Tools to Tie: Flower Characteristics, VOC Emission Profile, and Glandular Trichomes of Two Mexican Salvia Species to Attract Bees
Abstract
:1. Introduction
2. Results
2.1. Floral Traits and Pollinator Monitoring
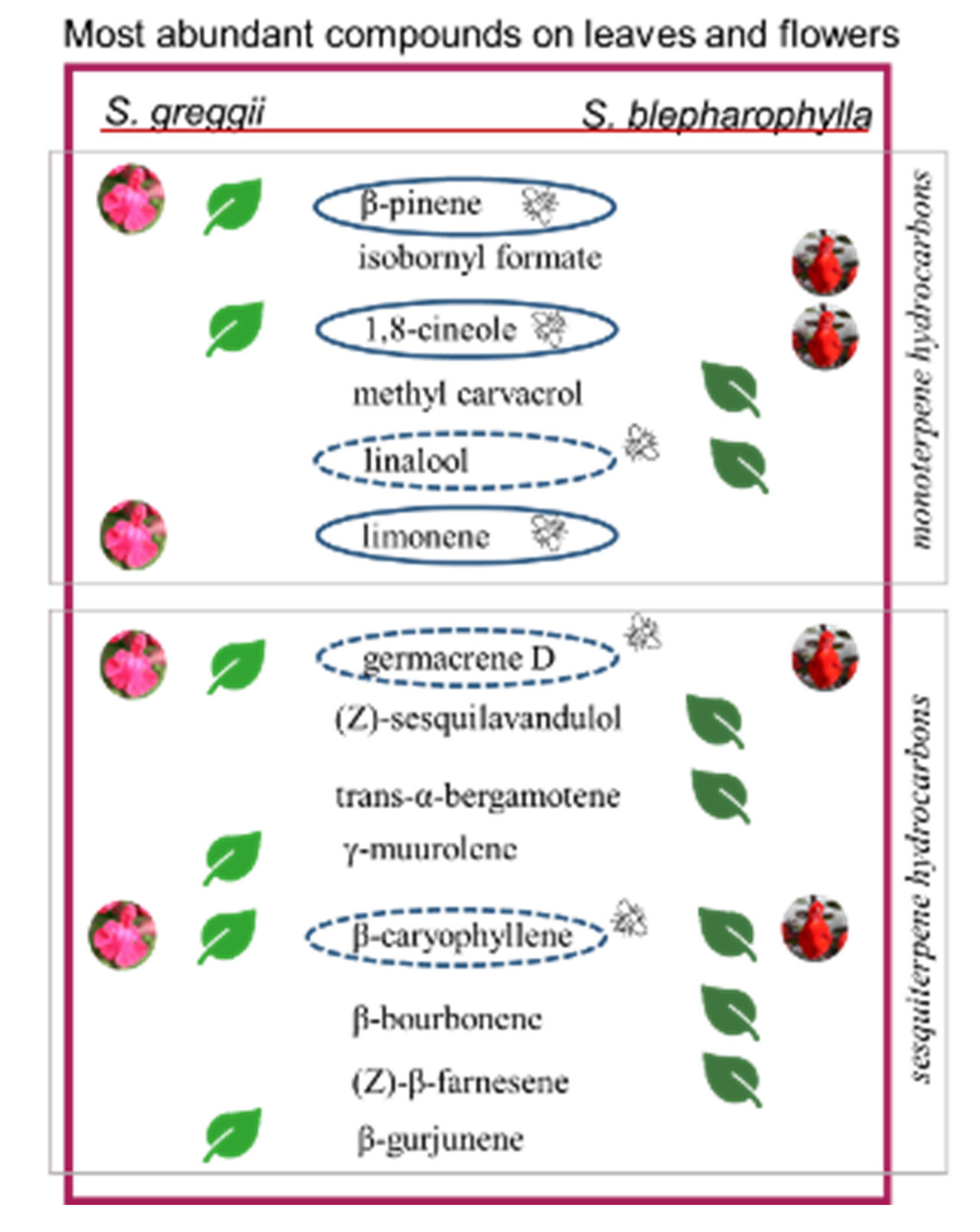
2.2. Glandular Indumenta and Volatile Organic Compounds (VOCs)
- -
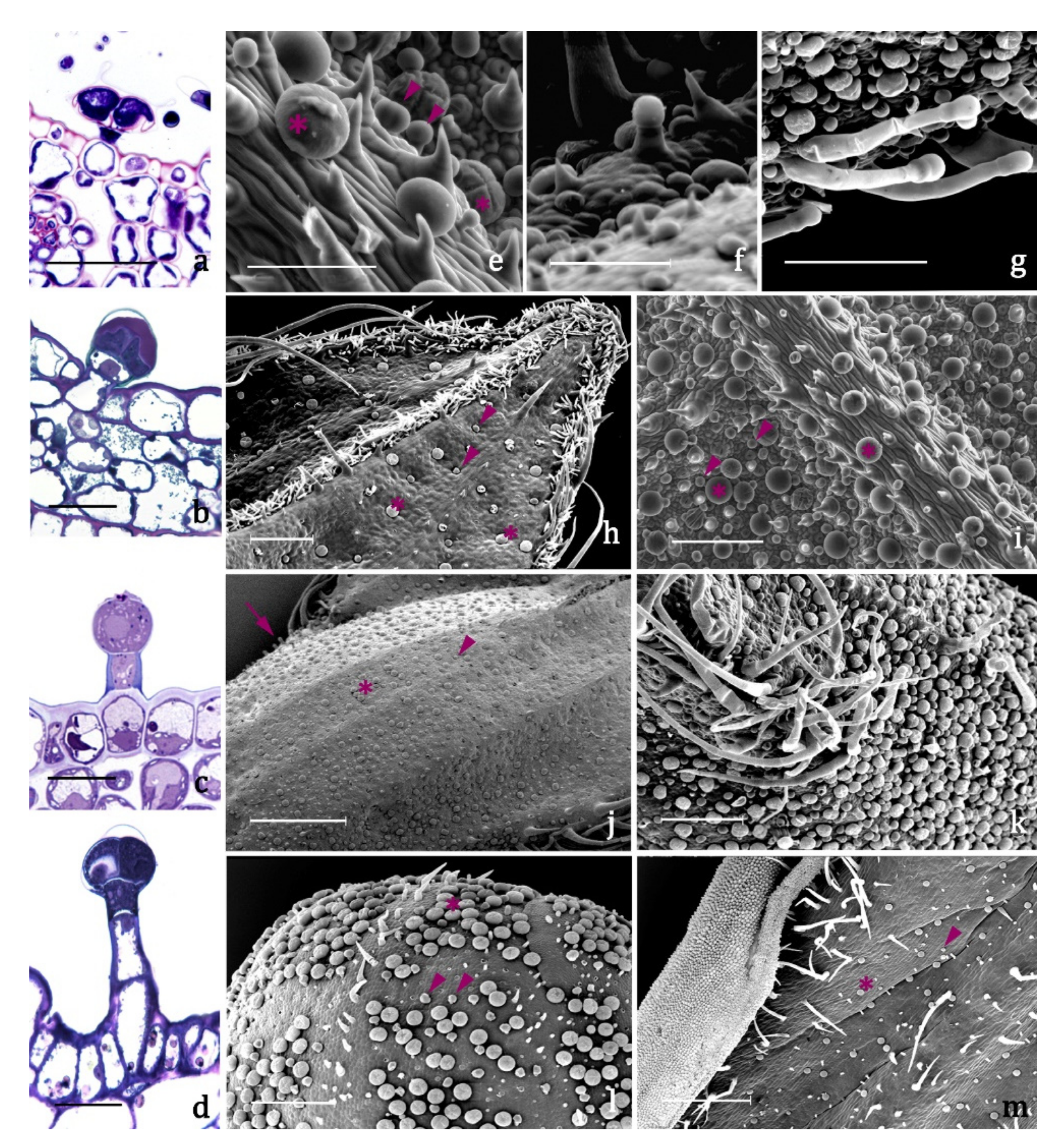
- type A (Figure 3a,e, Table 2), present on leaves and inflorescences of both species (Figure 3h–m), is a typical peltate trichome, constituted by a basal epidermal cell, a neck cell, and by a 4 –cellular glandular head surrounded by a large subcuticular space in which the secretion is stored. The responses to all the lipophilic stains were positive as well as to Ruthenium Red and AlCl3, indicating the presence of terpenes and of major polysaccharide and flavonoid derivatives (Table 3).
- -
- type B (Figure 3b,e, Table 2) is a short capitate trichome, widespread on both the vegetative and the reproductive organs of both examined species (Figure 3h–m). It is constituted by a basal epidermal cell, a neck-stalk cell, and by a glandular head of 2–4 cells surrounded by a wide subcuticular space. Generally, these trichomes present an exclusive polysaccharide secretion released through the intact cuticle (Table 3).
