Proline-Mediated Drought Tolerance in the Barley (Hordeum vulgare L.) Isogenic Line Is Associated with Lateral Root Growth at the Early Seedling Stage
Abstract
:1. Introduction
2. Results
2.1. Root and Shoot Growth Traits of Barley Lines under Water Stress
2.2. Root and Shoot Growth Traits of Barley Lines under Water Stress
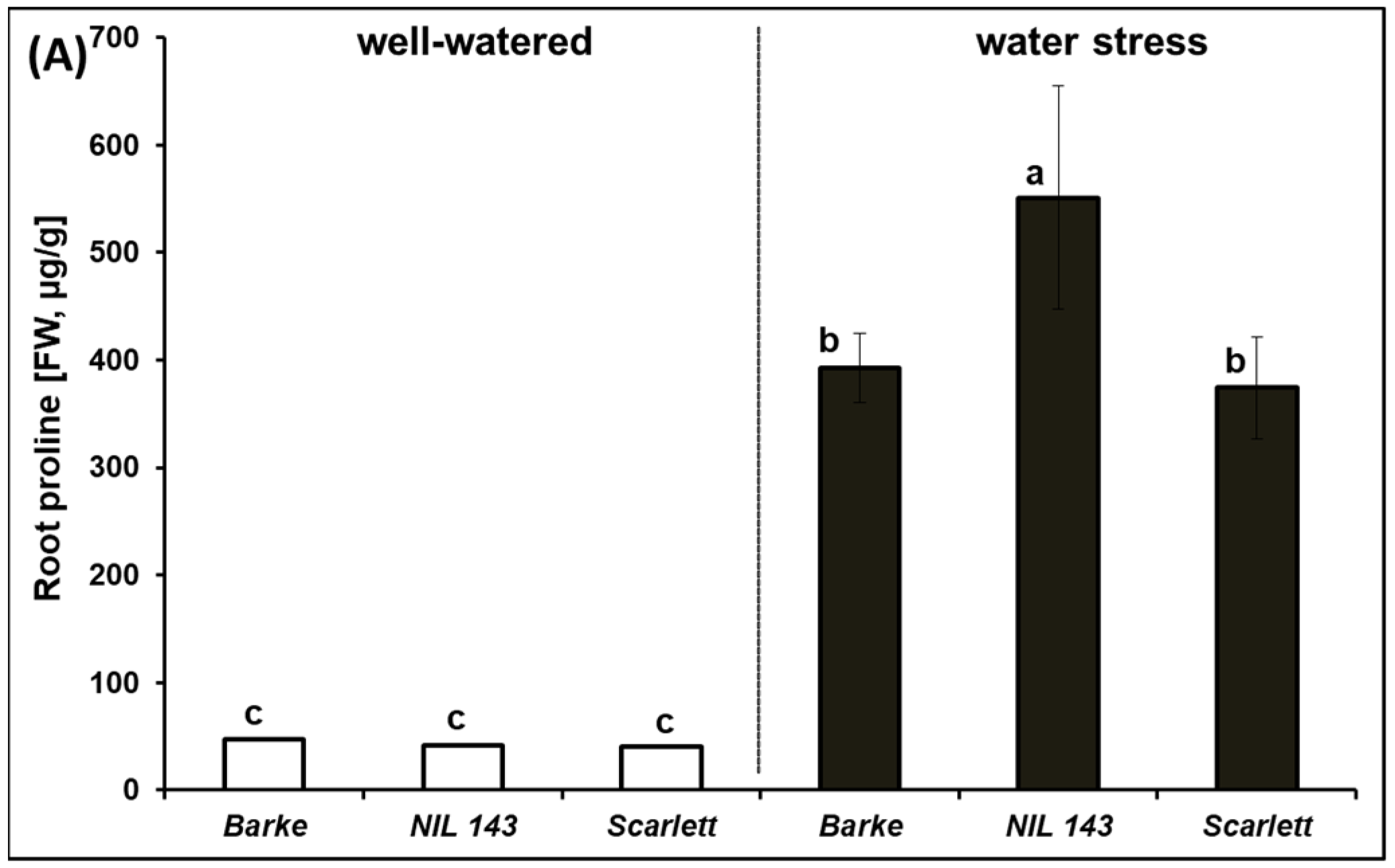
2.3. Effect of Water Stress on Proline in Barley Seedling Shoots and Roots
2.4. Barley Seedlings’ Root Architectural Traits under Water Stress
3. Discussion
3.1. Barley Seedlings’ Root System and Root Placement in Response to Water Stress
3.2. Organ-Dependent Proline Accumulation in Barley Seedlings Promotes Water Stress Tolerance
3.3. Proline Led to Changes in the Morpho-Physiological Traits of Barley under Water Stress
4. Materials and Methods
4.1. Growth Condition
4.2. Experimental Design
4.3. Root and Shoot Measurements
4.3.1. Gas Exchange and Chlorophyll Fluorescence
4.3.2. Plant Water Potential
4.3.3. Proline Determination
4.3.4. Chlorophyll Determination
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, L.; Basso, B. Impacts of climate variability and adaptation strategies on crop yields and soil organic carbon in the US Midwest. PLoS ONE 2020, 15, e0225433. [Google Scholar] [CrossRef] [PubMed]
- Ochieng, J.; Kirimi, L.; Mathenge, M. Effects of climate variability and change on agricultural production: The case of small scale farmers in Kenya. NJAS—Wagening. J. Life Sci. 2016, 77, 71–78. [Google Scholar] [CrossRef]
- Jiménez-Donaire, M.D.P.; Giráldez, J.V.; Vanwalleghem, T. Impact of climate change on agricultural droughts in Spain. Water 2020, 12, 3214. [Google Scholar] [CrossRef]
- Raza, A.; Razzaq, A.; Mehmood, S.S.; Zou, X.; Zhang, X.; Lv, Y.; Xu, J. Impact of climate change on crops adaptation and strategies to tackle its outcome: A review. Plants 2019, 8, 34. [Google Scholar] [CrossRef] [Green Version]
- Calleja-Cabrera, J.; Boter, M.; Oñate-Sánchez, L.; Pernas, M. Root Growth Adaptation to Climate Change in Crops. Front. Plant Sci. 2020, 11, 544. [Google Scholar] [CrossRef]
- Uga, Y.; Sugimoto, K.; Ogawa, S.; Rane, J.; Ishitani, M.; Hara, N.; Kitomi, Y.; Inukai, Y.; Ono, K.; Kanno, N.; et al. Control of root system architecture by DEEPER ROOTING 1 increases rice yield under drought conditions. Nat. Genet. 2013, 45, 1097–1102. [Google Scholar] [CrossRef]
- Arai-Sanoh, Y.; Takai, T.; Yoshinaga, S.; Nakano, H.; Kojima, M.; Sakakibara, H.; Kondo, M.; Uga, Y. Deep rooting conferred by DEEPER ROOTING 1 enhances rice yield in paddy fields. Sci. Rep. 2014, 4, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Tardieu, F.; Simonneau, T.; Muller, B. The Physiological Basis of Drought Tolerance in Crop Plants: A Scenario-Dependent Probabilistic Approach. Annu. Rev. Plant Biol. 2018, 69, 733–759. [Google Scholar] [CrossRef] [Green Version]
- Yamauchi, T.; Pedersen, O.; Nakazono, M.; Tsutsumi, N. Key root traits of Poaceae for adaptation to soil water gradients. New Phytol. 2021, 229, 3133–3140. [Google Scholar] [CrossRef] [PubMed]
