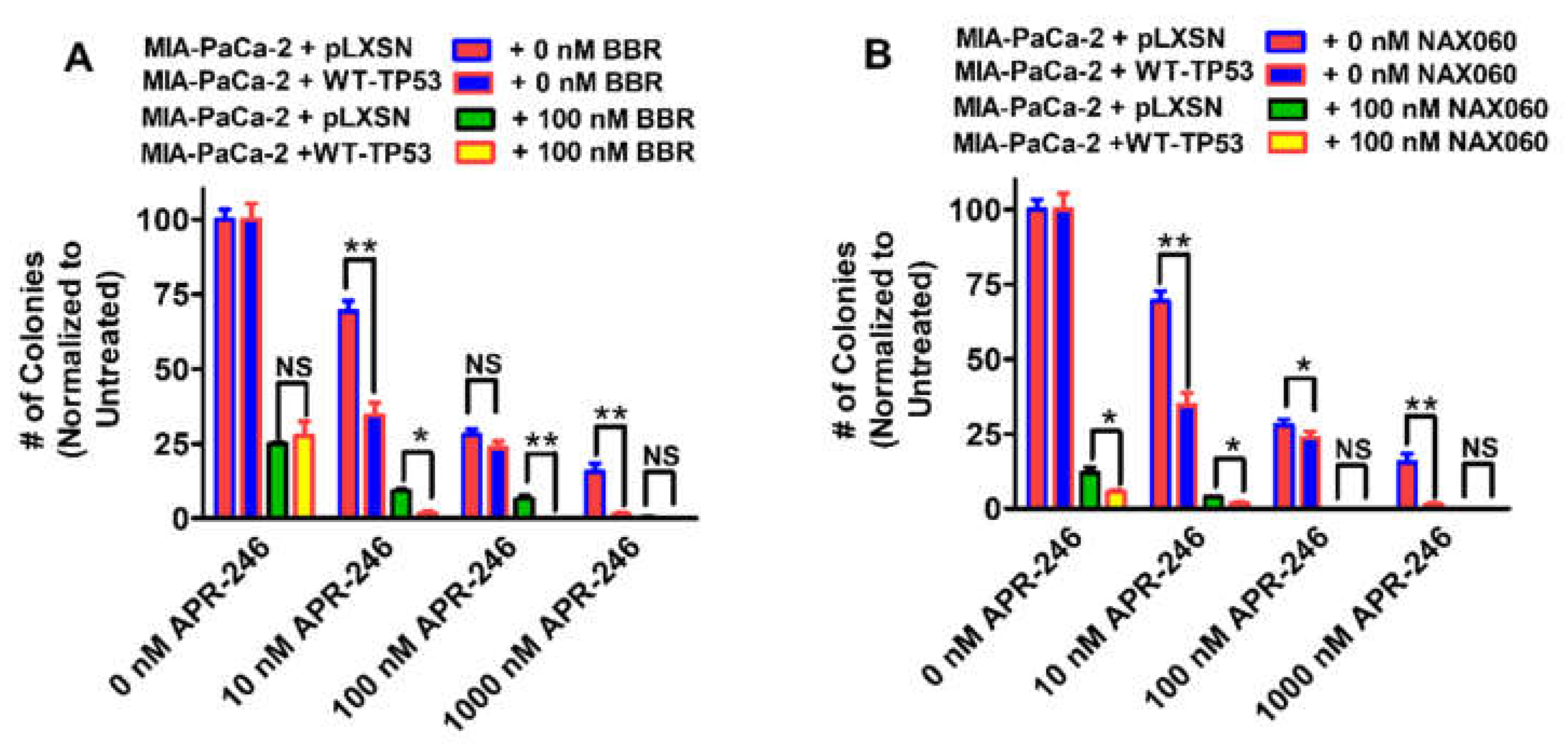
3.3. Abilities of Low Doses of BBR or NAX060 to Increase the Cytotoxicity of APR-246 and Decrease Clonogenicity of MIA-PaCa-2 Cells Containing and Lacking WT-TP53
3.4. Interactions between Berberine (BBR), Chemically Modified BBRs (NAX Compounds) and APR-246 in PANC-28 + pLXSN and PANC-28 + WT-TP53 Cells
3.5. Abilities of Low Doses of BBR or NAX060 to Increase the Cytotoxicity of APR-246 and Decrease Clonogenicity of PANC-28 Cells Containing and Lacking WT-TP53
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Muniraj, T.; Jamidar, P.A.; Aslanian, H.R. Pancreatic cancer: A comprehensive review and update. Dis. Month DM 2013, 59, 368–402. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics. CA Cancer J. Clin. 2013, 63, 11–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gurusamy, K.S.; Kumar, S.; Davidson, B.R.; Fusai, G. Resection versus other treatments for locally advanced pancreatic cancer. Cochrane Database Syst. Rev. 2014, 2, CD010244. [Google Scholar] [CrossRef] [PubMed]
- Kommalapati, A.; Tella, S.H.; Goyal, G.; Ma, W.W.; Mahipal, A. Contemporary management of localized resectable pancreatic cancer. Cancers 2018, 10, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruarus, A.; Vroomen, L.; Puijk, R.; Scheffer, H.; Meijerink, M. Locally advanced pancreatic cancer: A review of local ablative therapies. Cancers 2018, 10, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chin, V.; Nagrial, A.; Sjoquist, K.; O’Connor, C.A.; Chantrill, L.; Biankin, A.V.; Scholten, R.J.; Yip, D. Chemotherapy and radiotherapy for advanced pancreatic cancer. Cochrane Database Syst. Rev. 2018, 3, CD011044. [Google Scholar] [CrossRef]
- Müller, P.C.; Frey, M.C.; Ruzza, C.M.; Nickel, F.; Jost, C.; Gwerder, C.; Hackert, T.; Z’graggen, K.; Kessler, U. Neoadjuvant chemotherapy in pancreatic cancer: An appraisal of the current high-level evidence. Pharmacology 2021, 106, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Sinn, M.; Sinn, B.V.; Treue, D.; Keilholz, U.; Damm, F.; Schmuck, R.; Lohneis, P.; Klauschen, F.; Striefler, J.K.; Bahra, M.; et al. TP53 mutations predict sensitivity to adjuvant gemcitabine in patients with pancreatic ductal adenocarcinoma: Next-generation sequencing results from the CONKO-001 trial. Clin. Cancer Res. 2020, 26, 3732–3739. [Google Scholar] [CrossRef] [Green Version]
- Pu, N.; Chen, Q.; Gao, S.; Liu, G.; Zhu, Y.; Yin, L.; Hu, H.; Wei, L.; Wu, Y.; Maeda, S.; et al. Genetic landscape of prognostic value in pancreatic ductal adenocarcinoma microenvironment. Ann. Trans. Med. 2019, 7, 645. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Gong, Y.; Fan, Z.; Luo, G.; Huang, Q.; Deng, S.; Cheng, H.; Jin, K.; Ni, Q.; Yu, X.; et al. Molecular alterations and targeted therapy in pancreatic ductal adenocarcinoma. J. Hemat. Oncol. 2020, 13, 130. [Google Scholar] [CrossRef]
- Grant, T.J.; Hua, K.; Singh, A. Molecular pathogenesis of pancreatic cancer. Prog. Mole. Biol. Trans. Sci. 2016, 144, 241–275. [Google Scholar]
- Waters, A.M.; Der, C.J. KRAS: The critical driver and therapeutic target for pancreatic cancer. Cold Spring Harbor Pre. Med. 2018, 8, a031435. [Google Scholar] [CrossRef] [PubMed]
- Berthélemy, P.; Bouisson, M.; Escourrou, J.; Vaysse, N.; Rumeau, J.L.; Pradayrol, L. Identification of K-ras mutations in pancreatic juice in the early diagnosis of pancreatic cancer. Ann. Int. Med. 1995, 123, 188–191. [Google Scholar] [CrossRef] [PubMed]