- -
- type C (Figure 3c,f, Table 2) is a medium capitate trichome present only on the calyx of S. blepharophylla (Figure 3j). It is made up of one epidermal cell, one stalk cell, one neck cell and a globose head of 1–2 secretory cells surrounded by a storage chamber. The secretion tested positive to all the lipophilic stains, particularly the NADI reagent, indicating that they are exclusive terpene producers (Table 3).
- -
- type D (Figure 3d,g, Table 2) is a long capitate trichome occurring only on the calyx of S. greggii (Figure 3k). It is composed by 1–2 epidermal cells, two stalk cells, one neck cell and by a head of 2–4 secretory cells. The secreted material stored in the subcuticular space tested positive only to the lipophilic dyes, indicating the exclusive production of terpenes (Table 3).
3. Discussion
4. Materials and Methods
4.1. Plant Material, Floral Traits, and Pollinator Monitoring
4.1.1. Plant Material
4.1.2. Flower Traits
4.1.3. Pollinator Monitoring
4.2. Micro-Morphology of the Glandular Indumentum and Phytochemical Investigation (VOCs)
4.2.1. Scanning Electron Microscopy (SEM) and Light Microscopy (LM)
4.2.2. Headspace-Solid Phase Microextraction (HS-SPME) Analyses, Gas Chromatography–Mass Spectrometry (GC-MS) Analyses, and Peak Identification
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Pichersky, E.; Gershenzon, J. The formation and function of plant volatiles: Perfumes for pollinator attraction and defense. Curr. Opin. Plant Biol. 2002, 5, 237–243. [Google Scholar] [CrossRef]
- Herrera, C.M.; Pellmyr, O. Plant Animal Interactions: An Evolutionary Approach; Wiley-Blackwell: Oxford, UK, 2002. [Google Scholar]
- Maffei, M.E. Sites of synthesis, biochemistry and functional role of plant volatiles. S. Afr. J. Bot. 2010, 76, 612–631. [Google Scholar] [CrossRef] [Green Version]
- Berera, P.; Ronchi, A.; Fico, G. Seduzione e Repulsione, Quello che le Piante non Dicono; Grafo srl: Palazzago, Italy, 2016. [Google Scholar]
- LoPresti, E.F. Chemicals on plant surfaces as a heretofore unrecognized, but ecologically informative, class for investigations into plant defence. Biol. Rev. 2016, 91, 1102–1117. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, C.; Ascrizzi, R.; Lupi, D.; Tassera, G.; Santagostini, L.; Giovanetti, M.; Flamini, G.; Fico, G. Salvia verticillata: Linking glandular trichomes, volatiles and pollinators. Phytochemistry 2018, 155, 53–60. [Google Scholar] [CrossRef] [PubMed]
- War, A.R.; Paulraj, M.G.; Ahmad, T.; Buhro, A.A.; Hussain, B.; Ignacimuthu, S.; Sharma, H.C. Mechanisms of plant defense against insect herbivores. Plant Sign. Behav. 2012, 7, 1306–1320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wester, P.; Claßen-Bockhoff, R. Floral diversity and pollen transfer mechanisms in bird-pollinated Salvia species. Ann. Bot. 2007, 100, 401–421. [Google Scholar] [CrossRef] [Green Version]
- Bisio, A.; Corallo, A.; Gastaldo, P.; Romussi, G.; Ciarallo, G.; Fontana, N.; de Tommasi, N.; Profumo, P. Glandular hairs and secreted material in Salvia blepharophylla Brandegee ex Epling grown in Italy. Ann. Bot. 1999, 83, 441–452. [Google Scholar] [CrossRef] [Green Version]
- Giuliani, C.; Ascrizzi, R.; Corrà, S.; Maleci Bini, L.; Flamini, G.; Fico, G. Ultrastructural insight into terpene-producing trichomes and essential oil profile in Salvia greggii A. Gray. Flora 2017, 236, 107–114. [Google Scholar] [CrossRef]
- Giovanetti, M.; Giuliani, C.; Boff, S.; Fico, G.; Lupi, D. A botanic garden as a tool to combine public perception of nature and life-science investigations on native/exotic plants interactions with local pollinators. PLoS ONE 2020, 15, e0228965. [Google Scholar] [CrossRef] [Green Version]
- Wester, P.; Claßen-Bockhoff, R. Pollination syndromes of New World Salvia species with special reference to bird pollination. Ann. Mo. Bot. Gard. 2011, 98, 101–155. [Google Scholar] [CrossRef]