- Comas, L.H.; Becker, S.R.; Cruz, V.M.V.; Byrne, P.F.; Dierig, D.A. Root traits contributing to plant productivity under drought. Front. Plant Sci. 2013, 4, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weemstra, M.; Kiorapostolou, N.; van Ruijven, J.; Mommer, L.; de Vries, J.; Sterck, F. The role of fine-root mass, specific root length and life span in tree performance: A whole-tree exploration. Funct. Ecol. 2020, 34, 575–585. [Google Scholar] [CrossRef]
- Ostonen, I.; Püttsepp, Ü.; Biel, C.; Alberton, O.; Bakker, M.R.; Lõhmus, K.; Majdi, H.; Metcalfe, D.; Olsthoorn, A.F.M.; Pronk, A.; et al. Specific root length as an indicator of environmental change. Plant Biosyst. 2007, 141, 426–442. [Google Scholar] [CrossRef]
- Guan, C.; Cui, X.; Liu, H.; Li, X.; Li, M.; Zhang, Y. Proline Biosynthesis Enzyme Genes Confer Salt Tolerance to Switchgrass (Panicum virgatum L.) in Cooperation with Polyamines Metabolism. Front. Plant Sci. 2020, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Biancucci, M.; Mattioli, R.; Moubayidin, L.; Sabatini, S.; Costantino, P.; Trovato, M. Proline affects the size of the root meristematic zone in Arabidopsis. BMC Plant Biol. 2015, 15, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teixeira, W.F.; Soares, L.H.; Fagan, E.B.; da Costa Mello, S.; Reichardt, K.; Dourado-Neto, D. Amino Acids as Stress Reducers in Soybean Plant Growth under Different Water-Deficit Conditions. J. Plant Growth Regul. 2020, 39, 905–919. [Google Scholar] [CrossRef]
- Rady, M.M.; Taha, R.S.; Mahdi, A.H.A. Proline enhances growth, productivity and anatomy of two varieties of Lupinus termis L. grown under salt stress. S. Afr. J. Bot. 2016, 102, 221–227. [Google Scholar] [CrossRef]
- Ghaffari, H.; Tadayon, M.R.; Nadeem, M.; Cheema, M.; Razmjoo, J. Proline-mediated changes in antioxidant enzymatic activities and the physiology of sugar beet under drought stress. Acta Physiol. Plant. 2019, 41, 1–13. [Google Scholar] [CrossRef]
- Schmalenbach, I.; Körber, N.; Pillen, K. Selecting a set of wild barley introgression lines and verification of QTL effects for resistance to powdery mildew and leaf rust. Theor. Appl. Genet. 2008, 117, 1093–1106. [Google Scholar] [CrossRef] [Green Version]
- Naz, A.A.; Arifuzzaman, M.; Muzammil, S.; Pillen, K.; Leon, J. Wild barley introgression lines revealed novel QTL alleles for root and related shoot traits in the cultivated barley (Hordeum vulgare L.). BMC Genet. 2014, 15, 107. [Google Scholar] [CrossRef] [Green Version]
- Honsdorf, N.; March, T.J.; Berger, B.; Tester, M.; Pillen, K. High-throughput phenotyping to detect drought tolerance QTL in wild barley introgression lines. PLoS ONE 2014, 9, e97047. [Google Scholar] [CrossRef] [Green Version]
- Muzammil, S.; Shrestha, A.; Dadshani, S.; Pillen, K.; Siddique, S.; Léon, J.; Naz, A.A. An Ancestral Allele of Pyrroline-5-carboxylate synthase1 Promotes Proline Accumulation and Drought Adaptation in Cultivated Barley. Plant Physiol. 2018, 178, 771–782. [Google Scholar] [CrossRef] [Green Version]