- Siolas, D.; Vucic, E.; Kurz, E.; Hajdu, C.; Bar-Sagi, D. Gain-of-function p53R172H mutation drives accumulation of neutrophils in pancreatic tumors, promoting resistance to immunotherapy. Cell Rep. 2021, 36, 109578. [Google Scholar] [CrossRef] [PubMed]
- Voutsadakis, I.A. Mutations of p53 associated with pancreatic cancer and therapeutic implications. Ann. Hepato-Biliary-Panc. Surg. 2021, 25, 315–327. [Google Scholar] [CrossRef] [PubMed]
- Maddalena, M.; Mallel, G.; Nataraj, N.B.; Shreberk-Shaked, M.; Hassin, O.; Mukherjee, S.; Arandkar, S.; Rotkopf, R.; Kapsack, A.; Lambiase, G.; et al. TP53 missense mutations in PDAC are associated with enhanced fibrosis and an immunosuppressive microenvironment. Proc. Nat. Acad. USA 2021, 118, e2025631118. [Google Scholar] [CrossRef]
- Hashimoto, S.; Furukawa, S.; Hashimoto, A.; Tsutaho, A.; Fukao, A.; Sakamura, Y.; Parajuli, G.; Onodera, Y.; Otsuka, Y.; Handa, H.; et al. ARF6 and AMAP1 are major targets of KRAS and TP53 mutations to promote invasion, PD-L1 dynamics, and immune evasion of pancreatic cancer. Proc. Nat. Acad. Sci. USA 2019, 116, 17450–17459. [Google Scholar] [CrossRef] [Green Version]
- Tong, X.; Xu, D.; Mishra, R.K.; Jones, R.D.; Sun, L.; Schiltz, G.E.; Liao, J.; Yang, G.Y. Identification of a druggable protein-protein interaction site between mutant p53 and its stabilizing chaperone DNAJA1. J. Biol. Chem. 2021, 296, 100098. [Google Scholar] [CrossRef] [PubMed]
- Butera, G.; Brandi, J.; Cavallini, C.; Scarpa, A.; Lawlor, R.T.; Scupoli, M.T.; Marengo, E.; Cecconi, D.; Manfredi, M.; Donadelli, M. The mutant p53-driven secretome has oncogenic functions in pancreatic ductal adenocarcinoma cells. Biomolecules 2020, 10, 884. [Google Scholar] [CrossRef] [PubMed]
- Godfrey, J.D.; Morton, J.P.; Wilczynska, A.; Sansom, O.J.; Bushell, M.D. MiR-142-3p is downregulated in aggressive p53 mutant mouse models of pancreatic ductal adenocarcinoma by hypermethylation of its locus. Cell Death Dis. 2018, 9, 644. [Google Scholar] [CrossRef]
- Fiorini, C.; Cordani, M.; Padroni, C.; Blandino, G.; Di Agostino, S.; Donadelli, M. Mutant p53 stimulates chemoresistance of pancreatic adenocarcinoma cells to gemcitabine. Biochim. Biophys. Acta 2015, 1853, 89–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Escobar-Hoyos, L.F.; Penson, A.; Kannan, R.; Cho, H.; Pan, C.H.; Singh, R.K.; Apken, L.H.; Hobbs, G.A.; Luo, R.; Lecomte, N.; et al. Altered RNA splicing by mutant p53 activates oncogenic RAS signaling in pancreatic cancer. Cancer Cell 2020, 38, 198–211. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.P.; Li, X.; Deng, J.; Zhang, Y.; Dai, B.; Allton, K.L.; Hughes, T.G.; Siangco, C.; Augustine, J.J.; Kang, Y.; et al. Oncogenic KRAS recruits an expansive transcriptional network through mutant p53 to drive pancreatic cancer metastasis. Cancer Dis. 2021, 11, 2094–2111. [Google Scholar] [CrossRef] [PubMed]
- Bailey, J.M.; Hendley, A.M.; Lafaro, K.J.; Pruski, M.A.; Jones, N.C.; Alsina, J.; Younes, M.; Maitra, A.; McAllister, F.; Iacobuzio-Donahue, C.A.; et al. p53 mutations cooperate with oncogenic Kras to promote adenocarcinoma from pancreatic ductal cells. Oncogene 2016, 35, 4282–4288. [Google Scholar] [CrossRef]
- Bykov, V.J.; Issaeva, N.; Selivanova, G.; Wiman, K.G. Mutant p53-dependent growth suppression distinguishes PRIMA-1 from known anticancer drugs: A statistical analysis of information in the National Cancer Institute database. Carcinogenesis 2002, 23, 2011–2018. [Google Scholar] [CrossRef] [Green Version]
