Accumulation of TIP2;2 Aquaporin during Dark Adaptation Is Partially PhyA Dependent in Roots of Arabidopsis Seedlings
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effects of phyA Mutation on Fluorescent Intensity of TIP2;2-GFP under Light Conditions
2.1.1. Tissue-Specific Localization of TIP2;2-GFP in the Wild Type and phyA Mutant

2.1.2. Fluorescence of TIP2;2-GFP in Light-Grown Wild-Type and phyA Mutant Plants



| F2 plant | Number of plants classified with phyA mutation and fluorescent intensity *1 | χ2 values *2 Expected frequency | |||||
|---|---|---|---|---|---|---|---|
| PHYA | phyA | ||||||
| ++ | + | − | ++ | + | − | ||
| TIP2;2-GFP/ phyA 8-4 × Ler | 0 | 37 | 18 | 2 | 14 | 2 | 0.00456 [PHYA]: [phyA] = 3:1 1.54 [PHYA or GFP+]: [phyA gfp−] = 15:1 |
| TIP2;2-GFP/ phyA 8-4 ×phyA | 0 | 0 | 0 | 2 | 18 | 3 | 1.75 [GFP+]:[gfp−] = 3:1 |
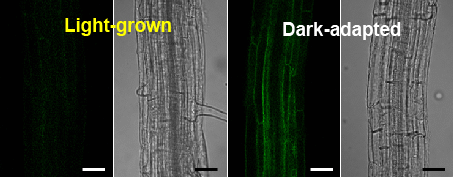
2.2. Dark-Induced Increase in TIP2;2-GFP

2.3. PhyA Regulation of Aquaporins
2.4. Why TIP2;2 Increases during Dark Adaptation
3. Experimental
3.1. Plant Material and Growth Conditions
3.2. Fluorescent Intensity Analysis
3.3. Protein Preparation
3.4. Immunoblotting
3.5. Generation of TIP2;2-GFP Transgenic Lines
3.6. Protoplast Preparation
3.7. RT-PCR
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Taiz, L. The plant vacuole. J. Exp. Biol. 1992, 172, 113–122. [Google Scholar]
- Marty, F. Plant vacuoles. Plant Cell 1999, 11, 587–600. [Google Scholar]
- Frigerio, L.; Hinz, G.; Robinson, D.G. Multiple vacuoles in plant cells: Rule or exception? Traffic 2008, 9, 1564–1570. [Google Scholar] [CrossRef]
- Martinoia, E.; Maeshima, M.; Neuhaus, H.E. Vacuolar transporters and their essential role in plant metabolism. J. Exp. Bot. 2007, 58, 83–102. [Google Scholar] [CrossRef]
- Maurel, C. Plant aquaporins: Novel functions and regulation properties. FEBS Lett. 2007, 581, 2227–2236. [Google Scholar] [CrossRef]
- Katsuhara, M.; Hanba, Y.T.; Shiratake, K.; Maeshima, M. Review: Expanding roles of plant aquaporins in plasma membranes and cell organelles. Funct. Plant Biol. 2008, 35, 1–14. [Google Scholar] [CrossRef]
- Maurel, C.; Santoni, V.; Luu, D.T.; Wudick, M.M.; Verdoucq, L. The cellular dynamics of plant aquaporin expression and functions. Curr. Opin. Plant Biol. 2009, 12, 690–698. [Google Scholar] [CrossRef]
- Johnson, K.D.; Herman, E.M.; Chrispeels, M.J. An abundant, highly conserved tonoplast protein in seeds. Plant Physiol. 1989, 91, 1006–1013. [Google Scholar] [CrossRef]
- Johanson, U.; Karlsson, M.; Johansson, I.; Gustavsson, S.; Sjövall, S.; Fraysse, L.; Weig, A.R.; Kjellbom, P. The complete set of genes encoding major intrinsic proteins in Arabidopsis provides a framework for a new nomenclature for major intrinsic proteins in plants. Plant Physiol. 2001, 126, 1358–1369. [Google Scholar] [CrossRef]
- Maurel, C.; Reizer, J.; Schroeder, J.I.; Chrispeels, M.J. The vacuolar membrane protein γ-TIP creates water specific channels in Xenopus oocytes. EMBO J. 1993, 12, 2241–2247. [Google Scholar]
- Kaldenhoff, R.; Fischer, M. Functional aquaporin diversity in plants. Biochim. Biophys. Acta 2006, 1758, 1134–1141. [Google Scholar] [CrossRef]
- Maurel, C.; Kado, R.T.; Guern, J.; Chrispeels, M.J. Phosphorylation regulates the water channel activity of the seed-specific aquaporin α-TIP. EMBO J. 1995, 14, 3028–3035. [Google Scholar]