- Honsdorf, N.; March, T.J.; Pillen, K. QTL controlling grain filling under terminal drought stress in a set of wild barley introgression lines. PLoS ONE 2017, 12, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Hernandez, J.; Meints, B.; Hayes, P. Introgression Breeding in Barley: Perspectives and Case Studies. Front. Plant Sci. 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Shrestha, A. Genetic and molecular analysis of drought stress adaptation in cultivated and wild barley Asis Shrestha. Ph.D. Thesis, University of Bonn, Bonn, Germany, 2020. [Google Scholar]
- Sharp, R.E.; Hsiao, T.C.; Silk, W.K. Growth of the maize primary root at low water potentials: II. Role of growth and deposition of hexose and potassium in osmotic adjustment. Plant Physiol. 1990, 93, 1337–1346. [Google Scholar] [CrossRef]
- Voetberg, G.S.; Sharp, R.E. Growth of the maize primary root at low water potentials: III. Role of increased proline deposition in osmotic adjustment. Plant Physiol. 1991, 96, 1125–1130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagel, K.A.; Putz, A.; Gilmer, F.; Heinz, K.; Fischbach, A.; Pfeifer, J.; Faget, M.; Blossfeld, S.; Ernst, M.; Dimaki, C.; et al. GROWSCREEN-Rhizo is a novel phenotyping robot enabling simultaneous measurements of root and shoot growth for plants grown in soil-filled rhizotrons. Funct. Plant Biol. 2012, 39, 891–904. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wasson, A.P.; Nagel, K.A.; Tracy, S.; Watt, M. Beyond Digging: Non-invasive Root and Rhizosphere Phenotyping. Trends Plant Sci. 2020, 25, 119–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avramova, V.; Nagel, K.A.; Abdelgawad, H.; Bustos, D.; Duplessis, M.; Fiorani, F.; Beemster, G.T.S. Screening for drought tolerance of maize hybrids by multi-scale analysis of root and shoot traits at the seedling stage. J. Exp. Bot. 2016, 67, 2453–2466. [Google Scholar] [CrossRef]
- Lynch, J. Root architecture and plant productivity. Plant Physiol. 1995, 109, 7–13. [Google Scholar] [CrossRef]
- Fry, E.L.; Evans, A.L.; Sturrock, C.J.; Bullock, J.M.; Bardgett, R.D. Root architecture governs plasticity in response to drought. Plant Soil 2018, 433, 189–200. [Google Scholar] [CrossRef] [Green Version]
- Faye, A.; Sine, B.; Chopart, J.L.; Grondin, A.; Lucas, M.; Diedhiou, A.G.; Gantet, P.; Cournac, L.; Min, D.; Audebert, A.; et al. Development of a model estimating root length density from root impacts on a soil profile in pearl millet (Pennisetum glaucum (L.) R. Br). Application to measure root system response to water stress in field conditions. PLoS ONE 2019, 14, e0214182. [Google Scholar] [CrossRef] [Green Version]
- Palta, J.; Watt, M. Chapter 13—Vigorous Crop Root Systems: Form and Function for Improving the Capture of Water and Nutrients. In Crop Physiology; Sadras, V., Calderini, D., Eds.; Academic Press: San Diego, CA, USA, 2009; pp. 309–325. ISBN 9780123744319. [Google Scholar]
