Photosynthetic Performance of Oil Palm Genotypes under Drought Stress
Abstract
:1. Introduction
2. Results
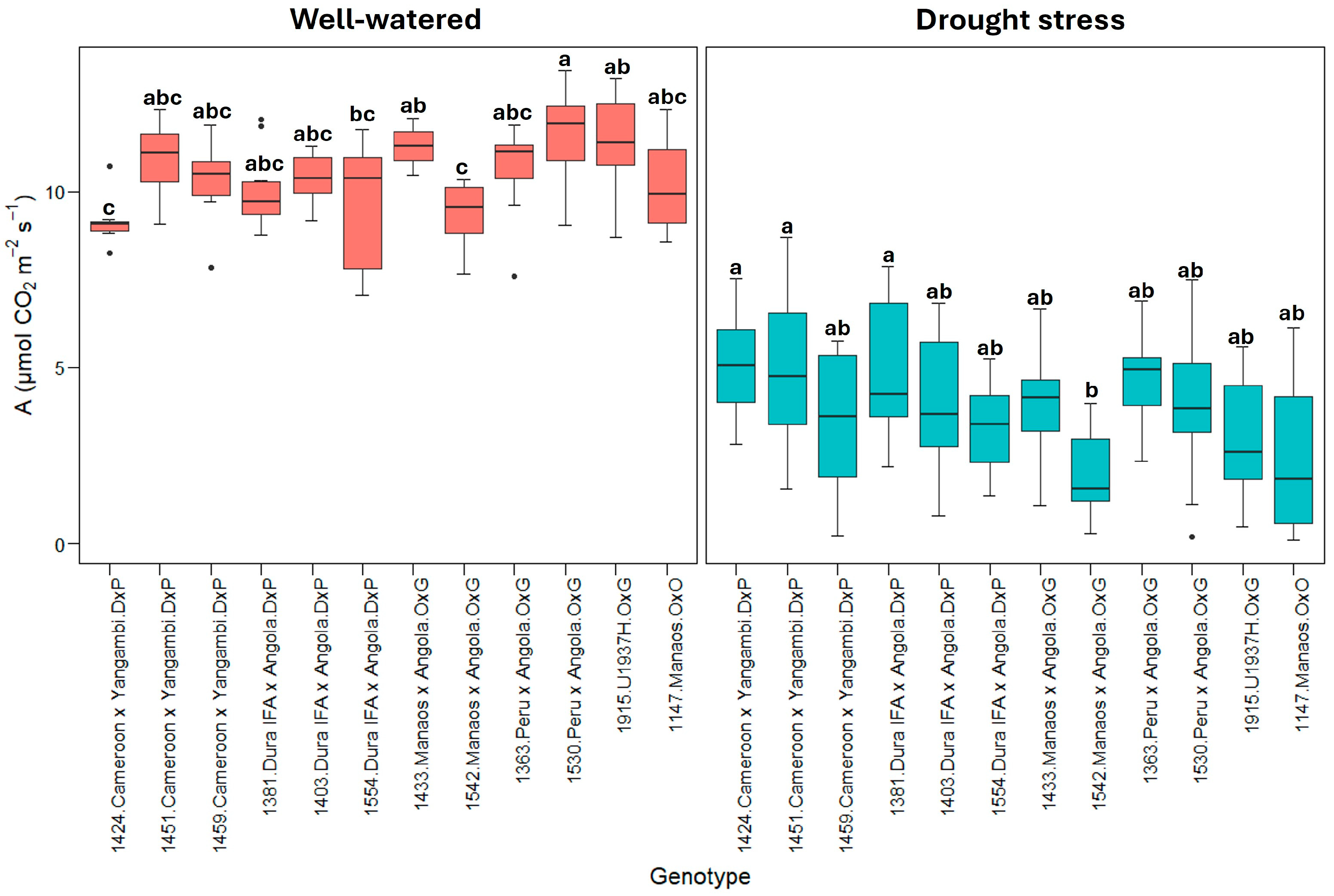
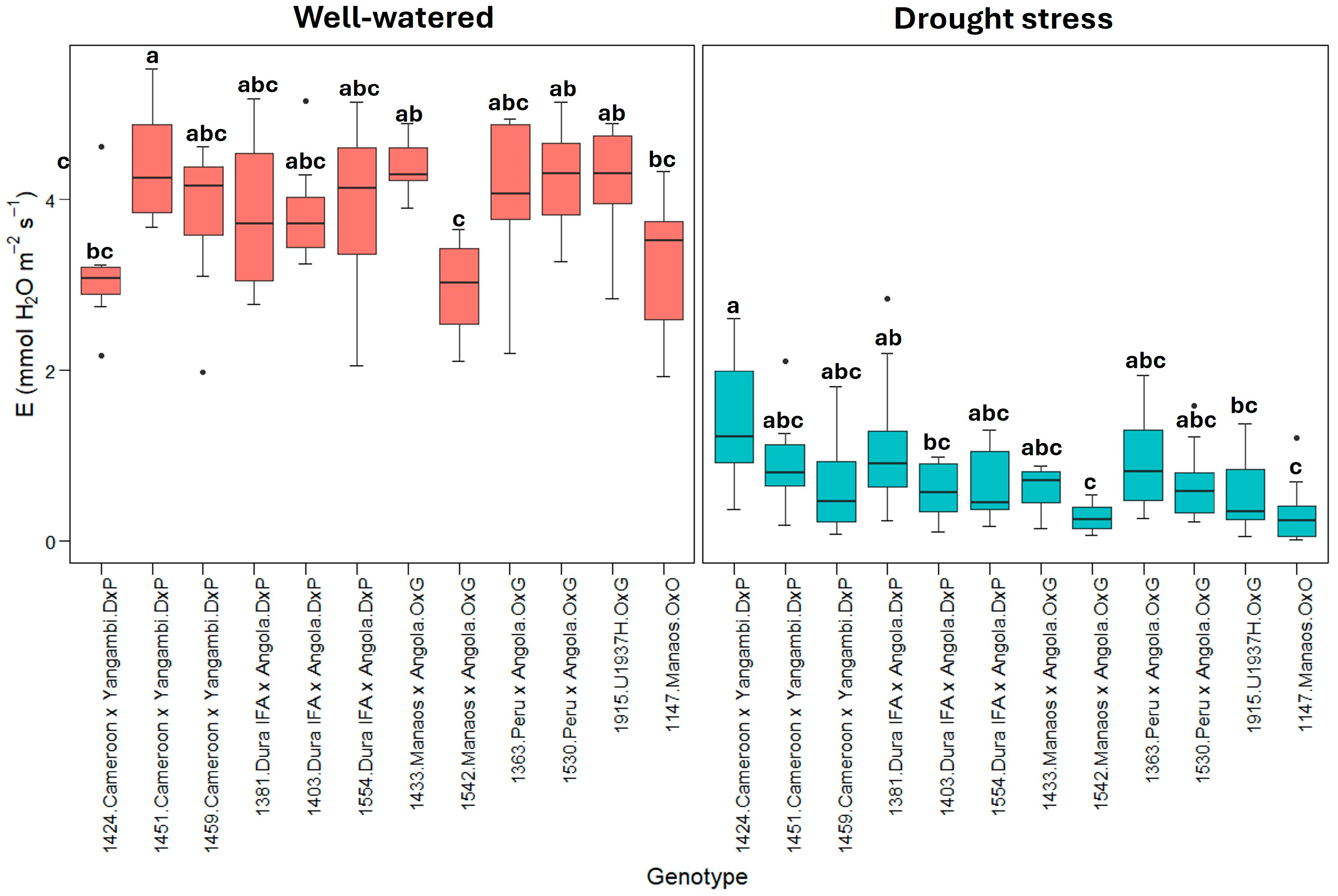
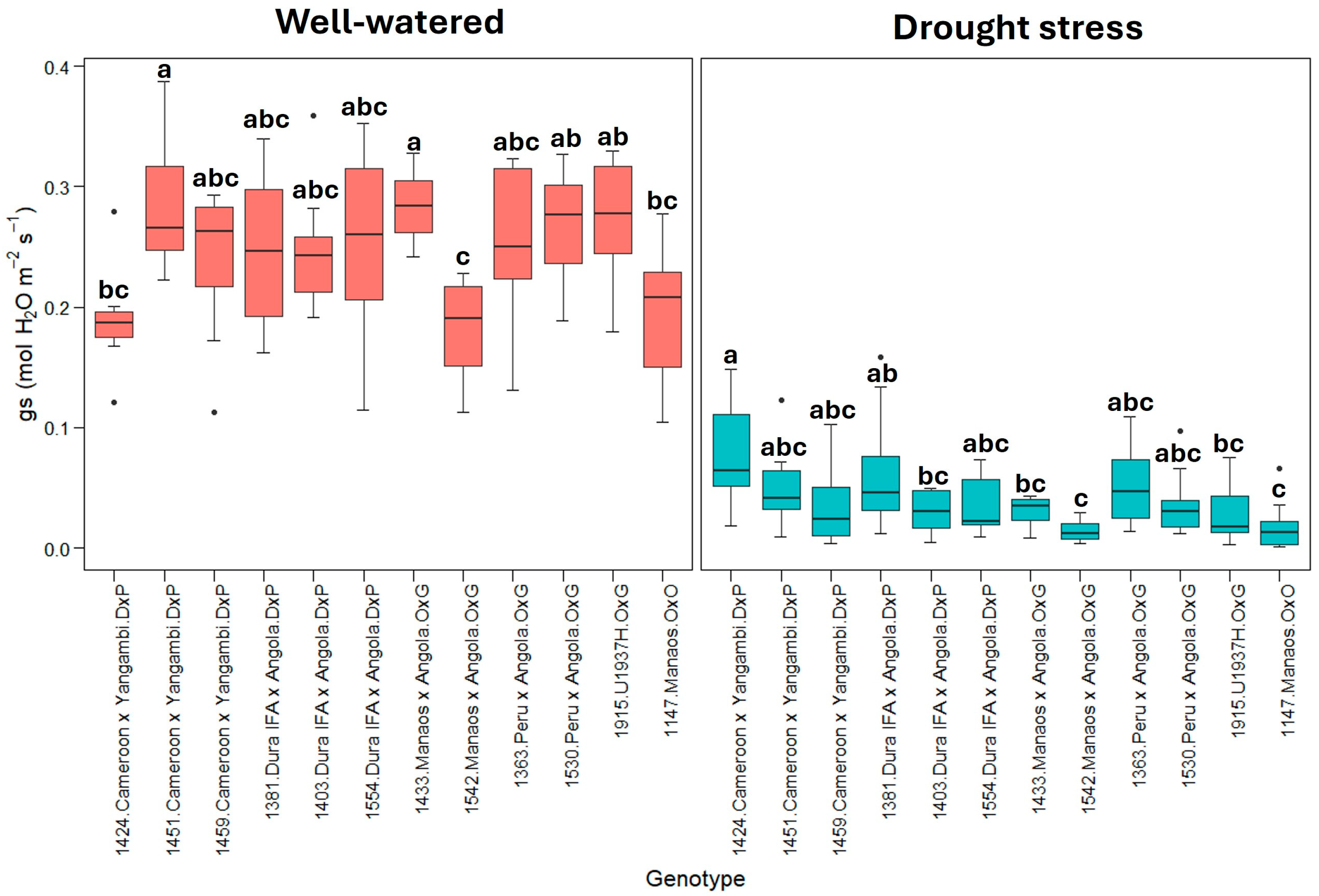
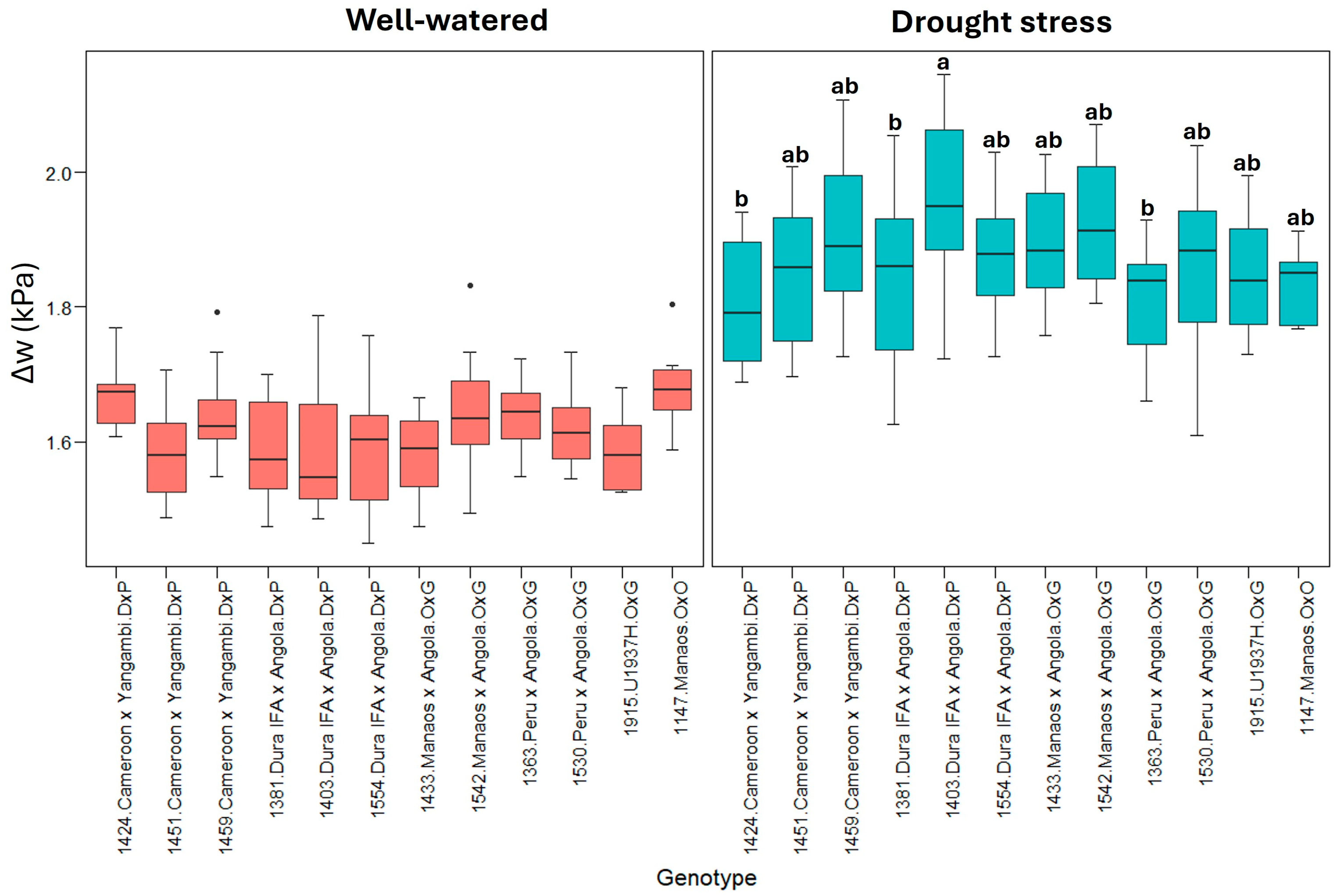
2.1. Physiological Parameters