- Leitão, L.; Prista, C.; Moura, T.F.; Loureiro-Dias, M.C.; Soveral, G. Grapevine aquaporins: Gating of a tonoplast intrinsic protein (TIP2;1) by cytosolic pH. PLoS One 2012, 7, e33219. [Google Scholar]
- Vera-Estrella, R.; Barkla, B.J.; Bohnert, H.J.; Pantoja, O. Novel regulation of aquaporins during osmotic stress. Plant Physiol. 2004, 135, 2318–2329. [Google Scholar] [CrossRef]
- Daniels, M.J.; Chaumont, F.; Mirkov, T.E.; Chrispeels, M.J. Characterization of a new vacuolar membrane aquaporin sensitive to mercury at a unique site. Plant Cell 1996, 8, 587–599. [Google Scholar]
- Loqué, D.; Ludewig, U.; Yuan, L.; von Wirén, N. Tonoplast intrinsic proteins AtTIP2;1 and AtTIP2;3 facilitate NH3 transport into the vacuole. Plant Physiol. 2005, 137, 671–680. [Google Scholar] [CrossRef]
- Jahn, T.P.; Møller, A.L.; Zeuthen, T.; Holm, L.M.; Klaerke, D.A.; Mohsin, B.; Kühlbrandt, W.; Schjoerring, J.K. Aquaporin homologues in plants and mammals transport ammonia. FEBS Lett. 2004, 574, 31–36. [Google Scholar] [CrossRef]
- Liu, L.H.; Ludewig, U.; Gassert, B.; Frommer, W.B.; von Wirén, N. Urea transport by nitrogen-regulated tonoplast intrinsic proteins in Arabidopsis. Plant Physiol. 2003, 133, 1220–1228. [Google Scholar] [CrossRef]
- Bienert, G.P.; Moller, A.L.; Kristiansen, K.A.; Schulz, A.; Moller, I.M.; Schjoerring, J.K.; Jahn, T.P. Specific aquaporins facilitate the diffusion of hydrogen peroxide across membranes. J. Biol. Chem. 2007, 282, 1183–1192. [Google Scholar] [CrossRef]
- Gerbeau, P.; Güçlü, J.; Ripoche, P.; Maurel, C. Aquaporin Nt-TIPa can account for the high permeability of tobacco cell vacuolar membrane to small neutral solutes. Plant J. 1999, 18, 577–587. [Google Scholar] [CrossRef]
- Jauh, G.Y.; Phillips, T.E.; Rogers, J.C. Tonoplast intrinsic protein isoforms as markers for vacuolar functions. Plant Cell 1999, 11, 1867–1882. [Google Scholar]
- Maeshima, M.; Hara-Nishimura, I.; Takeuchi, Y.; Nishimura, M. Accumulation of vacuolar H+-pyrophosphatase and H+-ATPase during reformation of the central vacuole in germinating pumpkin seeds. Plant Physiol. 1994, 106, 61–69. [Google Scholar]
- Gattolin, S.; Sorieul, M.; Hunter, P.R.; Khonsari, R.H.; Frigerio, L. In vivo imaging of the tonoplast intrinsic protein family in Arabidopsis roots. BMC Plant Biol. 2009, 9. [Google Scholar] [CrossRef]
- Gattolin, S.; Sorieul, M.; Frigerio, L. Mapping of tonoplast intrinsic proteins in maturing and germinating Arabidopsis seeds reveals dual localization of embryonic TIPs to the tonoplast and plasma membrane. Mol. Plant 2011, 4, 180–189. [Google Scholar] [CrossRef]
- Hunter, P.R.; Craddock, C.P.; di Benedetto, S.; Roberts, L.M.; Frigerio, L. Fluorescent reporter proteins for the tonoplast and the vacuolar lumen identify a single vacuolar compartment in Arabidopsis cells. Plant Physiol. 2007, 145, 1371–1382. [Google Scholar]
- Wudick, M.M.; Luu, D.T.; Maurel, C. A look inside: Localization patterns and functions of intracellular plant aquaporins. New Phytol. 2009, 184, 289–302. [Google Scholar] [CrossRef]
- Suga, S.; Komatsu, S.; Maeshima, M. Aquaporin isoforms responsive to salt and water stresses and phytohormones in radish seedlings. Plant Cell Physiol. 2002, 43, 1229–1237. [Google Scholar] [CrossRef]
- Maurel, C.; Javot, H.; Lauvergeat, V.; Gerbeau, P.; Tournaire, C.; Santoni, V.; Heyes, J. Molecular physiology of aquaporins in plants. Int. Rev. Cytol. 2002, 215, 105–148. [Google Scholar] [CrossRef]