- Zache, N.; Lambert, J.M.; Wiman, K.G.; Bykov, V.J. PRIMA-1MET inhibits growth of mouse tumors carrying mutant p53. Cell. Oncol. 2008, 30, 411–418. [Google Scholar]
- Bykov, V.J.; Zache, N.; Stridh, H.; Westman, J.; Bergman, J.; Selivanova, G.; Wiman, K.G. PRIMA-1(MET) synergizes with cisplatin to induce tumor cell apoptosis. Oncogene 2005, 24, 3484–3491. [Google Scholar] [CrossRef] [Green Version]
- Lehmann, S.; Bykov, V.J.; Ali, D.; Andrén, O.; Cherif, H.; Tidefelt, U.; Uggla, B.; Yachnin, J.; Juliusson, G.; Moshfegh, A.; et al. Targeting p53 in vivo: A first-in-human study with p53-targeting compound APR-246 in refractory hematologic malignancies and prostate cancer. J. Clin. Oncol. 2012, 30, 3633–3639. [Google Scholar] [CrossRef]
- Bykov, V.J.; Wiman, K.G. Mutant p53 reactivation by small molecules makes its way to the clinic. FEBS Let. 2014, 588, 2622–2627. [Google Scholar] [CrossRef] [Green Version]
- Deneberg, S.; Cherif, H.; Lazarevic, V.; Andersson, P.O.; von Euler, M.; Juliusson, G.; Lehmann, S. An open-label phase I dose-finding study of APR-246 in hematological Malignancies. Blood Cancer J. 2016, 6, e447. [Google Scholar] [CrossRef] [Green Version]
- Perdrix, A.; Najem, A.; Saussez, S.; Awada, A.; Journe, F.; Ghanem, G.; Krayem, M. PRIMA-1 and PRIMA-1Met (APR-246): From mutant/wild type p53 reactivation to unexpected mechanisms underlying their potent anti-tumor effect in combinatorial therapies. Cancers 2017, 9, 172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maslah, N.; Salomao, N.; Drevon, L.; Verger, E.; Partouche, N.; Ly, P.; Aubin, P.; Naoui, N.; Schlageter, M.H.; Bally, C.; et al. Synergistic effects of PRIMA-1Met (APR-246) and 5-azacitidine in TP53-mutated myelodysplastic syndromes and acute myeloid leukemia. Haematologica 2020, 105, 1539–1551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sallman, D.A.; DeZern, A.E.; Garcia-Manero, G.; Steensma, D.P.; Roboz, G.J.; Sekeres, M.A.; Cluzeau, T.; Sweet, K.L.; McLemore, A.; McGraw, K.L.; et al. Eprenetapopt (APR-246) and Azacitidine in TP53-mutant myelodysplastic syndromes. J. Clin. Oncol. 2021, 39, 1584–1594. [Google Scholar] [CrossRef] [PubMed]
- Synnott, N.C.; Madden, S.F.; Bykov, V.; Crown, J.; Wiman, K.G.; Duffy, M.J. The mutant p53-targeting compound APR-246 induces ROS-modulating genes in breast cancer cells. Trans. Oncol. 2018, 11, 1343–1349. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Besch-Williford, C.; Cook, M.T.; Belenchia, A.; Brekken, R.A.; Hyder, S.M. APR-246 alone and in combination with a phosphatidylserine-targeting antibody inhibits lung metastasis of human triple-negative breast cancer cells in nude mice. Breast Cancer 2019, 11, 249–259. [Google Scholar]
- Li, X.L.; Zhou, J.; Chan, Z.L.; Chooi, J.Y.; Chen, Z.R.; Chng, W.J. PRIMA-1met (APR-246) inhibits growth of colorectal cancer cells with different p53 status through distinct mechanisms. Oncotarget 2015, 6, 36689–36699. [Google Scholar] [CrossRef]
- Patyka, M.; Sharifi, Z.; Petrecca, K.; Mansure, J.; Jean-Claude, B.; Sabri, S. Sensitivity to PRIMA-1MET is associated with decreased MGMT in human glioblastoma cells and glioblastoma stem cells irrespective of p53 status. Oncotarget 2016, 7, 60245–60269. [Google Scholar] [CrossRef] [Green Version]
- Yin, Z.X.; Hang, W.; Liu, G.; Wang, Y.S.; Shen, X.F.; Sun, Q.H.; Li, D.D.; Jian, Y.P.; Zhang, Y.H.; Quan, C.S.; et al. PARP-1 inhibitors sensitize HNSCC cells to APR-246 by inactivation of thioredoxin reductase 1 (TrxR1) and promotion of ROS accumulation. Oncotarget 2018, 9, 1885–1897. [Google Scholar] [CrossRef] [Green Version]