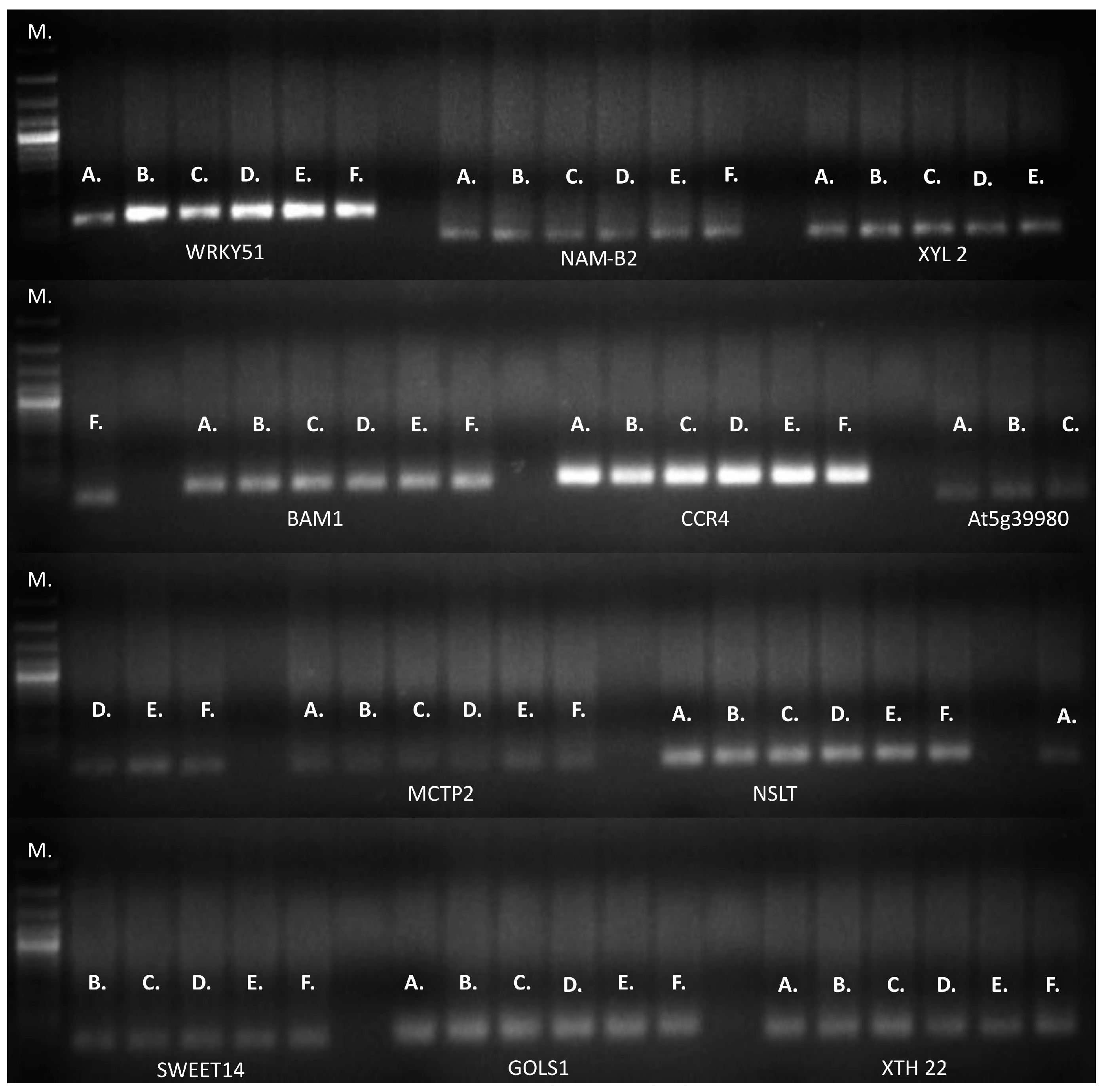
2.2. RT-qPCR Validation of Genes Involved in Drought Tolerance
3. Discussion
3.1. Physiological Parameters
3.2. RT-qPCR Validation of Genes Involved in Drought Tolerance
4. Materials and Methods
4.1. Location
4.2. Plant Materials and Experimental Design
4.3. Statistical Analysis
4.4. Physiological Parameters
4.5. RT-qPCR Validation of Genes Involved in Drought Tolerance
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United States Department of Agriculture. Oilseed: World Markets and Trade; United States Department of Agriculture: Washington, DC, USA, 2024.