- Kim, H.S.; Yu, Y.; Snesrud, E.C.; Moy, L.P.; Linford, L.D.; Haas, B.J.; Nierman, W.C.; Quackenbush, J. Transcriptional divergence of the duplicated oxidative stress-responsive genes in the Arabidopsis genome. Plant J. 2005, 41, 212–220. [Google Scholar]
- Liu, F.; Vantoai, T.; Moy, L.P.; Bock, G.; Linford, L.D.; Quackenbush, J. Global transcription profiling reveals comprehensive insights into hypoxic response in Arabidopsis. Plant Physiol. 2005, 137, 1115–1129. [Google Scholar] [CrossRef]
- Sato-Nara, K.; Nagasaka, A.; Yamashita, H.; Ishida, J.; Enju, A.; Seki, M.; Shinozaki, K.; Suzuki, H. Identification of genes regulated by dark adaptation and far-red light illumination in roots of Arabidopsis thaliana. Plant Cell Environ. 2004, 27, 1387–1394. [Google Scholar] [CrossRef]
- Culianez-Macia, F.A.; Martin, C. DIP: A member of the MIP family of membrane proteins that is expressed in mature seeds and dark-grown seedlings of Antirrhinum majus. Plant J. 1993, 4, 717–725. [Google Scholar]
- Sade, N.; Vinocur, B.J.; Diber, A.; Shatil, A.; Ronen, G.; Nissan, H.; Wallach, R.; Karchi, H.; Moshelion, M. Improving plant stress tolerance and yield production: Is the tonoplast aquaporin SlTIP2;2 a key to isohydric to anisohydric conversion? New Phytol. 2009, 181, 651–661. [Google Scholar] [CrossRef]
- Lin, W.L.; Peng, Y.H.; Li, G.W.; Arora, R.; Tang, Z.C.; Su, W.A.; Cai, W.M. Isolation and functional characterization of PgTIP1 a hormone-autotrophic cells-specific tonoplast aquaporin in ginseng. J. Exp. Bot. 2007, 58, 947–956. [Google Scholar] [CrossRef]
- Okubo-Kurihara, E.; Sano, T.; Higaki, T.; Kutsuna, N.; Hasezawa, S. Acceleration of vacuolar regeneration and cell growth by overexpression of an aquaporin NtTIP1;1 in tobacco BY-2 cells. Plant Cell Physiol. 2009, 50, 151–160. [Google Scholar] [CrossRef]
- Whitelam, G.C.; Halliday, K.J. Light and Plant Development; Blackwell Publisher Ltd.: Oxford, UK, 2007. [Google Scholar]
- Van Buskirk, E.K.; Decker, P.V.; Chen, M. Photobodies in light signaling. Plant Physiol. 2012, 158, 52–60. [Google Scholar] [CrossRef]
- Clack, T.; Mathews, S.; Sharrock, R.A. The phytochrome apoprotein family in Arabidopsis is encoded by five genes: The sequences and expression of PHYD and PHYE. Plant Mol. Biol. 1994, 25, 413–427. [Google Scholar] [CrossRef]
- Kami, C.; Lorrain, S.; Hornitschek, P.; Fankhauser, C. Light-regulated plant growth and development. Curr. Top. Dev. Biol. 2010, 91, 29–66. [Google Scholar] [CrossRef]
- Franklin, K.A.; Quail, P.H. Phytochrome functions in Arabidopsis development. J. Exp. Bot. 2010, 61, 11–24. [Google Scholar] [CrossRef]
- Botto, J.F.; Sánchez, R.A.; Whitelam, G.C.; Casal, J.J. Phytochrome A mediates the promotion of seed germination by very low fluences of light and canopy shade light in Arabidopsis. Plant Physiol. 1996, 110, 439–444. [Google Scholar]
- Hennig, L.; Büche, C.; Schäfer, E. Degradation of phytochrome A and the high irradiance response in Arabidopsis: A kinetic analysis. Plant Cell Environ. 2000, 23, 727–734. [Google Scholar]
- Correll, M.J.; Kiss, J.Z. The roles of phytochromes in elongation and gravitropism of roots. Plant Cell Physiol. 2005, 46, 317–323. [Google Scholar] [CrossRef]
- De Simone, S.; Oka, Y.; Inoue, Y. Effect of light on root hair formation in Arabidopsis thaliana phytochrome-deficient mutants. J. Plant Res. 2000, 113, 63–69. [Google Scholar] [CrossRef]