- Krayem, M.; Journe, F.; Wiedig, M.; Morandini, R.; Najem, A.; Salès, F.; van Kempen, L.C.; Sibille, C.; Awada, A.; Marine, J.C.; et al. p53 Reactivation by PRIMA-1(Met) (APR-246) sensitizes (V600E/K) BRAF melanoma to vemurafenib. Eur. J. Cancer 2016, 55, 98–110. [Google Scholar] [CrossRef]
- Fransson, Å.; Glaessgen, D.; Alfredsson, J.; Wiman, K.G.; Bajalica-Lagercrantz, S.; Mohell, N. Strong synergy with APR-246 and DNA-damaging drugs in primary cancer cells from patients with TP53 mutant high-grade serous ovarian cancer. J. Ovarian Res. 2016, 9, 27. [Google Scholar] [CrossRef] [Green Version]
- Mohell, N.; Alfredsson, J.; Fransson, Å.; Uustalu, M.; Byström, S.; Gullbo, J.; Hallberg, A.; Bykov, V.J.; Björklund, U.; Wiman, K.G. APR-246 overcomes resistance to cisplatin and doxorubicin in ovarian cancer cells. Cell Death Dis. 2015, 6, e1794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Izetti, P.; Hautefeuille, A.; Abujamra, A.L.; de Farias, C.B.; Giacomazzi, J.; Alemar, B.; Lenz, G.; Roesler, R.; Schwartsmann, G.; Osvaldt, A.B.; et al. PRIMA-1, a mutant p53 reactivator, induces apoptosis and enhances chemotherapeutic cytotoxicity in pancreatic cancer cell lines. Investig. New Drugs 2014, 32, 783–794. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yang, C.; Cheng, H.; Fan, Z.; Huang, Q.; Lu, Y.; Fan, K.; Luo, G.; Jin, K.; Wang, Z.; et al. Novel agents for pancreatic ductal adenocarcinoma: Emerging therapeutics and future directions. J. Hematol. Oncol. 2016, 11, 14. [Google Scholar] [CrossRef] [Green Version]
- Zandi, R.; Selivanova, G.; Christensen, C.L.; Gerds, T.A.; Willumsen, B.M.; Poulsen, H.S. PRIMA-1Met/APR-246 induces apoptosis and tumor growth delay in small cell lung cancer expressing mutant p53. Clin. Cancer Res. 2011, 17, 2830–2841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deben, C.; Lardon, F.; Wouters, A.; Op de Beeck, K.; Van den Bossche, J.; Jacobs, J.; Van Der Steen, N.; Peeters, M.; Rolfo, C.; Deschoolmeester, V.; et al. APR-246 (PRIMA-1(MET)) strongly synergizes with AZD2281 (olaparib) induced PARP inhibition to induce apoptosis in non-small cell lung cancer cell lines. Cancer Lett. 2016, 375, 313–322. [Google Scholar] [CrossRef]
- Haffo, L.; Lu, J.; Bykov, V.; Martin, S.S.; Ren, X.; Coppo, L.; Wiman, K.G.; Holmgren, A. Inhibition of the glutaredoxin and thioredoxin systems and ribonucleotide reductase by mutant p53-targeting compound APR-246. Sci. Rep. 2018, 8, 12671. [Google Scholar] [CrossRef]
- Zhang, Q.; Bykov, V.J.N.; Wiman, K.G.; Zawacka-Pankau, Z. APR-246 reactivates mutant p53 by targeting cysteines 124 and 277. Cell Death Dis. 2018, 9, 439. [Google Scholar] [CrossRef] [Green Version]
- Rökaeus, N.; Shen, J.; Eckhardt, I.; Bykov, V.J.; Wiman, K.G.; Wilhelm, M.T. PRIMA-1(MET)/APR-246 targets mutant forms of p53 family members p63 and p73. Oncogene 2010, 29, 6442–6451. [Google Scholar] [CrossRef] [Green Version]
- McCubrey, J.A.; Lertpiriyapong, K.; Steelman, L.S.; Abrams, S.L.; Yang, L.V.; Murata, R.M.; Rosalen, P.L.; Scalisi, A.; Neri, L.M.; Cocco, L.; et al. Effects of resveratrol, curcumin, berberine and other nutraceuticals on aging, cancer development, cancer stem cells and microRNAs. Aging 2017, 9, 1477–1536. [Google Scholar] [CrossRef] [Green Version]
- Neag, M.A.; Mocan, A.; Echeverría, J.; Pop, R.M.; Bocsan, C.I.; Crişan, G.; Buzoianu, A.D. Berberine: Botanical occurrence, traditional uses, extraction methods, and relevance in cardiovascular, metabolic, hepatic, and renal disorders. Front. Pharmacol. 2018, 9, 557. [Google Scholar] [CrossRef] [Green Version]
