1. Introduction
The cell wall is a protective structure surrounding the cell membrane of some types of eukaryotic and prokaryotic cells, including land plants and algae. In land plants, the structure, synthesis, and function of distinct types of cell and species have been identified extensively for decades [
1]. The fundamental roles of the plant cell wall involve morphogenesis and growth, providing mechanical strength, defending against biotic and abiotic stresses, adaptation, and so on [
2]. The plant cell wall mainly contains different types of polysaccharides, such as cellulose, hemicellulose, and pectin, and a small number of structural proteins. Many experimental results indicate that the cell wall is formed by the successive deposition of pectic substances, hemicellulose, cellulose, and lignin during cell wall differentiation, with the cell wall assembly process itself being irreversible, to form a dynamic network to bear the high intracellular turgor pressure. The polysaccharide in the plant cell wall was found to essentially involve in many facets of growth and development, such as cell expansion polarity and the thickening of cell walls through the deposition of polysaccharides [
2].
Like land plants, macroalgae such as red, green, and brown algae consist of a polysaccharide-rich cell wall, which is approximately 4–76% of algal dry weight [
3]. Among them, brown algae (Phaeophyta) represent the largest biomass-producing organism, containing more than 250 genera and 1500–2000 species, in the marine ecosystem [
4], and have different types of polysaccharides in common with plants (cellulose), animals (sulfated fucans), and some bacteria (alginates) [
1,
5]. The main polysaccharides in brown algal cell walls are fucoidan and alginates, which encompass up to 45% of algal dry weight, while cellulose only accounts for 1–8% of algal dry weight [
6]. Regarding the functionality of the main cell wall polysaccharide of brown algae, alginate fine structure likely contributes to cell wall rigidity, while sulfated polysaccharides, such as fucoidan, probably have a main role in the osmotic regulation [
6]. Furthermore, preceding studies of brown algae found that the cell wall contributed to cell adhesion [
7], cell expansion [
8], cell development with cell differentiation in growing filaments [
9], polar axis fixation, and cell fate [
10]. Although the cell wall was found to be involved in growth and development, little attention has been paid to date to the involvement of cell wall polysaccharides in growth.
Herburger et al. investigated the cell wall polysaccharide content in charophyte green algae changes during growth [
11]. For instance, the pectin content of
Zygnema and hemicelluloses content in
Klebsormidium increase with increasing cell age. Furthermore, laminarans and fucoidans content of brown algal cell wall was found to be significantly higher in mature
Laminaria cichorioides in autumn compared to unripe algae, suggesting that algal polysaccharides would play a key role in cell growth [
12].
Cladosiphon okamuranus, Okinawa Mozuku in Japanese, is one of the most important edible brown algae in Japan and an excellent source of fucoidan for all brown algae varieties. We found the presence of fucoidan as hemicellulose in the cell wall from
C. okamuranus, suggesting that this fucoidan might be involved in reinforcing cell wall structure by cross-linking to cellulose [
13]. Thus, we hypothesized that cell wall polysaccharides, especially the fucoidan as hemicellulose, had a significant effect on the growth.
Cladosiphon okamuranus is widely cultivated in Okinawa, Japan, from November to June, and its harvest season generally starts in January and ends in June, although this varies depending on the location. Here, we analyzed the cell wall structure of immature thalli of
C. okamuranus (young thalli), which was harvested at the beginning of the harvest season, and mature thalli (old thalli), which was harvested at peak season or later. Our present study focuses on how cell wall polysaccharides contribute to growth in brown algae, with special reference to