- Cronk, Q.; Ojeda, I. Bird-pollinated flowers in an evolutionary and molecular context. J. Exp. Bot. 2008, 59, 715–727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wester, P.; Claßen-Bockhoff, R. Hummingbird pollination in Salvia haenkei (Lamiaceae) lacking the typical lever mechanism. Plant Syst. Evol. 2006, 257, 133–146. [Google Scholar] [CrossRef]
- Maloof, J.E.; Inouye, D.W. Are nectar robbers cheaters or mutualists? Ecology 2000, 8, 2651–2661. [Google Scholar] [CrossRef]
- Benitez-Vieyra, S.; Fornoni, J.; Pérez-Alquicira, J.; Boege, K.; Domínguez, C.A. The evolution of signal–reward correlations in bee-and hummingbird-pollinated species of Salvia. Proc. R. Soc. Lond. B Biol. Sci. 2014, 281, 20132934. [Google Scholar] [CrossRef] [Green Version]
- Harder, L.D. Morphology as a predictor of flower choice by bumble bees. Ecology 1985, 66, 198–210. [Google Scholar] [CrossRef]
- Blanco-Pastor, J.L.; Ornosa, C.; Romero, D.; Liberal, I.M.; Gomez, J.M.; Vargas, P. Bees explain floral variation in a recent radiation of Linaria. J. Evol. Biol. 2015, 28, 851–863. [Google Scholar] [CrossRef]
- Rojas-Nossa, S.V.; Sánchez, J.M.; Navarro, L. Nectar robbing: A common phenomenon mainly determined by accessibility constraints, nectar volume and density of energy rewards. Oikos 2016, 125, 1044–1055. [Google Scholar] [CrossRef]
- Antunes, T.; Sevinate-Pinto, I. Glandular trichomes of Teucrium scorodonia L. Morphology and histochemistry. Flora 1991, 185, 65–70. [Google Scholar] [CrossRef]
- Miyake, T.; Yamaoka, R.; Yahara, T. Floral scents of hawkmoth-pollinated flowers in Japan. J. Plant Res. 1998, 111, 199–205. [Google Scholar] [CrossRef]
- Theis, N. Fragrance of Canada thistle (Cirsium arvense) attracts both floral herbivores and pollinators. J. Chem. Ecol. 2006, 32, 917–927. [Google Scholar] [CrossRef]
- Andersson, S.; Nilsson, L.A.; Groth, I.; Bergström, G. Floral scent in butterfly-pollinated plants: Possible convergence in chemical composition. Bot. J. Linn. Soc. 2002, 140, 129–153. [Google Scholar] [CrossRef] [Green Version]
- Dötterl, S.; Vereecken, N.J. The chemical ecology and evolution of bee-flower interactions: A review and perspectives. Can. J. Zool. 2010, 88, 668–697. [Google Scholar] [CrossRef]
- Lemaitre, A.B.; Pinto, C.F.; Niemeyer, H.M. Generalized pollination system: Are floral traits adapted to different pollinators? Arthropod Plant Interact. 2014, 8, 261–272. [Google Scholar] [CrossRef]
- Borg-Karlson, A.K.; Unelius, C.R.; Valterova, I.; Nilsson, A. Floral fragrance chemistry in the early flowering shrub Daphne mezereum. Phytochemistry 1996, 41, 1477–1483. [Google Scholar] [CrossRef]
- Williams, N.H.; Whitten, W.M. Orchid floral fragrances and male Euglossine bees: Methods and advances in the last sesquidecade. Biol. Bull. 1983, 164, 355–395. [Google Scholar] [CrossRef]
- Granero, A.M.; Guerra Sanz, J.M.G.M.; Gonzalez, F.J.E.; Martinez Vidal, J.L.; Dornhaus, A.; Ghani, J.; Serrano, A.R.; Chittka, L. Chemical compounds of the foraging recruitment pheromone in bumblebees. Naturwissenschaften 2005, 92, 371–374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Damon, A.; Roblero, P.S. A survey of pollination in remnant orchid populations in Soconusco Chiapas, Mexico. Trop. Ecol. 2007, 48, 1–14. [Google Scholar]
- Valterová, I.; Kunze, J.; Gumbert, A.; Luxovà, A.; Liblikas, I.; Kalinovà, B.; Karin, A.; Karlson, B. Male bumble bee pheromonal components in the scent of deceit pollinated orchids; unrecognized pollinator cues? Arthropod Plant Interact. 2007, 1, 137–145. [Google Scholar] [CrossRef]
- Blight, M.M.; Le Métayer, M.; Delègue, M.H.P.; Pickett, J.A.; Marion-Poll, F.; Wadhams, L.J. Identification of floral volatiles involved in recognition of oilseed rape flowers, Brassica napus by honeybees, Apis mellifera. J. Chem. Ecol. 1997, 23, 1715–1727. [Google Scholar] [CrossRef]
- Byers, K.J.R.P.; Bradshaw, H.D., Jr.; Riffell, J.A. Three floral volatiles contribute to differential pollinator attraction in monkeyflowers (Mimulus). J. Exp. Bot. 2014, 217, 614–623. [Google Scholar] [CrossRef] [Green Version]