- Park, S.H.; Sung, J.H.; Kim, E.J.; Chung, N. Berberine induces apoptosis via ROS generation in PANC-1 and MIA-PaCa2 pancreatic cell lines. Braz. J. Med. Biol. Res. 2015, 48, 111–119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agnarelli, A.; Natali, M.; Garcia-Gil, M.; Pesi, R.; Tozzi, M.G.; Ippolito, C.; Bernardini, N.; Vignali, R.; Batistoni, R.; Bianucci, A.M.; et al. Cell-specific pattern of berberine pleiotropic effects on different human cell lines. Sci. Rep. 2018, 8, 10599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patil, J.B.; Kim, J.; Jayaprakasha, G.K. Berberine induces apoptosis in breast cancer cells (MCF-7) through mitochondrial-dependent pathway. Eur. J. Pharmacol. 2010, 645, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, Y.; Du, X.; Ma, H.; Yao, J. The anti-cancer mechanisms of berberine: A review. Cancer Manag. Res. 2020, 12, 695–702. [Google Scholar] [CrossRef] [Green Version]
- Chidambara Murthy, K.N.; Jayaprakasha, G.K.; Patil, B.S. The natural alkaloid berberine targets multiple pathways to induce cell death in cultured human colon cancer cells. Eur. J. Pharmacol. 2012, 688, 14–21. [Google Scholar] [CrossRef]
- Ming, M.; Sinnett-Smith, J.; Wang, J.; Soares, H.P.; Young, S.H.; Eibl, G.; Rozengurt, E. Dose-dependent AMPK-dependent and independent mechanisms of berberine and metformin inhibition of mTORC1, ERK, DNA synthesis and proliferation in pancreatic cancer cells. PLoS ONE 2014, 9, e114573. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Li, H.; Wang, S.; Liu, R.; Wu, Z.; Wang, C.; Wang, Y.; Chen, M. Enhancing the antitumor activity of berberine hydrochloride by solid lipid nanoparticle encapsulation. AAPS Pharmscitech 2014, 15, 834–844. [Google Scholar] [CrossRef] [Green Version]
- Cordell, G.A.; Quinn-Beattie, M.L.; Farnsworth, N.R. The potential of alkaloids in drug discovery. Phytother. Res. 2001, 15, 183–205. [Google Scholar] [CrossRef]
- Tillhon, M.; Guamán Ortiz, L.M.; Lombardi, P.; Scovassi, A.I. Berberine: New perspectives for old remedies. Biochem. Pharmacol. 2012, 84, 1260–1267. [Google Scholar] [CrossRef]
- Remppis, A.; Bea, F.; Greten, H.J.; Buttler, A.; Wang, H.; Zhou, Q.; Preusch, M.R.; Enk, R.; Ehehalt, R.; Katus, H.; et al. Rhizoma Coptidis inhibits LPS-induced MCP-1/CCL2 production in murine macrophages via an AP-1 and NFkappaB-dependent pathway. Mediat. Inflamm. 2010, 2010, 194896. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Hua, B.; Saud, S.M.; Lin, H.; Hou, W.; Matter, M.S.; Jia, L.; Colburn, N.H.; Young, M.R. Berberine regulates AMP-activated protein kinase signaling pathways and inhibits colon tumorigenesis in mice. Mol. Carcinog. 2015, 54, 1096–1109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Z.; Liu, Q.; Xu, B.; Wu, J.; Guo, C.; Zhu, F.; Yang, Q.; Gao, G.; Gong, Y.; Shao, C. Berberine induces p53-dependent cell cycle arrest and apoptosis of human osteosarcoma cells by inflicting DNA damage. Mutat. Res. 2009, 662, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Ferraroni, M.; Bazzicalupi, C.; Papi, F.; Fiorillo, G.; Guamán-Ortiz, L.M.; Nocentini, A.; Scovassi, A.I.; Lombardi, P.; Gratteri, P. Solution and solid-state analysis of binding of 13-substituted berberine analogues to human telomeric G-quadruplexes. Chem. Asian J. 2016, 11, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Pierpaoli, E.; Arcamone, A.G.; Buzzetti, F.; Lombardi, P.; Salvatore, C.; Provinciali, M. Antitumor effect of novel berberine derivatives in breast cancer cells. Biofactors 2013, 39, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Bhowmik, D.; Hossain, M.; Buzzetti, F.; D’Auria, R.; Lombardi, P.; Kumar, G.S. Biophysical studies on the effect of the 13 position substitution of the anticancer alkaloid berberine on its DNA binding. J. Phys. Chem. B. 2012, 116, 2314–2324. [Google Scholar] [CrossRef]