- Kiss, J.Z.; Mullen, J.L.; Correll, M.J.; Hangarter, R.P. Phytochromes A and B mediate red-light-induced positive phototropism in roots. Plant Physiol. 2003, 131, 1411–1417. [Google Scholar] [CrossRef]
- Boursiac, Y.; Chen, S.; Luu, D.T.; Sorieul, M.; van den Dries, N.; Maurel, C. Early effects of salinity on water transport in Arabidopsis roots. Molecular and cellular features of aquaporin expression. Plant Physiol. 2005, 139, 790–805. [Google Scholar] [CrossRef]
- Higo, K.; Ugawa, Y.; Iwamoto, M.; Korenaga, T. Plant cis-acting regulatory DNA elements (PLACE) database: 1999. Nucleic Acids Res. 1999, 27, 297–300. [Google Scholar] [CrossRef]
- Castillon, A.; Shen, H.; Huq, E. Phytochrome Interacting Factors: Central players in phytochrome-mediated light signaling networks. Trends Plant Sci. 2007, 12, 514–521. [Google Scholar] [CrossRef]
- Terzaghi, W.B.; Cashmore, A.R. Light-regulated transcription. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1995, 46, 445–474. [Google Scholar] [CrossRef]
- Tatematsu, K.; Ward, S.; Leyser, O.; Kamiya, Y.; Nambara, E. Identification of cis-elements that regulate gene expression during initiation of axillary bud outgrowth in Arabidopsis. Plant Physiol. 2005, 138, 757–766. [Google Scholar] [CrossRef]
- Choi, H.; Hong, J.; Ha, J.; Kang, J.; Kim, S.Y. ABFs, a family of ABA-responsive element binding factors. J. Biol. Chem. 2000, 275, 1723–1730. [Google Scholar] [CrossRef]
- Ulmasov, T.; Hagen, G.; Guilfoyle, T.J. Dimerization and DNA binding of auxin response factors. Plant J. 1999, 19, 309–319. [Google Scholar] [CrossRef]
- Steudle, E. Water uptake by roots: Effects of water deficit. J. Exp. Bot. 2000, 51, 1531–1542. [Google Scholar] [CrossRef]
- Neuhäuser, B.; Dynowski, M.; Mayer, M.; Ludewig, U. Regulation of NH4+ transport by essential cross talk between AMT monomers through the carboxyl tails. Plant Physiol. 2007, 143, 1651–1659. [Google Scholar] [CrossRef]
- Vitart, V.; Baxter, I.; Doerner, P.; Harper, J.F. Evidence for a role in growth and salt resistance of a plasma membrane H+-ATPase in the root endodermis. Plant J. 2001, 27, 191–201. [Google Scholar] [CrossRef]
- Tamura, K.; Shimada, T.; Ono, E.; Tanaka, Y.; Nagatani, A.; Higashi, S.I.; Watanabe, M.; Nishimura, M.; Hara-Nishimura, I. Why green fluorescent fusion proteins have not been observed in the vacuoles of higher plants. Plant J. 2003, 35, 545–555. [Google Scholar] [CrossRef]
- Deng, X.W.; Quail, P.H. Signalling in light-controlled development. Semin. Cell Dev. Biol. 1999, 10, 121–129. [Google Scholar] [CrossRef]
- Buchanan-Wollaston, V.; Page, T.; Harrison, E.; Breeze, E.; Lim, P.O.; Nam, H.G.; Lin, J.F.; Wu, S.H.; Swidzinski, J.; Ishizaki, K.; et al. Comparative transcriptome analysis reveals significant differences in gene expression and signalling pathways between developmental and dark/starvation-induced senescence in Arabidopsis. Plant J. 2005, 42, 567–585. [Google Scholar] [CrossRef]
- Maurel, C. Aquaporins and water permeability of plant membranes. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1997, 48, 399–429. [Google Scholar] [CrossRef]
- Naito, S.; Hirai, M.Y.; Chino, M.; Komeda, Y. Expression of a soybean (Glycine max [L.] Merr.) seed storage protein gene in transgenic Arabidopsis thaliana and its response to nutritional stress and to abscisic acid mutations. Plant Physiol. 1994, 104, 497–503. [Google Scholar]
- EZ-C1 software, version 3.80, Nikon: Tokyo, Japan, 2007.