- Aronne, G.; Giovanetti, M.; Sacchi, R.; DeMicco, V. From flower to honey bouquet: Possible markers for the botanical origin of Robinia honey. Sci. World J. 2014, 547275. [Google Scholar] [CrossRef] [Green Version]
- Hetherington-Rauth, M.C.; Ramırez, S.R. Evolution and diversity of floral scent chemistry in the euglossine bee-pollinated orchid genus Gongora. Ann. Bot. 2016, 118, 135–148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lusebrink, I.; Girling, R.D.; Farthing, T.E.; Newman, A.; Chris, W.J.; Poppy, G.M. The effects of diesel exhaust pollution on floral volatiles and the consequences for honey bee olfaction. Chem. Ecol. 2015, 41, 904–912. [Google Scholar] [CrossRef] [PubMed]
- Le Metayer, M.; Marion-Poll, F.; Sandoz, J.C.; Pham-Delegue, M.H.; Blight, M.M.; Wadhams, L.J.; Masson, C.; Woodcock, C.M. Effect of conditioning on discrimination of oilseed rape volatiles by the honeybee: Use of a combined gas chromatography-proboscis extension behavioural assay. Chem. Senses 1997, 22, 391–398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grajales-Conesa, J.; Meléndez Ramírez, V.; Cruz-López, L.; Sánchez Guillén, D. Effect of Citrus floral extracts on the foraging behavior of the stingless bee Scaptotrigona pectoralis (Dalla Torre). Rev. Bras. Entomol. 2012, 56, 76–80. [Google Scholar] [CrossRef] [Green Version]
- Shanower, T.G. Trichomes and insects. In Encyclopedia of Entomology; Capinera, J.L., Ed.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 2333–2335. [Google Scholar]
- Karabourniotis, G.; Liakopoulos, G.; Nikolopoulos, D.; Bresta, P. Protective and defensive roles of non-glandular trichomes against multiple stresses: Structure–function coordination. J. For. Res. 2020, 31, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Theis, N.; Barber, N.A.; Gillespie, S.D.; Hazzard, R.V.; Adler, L.S. Attracting mutualists and antagonists: Plant trait variation explains the distribution of specialist floral herbivores and pollinators on crops and wild gourds. Am. J. Bot. 2014, 101, 1314–1322. [Google Scholar] [CrossRef] [Green Version]
- Ashton Acton, Q. Issues in Ecological Research and Application; Scholarly Editions: Atlanta, GA, USA, 2011. [Google Scholar]
- Köllner, T.G.; Lenk, C.; Schnee, C.; Köpke, S.; Lindemann, P.; Gershenzon, J.; Degenhardt, J. Localization of sesquiterpene formation and emission in maize leaves after herbivore damage. BMC Plant Biol. 2013, 13, 15. [Google Scholar] [CrossRef] [Green Version]
- Brandegee, T.S. Salvia blepharophylla. Repert. Spec. Nov. Regni Veg. Beih. 1939, 110, 314. [Google Scholar]
- Gray, A. Salvia greggii. Proc. Am. Acad. Arts Sci. 1872, 8, 369. [Google Scholar]
- Waites, A.R.; Agren, J.O.N. Pollinator visitation, stigmatic pollen loads and among-population variation in seed set in Lythrum salicaria. J. Ecol. 2004, 92, 512–526. [Google Scholar] [CrossRef]
- Jensen, W.A. Botanical Histochemistry: Principles and Practice; WH Freeman & Co: San Francisco, CA, USA, 1962. [Google Scholar]
- David, R.; Carde, J.P. Coloration différentielle des inclusions lipidiques et terpéniques des pseudophylles du Pin maritime au moyen du reactif. NADI. C. R. Biol. 1964, 258, 1338–1340. [Google Scholar]
- Beccari, N.; Mazzi, V. Manuale di Tecnica Microscopica; Società Editrice Libraria: Como, Italy, 1966. [Google Scholar]
- Greenspan, P.; Mayer, E.P.; Fowler, S.D. Nile red: A selective fluorescent stain for intracellular lipids droplets. J. Cell Biol. 1985, 100, 965–973. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giuliani, C.; Ascrizzi, R.; Tani, C.; Bottoni, M.; Maleci Bini, L.; Flamini, G.; Fico, G. Salvia uliginosa Benth.: Glandular trichomes as bio-factories of volatiles and essential oil. Flora 2017, 233, 12–21. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy; Allured Pub. Corp: Carol Stream, IL, USA, 2007. [Google Scholar]
- National Institute of Standards and Technology NIST/EPA/NIH Mass Spectral Library; The NIST Mass Spectrometry Data Center: Gaithersburg, MD, USA, 2014.


