1. Introduction
The Eurasian grapevine
Vitis vinifera L. [
1] is affected by numerous pests and diseases that greatly reduce production each year. The main diseases affecting grapevine, in the current climatic conditions and scenario projections for 2050, are downy and powdery mildews and gray mold, all caused by fungi [
2]. Together, these three diseases are responsible for the application of about 20 fungicide treatments in a relatively short period (April–September), from grape sprouting to harvest. Downy mildew caused by the oomycete
Plasmopara viticola (Berk. et Curt.) Berl. and De Toni is the most important grapevine disease in intermediate to hot climates with sub-humid to humid conditions, such as Northern Italian ones [
3,
4].
P. viticola is an obligate parasite that infects receptive grapevine tissues through stomata by forming an intercellular mycelium with haustoria for nutrition [
5]. The pathogen asexually reproduces by forming sporangiophores with sporangia, which in turn produce zoospores for new infection cycles, starting from 4–5 days of incubation at optimal temperatures (22–25 °C) and high relative humidity (>60%) [
6,
7].
Cultural practices alone are hardly effective to control grapevine downy mildew, and disease control mostly relies on the use of fungicides. However, the progressive reduction in the fungicide modes of action and the widespread presence of fungicide resistance in the pathogen populations are challenging disease management [
8].
The cultivation of resistant vines, with qualitative characteristics suitable for winemaking and direct and indirect consumption, would entail numerous advantages in eco-toxicological and economic terms. Traditionally, breeding programs for resistance to downy mildew are based on the crossing of
V. vinifera varieties with grapevine species that belong to areas where the host plants have co-evolved with
P. viticola or a similar species [
9]. The intense breeding activity carried out for over a century allowed breeders to obtain numerous varieties that couple the quality traits of
V. vinifera and the resistant traits of American or Asian species [
10]. At least 28 QTLs (quantitative trait loci) associated with
P. viticola resistance are known in several
Vitis species and hybrids (
https://www.vivc.de/loci, accessed on 9 May 2022). Of these, the
Rpv3 locus, found in North American wild species (
V. labrusca,
V. lincecumii,
V. riparia, and
V. rupestris) [
11], is the most widespread, since it can be found in most of the resistant cultivars registered on the Vitis International Variety Catalogue VIVC [
12].
Rpv3 resistance is linked to the occurrence of a hypersensitive response (HR) triggered by the products of two nearby TIR-NB-LRR genes and mediated by the synthesis of reactive oxygen species [
13,
14,
15]. Despite this very effective mechanism of resistance, very limited sporulation can sometimes be seen in this case. More recently, three new genomic loci (
Rpv29,
Rpv30, and
Rpv31) associated with a different mechanism of resistance, have been found in the
V. vinifera germplasm of Georgia (South Caucasus). These
loci induce partial resistance, where the pathogen growth and sporulation are significantly reduced compared to that in susceptible grapevine varieties [
16,
17].
The durability of resistance is a key factor for disease management, especially for tree crops, which must be productive for years and must be preserved by adopting suitable strategies, such as pyramiding different resistance genes in a single cultivar [
18]. A breakdown of resistance to
Rpv3 has been already found in
P. viticola strains from the Czech Republic, Italy, and Germany [
14,
19,
20,
21,
22]. In these reports, the resistant accession showed the same disease intensity as the susceptible reference. The characterization of pathogen populations is very important to deploy the different QTLs for plant genetic improvement [
23].
P. viticola produces both asexual (zoospores differentiated in sporangia) and sexual (oospores) spores for infection and survival, respectively, and possesses a polycyclic behavior, causing primary and secondary infection cycles within the same grapevine vegetative season. This favors the selection and diffusion of adapted pathogen strains.
The aim of this study was to gain an insight into the potential durability of resistance to two separate QTLs,
Rpv3-1 and
Rpv29, through the evaluation of adaptation of
P. viticola strains in northern Italy.
P. viticola strains (72) were isolated from different regions and characterized for several fitness parameters such as infectivity (latency period and disease incidence and severity), estimated from the presence of asexual reproductive structures (sporangiophores bearing sporangia), and sexual reproduction (oospores) on three cultivars with different genetic backgrounds and susceptibility profiles: Pinot Noir (
V. vinifera, susceptible), Bianca (resistant interspecific hybrid harboring
Rpv3-1 QTL), and Mgaloblishvili (
V. vinifera, resistant variety with
Rpv29 QTL) [
13,
15,
24]. The pathogenicity of each
P. viticola strain on the three different grapevine varieties was determined according to the leaf surface covered by sporulation at 5–13 days after inoculation. Furthermore, the production and viability of oospores (sexual spores produced by the pathogen for overwintering) by strains able to grow on resistant cultivars were assessed to evaluate whether survival of the pathogen is hampered in resistant varieties.