- Corley, R.H.V.; Tinker, P. The Oil Palm, 5th ed.; Wiley Blackwell: Hoboken, NJ, USA, 2016; ISBN 9781405189392. [Google Scholar]
- Navia, E.A.; Ávila, R.A.; Daza, E.E.; Restrepo, E.F.; Romero, H.M. Assessment of Tolerance to Bud Rot in Oil Palm under Field Conditions. Eur. J. Plant Pathol. 2014, 140, 711–720. [Google Scholar] [CrossRef]
- Torres, G.; Sarria, G.; Martinez, G.; Varon, F.; Drenth, A.; Guest, D. Bud Rot Caused by Phytophthora Palmivora: A Destructive Emerging Disease of Oil Palm. Phytopathology 2016, 106, 320–329. [Google Scholar] [CrossRef]
- Daza, E.; Ayala-Díaz, I.; Ruiz-Romero, R.; Romero, H.M. Effect of the Application of Plant Hormones on the Formation of Parthenocarpic Fruits and Oil Production in Oil Palm Interspecific Hybrids (Elaeis Oleifera Cortés × Elaeis Guineensis Jacq.). Plant Prod. Sci. 2020, 24, 354–362. [Google Scholar] [CrossRef]
- Romero, H.M.H.M.; Daza, E.; Ayala-Díaz, I.; Ruiz-Romero, R. High-Oleic Palm Oil (HOPO) Production from Parthenocarpic Fruits in Oil Palm Interspecific Hybrids Using Naphthalene Acetic Acid. Agronomy 2021, 11, 290. [Google Scholar] [CrossRef]
- Bayona-Rodríguez, C.; Romero, H.M. Drought Resilience in Oil Palm Cultivars: A Multidimensional Analysis of Diagnostic Variables. Plants 2024, 13, 1598. [Google Scholar] [CrossRef]
- Bayona-Rodriguez, C.J.; Romero, H.M. Physiological and Agronomic Behavior of Commercial Cultivars of Oil Palm (Elaeis Guineensis) and O×G Hybrids (Elaeis Oleifera × Elaeis Guineensis) at Rainy and Dry Seasons. Aust. J. Crop Sci. 2019, 13, 424–432. [Google Scholar] [CrossRef]
- Rivera Méndez, Y.D.; Moreno Chacón, L.; Bayona, C.J.; Romero, H.M. Physiological Response of Oil Palm Interspecific Hybrids (Elaeis Oleifera H.B.K. Cortes versus Elaeis Guineensis Jacq.) to Water Deficit. Braz. J. Plant Physiol. 2012, 24, 273–280. [Google Scholar] [CrossRef]
- Bräutigam, A.; Gowik, U. Photorespiration Connects C3 and C4 Photosynthesis. J. Exp. Bot. 2016, 67, 2953–2962. [Google Scholar] [CrossRef]
- Eisenhut, M.; Bräutigam, A.; Timm, S.; Florian, A.; Tohge, T.; Fernie, A.R.; Bauwe, H.; Weber, A.P.M. Photorespiration Is Crucial for Dynamic Response of Photosynthetic Metabolism and Stomatal Movement to Altered CO2 Availability. Mol. Plant 2017, 10, 47–61. [Google Scholar] [CrossRef]
- Apichatmeta, K.; Sudsiri, C.J.; Ritchie, R.J. Photosynthesis of Oil Palm (Elaeis Guineensis). Sci. Hortic. 2017, 214, 34–40. [Google Scholar] [CrossRef]
- Silva, P.A.; Cosme, V.S.; Rodrigues, K.C.B.B.; Detmann, K.S.C.C.; Leão, F.M.; Cunha, R.L.; Buselli, R.A.F.; DaMatta, F.M.; Pinheiro, H.A.; Festucci Buselli, R.A.; et al. Drought Tolerance in Two Oil Palm Hybrids as Related to Adjustments in Carbon Metabolism and Vegetative Growth. Acta Physiol. Plant. 2017, 39, 58. [Google Scholar] [CrossRef]
- Wang, L.; Lee, M.; Ye, B.; Yue, G.H. Genes, Pathways and Networks Responding to Drought Stress in Oil Palm Roots. Sci. Rep. 2020, 10, 21303. [Google Scholar] [CrossRef]
- Gervais, T.; Creelman, A.; Li, X.Q.; Bizimungu, B.; De Koeyer, D.; Dahal, K. Potato Response to Drought Stress: Physiological and Growth Basis. Front. Plant Sci. 2021, 12, 698060. [Google Scholar] [CrossRef]
- Cantó-Pastor, A.; Kajala, K.; Shaar-Moshe, L.; Manzano, C.; Timilsena, P.; De Bellis, D.; Gray, S.; Holbein, J.; Yang, H.; Mohammad, S.; et al. A Suberized Exodermis Is Required for Tomato Drought Tolerance. Nat. Plants 2024, 10, 118–130. [Google Scholar] [CrossRef]