| a | b | c | d | E | f | |
|---|---|---|---|---|---|---|
| S. blepharophylla | 17.79 1 (0.13) | 27.21 1 (0.42) | 10.60 1 (0.42) | 18.63 1 (0.38) | 16.61 1 (0.84) | 0.64 1 (0.05) |
| S. greggii | 10.50 2 (0.26) | 24.66 2 (0.77) | 10.44 1 (0.39) | 16.40 2 (0.35) | 14.22 2 (0.38) | 0.73 2 (0.01) |
| Trichome Morphotype | Leaf | Calyx | Corolla | ||||
|---|---|---|---|---|---|---|---|
| Abaxial | Adaxial | Abaxial | Adaxial | Abaxial | Adaxial | ||
| S. blepharophylla | A | + | + | + | - | + | + |
| B | + | + | + | + | + | + | |
| C | - | - | + | - | - | - | |
| S. greggii | A | + | + | + | + | + | + |
| B | + | + | + | + | + | + | |
| D | - | - | + | - | - | - | |
| Staining Procedure | Target Compounds | Observed Colour | S. blepharophylla | S. greggii | ||||
|---|---|---|---|---|---|---|---|---|
| Type A | Type B | Type C | Type A | Type B | Type D | |||
| Nile Red | Neutral lipids | Golden-yellow | ++ | - | ++ | ++ | - | ++ |
| Fluoral yellow-088 | Total lipids | Yellow to orange | ++ | - | ++ | ++ | - | ++ |
| NADI reagent | Terpenes | Violet-blue | ++ | - | ++ | ++ | - | ++ |
| FeCl3 | Polyphenols | Emerald-green | + | - | - | ++ | - | - |
| AlCl3 | Flavonoids | Blue-green | + | - | - | + | - | - |
| Ruthenium red | Acid polysaccharides | Pinkish to red | + | + | - | + | + | - |
| Alcian blue | Muco- polysaccharides | Pale-blue | + | + | - | + | + | - |
| Salvia blepharophylla | Salvia greggii | |||||
|---|---|---|---|---|---|---|
| l.r.i.a | Compounds | Relative Abundance (%) | Relative Abundance (%) | |||
| Flowers | Leaves | Flowers | Leaves | |||
| 1 | 941 | α-pinene | 2.14 | -b | 2.27 | 3.23 |
| 2 | 954 | camphene | - | - | - | 0.57 |
| 3 | 982 | β-pinene | 2.44 | - | 14.32 | 24.96 |
| 4 | 993 | myrcene | - | - | 1.90 | - |
| 5 | 1005 | α-phellandrene | - | - | 0.45 | - |
| 6 | 1032 | limonene | - | 1.66 | 55.20 | - |
| 7 | 1034 | 1,8-cineole | 45.68 | - | - | 19.56 |
| 8 | 1052 | (E)-β-ocimene | - | 3.71 | - | - |
| 9 | 1062 | γ-terpinene | 0.36 | 0.86 | 0.22 | - |
| 10 | 1070 | cis-sabinene hydrate | - | - | 0.25 | 0.34 |
| 11 | 1076 | trans-linalool oxide (furanoid) | - | - | - | 0.89 |
| 12 | 1088 | terpinolene | - | 0.44 | 0.42 | - |
| 13 | 1090 | cis-linalool oxide (furanoid) | - | - | - | 0.67 |
| 14 | 1101 | linalool | - | 5.07 | - | 2.11 |
| 15 | 1102 | nonanal | 0.56 | - | - | - |
| 16 | 1104 | α-thujone | - | 0.41 | - | - |
| 17 | 1134 | cis-limonene oxide | - | - | 0.20 | - |
| 18 | 1140 | nopinone | 0.75 | 0.67 | - | - |
| 19 | 1141 | trans-limonene oxide | - | - | 3.63 | 0.08 |
| 20 | 1143 | camphor | 2.95 | - | - | 2.09 |
| 21 | 1156 | isoborneol | - | - | - | 0.17 |
| 22 | 1158 | sabinaketone | - | 0.47 | - | - |
