1. Introduction
The extensively used formaldehyde is produced by using air and methanol as the raw materials. The reaction occurs in the reactor in the presence of a catalyst. The resulting products of the reaction are formaldehyde and water [
1]. Then the mixture of products and unreacted reactants goes to the absorption column where water is showered from the top. The bottom product is formalin i.e., a 37% aqueous solution of formaldehyde [
2]. The unreacted reaction mixture is removed from the top [
1,
3,
4,
5,
6,
7]. The extensive range of applications of formaldehyde makes it a valuable chemical. It may be used in different industries such as domestic, medical, cosmetics, and the textile industry [
8,
9,
10].
The consumption and demand of formaldehyde is increasing. Formaldehyde is the principal component for the production of resins, phenols, urea, and melamine [
11]. It is used for weather resistance i.e., in adhesives and wood coatings [
12]. In addition, it has a disinfectant property; it is present in soaps as a disinfectant. In medical fields, formaldehyde is used for the sterilization of the surgical instruments. It imparts the resistance to fabric against crumples. In cosmetic products, formaldehyde is used as a preservative since it enhances the effectiveness of products against different microorganisms. It is used in glue production for household use. Formaldehyde is used in the manufacturing of plastics, carpets, and vaccines, etc. In plastic utensils industry, it is the major component [
13].
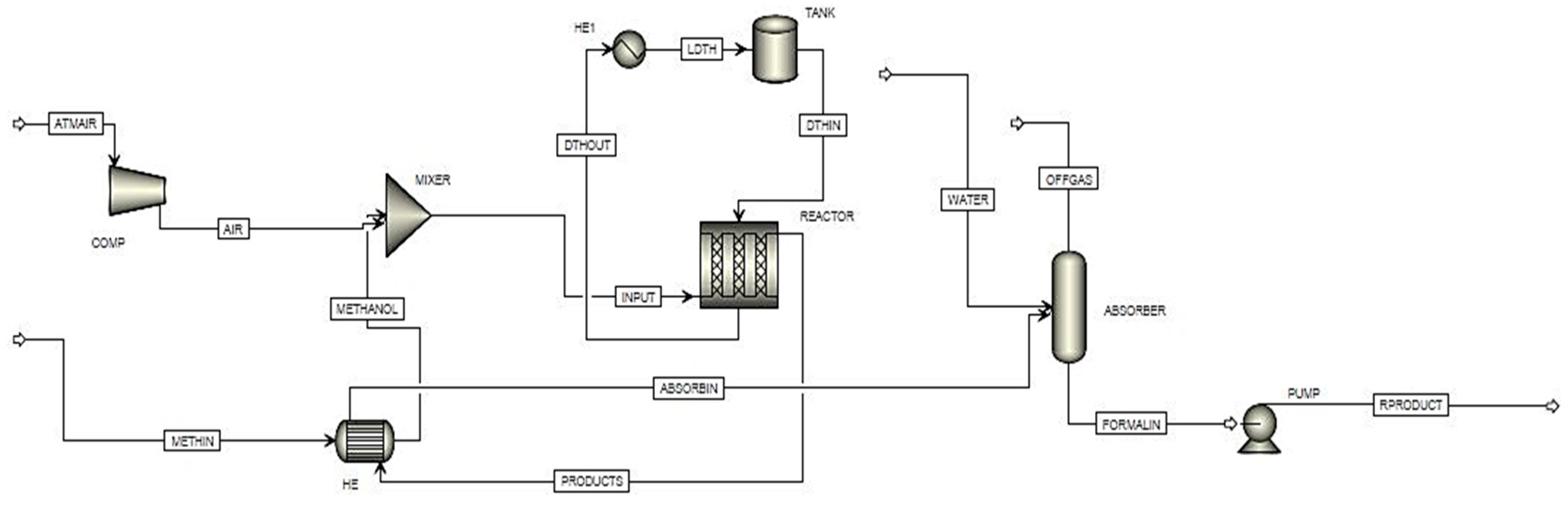
Commercially, formaldehyde is produced mostly from air and methanol as raw materials using three different methods. In the first method, formaldehyde is produced using air and methanol in the presence of molybdenum oxide catalyst present inside the tubes of shell and tube reactor [
14]. The reacting mixture enters at tube side to interact with catalyst forming the product [
15].