- Chen, J.; Liu, X.; Du, S.S.; Ma, Y.; Liu, L. Effects of Drought on the Relationship between Photosynthesis and Chlorophyll Fluorescence for Maize. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2021, 14, 11148–11161. [Google Scholar] [CrossRef]
- Herppich, W.B.; Gusovius, H.J.; Flemming, I.; Drastig, K. Effects of Drought and Heat on Photosynthetic Performance, Water Use and Yield of Two Selected Fiber Hemp Cultivars at a Poor-Soil Site in Brandenburg (Germany). Agronomy 2020, 10, 1361. [Google Scholar] [CrossRef]
- Galeano, E.; Vasconcelos, T.S.; De Oliveira, P.N.; Carrer, H. Physiological and Molecular Responses to Drought Stress in Teak (Tectona Grandis L.F.). PLoS ONE 2019, 14, e0221571. [Google Scholar] [CrossRef]
- Wang, L.; Yang, Y.; Zhang, S.; Che, Z.; Yuan, W.; Yu, D. GWAS Reveals Two Novel Loci for Photosynthesis-Related Traits in Soybean. Mol. Genet. Genom. 2020, 295, 705–716. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, L.; Zhang, D.; Cheng, H.; Wang, Q.; Yang, H.; Yu, D. GWAS Identifies Two Novel Loci for Photosynthetic Traits Related to Phosphorus Efficiency in Soybean. Mol. Breed. 2020, 40, 29. [Google Scholar] [CrossRef]
- Wu, Y.; Shi, H.; Yu, H.; Ma, Y.; Hu, H.; Han, Z.; Zhang, Y.; Zhen, Z.; Yi, L.; Hou, J. Combined GWAS and Transcriptome Analyses Provide New Insights Into the Response Mechanisms of Sunflower Against Drought Stress. Front. Plant Sci. 2022, 13, 847435. [Google Scholar] [CrossRef]
- Hamzelou, S.; Kamath, K.S.; Masoomi-aladizgeh, F.; Johnsen, M.M.; Atwell, B.J.; Haynes, P.A. Wild and Cultivated Species of Rice Have Distinctive Proteomic Responses to Drought. Int. J. Mol. Sci. 2020, 21, 5980. [Google Scholar] [CrossRef]
- Magwanga, R.O.; Lu, P.; Kirungu, J.N.; Cai, X.; Zhou, Z.; Agong, S.G.; Wang, K.; Liu, F. Identification of QTLs and Candidate Genes for Physiological Traits Associated with Drought Tolerance in Cotton. J. Cott. Res. 2020, 3, 3. [Google Scholar] [CrossRef]
- Ghatak, A.; Chaturvedi, P.; Weckwerth, W. Cereal Crop Proteomics: Systemic Analysis of Crop Drought Stress Responses Towards Marker-Assisted Selection Breeding. Front. Plant Sci. 2017, 8, 757. [Google Scholar] [CrossRef]
- Bakala, H.S.; Devi, J.; Singh, G.; Singh, I. Drought and Heat Stress: Insights into Tolerance Mechanisms and Breeding Strategies for Pigeonpea Improvement. Planta 2024, 259, 123. [Google Scholar] [CrossRef]
- Alseekh, S.; Kostova, D.; Bulut, M.; Fernie, A.R. Genome-Wide Association Studies: Assessing Trait Characteristics in Model and Crop Plants. Cell. Mol. Life Sci. 2021, 78, 5743–5754. [Google Scholar] [CrossRef]
- Mejía-Alvarado, F.S.; Caicedo-zambrano, A.F.; Botero-rozo, D.; Araque, L.; Bayona-Rodríguez, C.J.; Jazayeri, S.M.; Montoya, C.; Ayala-Díaz, I.; Ruiz-romero, R.; Romero, H.M. Integrative Analysis of Transcriptomic Profiles and Physiological Responses Provide New Insights into Drought Stress Tolerance in Oil Palm (Elaeis Guineensis Jacq.). Int. J. Mol. Sci. 2024, 25, 8761. [Google Scholar] [CrossRef]
- Chen, Q.; Hu, T.; Li, X.; Song, C.-P.; Zhu, J.-K.; Chen, L.; Zhao, Y. Phosphorylation of SWEET Sucrose Transporters Regulates Plant Root:Shoot Ratio under Drought. Nat. Plants 2022, 8, 68–77. [Google Scholar] [CrossRef]
- Ye, Z.; Du, B.; Zhou, B.; Cao, Y.; Zhang, L. Camellia Oleifera CoSWEET10 Is Crucial for Seed Development and Drought Resistance by Mediating Sugar Transport in Transgenic Arabidopsis. Plants 2023, 12, 2818. [Google Scholar] [CrossRef]
- Chen, J.; Wan, H.; Zhao, H.; Dai, X.; Wu, W.; Liu, J.; Xu, J.; Yang, R.; Xu, B.; Zeng, C.; et al. Identification and Expression Analysis of the Xyloglucan Transglycosylase/Hydrolase (XTH) Gene Family under Abiotic Stress in Oilseed ( Brassica Napus L.). BMC Plant Biol. 2024, 24, 400. [Google Scholar] [CrossRef]