| 23 | 1162 | trans-pinocamphone | - | - | 0.15 | 0.26 |
| 24 | 1167 | borneol | - | - | - | 0.26 |
| 25 | 1170 | δ-terpineol | - | - | - | 0.05 |
| 26 | 1178 | 4-terpineol | 1.23 | - | 0.19 | - |
| 27 | 1187 | (Z)-3-hexenyl-butyrate | 0.97 | - | - | - |
| 28 | 1192 | methyl salicylate | - | - | - | 0.14 |
| 29 | 1195 | γ-terpineol | - | - | 0.20 | - |
| 30 | 1202 | trans-dihydro carvone | - | - | 0.19 | - |
| 31 | 1204 | decanal | 0.63 | 1.88 | - | 0.23 |
| 32 | 1232 | isobornyl formate | 8.56 | 0.73 | - | - |
| 33 | 1241 | methyl carvacrol | 5.32 | 10.68 | 0.61 | 0.23 |
| 34 | 1259 | linalool acetate | - | 0.80 | - | - |
| 35 | 1272 | n-decanol | - | 0.08 | - | - |
| 36 | 1283 | (E)-anethole | - | 0.92 | - | - |
| 37 | 1285 | isobornyl acetate | 1.91 | - | - | - |
| 38 | 1300 | n-tridecane | - | 0.56 | - | - |
| 39 | 1340 | δ-elemene | - | 0.65 | - | - |
| 40 | 1351 | α-cubebene | - | - | - | 0.05 |
| 41 | 1368 | cyclosativene | - | - | 0.20 | 0.24 |
| 42 | 1376 | α-copaene | 0.86 | - | 0.53 | 2.6 |
| 43 | 1384 | β-bourbonene | 1.20 | 10.43 | 0.83 | 2.74 |
| 44 | 1390 | β-cubebene | 0.28 | - | 0.23 | 0.4 |
| 45 | 1391 | 7-epi-sesquithujene | - | 0.77 | - | - |
| 46 | 1392 | β-elemene | - | 1.05 | 0.15 | 0.55 |
| 47 | 1400 | n-tetradecane | - | 0.23 | - | - |
| 48 | 1403 | longifolene | 0.41 | - | - | 0.16 |
| 49 | 1409 | α-cedrene | 0.68 | 0.28 | - | - |
| 50 | 1420 | β-caryophyllene | 6.84 | 11.07 | 5.73 | 5.59 |
| 51 | 1429 | β-copaene | 0.48 | 1.33 | 0.39 | 0.65 |
| 52 | 1432 | β-gurjunene | - | - | 0.41 | 6.74 |
| 53 | 1438 | trans-α-bergamotene | - | 6.89 | - | - |
| 54 | 1439 | α-guaiene | - | - | - | 0.12 |
| 55 | 1441 | aromadendrene | - | - | 0.35 | 0.15 |
| 56 | 1445 | (Z)-β-farnesene | - | 6.82 | - | - |
| 57 | 1455 | (E)-geranyl acetone | 0.41 | 0.41 | - | - |
| 58 | 1456 | α-humulene | 0.76 | 2.34 | - | 0.45 |
| 59 | 1461 | alloaromadendrene | - | 1.40 | 0.16 | 0.93 |
| 60 | 1462 | cis-muurola-4(14),5-diene | - | 0.20 | 0.27 | 0.21 |
| 61 | 1470 | trans-cadina-1(6),4-diene | 2.65 | - | - | - |
| 62 | 1477 | γ-muurolene | 0.79 | - | 1.48 | 10.2 |
| 63 | 1480 | γ-curcumene | - | 0.06 | - | - |
| 64 | 1481 | germacrene D | 5.01 | 4.22 | 6.37 | 7.22 |
| 65 | 1490 | (E,Z)-α-farnesene | - | 0.80 | - | - |
| 66 | 1491 | trans-muurola-4(14),5-diene | - | - | 0.16 | - |
| 67 | 1492 | valencene | 0.46 | - | - | 0.51 |
| 68 | 1495 | bicyclogermacrene | - | 2.38 | - | - |
| 69 | 1496 | γ-amorphene | - | - | - | 0.13 |
| 70 | 1498 | α-muurolene | - | - | 0.30 | 1.08 |
| 71 | 1500 | n-pentadecane | - | 0.32 | - | - |
| 72 | 1502 | γ-patchoulene | 0.52 | 0.10 | 0.29 | - |
| 73 | 1507 | (E,E)-α-farnesene | - | 2.42 | 0.29 | 0.82 |
| 74 | 1513 | trans-γ-cadinene | 0.98 | - | 0.70 | 0.43 |