- Placido, D.F.; Campbell, M.T.; Folsom, J.J.; Cui, X.; Kruger, G.R.; Baenziger, P.S.; Walia, H. Introgression of novel traits from a wild wheat relative improves drought adaptation in wheat. Plant Physiol. 2013, 161, 1806–1819. [Google Scholar] [CrossRef] [Green Version]
- Carvalho, P.; Foulkes, M.J. Roots and Uptake of Water and Nutrients. In Encyclopedia of Sustainability Science and Technology; Meyers, R.A., Ed.; Springer: New York, NY, USA, 2018; pp. 1–24. ISBN 9781493924936. [Google Scholar]
- McCully, M.E. Roots in soil: Unearthing the complexities of roots and their rhizospheres. Annu. Rev. Plant Biol. 1999, 50, 695–718. [Google Scholar] [CrossRef]
- Boudiar, R.; Casas, A.M.; Gioia, T.; Fiorani, F.; Nagel, K.A.; Igartua, E. Effects of Low Water Availability on Root Placement and Shoot Development in Landraces and Modern Barley Cultivars. Agronomy 2020, 10, 134. [Google Scholar] [CrossRef] [Green Version]
- Han, E.; Kautz, T.; Köpke, U. Precrop root system determines root diameter of subsequent crop. Biol. Fertil. Soils 2016, 52, 113–118. [Google Scholar] [CrossRef]
- Gupta, A.; Rico-Medina, A.; Caño-Delgado, A.I. The physiology of plant responses to drought. Science 2020, 368, 266–269. [Google Scholar] [CrossRef]
- Mirza, H.; Kamrun, N.; Masayuki, F.; Hirosuke, O.; Islam, T.M.; Shahzad, B.; Fahad, S.; Tanveer, M.; Saud, S.; Khan, I.A. Plant Responses and Tolerance to Salt Stress. In Approaches for Enhancing Abiotic Stress Tolerance in Plants; CRC Press: Boca Raton, FL, USA, 2019; pp. 61–78. [Google Scholar] [CrossRef]
- Quilambo, O.A. Proline content, water retention capability and cell membrane integrity as parameters for drought tolerance in two peanut cultivars. S. Afr. J. Bot. 2004, 70, 227–234. [Google Scholar] [CrossRef] [Green Version]
- Mattioli, R.; Palombi, N.; Funck, D.; Trovato, M. Proline Accumulation in Pollen Grains as Potential Target for Improved Yield Stability Under Salt Stress. Front. Plant Sci. 2020, 11, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Székely, G.; Ábrahám, E.; Cséplo, Á.; Rigó, G.; Zsigmond, L.; Csiszár, J.; Ayaydin, F.; Strizhov, N.; Jásik, J.; Schmelzer, E.; et al. Duplicated P5CS genes of Arabidopsis play distinct roles in stress regulation and developmental control of proline biosynthesis. Plant J. 2008, 53, 11–28. [Google Scholar] [CrossRef] [Green Version]
- Sharma, S.; Villamor, J.G.; Verslues, P.E. Essential role of tissue-specific proline synthesis and catabolism in growth and redox balance at low water potential. Plant Physiol. 2011, 157, 292–304. [Google Scholar] [CrossRef] [Green Version]
- Ogawa, A.; Yamauchi, A. Root Osmotic Adjustment under Osmotic Stress in Maize Seedlings 1. Transient Change of Growth and Water Relations in Roots in Response to Osmotic Stress. Plant Prod. Sci. 2006, 9, 27–38. [Google Scholar] [CrossRef]
- Furlan, A.L.; Bianucci, E.; Castro, S.; Dietz, K.-J. Metabolic features involved in drought stress tolerance mechanisms in peanut nodules and their contribution to biological nitrogen fixation. Plant Sci. 2017, 263, 12–22. [Google Scholar] [CrossRef]