C. okamuranus, which is identified as an abundant source of fucoidan for all brown algae.
3. Discussion
Many studies have proven that the polysaccharides in plant cell walls are involved in many essential facets of growth and development, such as cell expansion polarity and the thickening of cell walls through the deposition of polysaccharides [
2]. However, little is known about the involvement of polysaccharides in the growth and development of algae. The present study provides new insights into the growth of brown algal cell walls by showing that the cell wall polysaccharides play a leading role in the process of growth, considering the physicochemical characteristics of young and old
C. okamuranus. Firstly, we examined the physical characteristics and identified the several variations between young and old thalli. The young thalli were lighter in color and felt slimier to the touch compared to the old thalli. It was found that the molecular weight and sulfate content of the fucoidan might be the reason for the variation in the sliminess of thalli harvested at different time [
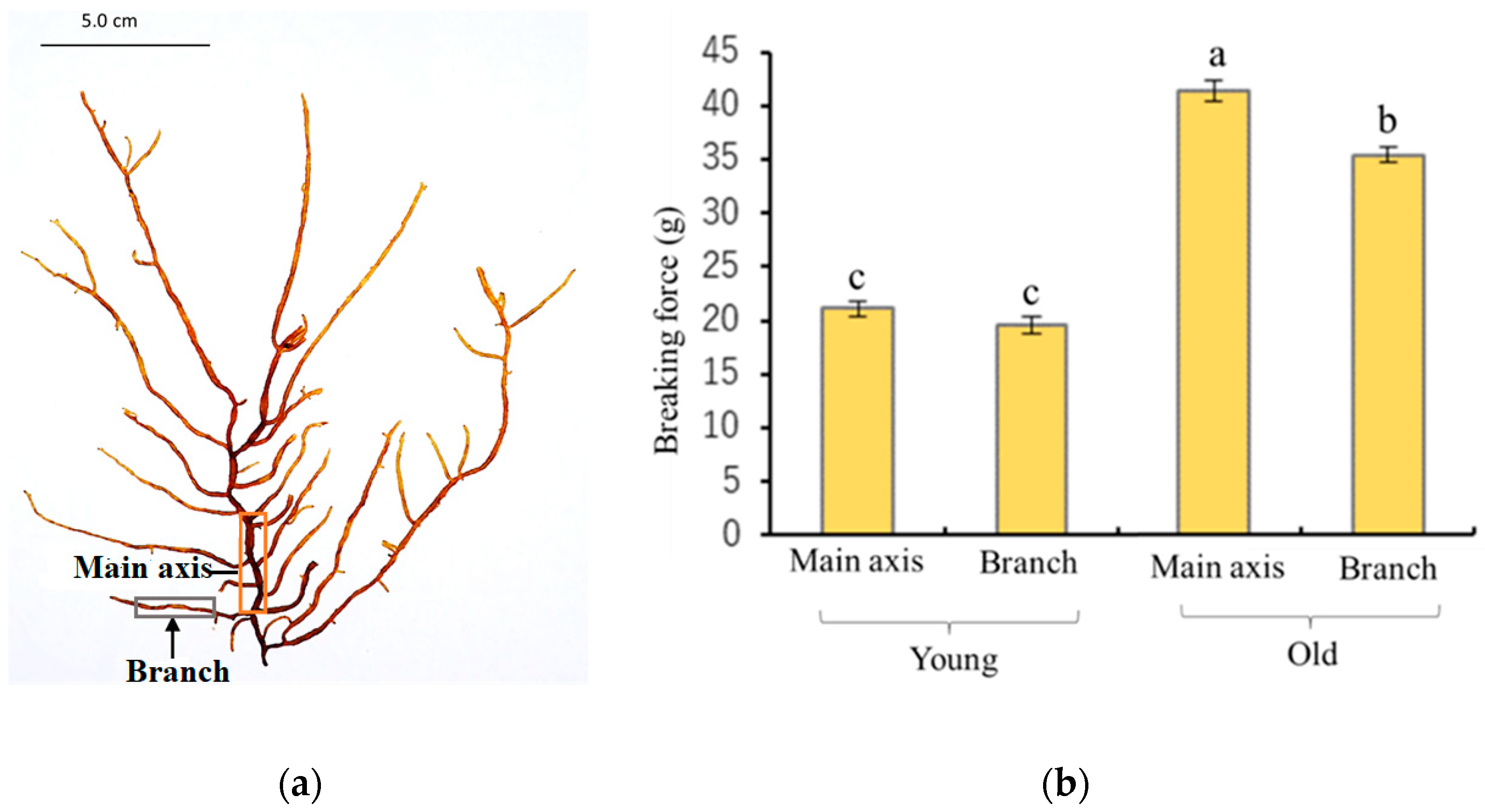
14]. Furthermore, the breaking strength of the main axis and lateral branches of old thalli were significantly higher than that of young thalli (
Figure 1b). In anatomical analysis, it was confirmed that the basic structures of the young and old thalli were almost the same except for the assimilatory filaments, suggesting that the generation of tension strength may be influenced by microscale factors such as the thickness and/or composition of the cell wall (
Figure 2). Preceding studies have consistently found that old algal tissue was significantly stronger than newly formed tissue, which has been suggested to be due to the differences in tissue composition or structure [