| 75 | 1524 | β-sesquiphellandrene | - | 1.73 | - | - |
| 76 | 1524 | δ-cadinene | 0.69 | - | 0.24 | 0.40 |
| 77 | 1549 | elemol | - | 0.25 | - | - |
| 78 | 1565 | (E)-nerolidol | - | 1.29 | - | - |
| 79 | 1575 | germacrene D-4-ol | - | - | - | 0.16 |
| 80 | 1576 | spathulenol | - | 2.16 | - | - |
| 81 | 1593 | (Z)-sesquilavandulol | - | 9.63 | - | - |
| 82 | 1595 | guaiol | - | - | - | 0.76 |
| 83 | 1600 | n-hexadecane | - | 1.28 | - | - |
| 84 | 1606 | humulene epoxide II | - | 0.14 | - | - |
| 85 | 1640 | epi-α-cadinol | - | - | 0.18 | - |
| 86 | 1693 | juniperol acetate | - | 0.33 | - | - |
| 87 | 1700 | n-heptadecane | - | 0.08 | - | - |
| Monoterpene hydrocarbons | 4.94 | 6.67 | 74.78 | 28.76 | ||
| Oxygenated monoterpenes | 66.40 | 18.83 | 5.42 | 26.71 | ||
| Sesquiterpene hydrocarbons | 22.61 | 54.95 | 19.08 | 42.37 | ||
| Oxygenated sesquiterpenes | - | 13.80 | 0.18 | 0.92 | ||
| Phenylpropanoids | - | 0.92 | - | - | ||
| Apocarotenoids | 0.41 | 0.41 | - | - | ||
| Other non-terpene derivatives | 2.16 | 4.43 | - | 0.37 | ||
| Total identified (%) | 96.52 | 100.00 | 99.46 | 99.13 | ||
| Compound | Bee Species | Reference |
|---|---|---|
| 1,8-Cineole | Euglossini, Bombus terrestris; Bombus vorticosus | [26,27,28,29] |
| α-Pinene | Euglossini, Apis mellifera; Honeybees | [26,30] |
| β-Pinene | Bombus; Honeybees | [31,32,33] |
| Limonene | Bombus, Honeybees; B. terrestris; B. vorticosus | [29,31] |
| β-Caryophyllene | Apis mellifera; | [29,34] |
| α-Farnesene | [24] | |
| (E,E)-α-Farnesene | B. terrestris; B. vorticosus; Apis mellifera | [29,31,35] |
| Linalool | Colletidae bees; Apidae; Lasioglossum spp. | [36] |
| (E)-β-Ocimene | Colletidae bees; Apidae; Lasioglossum spp. | [36] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giuliani, C.; Giovanetti, M.; Lupi, D.; Mesiano, M.P.; Barilli, R.; Ascrizzi, R.; Flamini, G.; Fico, G. Tools to Tie: Flower Characteristics, VOC Emission Profile, and Glandular Trichomes of Two Mexican Salvia Species to Attract Bees. Plants 2020, 9, 1645. https://doi.org/10.3390/plants9121645
Giuliani C, Giovanetti M, Lupi D, Mesiano MP, Barilli R, Ascrizzi R, Flamini G, Fico G. Tools to Tie: Flower Characteristics, VOC Emission Profile, and Glandular Trichomes of Two Mexican Salvia Species to Attract Bees. Plants. 2020; 9(12):1645. https://doi.org/10.3390/plants9121645
Chicago/Turabian StyleGiuliani, Claudia, Manuela Giovanetti, Daniela Lupi, Marco Palamara Mesiano, Renata Barilli, Roberta Ascrizzi, Guido Flamini, and Gelsomina Fico. 2020. "Tools to Tie: Flower Characteristics, VOC Emission Profile, and Glandular Trichomes of Two Mexican Salvia Species to Attract Bees" Plants 9, no. 12: 1645. https://doi.org/10.3390/plants9121645







