Effects of Graphene Oxide on the Growth and Photosynthesis of the Emergent Plant Iris pseudacorus
Abstract
:1. Introduction
2. Results and Discussion
2.1. Biomass and Photosynthetic Pigment Contents of I. pseudacorus
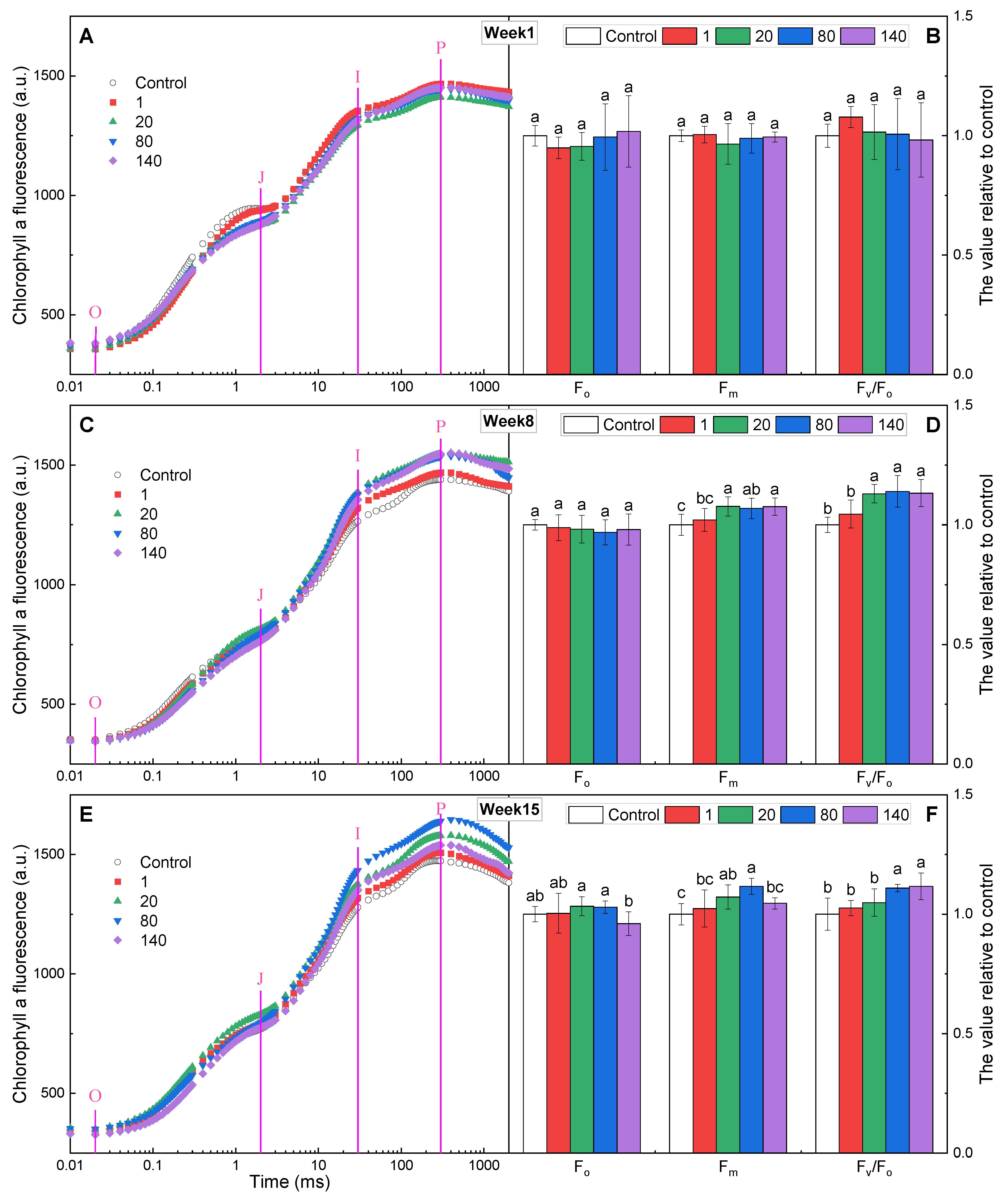
2.2. Chlorophyll Fluorescence Rise Kinetics OJIP Curves
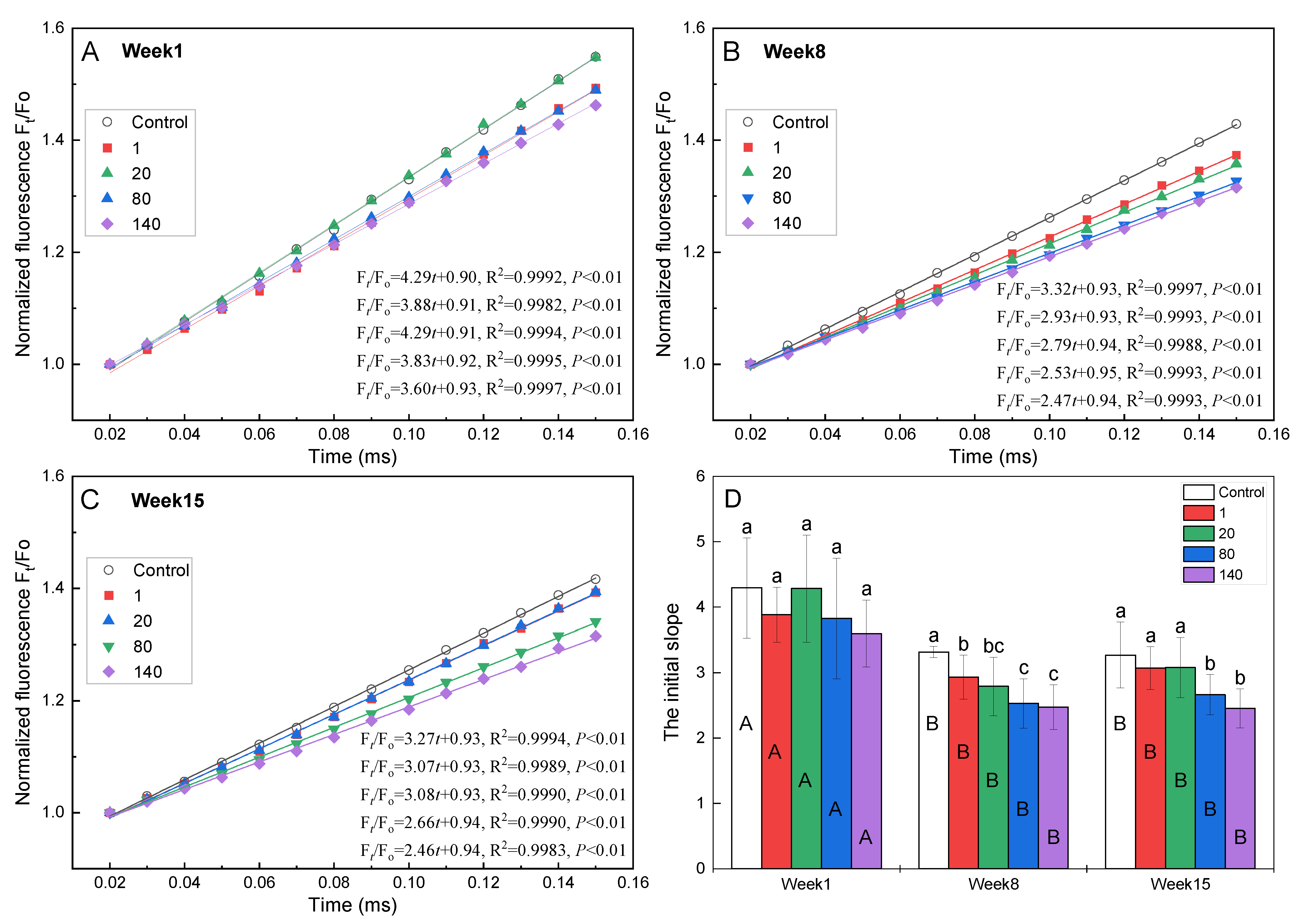
2.3. The Initial Slope of the Standardized Fluorescence Transient Ft/FO
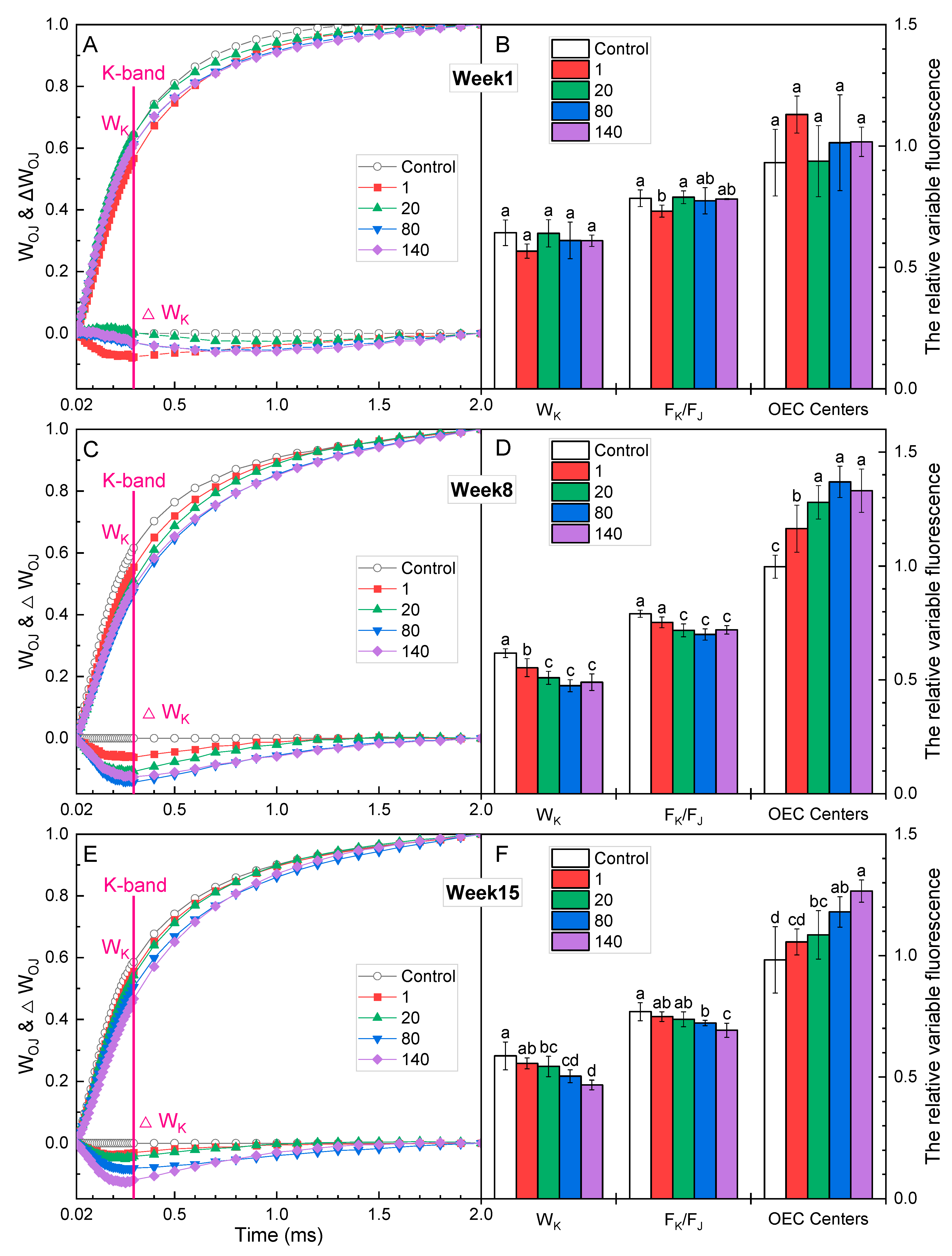
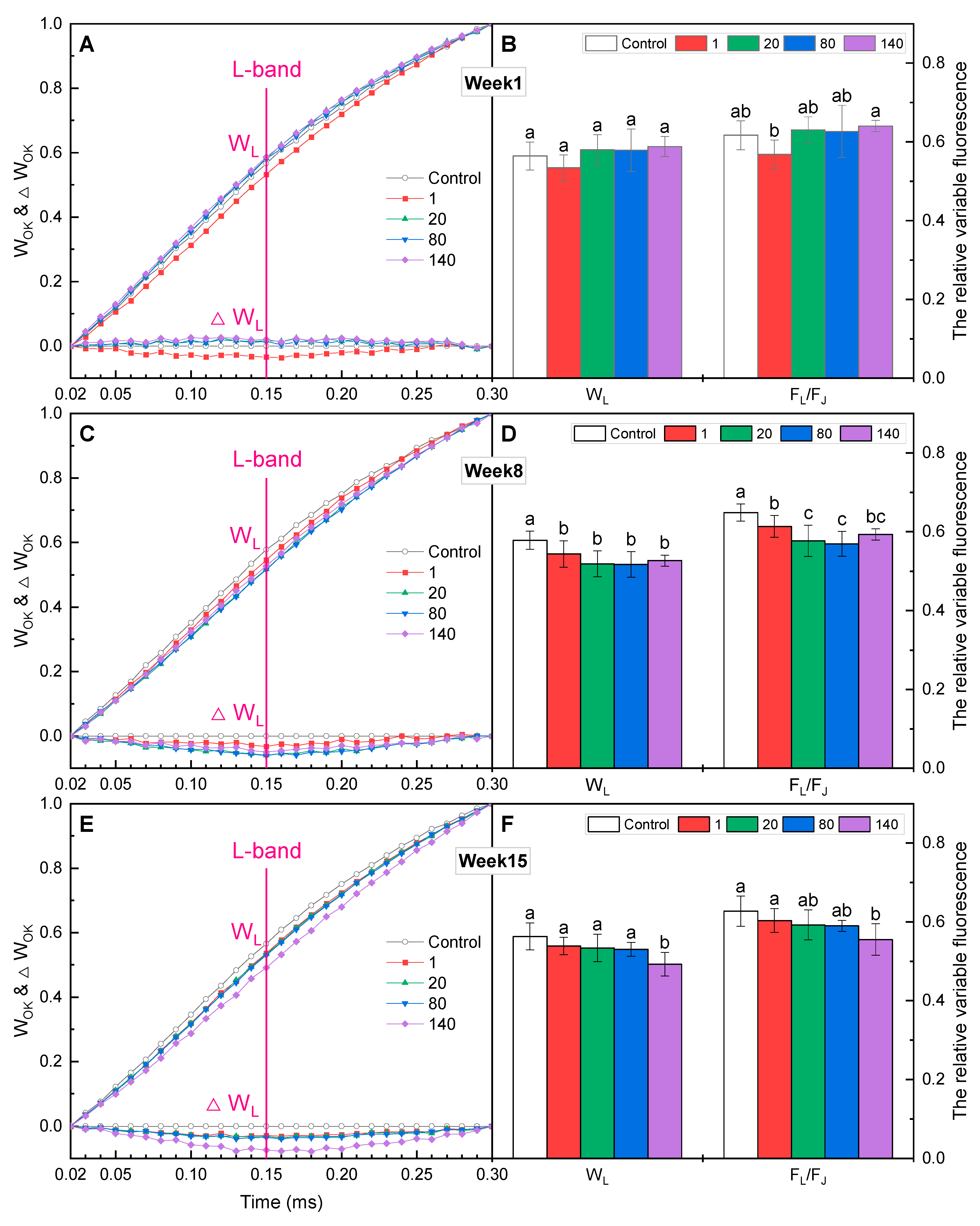
2.4. K-Band and L-Band
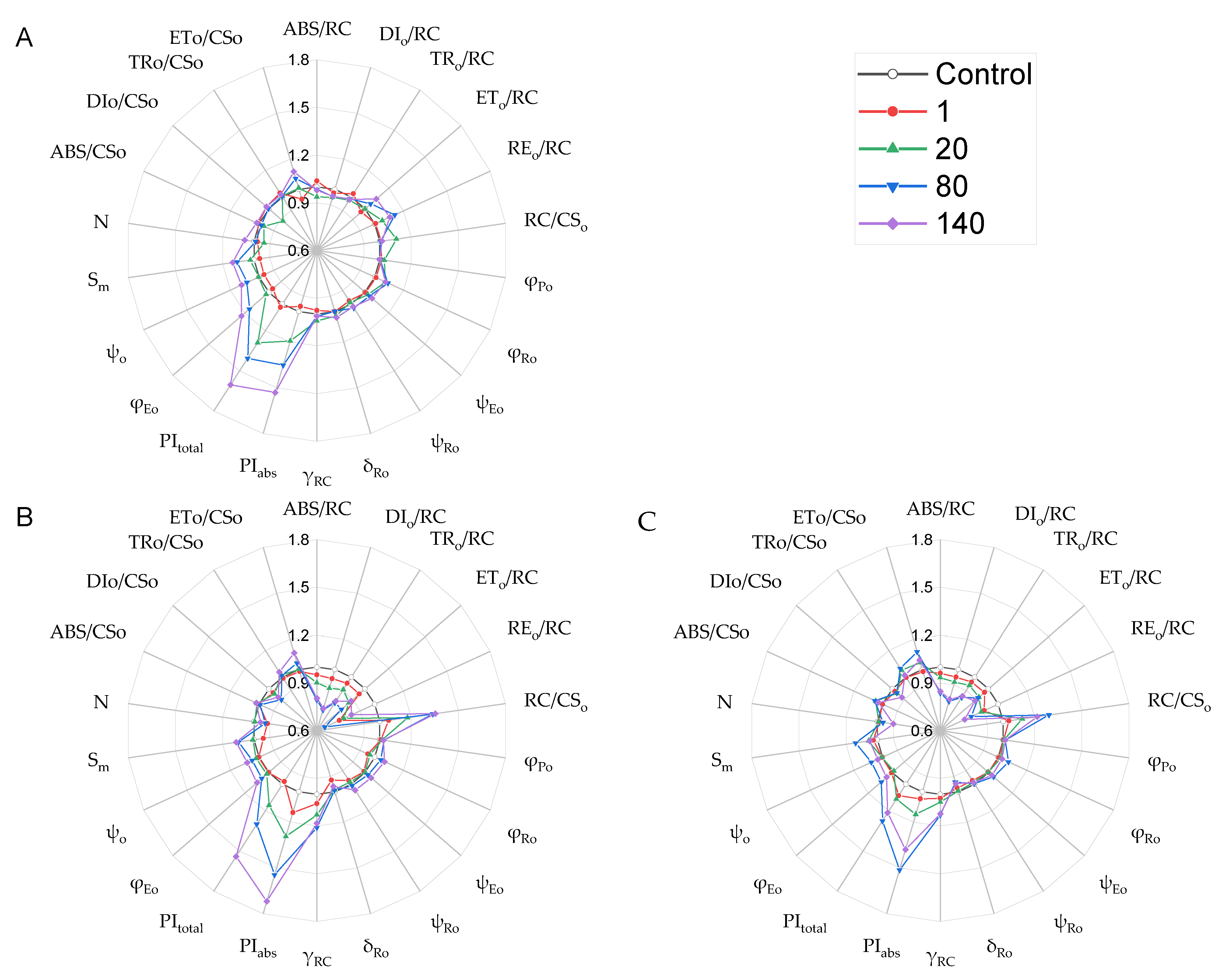
2.5. Chlorophyll a Fluorescence Parameters
2.6. Mineral Element Contents in I. pseudacorus
3. Materials and Methods
3.1. Materials and Treatments
3.2. Analysis of Biomass and Photosynthetic Pigment Contents of I. pseudacorus
3.3. Measurement of Chlorophyll a Fluorescence in I. pseudacorus
3.4. Analysis of Nutrient Levels of I. pseudacorus
3.5. Data Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dimiev, A.; Kosynkin, D.V.; Alemany, L.B.; Chaguine, P.; Tour, J.M. Pristine graphite oxide. J. Am. Chem. Soc. 2012, 134, 2815–2822. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Chen, L.; Cheng, S.; Zhu, L.; Liu, L.; Wen, P.; Zhou, L.; Xue, W.; Lu, S.; Zhang, W.; et al. An overview of light-mediated impact of graphene oxide on algae: Photo-transform, toxicity and mechanism. Water 2022, 14, 2997. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, Y.; Zhang, X.; Liao, W. Environmental transformation of graphene oxide in the aquatic environment. Chemosphere 2021, 262, 127885. [Google Scholar] [CrossRef] [PubMed]
- Yao, N.; Li, C.; Yu, J.; Xu, Q.; Wei, S.; Tian, Z.; Yang, Z.; Yang, W.; Shen, J. Insight into adsorption of combined antibiotic-heavy metal contaminants on graphene oxide in water. Sep. Purif. Technol. 2020, 236, 116278. [Google Scholar] [CrossRef]