3. Discussion
The most widespread
V. vinifera cultivars are commonly susceptible to
P. viticola and only recently has the partial investigation of the Georgian germplasm allowed the identification of the resistant variety MG and the first QTLs associated with downy mildew resistance (
Rpv29-31) within the species [
16,
24]. The unique mechanism of resistance of MG [
17,
25] makes the cultivar suitable for genetic improvement of grapevine for downy mildew resistance, where stacking different resistance
loci is fundamental to avoid resistance breakdown (adaptation) by
P. viticola [
10]. Before exploiting resistance
loci, it is important to characterize the pathogen population for the presence of adapted pathogen strains in the area where the plants will be grown [
23]. Here, the ability to infect grapevine accessions with different susceptibility profiles and several fitness components (disease incidence and severity, latency period, t
50, production, and viability of resting spores) were investigated in
P. viticola isolates of Northern Italy, an area where downy mildew frequently causes severe damage.
The experimental inoculation of the 72
P. viticola isolates on PN (susceptible
V. vinifera reference), B (interspecific hybrid harboring the
Rpv3.1 locus), and MG (resistant
V. vinifera possessing the
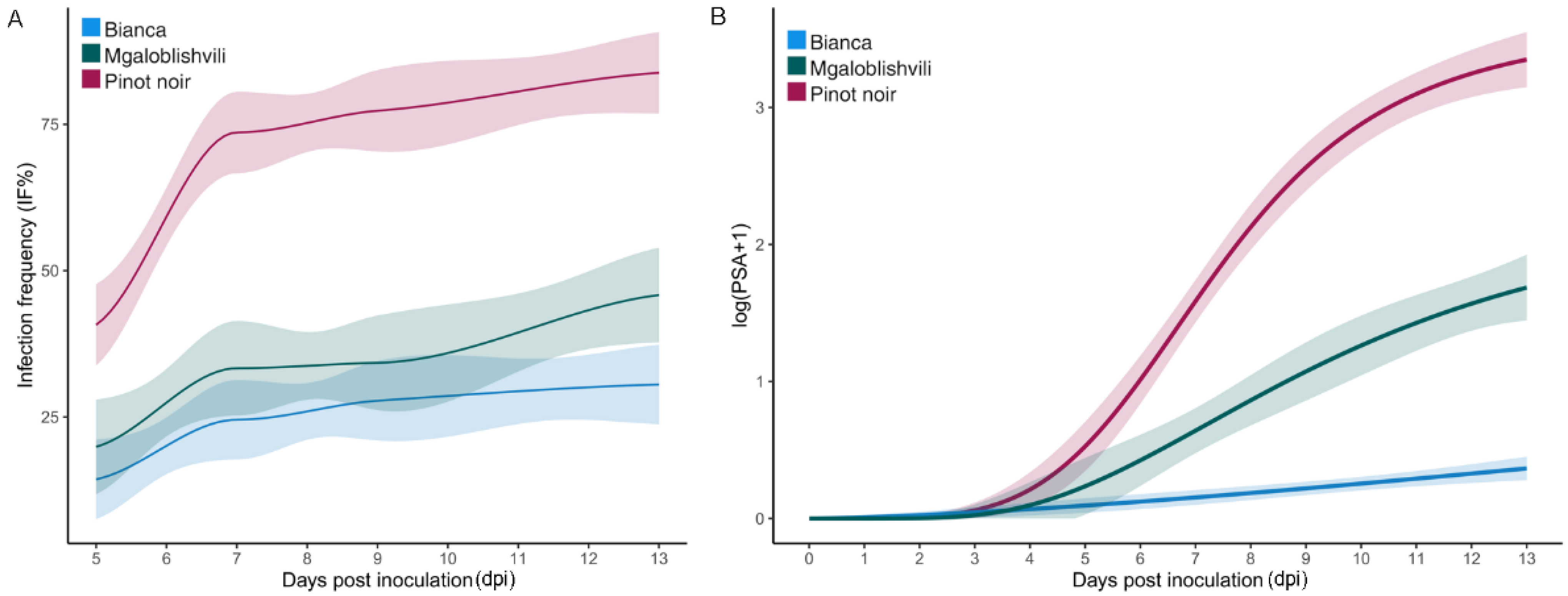
Rpv29 locus) resulted in a reduced infection frequency (IF) and disease severity (PSA) on the two resistant accessions compared to the susceptible one. Nevertheless, while the IF values on B and MG were analogous, the PSA values on MG were higher than those observed on B. IF is the parameter that better unifies the two resistant cultivars and it is also the most indicative parameter for comparing susceptible and resistant accessions; in fact, as a disease incidence index, it quantifies the ability to infect the host as a proportion or percentage of diseased individuals out of the total assessed [
26]. By contrast, the disease severity expresses the proportion or percentage of plant tissue affected by the disease. As evaluated in this study, severity measures the extent of the secondary inoculum produced by the pathogen and, indirectly, the extent of colonization.