- Guamán Ortiz, L.M.; Tillhon, M.; Parks, M.; Dutto, I.; Prosperi, E.; Savio, M.; Arcamone, A.G.; Buzzetti, F.; Lombardi, P.; Scovassi, A.I. Multiple effects of berberine derivatives on colon cancer cells. Biomed. Res. Int. 2014, 2014, 924585. [Google Scholar] [CrossRef] [Green Version]
- Guamán Ortiz, L.M.; Croce, A.L.; Aredia, F.; Sapienza, S.; Fiorillo, G.; Syeda, T.M.; Buzzetti, F.; Lombardi, P.; Scovassi, A.I. Effect of new berberine derivatives on colon cancer cells. Acta. Biochim. Biophys. Sin. 2015, 47, 824–833. [Google Scholar] [CrossRef] [Green Version]
- Marverti, G.; Ligabue, A.; Lombardi, P.; Ferrari, S.; Monti, M.G.; Frassineti, C.; Costi, M.P. Modulation of the expression of folate cycle enzymes and polyamine metabolism by berberine in cisplatin-sensitive and -resistant human ovarian cancer cells. Int. J. Oncol. 2013, 43, 1269–1280. [Google Scholar] [CrossRef]
- Chatterjee, S.; Mallick, S.; Buzzetti, F.; Fiorillo, G.; Syeda, T.M.; Lombardi, P.; Das Sahab, K.; Kumar, G.S. New 13-pyridinealkyl berberine analogues intercalate to DNA and induce apoptosis in HepG2 and MCF-7 cells through ROS mediated p53 dependent pathway: Biophysical, biochemical and molecular modeling studies. RSC Adv. 2015, 5, 90632. [Google Scholar] [CrossRef]
- Pierpaoli, E.; Damiani, E.; Orlando, F.; Lucarini, G.; Bartozzi, B.; Lombardi, P.; Salvatore, C.; Geroni, C.; Donati, A.; Provinciali, M. Antiangiogenic and antitumor activities of berberine derivative NAX014 compound in a transgenic murine model of HER2/neu-positive mammary carcinoma. Carcinogenesis 2015, 36, 1169–1179. [Google Scholar] [CrossRef] [Green Version]
- Deer, E.L.; González-Hernández, J.; Coursen, J.D.; Shea, J.E.; Ngatia, J.; Scaife, C.L.; Firpo, M.A.; Mulvihill, S.J. Phenotype and genotype of pancreatic cancer cell lines. Pancreas 2010, 39, 425–435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klemke, L.; Fehlau, C.F.; Winkler, N.; Toboll, F.; Singh, S.K.; Moll, U.M.; Schulz-Heddergott, R. The gain-of-function p53 R248W mutant promotes migration by STAT3 deregulation in human pancreatic cancer cells. Front. Oncol. 2021, 11, 642603. [Google Scholar] [CrossRef] [PubMed]
- Frazier, M.L.; Fernández, E.; de Llorens, R.; Brown, N.M.; Pathak, S.; Cleary, K.R.; Abbruzzese, J.L.; Berry, K.; Olive, M.; Le Maistre, A.; et al. Pancreatic adenocarcinoma cell line, MDAPanc-28, with features of both acinar and ductal cells. Int. J. Pancreatol. 1996, 19, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Abbruzzese, J.L.; Izzo, J.; Hittelman, W.N.; Li, D. AURKA amplification, chromosome instability, and centrosome abnormality in human pancreatic carcinoma cells. Cancer Genet. Cytogenet. 2005, 159, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Abrams, S.L.; Akula, S.M.; Meher, A.K.; Steelman, L.S.; Gizak, A.; Duda, P.; Rakus, D.; Martelli, A.M.; Ratti, S.; Cocco, L.; et al. GSK-3β can regulate the sensitivity of MIA-PaCa-2 pancreatic and MCF-7 breast cancer cells to chemotherapeutic drugs, targeted therapeutics and nutraceuticals. Cells 2021, 10, 816. [Google Scholar] [CrossRef] [PubMed]