- Yan, C.; Huang, J.; Cao, C.; Li, X.; Lin, X.; Wang, Y.; Qian, X. Iris pseudacorus as precursor affecting ecological transformation of graphene oxide and performance of constructed wetland. J. Hazard. Mater. 2022, 436, 129164. [Google Scholar] [CrossRef]
- Martin-Folgar, R.; Esteban-Arranz, A.; Negri, V.; Morales, M. Toxicological effects of three different types of highly pure graphene oxide in the midge Chironomus riparius. Sci. Total Environ. 2022, 815, 152465. [Google Scholar] [CrossRef]
- Paital, B.; Guru, D.; Mohapatra, P.; Panda, B.; Parida, N.; Rath, S.; Kumar, V.; Saxena, P.S.; Srivastava, A. Ecotoxic impact assessment of graphene oxide on lipid peroxidation at mitochondrial level and redox modulation in fresh water fish Anabas testudineus. Chemosphere 2019, 224, 796–804. [Google Scholar] [CrossRef]
- Dasmahapatra, A.K.; Tchounwou, P.B. Histopathological evaluation of the interrenal gland (adrenal homolog) of Japanese medaka (Oryzias latipes) exposed to graphene oxide. Environ. Toxicol. 2022, 37, 2460–2482. [Google Scholar] [CrossRef]
- Cruces, E.; Barrios, A.C.; Cahue, Y.P.; Januszewski, B.; Gilbertson, L.M.; Perreault, F. Similar toxicity mechanisms between graphene oxide and oxidized multi-walled carbon nanotubes in Microcystis aeruginosa. Chemosphere 2021, 265, 129137. [Google Scholar] [CrossRef]
- Zhao, J.; Cao, X.; Wang, Z.; Dai, Y.; Xing, B. Mechanistic understanding toward the toxicity of graphene-family materials to freshwater algae. Water Res. 2017, 111, 18–27. [Google Scholar] [CrossRef]
- Banchi, E.; Candotto Carniel, F.; Montagner, A.; Bosi, S.; Bramini, M.; Crosera, M.; León, V.; Martín, C.; Pallavicini, A.; Vázquez, E.; et al. Graphene-based materials do not impair physiology, gene expression and growth dynamics of the aeroterrestrial microalga Trebouxia gelatinosa. Nanotoxicology 2019, 13, 492–509. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Fan, W.; Du, J.; Feng, W.; Dong, Z.; Liu, Y.; Zhou, T. The toxicity of graphene oxide affected by algal physiological characteristics: A comparative study in cyanobacterial, green algae, diatom. Environ. Pollut. 