Concerning the overall disease progress of the pathogen strains, the two resistant accessions did not differ from PN for LP, indicating that when infection occurs, the time to first sporulation is not affected in the selected resistant cultivars, contrarily to what was observed in other studies [
27]. What changed between PN and B was the slope of the curve and the disease severity, indicating that the pathogen development was both slowed down and limited. However, the slope of the curve and t
50 (the time to reach 50% of the total PSA) of MG were similar to those of PN, even if the final disease severity was three times reduced. This indicates that the response of MG is based on a reduction rather than a slowdown of the pathogen infection. Indeed, the time-course investigation of
P. viticola development in leaf tissues previously showed that the development of the pathogen is identical in PN and MG until 3 dpi, and it was only from this time-point onwards that the vegetative growth of the pathogen was hampered [
17]. The lower PSA and slope of the disease progress curve observed in B indicate a stronger limitation of the rate of disease development after the initial infection. This can be explained by the different characteristics of the two resistant varieties: the mechanism of resistance of B is based on the hypersensitive response (HR) and the presence of very low sporulation in the infected areas [
12,
15,
17]; MG response to
P. viticola, instead, does not lead to HR but to a limitation of the pathogen growth starting from 3 dpi [
17].
RP can be proposed as a new parameter for the evaluation of
P. viticola adaptation to plant resistance in addition to disease severity, which is commonly used as a phenotyping tool for the identification of resistant traits in grapevine [
28]. In fact, it easily estimates the measure in which the growth of the strain is affected by the plant considering, at the same time, the virulence level of the strain evaluated on a susceptible reference. Indeed, as visible in the PSA values achieved on PN, the different strains can be characterized by different infection capabilities (virulence). Looking at the disease severity (PSA) values of a strain on a resistant accession without considering its virulence on a susceptible reference can lead to a misidentification of an adapted strain. RP, on the contrary, enables the easy identification of potentially adapted strains. For instance, if a strain possesses similar PSA values on both susceptible and resistant plants (low RP value) it can be a candidate-adapted strain. On the contrary, if a strain possesses a high PSA on the susceptible plant and a low PSA on a resistant plant (high RP value), then it is likely a non-adapted strain. Furthermore, a simple bubble-chart representation of these values such as that performed in this study, easily shows whether the strain possesses a low or high virulence.
It could be argued that MG is not suitable for the genetic improvement of grapevine for downy mildew resistance since the disease severity is higher than that on B. However, it must be considered that B is an interspecific hybrid harboring the resistance genes of the American vines that co-evolved with
P. viticola, while MG is a pure
V. vinifera variety that met the pathogen only after its introduction into Europe at the end of the XIX century [
29]. The reason for the presence of resistance traits in MG has not been elucidated yet. What is known is that during the interaction with
P. viticola, MG overexpresses genes encoding for DAMP, PAMP and an effector receptor and other genes associated with signal transduction and synthesis of chemical (terpenes) and physical (cell wall thickening) barriers [
17,
25,
30]. The possibility of exploiting MG genes for resistance to
P. viticola has two advantages: the first is that crossings are easily made within the species (and not between species, as happens with other
Vitis species); the second is that MG can be used for stacking new traits into already obtained crosses, contributing to the achievement of a durable resistance [
31].