- Sokolosky, M.; Chappell, W.H.; Stadelman, K.; Abrams, S.L.; Davis, N.M.; Steelman, L.S.; McCubrey, J.A. Inhibition of GSK-3β activity can result in drug and hormonal resistance and alter sensitivity to targeted therapy in MCF-7 breast cancer cells. Cell Cycle 2014, 13, 20–33. [Google Scholar] [CrossRef]
- Abrams, S.L.; Follo, M.Y.; Steelman, L.S.; Lertpiriyapong, K.; Cocco, L.; Ratti, S.; Martelli, A.M.; Candido, S.; Libra, M.; Murata, R.M.; et al. Abilities of berberine and chemically modified berberines to inhibit proliferation of pancreatic cancer cells. Adv. Biol. Regul. 2019, 71, 172–182. [Google Scholar] [CrossRef]
- Akula, S.M.; Candido, S.; Libra, M.; Abrams, S.L.; Steelman, L.S.; Lertpiriyapong, K.; Ramazzotti, G.; Ratti, S.; Follo, M.Y.; Martelli, A.M.; et al. Abilities of berberine and chemically modified berberines to interact with metformin and inhibit proliferation of pancreatic cancer cells. Adv. Biol Reg. 2019, 73, 100633. [Google Scholar] [CrossRef]
- McCubrey, J.A.; Meher, A.K.; Akula, S.M.; Abrams, S.L.; Steelman, L.S.; LaHair, M.L.; Franklin, R.A.; Martelli, A.M.; Ratti, S.; Lucio Cocco, L.; et al. Wild type and gain of function mutant TP53 can regulate the sensitivity of pancreatic cells to chemotherapeutic drugs, EGFR/Ras/Raf/MEK, and PI3K/mTORC1/GSK-3 pathway inhibitors, nutraceuticals and alter metabolic properties. Aging 2022, (in press). [Google Scholar]
- Eliyahu, D.; Michalovitz, D.; Eliyahu, S.; Pinhasi-Kimhi, O.; Oren, M. Wild-type p53 can inhibit oncogene-mediated focus formation. Proc. Natl. Acad. Sci. USA 1989, 86, 8763–9767. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, A.D.; Rosman, G.J. Improved retroviral vectors for gene transfer and expression. Biotechniques 1989, 7, 980–982. [Google Scholar] [PubMed]
- Abrams, S.L.; Lertpiriyapong, K.; Yang, L.V.; Martelli, A.M.; Cocco, L.; Ratti, S.; Falasca, M.; Murata, R.M.; Rosalen, P.L.; Lombardi, P.; et al. Introduction of WT-TP53 into pancreatic cancer cells alters sensitivity to chemotherapeutic drugs, targeted therapeutics and nutraceuticals. Adv. Biol. Regul. 2018, 69, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Abrams, S.L.; Akula, S.M.; Martelli, A.M.; Cocco, L.; Ratti, S.; Libra, M.; Candido, S.; Montalto, G.; Cervello, M.; Gizak, A.; et al. Sensitivity of pancreatic cancer cells to chemotherapeutic drugs, signal transduction inhibitors and nutraceuticals can be regulated by WT-TP53. Adv. Biol. Regul. 2021, 79, 100780. [Google Scholar] [CrossRef] [PubMed]
- Abrams, S.L.; Akula, S.M.; Steelman, L.S.; Follo, M.L.; Cocco, L.; Ratti, S.; Martelli, A.M.; Libra, M.; Falzone, L.; Candido, S.; et al. Effects of the MDM2 inhibitor Nutlin-3a on sensitivity of pancreatic cancer cells to berberine and modified berberines in the presence and absence of WT-TP53. Adv. Biol. Regul. 2021, 83, 100840. [Google Scholar] [CrossRef] [PubMed]

















| Berberine or NAX Compound | MIA-PaCa-2 + pLXSN (-APR-246) | MIA-PaCa-2 + pLXSN (+12.5 nM APR-246) | p Value and Statistical Significance Symbol | Fold Change +/− APR-246 | MIA-PaCa-2 + WT-TP53 (-APR-246) | MIA-PaCa-2 + WT-TP53 (+12.5 nM APR-246) | p Value and Statistical Significance Symbol | Fold Change +/− APR-246 |
| Berberine | 1200 nM | 1100 nM | p = 0.047, * | 1.1 × ↓ | 1100 nM | 1000 nM | p = 1, NS | 1.1 × ↓ |
| NAX012 | 1000 nM | 1000 nM | p = 1, NS | 1 × | 700 nM | 700 nM | p = 1, NS | 1 × |
| NAX014 | 1100 nM | 1000 nM | p = 0.048, * | 1.1 × ↓ | 700 nM | 160 nM | p < 0.0001, *** | 4.4 × ↓ |