2020, 260, 113847. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Singh, S.; Singh, V.; Joung, D.; Dowding, J.M.; Reid, D.; Anderson, J.; Zhai, L.; Khondaker, S.I.; Self, W.T.; et al. Oxygenated functional group density on graphene oxide: Its effect on cell toxicity. Part. Part. Syst. Charact. 2013, 30, 148–157. [Google Scholar] [CrossRef]
- Zhang, P.; Guo, Z.; Luo, W.; Monikh, F.A.; Xie, C.; Valsami-Jones, E.; Lynch, I.; Zhang, Z. Graphene oxide-induced pH alteration, iron overload, and subsequent oxidative damage in rice (Oryza sativa L.): A new mechanism of nanomaterial phytotoxicity. Environ. Sci. Technol. 2020, 54, 3181–3190. [Google Scholar] [CrossRef] [PubMed]
- Malina, T.; Lamaczová, A.; Maršálková, E.; Zbořil, R.; Maršálek, B. Graphene oxide interaction with Lemna minor: Root barrier strong enough to prevent nanoblade-morphology-induced toxicity. Chemosphere 2022, 291, 132739. [Google Scholar] [CrossRef]
- Castro, V.L.; Clemente, Z.; Jonsson, C.; Silva, M.; Vallim, J.H.; de Medeiros, A.M.Z.; Martinez, D.S.T. Nanoecotoxicity assessment of graphene oxide and its relationship with humic acid. Environ. Toxicol. Chem. 2018, 37, 1998–2012. [Google Scholar] [CrossRef]
- Yan, C.; Huang, J.; Lin, X.; Wang, Y.; Cao, C.; Qian, X. Performance of constructed wetlands with different water level for treating graphene oxide wastewater: Characteristics of plants and microorganisms. J. Environ. Manag. 2023, 334, 117432. [Google Scholar] [CrossRef]
- Knauert, S.; Singer, H.; Hollender, J.; Knauer, K. Phytotoxicity of atrazine, isoproturon, and diuron to submersed macrophytes in outdoor mesocosms. Environ. Pollut. 2010, 158, 167–174. [Google Scholar] [CrossRef]
- Minuti, G.; Stiers, I.; Coetzee, J.A. Climatic suitability and compatibility of the invasive Iris pseudacorus L. (Iridaceae) in the Southern Hemisphere: Considerations for biocontrol. Biol. Control 2022, 169, 104886. [Google Scholar] [CrossRef]