Looking in detail at the behavior of the single isolates on the resistant cultivars, it was possible to see that about 40% of the strains show no signatures of adaptation, since they were not able to infect B nor MG. Of the remaining strains, only 12.5% showed adaptation to MG and 5.6% to B, as demonstrated by the RP values below 25% for MG and 85% for B. Only one strain (CAST7) showed adaptation to both B and MG and should be better characterized because it groups in PC4. Almost all the adapted strains were not very aggressive, as testified by the reduced growth on the reference cultivar PN, and therefore they could possess fitness penalties compared to non-adapted strains within the pathogen population in the field. The studies on the effector array of
P. viticola during the interaction with
vinifera and non-
vinifera species are under constant evolution [
25,
32,
33,
34]. It would be interesting in the future to identify the effectors expressed by these two strains.
Pathogenicity tests showed that most of the isolates (74%) were able to infect two (PC2 and PC3 strains) or three (PC4 strains) cultivars, while only 26% of the isolates (PC1) were able to infect PN alone. Even if PC3-4 strains were able to infect the two resistant cultivars, it must be pointed out that they induced lower disease intensities. Indeed, PC2-4 showed a reduced PSA on B and MG compared to PN, and PC4 was also characterized by a reduced IF on B. Considering the results obtained at each time-point on the individual cultivars, no significant differences could be found among the different pathogenicity classes; for instance, PC1-4 showed analogous LP length, IF, and PSA values. Since the different PCs had analogous disease parameters on the individual cultivars, we can assume that PCs have similar fitness and that the lower PSA on B and MG compared to PN is a consequence of the resistant response of the plants. The only significant difference among PCs was found in the IF values on PN, where PC3 individuals showed a 20–33% significantly lower disease incidence than PC1, with PC2 and PC4 in between. This could indicate that the strains able to infect plants with Rpv29 possess a slight fitness penalty that leads to a limited number of infections on PN.
The oospore density achieved by experimentally inoculating
P. viticola mating pairs on detached grapevine leaves in this study (5–8 oospores/cm
2) was quite a lot lower than that obtained from naturally infected leaves in vineyards in a previous study (308.5 oospores/cm
2) [
35]. This difference could be due to the different plant tissues’ characteristics; in the laboratory, we inoculated detached leaf tissues that were kept in Petri dishes for 21 days, while in the field the oospores initially differentiate on living leaves that receive nutrients from the plant and probably, in these conditions, the pathogen has more energy for the sexual reproduction process. The very low number of oospores retrieved from the laboratory samples did not allow us to assess their germinability and infectivity. Indeed, the oospores normally germinate at a relatively low percentage (0.1–22%) [
36,
37,
38], and probably a higher number of oospores could have helped us in assessing this very important parameter. Despite this, the results obtained in this study highlight that
P. viticola had potential penalties in the oospore production on resistant cultivars in one crossing (CASB11xCAST7) out of three. This is partly in line with the results reported in a previous study on samples collected in the field, where the oospore production was four times higher on
V. vinifera cultivars than on plants harboring
Rpv1 and
Rpv3 loci [
39]. It must be pointed out that, in our case, we used single isolates weekly propagated on detached leaves to produce oospores and the absence of significant differences in the density on the different cultivars could be originated either from the low amount of oospores produced or the different characteristics of the isolates, which should be better investigated. The viability of the oospores differentiated in vineyards and overwintered in the same controlled conditions adopted in this study, ranged from 10–40% [
36]. The very high values (67–93%) obtained in the viability tests in laboratory conditions showed that most of the oospores successfully completed their maturation period, reaching a stable structure, while few oospores degenerated. This could be due to the fact that the oospores were kept in controlled environmental conditions, while in the field the oospores undergo variable environmental conditions that can negatively affect their maturation process and viability at the initial stages. Since the oospores formed by the different mating pairs possessed high and analogous viability on all the cultivars, our results confirm that resistant cultivars seem to act on the differentiation more than on the viability of the oospores, as already observed [
39]. In the future, it would be useful to enlarge the collection and characterization of
P. viticola oospores, differentiated both in the field and laboratory, for their amount, viability, germinability, and infectivity. The study of field-differentiated oospores has the advantage of investigating the response of the different cultivars to natural conditions, while laboratory crosses allow us to precisely evaluate the effect of the cultivars on the individual mating pairs. An improvement in the protocol for the experimental production of oospores in the laboratory (e.g., by inoculating
P. viticola strains on plants or increasing the number of inoculated leaves) is, however, necessary to achieve enough material for all such experimental activities.