15,
16,
17]. These biomechanical characteristics of the different tissues would corelate with prominent differences in moisture content, cortical layer thickness, cell wall thickness, and the number of insoluble polysaccharides such as cellulose, hemicellulose, and fucoidan [
15]. According to the results of our study, the moisture content was lower in old thalli (91.4%) than young (93.2%), while yield of AIR was higher in old thalli (5.5%) than young thalli (2.8%) (
Table 1), suggesting that cell wall material content increases and moisture content decreases during growth. A similar trend was reported in moisture content and crude fucoidan content after March in
C. okamuranus and kelp species such as
L. digitata,
L. hyperborea,
Saccharina latissima, and
Alaria esculenta [
14,
18]. Therefore, it can be proven that algal tissue becomes stronger in line with the deposition of cell wall materials and the reduction in water content during the growth process.
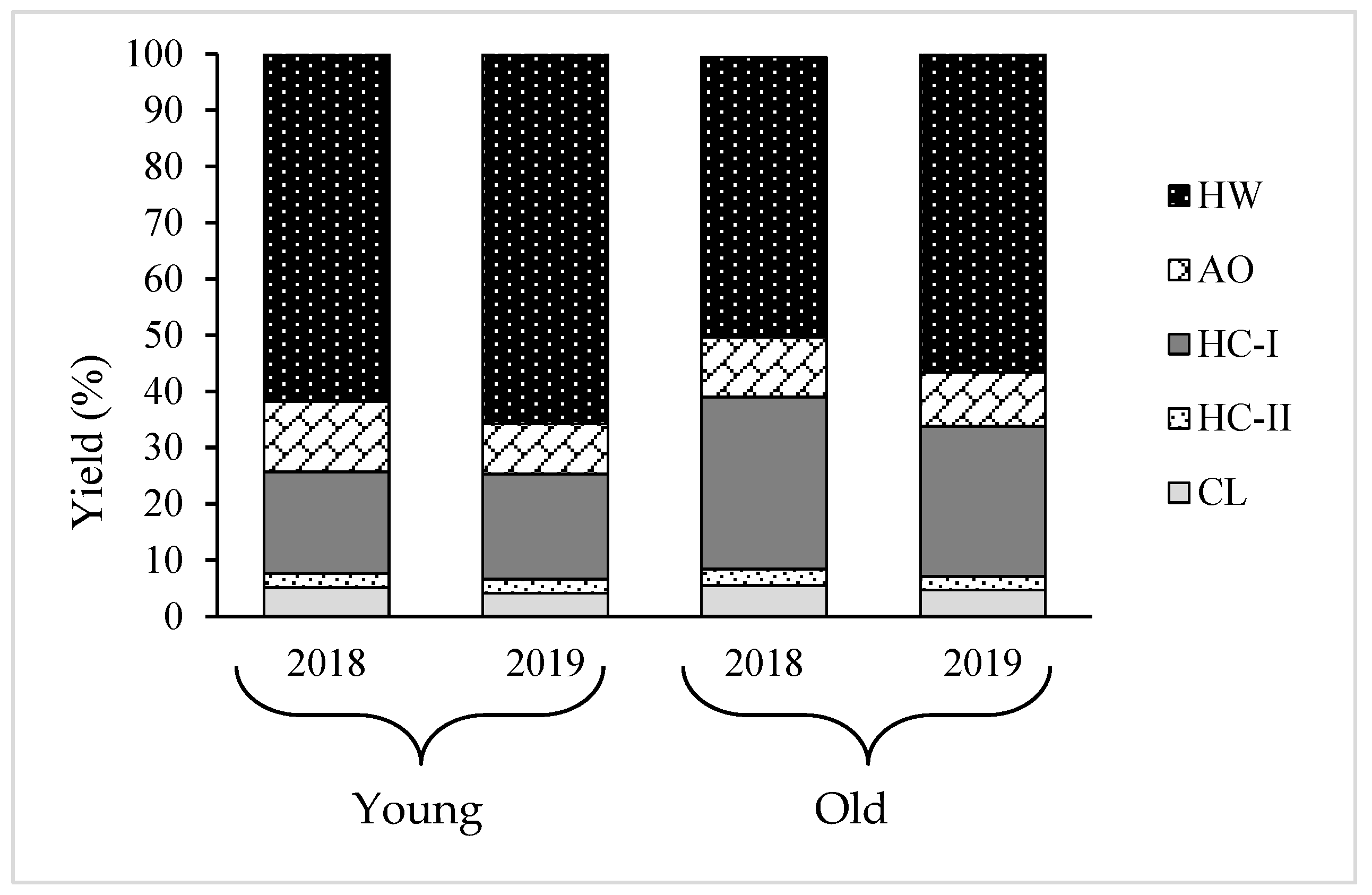
Since increase in the cell wall material was observed with the growth of thalli, the variation in the cell wall composition needs to be analyzed to understand which polysaccharides mostly engage with the process of growth. Therefore, AIR was fractionated into five fractions: HW, AO, HC-I, HC-II, and CL. The results show that almost 80% of the total recovery cell wall from AIR was HW and HC-I, and the amount of HC-I increased, while HW decreased during growth (
Table 2 and
Table 3). Our previous study found that HW and HC-I mainly contained fucoidan composed of Fuc, GlcA, and sulfate in molar ratios of 1.0:0.3:0.9 and 1.0:0.2:0.3, respectively, and their structure differed in terms of contents of sulfate and Xyl, MW, and profile of the small angle x-ray scattering [