- Mohsin, M.; Nawrot, N.; Wojciechowska, E.; Kuittinen, S.; Szczepańska, K.; Dembska, G.; Pappinen, A. Cadmium accumulation by Phragmites australis and Iris pseudacorus from stormwater in floating treatment wetlands microcosms: Insights into plant tolerance and utility for phytoremediation. J. Environ. Manag. 2023, 331, 117339. [Google Scholar] [CrossRef]
- Yadav, N.; Govindwar, S.P.; Rane, N.; Ahn, H.; Xiong, J.; Jang, M.; Kim, S.H.; Jeon, B. Insights on the role of periphytic biofilm in synergism with Iris pseudacorus for removing mixture of pharmaceutical contaminants from wastewater. J. Hazard. Mater. 2021, 418, 126349. [Google Scholar] [CrossRef] [PubMed]
- Ai, Y.; Wang, Y.; Song, L.; Hong, W.; Zhang, Z.; Li, X.; Zhou, S.; Zhou, J. Effects of biochar on the physiology and heavy metal enrichment of Vetiveria zizanioides in contaminated soil in mining areas. J. Hazard. Mater. 2023, 448, 130965. [Google Scholar] [CrossRef] [PubMed]
- Young, A.J. The photoprotective role of carotenoids in higher plants. Physiol. Plant. 1991, 83, 702–708. [Google Scholar] [CrossRef]
- Yan, C.; Huang, J.; Cao, C.; Wang, Y.; Lin, X.; Qian, X. Response of constructed wetland for wastewater treatment to graphene oxide: Perspectives on plant and microbe. J. Hazard. Mater. 2022, 422, 126911. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Lu, Y.; Goltsev, V.; Strasser, R.J.; Kalaji, H.M.; Wang, H.; Wang, X.; Chen, S.; Qiang, S. Comparative effect of tenuazonic acid, diuron, bentazone, dibromothymoquinone and methyl viologen on the kinetics of Chl a fluorescence rise OJIP and the MR820 signal. Plant Physiol. Biochem. 2020, 156, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Lazár, D. The polyphasic chlorophyll a fluorescence rise measured under high intensity of exciting light. Funct. Plant Biol. 2006, 33, 9–30. [Google Scholar] [CrossRef] [PubMed]