- Furlan, A.L.; Bianucci, E.; Giordano, W.; Castro, S.; Becker, D.F. Proline metabolic dynamics and implications in drought tolerance of peanut plants. Plant Physiol. Biochem. 2020, 151, 566–578. [Google Scholar] [CrossRef] [PubMed]
- Bandurska, H.; Niedziela, J.; Pietrowska-Borek, M.; Nuc, K.; Chadzinikolau, T.; Radzikowska, D. Regulation of proline biosynthesis and resistance to drought stress in two barley (Hordeum vulgare L.) genotypes of different origin. Plant Physiol. Biochem. 2017, 118, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Boussora, F.; Allam, M.; Guasmi, F.; Ferchichi, A.; Rutten, T.; Hansson, M.; Youssef, H.M.; Börner, A. Spike developmental stages and ABA role in spikelet primordia abortion contribute to the final yield in barley (Hordeum vulgare L.). Bot. Stud. 2019, 60, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verslues, P.E.; Sharp, R.E. Proline accumulation in maize (Zea mays L.) primary roots at low water potentials. II. Metabolic source of increased proline deposition in the elongation zone. Plant Physiol. 1999, 119, 1349–1360. [Google Scholar] [CrossRef] [Green Version]
- Bandurska, H.; Stroiński, A. ABA and proline accumulation in leaves and roots of wild (Hordeum spontaneum) and cultivated (Hordeum vulgare ‘Maresi’) barley genotypes under water deficit conditions. Acta Physiol. Plant. 2003, 25, 55–61. [Google Scholar] [CrossRef]
- Forde, B.G. Glutamate signalling in roots. J. Exp. Bot. 2014, 65, 779–787. [Google Scholar] [CrossRef] [Green Version]
- Forde, B.G.; Cutler, S.R.; Zaman, N.; Krysan, P.J. Glutamate signalling via a MEKK1 kinase-dependent pathway induces changes in Arabidopsis root architecture. Plant J. 2013, 75, 1–10. [Google Scholar] [CrossRef]
- Ahmed, I.M.; Bibi, N.; Nadira, U.A.; Zhang, G.P. Tolerance to Combined Stress of Drought and Salinity in Barley; Mahalingam, R., Ed.; Springer International Publishing: Cham, Switzerland, 2015; ISBN 9783319078991. [Google Scholar]
- Iqbal, N.; Nazar, R.; Khan, N.A. Osmolytes and Plants Acclimation to Changing Environment: Emerging Omics Technologies; Springer: New Delhi, India, 2015; pp. 1–170. [Google Scholar] [CrossRef]
- Verslues, P.E.; Sharma, S. Proline Metabolism and Its Implications for Plant-Environment Interaction. Arab. Book/Am. Soc. Plant Biol. 2010, 8, e0140. [Google Scholar] [CrossRef] [Green Version]
- Mafakheri, A.; Siosemardeh, A.; Bahramnejad, B.; Struik, P.C.; Sohrabi, E. Effect of drought stress on yield, proline and chlorophyll contents in three chickpea cultivars. Aust. J. Crop Sci. 2010, 4, 580–585. [Google Scholar]
- Hayat, S.; Hayat, Q.; Alyemeni, M.N.; Wani, A.S.; Pichtel, J.; Ahmad, A. Role of proline under changing environments: A review. Plant Signal. Behav. 2012, 7, 1456–1466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szabados, L.; Savouré, A. Proline: A multifunctional amino acid. Trends Plant Sci. 2010, 15, 89–97. [Google Scholar] [CrossRef]