13]. Highly sulfated polysaccharides in HW were typically fucoidan, and have been suggested to be weakly held in the cell wall matrix and possibly involved in osmotic regulation in brown algae [
19], while HC-I contained 1,4-linked Xyl and 1,4-linked Fuc, as well as components of typical fucoidan, suggesting that these are likely to be 1,4-xylan and/or 1,4-fucan, which may be involved in reinforcing cell wall structure by cross-linking to cellulose as hemicellulose in terrestrial plants, keeping CL microfibrils separated, and controlling cell wall expansion [
20]. This hypothesis is further supported by the results of this study. The young thalli are rich in matrix polysaccharides; HW and the old thalli possessed a larger amount of fibrillar polysaccharides, HC-I, compared to young thalli, suggesting that cell wall thickening in
Cladosiphon okamuranus during growth is characterized by an increase in the HC-I content. A similar scenario was reported in land plants, but with different types of polysaccharide. For instance, pectic substances are mostly deposited at a young stage, while the contents of compositional sugars from cellulose and hemicelluloses increase with the maturation of the secondary wall [
21]. However, substantial variation was not found in the yield of other minor fractions, AO, HC-II, and CL, between old and young. In contrast, variations in the cellulose amount have been shown to cause changes in both the strength and rigidity of plant tissues [
22,
23]. Cellulose is the most abundant major polysaccharide, representing up to 50% of terrestrial plants’ cell walls, but only makes up 5% of the cell wall in brown algae, both young and old thalli, in this study [
6].