- Hassannejad, S.; Lotfi, R.; Ghafarbi, S.P.; Oukarroum, A.; Abbasi, A.; Kalaji, H.M.; Rastogi, A. Early identification of herbicide modes of action by the use of chlorophyll fluorescence measurements. Plants 2020, 9, 529. [Google Scholar] [CrossRef]
- Clairvil, E.; Martins, J.P.R.; Braga, P.C.S.; Moreira, S.W.; Conde, L.T.; Cipriano, R.; Falqueto, A.R.; Gontijo, A.B.P.L. Zinc and cadmium as modulating factors of the morphophysiological responses of Alternanthera tenella Colla (Amaranthaceae) under in vitro conditions. Photosynthetica 2021, 59, 652–663. [Google Scholar] [CrossRef]
- Xia, J.; Li, Y.; Zou, D. Effects of salinity stress on PSII in Ulva lactuca as probed by chlorophyll fluorescence measurements. Aquat. Bot. 2004, 80, 129–137. [Google Scholar] [CrossRef]
- Sun, C.; Xu, Y.; Hu, N.; Ma, J.; Sun, S.; Cao, W.; Klobučar, G.; Hu, C.; Zhao, Y. To evaluate the toxicity of atrazine on the freshwater microalgae Chlorella sp. using sensitive indices indicated by photosynthetic parameters. Chemosphere 2020, 244, 125514. [Google Scholar] [CrossRef]
- Strasser, R.J.; Tsimilli-Michael, M.; Srivastava, A. Analysis of the chlorophyll a fluorescence transient. In Chlorophyll a Fluorescence: A Signature of Photosynthesis; Papageorgiou, G.C., Govindjee, Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 321–362. [Google Scholar]
- Volgusheva, A.A.; Kukarskikh, G.P.; Antal, T.K.; Lavrukhina, O.G.; Krendeleva, T.E.; Rubin, A.B. Effect of dibromothymoquinone on Chlorophyll a Fluorescence in Chlamydomonas reinhardtii cells incubated in complete or sulfur-depleted medium. Biophysics 2008, 53, 378–385. [Google Scholar] [CrossRef]
- Jin, L.; Che, X.; Zhang, Z.; Gao, H. The relationship between the changes in Wk and different damage degree of PSII donor side and acceptor side under high temperature with high light in Cucumber. Plant Physiol. J. 2015, 51, 969–976. [Google Scholar]
- Tikkanen, M.; Mekala, N.R.; Aro, E. Photosystem II photoinhibition-repair cycle protects Photosystem I from irreversible damage. Biochim. Biophys. Acta 2014, 1837, 210–215. [Google Scholar] [CrossRef]