- Photoshop CS5 extended software, version 12.1, Adobe Systems: San Jose, CA, USA, 2011.
- Maeshima, M. Characterization of the major integral protein of vacuolar membrane. Plant Physiol. 1992, 98, 1248–1254. [Google Scholar] [CrossRef]
- MultiGauge software, version 3.0, Fujifilm: Tokyo, Japan, 2004.
- Kaufmann, S.H.; Kellner, U. Erasure of western blots after autoradiographic or chemiluminescent detection. In Immunochemical Protocols: Methods in Molecular Biology; Pound, J.D., Ed.; Humanna Press: Totowa, NJ, USA, 1998; Volumr 80, pp. 223–235. [Google Scholar]
- Nakagawa, T.; Kurose, T.; Hino, T.; Tanaka, K.; Kawamukai, M.; Niwa, Y.; Toyooka, K.; Matsuoka, K.; Jinbo, T.; Kimura, T. Development of series of gateway binary vectors, pGWBs, for realizing efficient construction of fusion genes for plant transformation. J. Biosci. Bioeng. 2007, 104, 34–41. [Google Scholar] [CrossRef]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 1998, 16, 735–743. [Google Scholar] [CrossRef]
- Nagatani, A.; Reed, J.W.; Chory, J. Isolation and initial characterization of Arabidopsis mutants that are deficient in phytochrome A. Plant Physiol. 1993, 102, 269–277. [Google Scholar] [CrossRef]
- Suga, S.; Murai, M.; Kuwagata, T.; Maeshima, M. Differences in aquaporin levels among cell types of radish and measurement of osmotic water permeability of individual protoplasts. Plant Cell Physiol. 2003, 44, 277–286. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Uenishi, Y.; Nakabayashi, Y.; Tsuchihira, A.; Takusagawa, M.; Hashimoto, K.; Maeshima, M.; Sato-Nara, K. Accumulation of TIP2;2 Aquaporin during Dark Adaptation Is Partially PhyA Dependent in Roots of Arabidopsis Seedlings. Plants 2014, 3, 177-195. https://doi.org/10.3390/plants3010177
Uenishi Y, Nakabayashi Y, Tsuchihira A, Takusagawa M, Hashimoto K, Maeshima M, Sato-Nara K. Accumulation of TIP2;2 Aquaporin during Dark Adaptation Is Partially PhyA Dependent in Roots of Arabidopsis Seedlings. Plants. 2014; 3(1):177-195. https://doi.org/10.3390/plants3010177
Chicago/Turabian StyleUenishi, Yumi, Yukari Nakabayashi, Ayako Tsuchihira, Mari Takusagawa, Kayo Hashimoto, Masayoshi Maeshima, and Kumi Sato-Nara. 2014. "Accumulation of TIP2;2 Aquaporin during Dark Adaptation Is Partially PhyA Dependent in Roots of Arabidopsis Seedlings" Plants 3, no. 1: 177-195. https://doi.org/10.3390/plants3010177