The further structural analysis of HW and HC-I was conducted related to young and old thalli. Sugar composition analysis showed that HW and HC-I in young and old thalli contained mainly fucoidan composed of Fuc, GlcA, and sulfate in molar ratios of 1.0:0.3:0.6~0.7 and 1.0:0.3:0.2~0.3, respectively, with a trace amount of Gal, Glc, and Xyl. Although there were variations in the sulfate and xylose content between HW and HC-I, there were no significant differences observed in the sugar composition and sulfate content between young and old thalli. However, fucoidan appears to be more strongly bound to cellulose in the old thalli cell wall compared to the young thalli cell wall according to the Glc:Fuc molar ratio in the CL fraction (
Table 4 and
Table 5). We believe that a large quantity of the fucoidan in HC-I in old thalli might be bound strongly to the cellulose. Moreover, HW and HC-I in young and old thalli were further characterized by the determination of their molecular weight, which was found to be larger in old thalli than young. Zvyagintseva et al. also found that the molecular weight of fucoidan in 2-year-old
L. cichorioides was significantly higher (20–30 kDa) than a 0.8-year-old sample (8–10 kDa) [
12]. It appears that the high molecular weight of old thalli is to be due to an increase in its degree of polymerization during growth [
24]. Further fractionation was carried out for HW and HC-I in both young and old thalli using anion exchange chromatography to explore their purity and heterogeneity based on their charge density. These results further indicated that purified HW contains two main fractions, HW-FT and HW-NaCl, but that the yield of HW-NaCl fractions in young thalli were higher than that of old, while HC-I eluted only HC-I-NaCl, and both young and old thalli appeared to be similar (
Table 6). These results confirmed that there were many structural variations in the main fractions, HW and HC-I, which may be related to the localization of young and old thalli, which is involved in the process of growth.
4. Materials and Methods
4.1. Algal Sample
Cladosiphon okamuranus, cultivated in Chinen, Nanjo City, Okinawa Prefecture (26°08′05′′ N, 127°48′19′′ E), was harvested after about 40 days (young thalli) and about 90 days (old thalli), after planting in the sea, in 2018 and 2019, respectively.
4.2. Measurement of Moisture Content
About 0.1 g of sample was taken after harvesting, and moisture content was measured using a moisture content analyzer (MOC-63U SHIMADZU, Kyoto, Japan).
4.3. Measurement of Tensile Strength
The main axis was defined as the thickest branch growing from the base of the thalli, and the lateral branches were defined as branches departing from the main axis, as shown in
Figure 1a. The tensile strength of the main axis and lateral branches was determined using a tensile tester (STB1225S, A&D Co., Ltd., Tokyo, Japan). About 10 mm of thalli was fixed between two clamps and stretched by raising the upper clamp at a speed of 20 mm/min until the thalli broke.
4.4. Light Microscopy
The samples fixed with 100% ethanol were used for making cross sections without washing. The samples were observed through a light microscope (BX53F2, OLYMPUS, Tokyo, Japan) equipped with a digital camera (WRAYCUM NOA2000, WRAYMER, Osaka, Japan).
4.5. Preparation of Alcohol Insoluble Residue (AIR)
Each algal sample (50 g) was ground using a blender in 4 volumes of ethanol (200 mL), followed by centrifugation at 8000×
g at 25 °C for 15 min; then, the precipitate was sequentially treated with 80% ethanol, 100% ethanol, and methanol:chloroform (1:1, v:v), followed by acetone. Suction filtration was performed and residue was dried at room temperature and used as AIR [
13].
4.6. Fractionation of Cell Wall Polysaccharides
The fractionation procedure was based on the different solubilities of the polysaccharides from brown seaweeds, as described in our previous study [
14]. Briefly, AIR was sequentially treated with hot water (HW), 0.25% ammonium oxalate (AO), 4% KOH, and 24% KOH to produce 4 fractions: HW, AO, hemicellulose-I (HC-I), and HC-II. The final residue was collected as cellulose (CL). After neutralizing HC-I and HC-II using acetic acid, AO, HC-I, and HC-II were dialyzed, lyophilized, and used for analysis. The CL was washed with water after neutralizing with acetic acid, and then lyophilized.
4.7. Chemical Composition Analysis
Total sugar and UA were determined using the phenol–sulfuric acid method using Fuc as a standard [
25], and the
m-hydroxybiphenyl method using GlcA as a standard [
26], respectively. Total polyphenols were quantified using the Folin–Ciocalteu method, using gallic acid as a standard [
27]. Protein content was measured using bicinchoninic acid assay (BCA) following the manufacturer’s instructions of a BCA Protein Assay Kit (Takara Bio Inc., Shiga, Japan). The calibration curve was prepared using bovine serum albumin.