| NAX035 | 500 nM | 450 nM | p = 0.035, * | 1.1 × ↓ | 300 nM | 16 nM | p < 0.0001, *** | 18.8 × ↓ |
| NAX038 | 600 nM | 500 nM | p < 0.0001, *** | 1.2 × ↓ | 380 nM | 300 nM | p = 0.004, ** | 1.3 × ↓ |
| NAX042 | 380 nM | 300 nM | p = 0.005, * | 1.3 × ↓ | 300 nM | 13 nM | p < 0.0001, *** | 23 × ↓ |
| NAX053 | 300 nM | 200 nM | p = 0.0002, ** | 1.5 × ↓ | 200 nM | 2 nM | p < 0.0001, *** | 100 × ↓ |
| NAX054 | >2000 nM | >2000 nM | p = 1, NS | 1 × | >2000 nM | 1000 nM | p < 0.0001, *** | >2 × ↓ |
| NAX060 | 800 nM | 400 nM | p < 0.0001, *** | 2 × ↓ | 220 nM | 2 nM | p < 0.0001, *** | 110 × ↓ |
| NAX075 | >2000 nM | 500 nM | p < 0.0001, *** | >4 × ↓ | 1000 nM | 2.8 nM | p < 0.0001, *** | 357 × ↓ |
| NAX077 | >2000 nM | 800 nM | p < 0.0001, *** | >2.5 × ↓ | 1900 nM | 1100 nM | p = 0.0052, * | 1.7 × ↓ |
| NAX111 | 300 nM | 100 nM | p < 0.0001, *** | 3 × ↓ | 250 nM | 200 nM | p < 0.0001, *** | 1.3 × ↓ |
| Berberine or NAX Compound | PANC-28 + pLXSN (-APR-246) | PANC-28 + pLXSN (+12.5 nM APR-246) | p Value and Statistical Significance Symbol | Fold Change +/−APR-246 | PANC-28 + WT-TP53 (-APR-246) | PANC-28 + WT-TP53 (-APR-246) | p Value and Statistical Significance Symbol | Fold Change +/− APR-246 |
| Berberine | 1600 nM | 1500 nM | p = 0.119, NS | 1.1 × ↓ | 1000 nM | 18 nM | p < 0.0001, *** | 55.6 × ↓ |
| NAX012 | 2000 nM | 500 nM | p < 0.0001, *** | 4 × ↓ | 800 nM | 5.5 nM | p < 0.0001, *** | 145 × ↓ |
| NAX014 | 1200 nM | 700 nM | p = 0.0002, ** | 1.7 × ↓ | 500 nM | 2 nM | p < 0.0001, *** | 250 × ↓ |
| NAX035 | 700 nM | 700 nM | p = 1, NS | 1 × | 50 nM | 4 nM | p < 0.0001, *** | 12.5 × ↓ |
| NAX038 | 2000 nM | 1000 nM | p < 0.0001, *** | 2 × ↓ | 120 nM | 2.2 nM | p < 0.0001, *** | 54.5 × ↓ |
| NAX042 | 1500 nM | 1000 nM | p < 0.0001, *** | 1.5 × ↓ | 32 nM | 7 nM | p < 0.0001, *** | 4.6 × ↓ |
| NAX053 | 600 nM | 600 nM | p = 1, NS | 1 × | 65 nM | 4 nM | p < 0.0001, *** | 16.3 × ↓ |
| NAX054 | >2000 nM | >2000 nM | p = 1, NS | 1 × | >2000 nM | >2000 nM | p = 1, NS | 1 × |
| NAX060 | 450 nM | 200 nM | p < 0.0001, *** | 2.3 × ↓ | 120 nM | 9 nM | p < 0.0001, *** | 13.3 × ↓ |
| NAX075 | 1200 nM | 1200 nM | p = 1, NS | 1 × | 1000 nM | 45 nM | p < 0.0001, *** | 22.2 × ↓ |
| NAX077 | 1700 nM | 1700 nM | p = 1, NS | 1 × | 1500 nM | 1.5 nM | p < 0.0001, *** | 1000 × ↓ |
| NAX111 | 1000 nM | 1000 nM | p = 1, NS | 1 × | 150 nM | 6 nM | p < 0.0001, *** | 25 × ↓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McCubrey, J.A.; Abrams, S.L.; Steelman, L.S.; Cocco, L.; Ratti, S.; Martelli, A.M.; Lombardi, P.; Gizak, A.; Duda, P. APR-246—The Mutant TP53 Reactivator—Increases the Effectiveness of Berberine and Modified Berberines to Inhibit the Proliferation of Pancreatic Cancer Cells. Biomolecules 2022, 12, 276. https://doi.org/10.3390/biom12020276
McCubrey JA, Abrams SL, Steelman LS, Cocco L, Ratti S, Martelli AM, Lombardi P, Gizak A, Duda P. APR-246—The Mutant TP53 Reactivator—Increases the Effectiveness of Berberine and Modified Berberines to Inhibit the Proliferation of Pancreatic Cancer Cells. Biomolecules. 2022; 12(2):276. https://doi.org/10.3390/biom12020276
Chicago/Turabian StyleMcCubrey, James Andrew, Stephen L. Abrams, Linda S. Steelman, Lucio Cocco, Stefano Ratti, Alberto M. Martelli, Paolo Lombardi, Agnieszka Gizak, and Przemysław Duda. 2022. "APR-246—The Mutant TP53 Reactivator—Increases the Effectiveness of Berberine and Modified Berberines to Inhibit the Proliferation of Pancreatic Cancer Cells" Biomolecules 12, no. 2: 276. https://doi.org/10.3390/biom12020276