- Gao, J.; Li, P.; Ma, F.; Goltsev, V. Photosynthetic performance during leaf expansion in Malus micromalus probed by chlorophyll a fluorescence and modulated 820 nm reflection. J. Photoch. Photobio. B 2014, 137, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Le, Y.; Wang, C.; Zhu, X.; Wang, Z.; Xing, B. Photosynthetic response mechanisms in typical C3 and C4 plants upon La2O3 nanoparticle exposure. Environ. Sci. Nano 2020, 7, 81–92. [Google Scholar] [CrossRef]
- Goltsev, V.; Zaharieva, I.; Chernev, P.; Kouzmanova, M.; Kalaji, H.M.; Yordanov, I.; Krasteva, V.; Alexandrov, V.; Stefanov, D.; Allakhverdiev, S.I.; et al. Drought-induced modifications of photosynthetic electron transport in intact leaves: Analysis and use of neural networks as a tool for a rapid non-invasive estimation. Biochim. Biophys. Acta 2012, 1817, 1490–1498. [Google Scholar] [CrossRef]
- Zhou, X.; Zeng, Y.; Tang, Y.; Huang, Y.; Lv, F.; Liu, L.; Wang, S. Artificial regulation of state transition for augmenting plant photosynthesis using synthetic light-harvesting polymer materials. Sci. Adv. 2020, 6, c5237. [Google Scholar] [CrossRef]
- Yang, H.; Zhang, J.; Zhang, H.; Xu, Y.; An, Y.; Wang, L. Effect of 5-aminolevulinic acid (5-ALA) on leaf chlorophyll fast fluorescence characteristics and mineral element content of Buxus megistophylla grown along urban roadsides. Horticulturae 2021, 7, 95. [Google Scholar] [CrossRef]
- Zhang, X.; Cao, H.; Zhao, J.; Wang, H.; Xing, B.; Chen, Z.; Li, X.; Zhang, J. Graphene oxide exhibited positive effects on the growth of Aloe vera L. Physiol. Mol. Biol. Plants 2021, 27, 815–824. [Google Scholar] [CrossRef]
- Makino, A.; Mae, T.; Ohira, K. Effect of nitrogen, phosphorus or potassium on the photosynthetic rate and ribulose-1,5-bisphosphate carboxylase content in rice leaves during expansion. Soil Sci. Plant Nutr. 1984, 30, 63–70. [Google Scholar] [CrossRef]