- Frimpong, F.; Windt, C.; van Dusschoten, D.; Naz, A.; Frei, M.; Fiorani, F. A wild allele of Pyrroline-5-carboxylate synthase1 leads to proline accumulation in spikes and leaves of barley contributing to improved performance under reduced water availability. Front. Plant Sci. 2021, 12, 180. [Google Scholar] [CrossRef]
- Nikolaeva, M.K.; Maevskaya, S.N.; Shugaev, A.G.; Bukhov, N.G. Effect of drought on chlorophyll content and antioxidant enzyme activities in leaves of three wheat cultivars varying in productivity. Russ. J. Plant Physiol. 2010, 57, 87–95. [Google Scholar] [CrossRef]
- Parkash, V.; Singh, S. A review on potential plant-basedwater stress indicators for vegetable crops. Sustainability 2020, 12, 3945. [Google Scholar] [CrossRef]
- Wang, Z.; Li, G.; Sun, H.; Ma, L.; Guo, Y.; Zhao, Z.; Gao, H.; Mei, L. Effects of drought stress on photosynthesis and photosynthetic electron transport chain in young apple tree leaves. Biol. Open 2018, 7, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, U.S. Drought Tolerance. In Physiology of Stressed Crops; CRC Press: Boca Raton, FL, USA, 2019; pp. 62–104. [Google Scholar] [CrossRef]
- Sharma, S.; Verslues, P.E. Mechanisms independent of abscisic acid (ABA) or proline feedback have a predominant role in transcriptional regulation of proline metabolism during low water potential and stress recovery. Plant Cell Environ. 2010, 33, 1838–1851. [Google Scholar] [CrossRef] [PubMed]
- Maggio, A.; Miyazaki, S.; Veronese, P.; Fujita, T.; Ibeas, J.I.; Damsz, B.; Narasimhan, M.L.; Hasegawa, P.M.; Joly, R.J.; Bressan, R.A. Does proline accumulation play an active role in stress-induced growth reduction? Plant J. 2002, 31, 699–712. [Google Scholar] [CrossRef] [Green Version]
- Signorelli, S. The fermentation analogy: A point of view for understanding the intriguing role of proline accumulation in stressed plants. Front. Plant Sci. 2016, 7, 1–6. [Google Scholar] [CrossRef] [Green Version]
- White, F.F.; Taylor, B.H.; Huffman, G.A.; Gordon, M.P.; Nester, E.W. Molecular and genetic analysis of the transferred DNA regions of the root-inducing plasmid of Agrobacterium rhizogenes. J. Bacteriol. 1985, 164, 33–44. [Google Scholar] [CrossRef] [Green Version]
- Genuchten, V. A Closed-form Equation for Predicting the Hydraulic Conductivity of Unsaturated Soils 1. Soil Sci. Soc. Am. J. 1980, 44, 892–898. [Google Scholar] [CrossRef] [Green Version]
- Tahara, M.; Carver, B.F.; Johnson, R.C.; Smith, E.L. Relationship between relative water content during reproductive development and winter wheat grain yield. Euphytica 1990, 49, 255–262. [Google Scholar] [CrossRef]
- Nagel, K.A.; Kastenholz, B.; Jahnke, S.; Van Dusschoten, D.; Aach, T.; Mühlich, M.; Truhn, D.; Scharr, H.; Terjung, S.; Walter, A.; et al. Temperature responses of roots: Impact on growth, root system architecture and implications for phenotyping. Funct. Plant Biol. 2009, 36, 947–959. [Google Scholar] [CrossRef]