- Sun, D.; Ji, X.; Jia, Y.; Huo, D.; Si, S.; Zeng, L.; Zhang, Y.; Niu, L. LreEF1A4, a Translation Elongation Factor from Lilium Regale, Is Pivotal for Cucumber Mosaic Virus and Tobacco Rattle Virus Infections and Tolerance to Salt and Drought. Int. J. Mol. Sci. 2020, 21, 2083. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Li, X.; Gao, S.; Nie, N.; Zhang, H.; Yang, Y.; He, S.; Liu, Q.; Zhai, H. A Novel WRKY Transcription Factor from Ipomoea Trifida, ItfWRKY70, Confers Drought Tolerance in Sweet Potato. Int. J. Mol. Sci. 2022, 23, 686. [Google Scholar] [CrossRef]
- Shaar-Moshe, L.; Hübner, S.; Peleg, Z. Identification of Conserved Drought-Adaptive Genes Using a Cross-Species Meta-Analysis Approach. BMC Plant Biol. 2015, 15, 111. [Google Scholar] [CrossRef]
- Wei, B.; Hou, K.; Zhang, H.; Wang, X.; Wu, W. Integrating Transcriptomics and Metabolomics to Studies Key Metabolism, Pathways and Candidate Genes Associated with Drought-Tolerance in Carthamus Tinctorius L. Under Drought Stress. Ind. Crops Prod. 2020, 151, 112465. [Google Scholar] [CrossRef]
- Saidi, M.N.; Mergby, D.; Brini, F. Identification and Expression Analysis of the NAC Transcription Factor Family in Durum Wheat (Triticum Turgidum L. Ssp. Durum). Plant Physiol. Biochem. 2017, 112, 117–128. [Google Scholar] [CrossRef]
- Zhao, Y.; Qin, Q.; Chen, L.; Long, Y.; Song, N.; Jiang, H.; Si, W. Characterization and Phylogenetic Analysis of Multiple C2 Domain and Transmembrane Region Proteins in Maize. BMC Plant Biol. 2022, 22, 388. [Google Scholar] [CrossRef]
- Wang, P.; Sun, W.; Li, L.; Xu, C.; Li, D.; Li, H.; Zhou, J.; Zhuge, Q. Characterization, Expression Profiling, and Functional Analysis of Poplar Drought Tolerance of the CCR4 Association Factor 1 Gene Family. Ind. Crops Prod. 2022, 188, 115639. [Google Scholar] [CrossRef]
- Ma, X.; Wang, D.; Xue, G.; Zheng, X.; Lu, Y.; Shi, J.; Hao, Z.; Chen, J. Characterization of the Liriodendron Chinense Pentatricopeptide Stress Response. Genes 2023, 14, 1125. [Google Scholar] [CrossRef]
- Santos-Silva, C.A.d.; Ferreira-Neto, J.R.C.; Amador, V.C.; Bezerra-Neto, J.P.; Vilela, L.M.B.; Binneck, E.; Rêgo, M.d.S.; da Silva, M.D.; Mangueira de Melo, A.L.T.; da Silva, R.H.; et al. From Gene to Transcript and Peptide: A Deep Overview on Non-Specific Lipid Transfer Proteins (NsLTPs). Antibiotics 2023, 12, 939. [Google Scholar] [CrossRef]
- Hernández-Lao, T.; Tienda-parrilla, M.; Labella-ortega, M.; Guerrero-Sánchez, V.; Rey, M.; Jorrín-Novo, J.; Castillejo-Sánchez, M. Proteomic and Metabolomic Analysis of the Quercus Ilex—Phytophthora Cinnamomi Pathosystem Reveals a Population-Specific Response, Independent of Co-Occurrence of Drought. Biomolecules 2024, 14, 160. [Google Scholar] [CrossRef]
- Rival, A. Breeding the Oil Palm (Elaeis Guineensis Jacq.) for Climate Change. Oilseeds Fats Crops Lipids 2017, 24, D107. [Google Scholar] [CrossRef]
- Paterson, R.R.M.; Lima, N. Climate Change Affecting Oil Palm Agronomy, and Oil Palm Cultivation Increasing Climate Change, Require Amelioration. Ecol. Evol. 2018, 8, 452–461. [Google Scholar] [CrossRef]
- Hatfield, J.L.; Dold, C. Water-Use Efficiency: Advances and Challenges in a Changing Climate. Front. Plant Sci. 2019, 10, 00103. [Google Scholar] [CrossRef]
- Cooper, M.; Messina, C.D. Breeding Crops for Drought-Affected Environments and Improved Climate Resilience. Plant Cell 2023, 35, 162–186. [Google Scholar] [CrossRef]