- Nekoukhou, M.; Fallah, S.; Pokhrel, L.R.; Abbasi-Surki, A.; Rostamnejadi, A. Foliar enrichment of copper oxide nanoparticles promotes biomass, photosynthetic pigments, and commercially valuable secondary metabolites and essential oils in dragonhead (Dracocephalum moldavica L.) under semi-arid conditions. Sci. Total Environ. 2023, 863, 160920. [Google Scholar] [CrossRef]
- Li, S.; Wang, S.; Song, Z.; Wang, P.; Lv, F.; Yang, R.; Li, Y. The oxidative damage of the Lagerstroemia indica chlorosis mutant gl1 involves in ferroptosis. J. Plant Physiol. 2023, 280, 153886. [Google Scholar] [CrossRef] [PubMed]
- Weng, Y.; You, Y.; Lu, Q.; Zhong, A.; Liu, S.; Liu, H.; Du, S. Graphene oxide exposure suppresses nitrate uptake by roots of wheat seedlings. Environ. Pollut. 2020, 262, 114224. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhu, X.; Mou, M.; Wang, Z.; Duo, L. Assessment of graphene oxide toxicity on the growth and nutrient levels of white clover (Trifolium repens L.). Ecotox. Environ. Saf. 2022, 234, 113399. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Wang, W.; Chen, X.; Gao, Y.; Wu, X.; Ding, M.; Duo, L. Graphene oxide affected root growth, anatomy, and nutrient uptake in alfalfa. Ecotox. Environ. Saf. 2023, 250, 114483. [Google Scholar] [CrossRef]
- Wang, Q.; Xie, D.; Peng, L.; Chen, C.; Li, C.; Que, X. Phytotoxicity of atrazine combined with cadmium on photosynthetic apparatus of the emergent plant species Iris pseudacorus. Environ. Sci. Pollut. Res. 2022, 29, 34798–34812. [Google Scholar] [CrossRef]


| GO Concentration mg·L−1 | Chlorophyll a mg·g−1 | Chlorophyll b mg·g−1 | Chlorophyll a + b mg·g−1 | Chlorophyll a/b | Carotenoid mg·g−1 |
|---|---|---|---|---|---|
| 0 | 0.3805 ± 0.0223 c* | 0.2282 ± 0.0079 c | 0.6087 ± 0.0299 c | 1.6665 ± 0.0452 c | 0.0502 ± 0.0038 c |
| 1 | 0.4508 ± 0.1008 bc | 0.2434 ± 0.0303 c | 0.6942 ± 0.1312 bc | 1.8345 ± 0.1865 bc | 0.0658 ± 0.0217 bc |