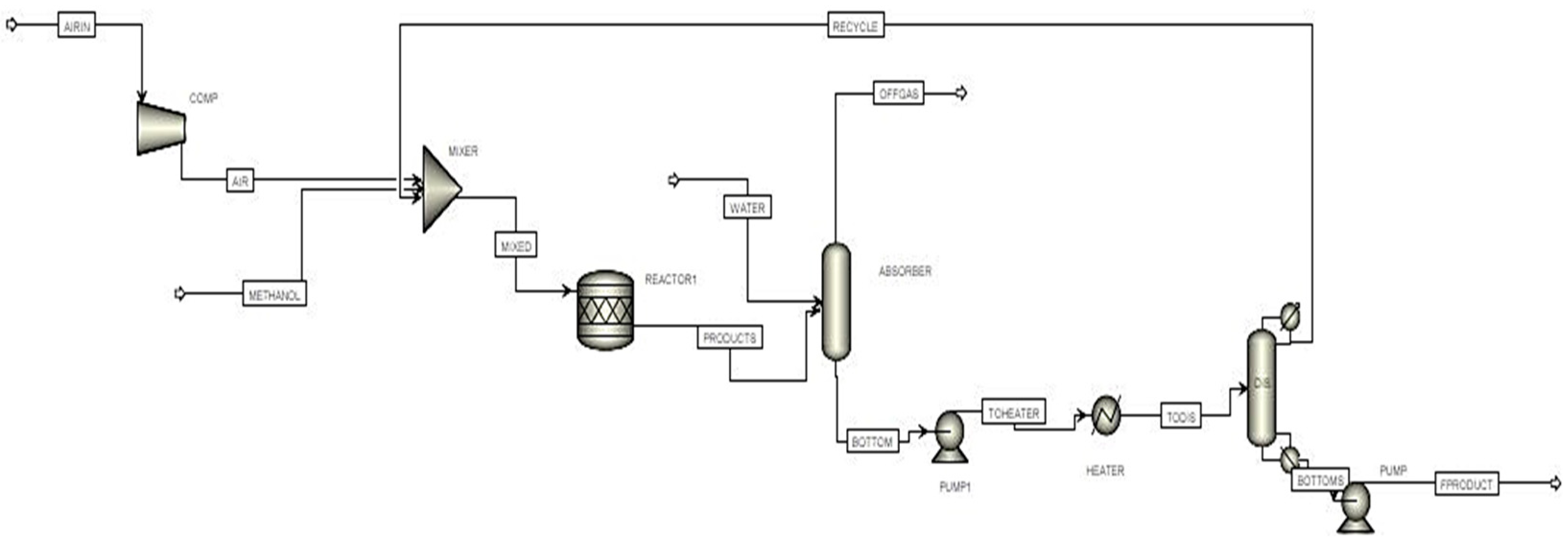
The second method involves the production of formaldehyde in the presence of silver oxide catalyst present in fixed catalytic bed reactor [
16].
In third method, formaldehyde is produced using oxidation of methane and other hydrocarbons [
17]. The separation processes and reaction mechanism in the above three methods are almost the same. For commercial production of formaldehyde, process optimization is required.
Lefferts et al. studied the production process of formaldehyde through oxidative hydrogenation of methanol in the presence of silver catalyst [
18]. They studied the effect of temperature, gas velocity, and concentration of both reactants on the production process. They developed the reaction model based on the experimental data and explained the impact of form and composition of silver catalyst over methanol conversion. Yang et al. used molybdenum oxide catalyst supported over silica for the oxidation of methanol to formaldehyde [
19]. Their study was based on the selectivity and activity of N
2O and O
2 used as oxidants. They observed that N
2O is responsible for the oxidation of carbon monoxide. Moreover, the supported molybdenum catalyst has higher activity than the non-supported catalyst. Qian et al. explained the formaldehyde synthesis process using polycrystalline silver catalyst [
20]. They compared the water ballast process with the methanol ballast process and observed an increased selectivity of formaldehyde in the absence of water. Moreover, the selectivity of the product is highly temperature dependent. Moreover, Waterhouse et al. used SEM techniques to determine the relationship between morphology of silver catalyst and its performance [
21].
In this study, a performance comparison of the industrially produced formaldehyde using two different catalysts is presented. Real-time industrial data are collected from a local industry in Pakistan, and material and energy balances, simulations, and cost analysis are executed. We have compared the two different catalysts based on material and energy balances, the size of the plant, the installation, and utility cost.
4. Catalyst Properties
The properties of the molybdenum oxide and silver oxide catalyst used in the production process are shown in
Table 1. It can be observed that the conversion of the molybdenum oxide-based plant is 99% and that of the silver oxide-based plant is 85%. The life of the molybdenum oxide catalyst is 12–18 months and that of the silver oxide-based process is 3–8 months. The porosity of the molybdenum oxide catalyst is 0.7 and that of the silver oxide catalyst is 0.5.
The life of the silver catalyst is highly dependent upon the operating conditions of the formaldehyde reactor. Silver catalysts in the formaldehyde plant are typically used for a period of a few months to a year depending on the reaction temperature and pressure [
22]. Generally, sintering occurs in the silver catalyst due to reaction temperatures, which results in high pressure drop over the bed, which decreases the performance of catalyst. It is observed that the water ballast process prolongs the life of catalyst by introducing water with its associated high heat capacity that equally distributes the heat over the catalyst bed resulting in minimizing coke formation and sintering [
23,
24]. Our objective is the performance comparison of both processes based on the plant size, installation and utility cost, and material and energy balance.
6. Conclusions
The performance of two catalysts was studied simultaneously and the results were compared. The molybdenum-based plant has a larger plant size, high utility cost, but low cost of installation. Moreover, it has higher conversion of methanol to product i.e., 99%. The silver-based plant requires half the amount of air as compared to the molybdenum-based process, so it has compact plant size, the installation cost is high, but utility cost is low. However, the conversion is less in this process i.e., 75–85% as compared to the molybdenum-based process. Based on the comparative study and calculations, the silver-based process for formaldehyde production is better on an industrial scale.