- Bayona-Rodríquez, C.J.; Ochoa-Cadavid, I.; Romero, H.M. Impacts of the Dry Season on the Gas Exchange of Oil Palm (Elaeis Guineensis) and Interspecific Hybrid (Elaeis Oleifera x Elaeis Guineensis) Progenies under Field Conditions in Eastern Colombia. Agron. Colomb. 2016, 34, 329–335. [Google Scholar] [CrossRef]
- Jazayeri, S.M.; Rivera, Y.D.; Camperos-Reyes, J.E.; Romero, H.M. Physiological Effects of Water Deficit on Two Oil Palm (Elaeis Guineensis Jacq.) Genotypes. Agron. Colomb. 2015, 33, 164–173. [Google Scholar] [CrossRef]
- Arias, D.; González, M.M.; Romero, H.M.; Prada, F.; Ayala-Diaz, I.; Montoya, C.; Daza, E.; Romero, H.M. Genetic and Phenotypic Diversity of Natural American Oil Palm (Elaeis Oleifera (H.B.K.) Cortés) Accessions. Tree Genet. Genomes 2015, 11, 122. [Google Scholar] [CrossRef]
- Mosquera-Montoya, M.; Camperos, J.E.; Ruiz, E.; Hernández, D.; García, A.; Vargas, L.E.; Mesa, E.; Munévar, D.; Sinisterra, K. Evidence of Sustainable Intensification in the Production of Palm Oil from Crops Planted with Elaeis Oleifera x Elaeis Guineensis in Colombia. Front. Sustain. Food Syst. 2023, 7, 1217653. [Google Scholar] [CrossRef]
- Tezara, W.; Domínguez, T.S.T.; Loyaga, D.W.; Ortiz, R.N.; Chila, V.H.R.; Ortega, M.J.B. Photosynthetic Activity of Oil Palm (Elaeis Guineensis) and Interspecific Hybrid Genotypes (Elaeis Oleifera × Elaeis Guineensis), and Response of Hybrids to Water Deficit. Sci. Hortic. 2021, 287, 110263. [Google Scholar] [CrossRef]
- Lopes Filho, W.R.L.; Rodrigues, F.H.S.; Ferreira, I.V.L.; Correa, L.O.; Cunha, R.L.; Pinheiro, H.A. Physiological Responses of Young Oil Palm (Elaeis Guineensis Jacq.) Plants to Repetitive Water Deficit Events. Ind. Crops Prod. 2021, 172, 114052. [Google Scholar] [CrossRef]
- Lopes Filho, W.R.L.; Rodrigues, F.H.S.; Chaves, R.P.F.; Cunha, R.L.; Costa, L.C.; Pinheiro, H.A. Repeated Water Deficit Events Trigger Adjustments in Enzymatic Antioxidant System in Oil Palm. Theor. Exp. Plant Physiol. 2024, 36, 235–250. [Google Scholar] [CrossRef]
- Valifard, M.; Le Hir, R.; Müller, J.; Scheuring, D.; Neuhaus, H.E.; Pommerrenig, B. Vacuolar Fructose Transporter SWEET17 Is Critical for Root Development and Drought Tolerance. Plant Physiol. 2021, 187, 2716–2730. [Google Scholar] [CrossRef] [PubMed]
- Haque, M.S.; Saha, N.R.; Islam, M.T.; Islam, M.M.; Kwon, S.J.; Roy, S.K.; Woo, S.H. Screening for Drought Tolerance in Wheat Genotypes by Morphological and SSR Markers. J. Crop Sci. Biotechnol. 2020, 24, 27–39. [Google Scholar] [CrossRef]
- Maheswari, M.; Tekula, V.L.; Yellisetty, V.; Sarkar, B.; Yadav, S.K.; Singh, J.; G., S.B.; Kumar, A.; Amirineni, S.; Narayana, J.; et al. Functional Mechanisms of Drought Tolerance in Maize through Phenotyping and Genotyping under Well Watered and Water Stressed Conditions. Eur. J. Agron. 2016, 79, 43–57. [Google Scholar] [CrossRef]

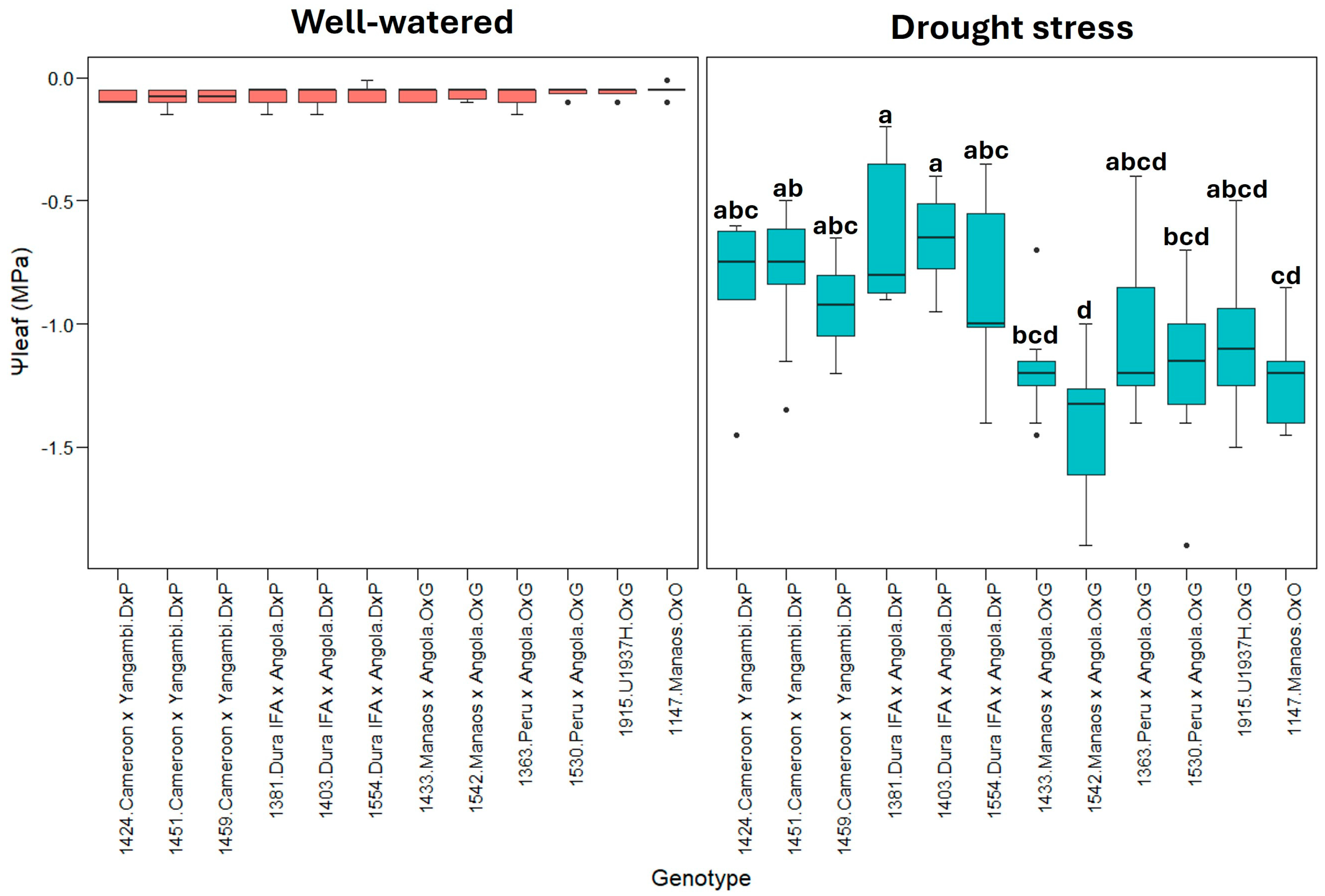
| Treatment | A | E | gs | Δw | WUE | Ψleaf |
|---|---|---|---|---|---|---|
| Well-watered treatment | 10.4 ± 1.37 * | 3.84 ± 0.84 | 0.24 ± 0.06 | 1.61 ± 0.08 | 2.79 ± 0.45 | −0.07 ± 0.03 |
| Drought stress | 3.80 ± 2.03 | 0.71 ± 0.56 | 0.03 ± 0.03 | 1.87 ± 0.11 | 6.49 ± 2.41 | −1.0 ± 0.36 |
| Condition | Parameter | Sum Sq | Mean Sq | F Value | Pr (>F) * |
|---|---|---|---|---|---|
| Well-watered | A | 66.42 | 6038 | 4.251 | 3.63 × 10−5 *** |
| E | 22.69 | 20631 | 3.805 | 0.000143 *** | |
| gs | 0.12271 | 0.011155 | 3.657 | 0.000227 *** | |
| Δw | 0.1289 | 0.011719 | 2.229 | 0.01843 * | |
| WUE | 5.630 | 0.5118 | 2.940 | 0.0021 ** | |