- Scholander, P.F.; Hammel, H.T.; Hemmingsen, E.A.; Bradstreet, E.D. Hydrostatic Pressure and Osmotic Potential in Leaves of Mangroves and Some Other Plants. Proc. Natl. Acad. Sci. USA 1964, 52, 119–125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meier, U. Growth Stages of Mono-and Dicotyledonous Plants, 2nd ed.; Blackwell Wissenschafts-Verlag: Berlin, Germany, 2001. [Google Scholar]
- Bates, L.; Waldren, R.; Teare, I. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 207, 205–207. [Google Scholar] [CrossRef]
- Markwell, J.P.; Danko, S.J.; Bauwe, H.; Osterman, J.; Gorz, H.J.; Haskins, F.A. A Temperature-Sensitive Chlorophyll b -Deficient Mutant of Sweetclover (Melilotus alba). Plant Physiol. 1986, 81, 329–334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Porra, R.J.; Thompson, W.A.; Kriedemann, P.E. Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: Verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. Biophys. Acta 1989, 3, 384–394. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]





| Trait | Treatment | ||||
|---|---|---|---|---|---|
| Genotype | WW | WS | p-Values | % Change | |
| Shoot height (cm) | Barke | 22.8 ± 1.8 ab | 16.3 ± 1.3 c | Genotype NS | −29 |
| Scarlett | 20.8 ± 0.9 abc | 17.6 ± 1.3 bc | Treatment *** | −9 | |
| NIL 143 | 25.8 ± 1.6 a | 16.2 ± 1.6 c | G × T NS | −30 | |
| Leaf number | Barke | 3.2 ± 0.2 ab | 2.5 ± 0.2 b | Genotype NS | −21 |
| Scarlett | 3.5 ± 0.2 ab | 2.7 ± 0.2 b | Treatment *** | −24 | |
| NIL 143 | 4.0 ± 0.3 a | 2.8 ± 0.3 b | G × T NS | −30 | |
| Leaf length (cm) | Barke | 11.4 ± 0.9 a | 8.8 ± 0.3 ab | Genotype NS | −23 |
| Scarlett | 9.4 ± 1.0 ab | 9.35 ± 0.4 ab | Treatment NS | −5 | |
| NIL 143 | 8.8 ± 1.7 ab | 8.8 ± 0.9 b | G × T NS | −27 | |
| Max-Leaf width (cm) | Barke | 0.73 ± 0.1 a | 0.66 ± 0.01 bc | Genotype *** | −9 |
| Scarlett | 0.57 ± 0.0 ab | 0.52 ± 0.02 c | Treatment * | −10 | |
| NIL 143 | 0.62 ± 0.0 abc | 0.53 ± 0.03 c | G × T NS | −16 | |
| Leaf area (cm2) | Barke | 23.9 ± 2.5 a | 13.3 ± 2.8 bc | Genotype NS | −45 |
| Scarlett | 16.8 ± 2.5 ab | 11.1 ± 2.5 c | Treatment *** | −34 | |
| NIL 143 | 25.0 ± 2.5 a | 8.9 ± 2.5 c | G × T NS | −64 | |
| SFW (g) | Barke | 1.28 ± 0.1 a | 0.48 ± 0.1 b | Genotype NS | −60 |
| Scarlett | 0.90 ± 0.1 a | 0.45 ± 0.1 b | Treatment *** | −46 | |
| NIL 143 | 1.33 ± 0.2 ab | 0.48 ± 0.1 b | G × T NS | −57 | |
| SDW (g) | Barke | 0.14 ± 0.01 a | 0.07 ± 0.0 bc | Genotype NS | −49 |
| Scarlett | 0.10 ± 0.01 ab | 0.06 ± 0.0 bc | Treatment *** | −40 | |
| NIL 143 | 0.14 ± 0.02 a | 0.05 ± 0.0 c | G × T NS | −60 | |
| RDW (g) | Barke | 0.035 ± 0.002 ab | 0.020 ± 0.01 c | Genotype ** | −75 |
| Scarlett | 0.023 ± 0.002 c | 0.018 ± 0.01 c | Treatment *** | −28 | |
| NIL 143 | 0.041 ± 0.002 a | 0.018 ± 0.01 c | G × T * | −128 | |
| Root/Shoot ratio | Barke | 0.25 ± 0.021 ab | 0.32 ± 0.02 a | Genotype NS | +28 |
| Scarlett | 0.22 ± 0.021 b | 0.28 ± 0.02 ab | Treatment *** | +27 | |
| NIL 143 | 0.25 ± 0.01 ab | 0.28 ± 0.003 ab | G × T NS | +12 | |
| Parameter | Treatment | ||||
|---|---|---|---|---|---|
| Genotype | WW | WS | p-Values | % Change | |