To estimate SO
3−, the sample was hydrolyzed in 2 M trifluoroacetic acid at 121 °C for 1 h, hydrolysate was subjected to high-performance liquid chromatography with an AS4A-SC column (4 mm × 250 mm, Dionex Co., Tokyo, Japan). The column was eluted at 1.5 mL/min at room temperature with a buffer containing 1.7 mM NaHCO
3 and 1.8 mM Na
2CO
3 [
28]. The SO
3− content in the sample was calculated from a calibration curve using Na
2SO
4 as a standard [
13].
4.8. Sugar Composition Analysis
To analyze sugar composition, samples were hydrolyzed to be used for SO
3− estimation, while the AIR and CL were treated with ice-cold 72% (
w/
w) H
2SO
4 at 4 °C for 1 h with sonication, followed by hydrolysis with 2 N H
2SO
4 at 121 °C for 1 h [
29,
30]. Monosaccharide in the hydrolysate was analyzed using high-performance anion exchange chromatography coupled with a pulsed amperometric detector (HPAEC-PAD), using a Carbo Pac PA1 column (4 mm × 250 mm, Thermo Fisher Scientific, MA, USA). The column was eluted at a flow rate of 1 mL/min at 35 °C with 14 mM NaOH for neutral sugar, followed by a linear gradient program of 0–250 mM CH
3COONa in 100 mM NaOH for UA.
4.9. Determination of Molecular Weight (MW)
The MW of HW and HC-I were determined by size-exclusion chromatography (LC-6A; Shimadzu Co., Kyoto, Japan) equipped with a TSKgel G5000 PWXL column (7.8 mm × 300 mm, Tosoh Co., Tokyo, Japan) and a refractive index detector RID-10A [
31]. The column was eluted by 0.2 M NaCl at a flow rate of 0.3 mL/min at 40 °C. Pullulan P-10 (MW = 0.96 × 10
4), P-50 (4.71 × 10
4), P-200 (20.0 × 10
4), and P-800 (70.8 × 10
4) (Showa Denko Co., Tokyo, Japan) were used as standards.
4.10. Anion Exchange Chromatography
For further analysis of HW and HC-I extracted from young and old thalli, anion exchange chromatography was used. A total of 50 mg of each fraction was dissolved in 20 mM Tris-HCl (pH 7.4) and applied onto a DEAE–Sephacel column (2.5 × 10 cm, GE Healthcare, Uppsala, Sweden) equilibrated with the same buffer. The column was washed with 20 mM Tris-HCl (pH 7.4) and then eluted with a linear gradient of 0–3.5 M NaCl in 20 mM Tris-HCl (pH 7.4), followed with 3.5 M NaCl in the same buffer.
4.11. Statistical Analysis
Data were expressed as mean ± standard deviation of three determinations. Statistical comparison was performed via a one-way analysis of variance followed by Tukey’s test. Probability values of less than 0.05 (p < 0.05) were considered significant.
5. Conclusions
In conclusion, differences in the physical and chemical composition of young and old thalli of C. okamuranus were observed related to their growth. Regarding the characteristics of thalli, the old thalli were stronger, darker in color, and felt less slimy to the touch compared to the young thalli. The moisture content was lower in old thalli than young thalli, while the yield of AIR was higher in old thalli than young thalli. Almost 80% of the total recovery cell wall from both young and old thalli was HW, and HC-I contained mainly fucoidan composed of Fuc, GlcA, and sulfate in molar ratios of 1.0:0.3:0.6~0.7 and 1.0:0.3:0.2~0.3, respectively. However, the ratio of HW and HC-I was different between young and old thalli; in old thalli, the yield of HC-I increased while the yield of HW decreased. These results suggested that the cell wall structure of C. okamuranus changes during growth, such as the ratios of the two main components, HW and HC-I. Furthermore, it was suggested that the strength of thalli during growth was significantly affected by HC-I.