| Ψleaf | 0.00692 | 0.0006291 | 0.755 | 0.6838 | |
| Drought stress | A | 109.3 | 9.932 | 3.009 | 0.00172 ** |
| E | 9.885 | 0.8986 | 3.652 | 0.000234 *** | |
| gs | 0.03251 | 0.0029550 | 3.661 | 0.000228 *** | |
| Δw | 0.1998 | 0.01816 | 2.931 | 0.00218 ** | |
| WUE | 80.3 | 7.301 | 1.255 | 0.262 | |
| Ψleaf | 6.139 | 0.5581 | 7.139 | 8.63 × 10−9 *** |
| Species/Genotype | Female Parent | Male Parent | Code Genotype |
|---|---|---|---|
| Elaeis oleifera (O×O) | Manaos | Manaos | 1147 |
| Elaeis guineensis (D×P) | Dura IFA | Angola | 1381 |
| 1403 | |||
| 1554 | |||
| Cameroon | Yangambi | 1424 | |
| 1459 | |||
| 1451 | |||
| E. oleifera × E. guineensis (O×G) | Peru | Angola | 1530 |
| 1363 | |||
| Manaos | Angola | 1433 | |
| 1542 | |||
| E. oleifera | Congo Mixto | 1915(U1937H) |
| Anotation | 5’→3’ | Sequence | Tm | Expected Size (pb) | References |
|---|---|---|---|---|---|
| WRKY transcription factor 51 | F | TTATGTGAAGACCGACCCATC | 59.4 | 140 | [32] |
| R | CAGGACAGGAAGGAGCAAAG | 60.5 | |||
| NAC transcription factor NAM-B2-like_ NAM-B2 | F | CGTCCTCTCACTTCCCCATAC | 63.3 | 127 | [35] |
| R | CGCTGCTGGTGTTGTTGTT | 57.3 | |||
| beta-xylosidase alpha-L-arabinofuranosidase 2-like OsI_08964_ XYL2 | F | CTTCAAACCCATCCACCAAG | 58.4 | 128 | [41] |
| R | AGGGGCACTATCACCCAGTT | 60.5 | |||
| Leucine-rich repeat receptor-like serine_ At1g17230 | F | CATCATCACAAGCACAAGCA | 56.4 | 121 | [22] |
| R | TCTCCAAGAATGCGAGGTGT | 58.4 | |||
| Multiple C2 and transmembrane domain-containing protein 2-like | F | GGATACAGACGGTGGTAGGG | 62.5 | 105 | [36] |
| R | GGCAGAACATCACAAACAGG | 58.4 | |||
| Nonspecific lipid-transfer protein 2-like | F | GTCCTCTGCTTGCTGTGCTA | 60.5 | 112 | [23,40] |
| R | AACCACTTTCTGGCTGTTGG | 58.4 | |||
| Pentatricopeptide repeat-containing protein_At5g39980 | F | AGCGATGTCTCAGTGCTTCTC | 61.3 | 94 | [39] |
| R | CTTCCTCCTGTTCCTCTCCA | 60.5 | |||
| Serine, threonine-protein kinase-like protein_CCR4 | F | ACAGCACCAGCAAGGAGAGA | 60.5 | 91 | [38] |
| R | GCTTTGAGGAGGGTTTCCA | 57.3 | |||
| Bidirectional sugar transporter SWEET14-like | F | AACGTGGTGCTATTCGGGTT | 61.1 | 93 | [29] |
| R | ACTCACACAGACCCATCCAAG | 62.4 | |||
| Galactinol synthase 1-like_ GOLS1 | F | TGCGGTGATGGATTGCTTCT | 61.2 | 142 | [35] |
| R | ACCAGCGTTGAAGTACAGGG | 62.1 | |||
| Xyloglucan endotransglucosylase/hydrolase protein 22-like XTH22 | F | TCCTCTGCTTCCCCTCTACC | 65.2 | 120 | [31] |
| R | CGCCCCAAGTGATCTGGAAA | 63.6 | |||
| Glyceraldehyde-3-phosphate dehydrogenase GAPDH | F | CAACCAACTGTCTTGCTCCTT | 59.4 | 138 | [25] |
| R | CTCCTCGCCAATCTTTCATT | 56.4 | |||
| Elongation factor 1-alpha_EF1-a1 | F | CCTTCTTGCTTTCACCCTTG | 58.4 | 143 | [31] |
| R | GGTTGTAGCCGACCTTCTTG | 60.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Montoya, C.; Daza, E.; Mejía-Alvarado, F.S.; Caicedo-Zambrano, A.F.; Ayala-Díaz, I.; Ruiz-Romero, R.; Romero, H.M. Photosynthetic Performance of Oil Palm Genotypes under Drought Stress. Plants 2024, 13, 2705. https://doi.org/10.3390/plants13192705
Montoya C, Daza E, Mejía-Alvarado FS, Caicedo-Zambrano AF, Ayala-Díaz I, Ruiz-Romero R, Romero HM. Photosynthetic Performance of Oil Palm Genotypes under Drought Stress. Plants. 2024; 13(19):2705. https://doi.org/10.3390/plants13192705
Chicago/Turabian StyleMontoya, Carmenza, Edison Daza, Fernan Santiago Mejía-Alvarado, Arley Fernando Caicedo-Zambrano, Iván Ayala-Díaz, Rodrigo Ruiz-Romero, and Hernán Mauricio Romero. 2024. "Photosynthetic Performance of Oil Palm Genotypes under Drought Stress" Plants 13, no. 19: 2705. https://doi.org/10.3390/plants13192705