| Chlorophyll-a (FW, µgmL−1) | Barke | 2.37 ± 0.3 a | 1.07 ± 0.1 b | Genotype *** | −24 |
| Scarlett | 2.14 ± 0.0 a | 1.17 ± 0.0 b | Treatment *** | −19 | |
| NIL 143 | 2.89 ± 0.2 a | 2.69 ± 0.0 a | G × T * | −7 | |
| Fv/Fm | Barke | 0.8 ± 0.0 a | 0.81 ± 0.0 a | Genotype NS | 0 |
| Scarlett | 0.81 ± 0.0 a | 0.81 ± 0.0 a | Treatment NS | 0 | |
| NIL 143 | 0.81 ± 0.0 a | 0.81 ± 0.0 a | G × T NS | 0 | |
| A (µmol m−2 s−1) | Barke | 32.3 ± 0.3 a | 12.0 ± 0.6 d | Genotype *** | −63 |
| Scarlett | 27.5 ± 1.9 b | 14.4 ± 0.3 d | Treatment *** | −48 | |
| NIL 143 | 30.4 ± 0.9 ab | 19.4 ± 0.4 c | G × T *** | −36 | |
| gsw (mol m−2 s−1) | Barke | 0.602 ± 0.06 a | 0.086 ± 0.01 c | Genotype * | −86 |
| Scarlett | 0.533 ± 0.02 a | 0.105 ± 0.02 c | Treatment *** | −80 | |
| NIL 143 | 0.530 ± 0.02 a | 0.183 ± 0.02 b | G × T *** | −65 | |
| E (mol m−2 s−1) | Barke | 8.2 × 10−3 ± 1.7 × 10−4 a | 1.9 × 10−3 ± 2.3 × 10−4 c | Genotype ** | −77 |
| Scarlett | 7.2 × 10−3 ± 1.5 × 10−4 a | 2.2 × 10−3 ± 4.9 × 10−4 c | Treatment *** | −69 | |
| NIL 143 | 7.8 × 10−3 ± 2.3 × 10−4 a | 3.7 × 10−3 ± 1.7 × 10−4 b | G × T *** | −53 | |
| iWUE (µmol CO2 mol−1 H2O) | Barke | 52.6 ± 3.14 c | 134.41 ± 3.14 a | Genotype ** | −155 |
| Scarlett | 54.5 ± 3.14 c | 122.5 ± 3.14 a | Treatment *** | −125 | |
| NIL 143 | 56.4 ± 3.44 c | 104.2 ± 3.14 b | G × T *** | −85 | |
| Ci (μmol mol−1) | Barke | 277.9 ± 3.3 a | 171.7 ± 7.1 c | Genotype *** | −178 |
| Scarlett | 284.6 ± 5.1 a | 183.4 ± 7.7 bc | Treatment * | −185 | |
| NIL 143 | 279.0 ± 2.9 a | 208.0 ± 6.9 b | G × T * | −179 | |
| Plant water potential, Ψ (Mpa) | Barke | −0.394 ± 0.2 ab | −1.23 ± 0.11 c | Genotype ** | −75 |
| Scarlett | −0.293 ± 0.2 ab | −1.26 ± 0.11 c | Treatment *** | −121 | |
| NIL 143 | −0.170 ± 0.2 a | −0.17 ± 0.11 bc | G × T NS | −78 | |
| % RWC | Barke | 98 ± 2.0 a | 46 ± 1.8 c | Genotype * | −51 |
| Scarlett | 88 ± 2.0 ab | 41 ± 1.8 c | Treatment *** | −53 | |
| NIL 143 | 95 ± 2.2 a | 59 ± 1.7 b | G × T * | −41 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frimpong, F.; Anokye, M.; Windt, C.W.; Naz, A.A.; Frei, M.; Dusschoten, D.v.; Fiorani, F. Proline-Mediated Drought Tolerance in the Barley (Hordeum vulgare L.) Isogenic Line Is Associated with Lateral Root Growth at the Early Seedling Stage. Plants 2021, 10, 2177. https://doi.org/10.3390/plants10102177
Frimpong F, Anokye M, Windt CW, Naz AA, Frei M, Dusschoten Dv, Fiorani F. Proline-Mediated Drought Tolerance in the Barley (Hordeum vulgare L.) Isogenic Line Is Associated with Lateral Root Growth at the Early Seedling Stage. Plants. 2021; 10(10):2177. https://doi.org/10.3390/plants10102177
Chicago/Turabian StyleFrimpong, Felix, Michael Anokye, Carel W. Windt, Ali A. Naz, Michael Frei, Dagmar van Dusschoten, and Fabio Fiorani. 2021. "Proline-Mediated Drought Tolerance in the Barley (Hordeum vulgare L.) Isogenic Line Is Associated with Lateral Root Growth at the Early Seedling Stage" Plants 10, no. 10: 2177. https://doi.org/10.3390/plants10102177