| 20 | 0.5790 ± 0.0905 b | 0.2871 ± 0.0248 b | 0.8662 ± 0.1153 b | 2.0072 ± 0.1517 b | 0.0931 ± 0.0207 b |
| 80 | 0.7966 ± 0.1488 a | 0.3446 ± 0.0480 a | 1.1412 ± 0.1967 a | 2.3001 ± 0.1097 a | 0.1396 ± 0.0331 a |
| 140 | 0.7305 ± 0.0381 a | 0.3263 ± 0.0142 ab | 1.0569 ± 0.0519 a | 2.2379 ± 0.0345 a | 0.1304 ± 0.0151 a |
| GO Level mg L−1 | Cu µg g−1 | Fe mg g−1 | Zn µg g−1 | Mg mg g−1 | Mn mg g−1 | K mg g−1 | N mg g−1 | P mg g−1 |
|---|---|---|---|---|---|---|---|---|
| 0 | 1.32 ± 0.23 c* | 8.44 ± 0.62 b | 9.75 ± 1.82 a | 3.60 ± 0.30 ab | 0.29 ± 0.02 b | 18.95 ± 0.50 a | 6.01 ± 0.86 b | 1.80 ± 0.19 a |
| 1 | 1.78 ± 0.37 bc | 9.34 ± 1.59 ab | 11.91 ± 0.45 a | 3.80 ± 0.11 ab | 0.33 ± 0.05 ab | 19.99 ± 2.13 a | 8.95 ± 1.21 a | 1.99 ± 0.38 a |
| 20 | 1.93 ± 0.27 b | 11.02 ± 0.98 a | 10.99 ± 1.39 a | 3.99 ± 0.34 a | 0.32 ± 0.04 ab | 19.78 ± 3.44 a | 9.76 ± 0.99 a | 2.06 ± 0.17 a |
| 80 | 2.42 ± 0.23 a | 11.67 ± 2.06 a | 10.81 ± 3.04 a | 3.96 ± 0.26 a | 0.37 ± 0.04 a | 18.23 ± 2.49 a | 10.12 ± 1.41 a | 2.00 ± 0.34 a |
| 140 | 2.44 ± 0.28 a | 10.09 ± 1.24 ab | 11.56 ± 1.25 a | 3.41 ± 0.30 b | 0.34 ± 0.01 ab | 17.48 ± 3.09 a | 9.41 ± 0.08 a | 1.90 ± 0.21 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, Z.; Li, J.; Li, C.; Guo, Q.; Hou, X.; Zhao, C.; Wang, Y.; Chen, C.; Wang, Q. Effects of Graphene Oxide on the Growth and Photosynthesis of the Emergent Plant Iris pseudacorus. Plants 2023, 12, 1738. https://doi.org/10.3390/plants12091738
Zhou Z, Li J, Li C, Guo Q, Hou X, Zhao C, Wang Y, Chen C, Wang Q. Effects of Graphene Oxide on the Growth and Photosynthesis of the Emergent Plant Iris pseudacorus. Plants. 2023; 12(9):1738. https://doi.org/10.3390/plants12091738
Chicago/Turabian StyleZhou, Zixin, Jiaxin Li, Cui Li, Qiang Guo, Xincun Hou, Chunqiao Zhao, Yu Wang, Chuansheng Chen, and Qinghai Wang. 2023. "Effects of Graphene Oxide on the Growth and Photosynthesis of the Emergent Plant Iris pseudacorus" Plants 12, no. 9: 1738. https://doi.org/10.3390/plants12091738






