Ripening Changes of the Chemical Composition, Proteolysis, and Lipolysis of a Hair Sheep Milk Mexican Manchego-Style Cheese: Effect of Nano-Emulsified Curcumin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Milk and NEC
2.2. Manufacturing of Mexican Manchego-Style Cheese
2.3. Chemical Composition of Cheeses
2.4. Total Phenolic Content (TPC) and Antioxidant Activity (AA) in Cheeses
2.5. Color in Manchego-Style Cheeses
2.6. Fatty Acid Composition of Cheeses
2.7. Lipolysis
2.8. Proteolysis
2.9. Data Analysis
3. Results and Discussion
3.1. Chemical Composition of Cheeses
3.2. Total Phenolic Content (TPC) and Antioxidant Activity (AA) in Cheeses
| Cheese Samples | Ripening Time (days) | Total Phenolic Content | Antioxidant Activity by DPPH | Antioxidant Activity by FRAP |
|---|---|---|---|---|
| B 10 ppm | 0 | 104.62 ± 4.37 a1 | 10.24 ± 0.08 a1 | 2.67 ± 0.18 a1 |
| 20 | 171.95 ± 7.71 b1 | 10.79 ± 0.23 ab1 | 2.78 ± 0.21 a1 | |
| 40 | 206.30 ± 3.33 c1 | 10.95 ± 0.17 b1 | 3.33 ± 0.19 b1 | |
| 60 | 213.49 ± 3.56 c1 | 11.66 ± 0.81 c1 | 3.45 ± 0.03 bc1 | |
| 80 | 251.53 ± 13.81 d1 | 10.62 ± 0.42 ab1 | 3.61 ± 0.12 c1 | |
| C 5 ppm | 0 | 111.96 ± 2.98 a12 | 10.57 ± 0.37 a12 | 2.98 ± 0.05 a2 |
| 20 | 183.62 ± 3.31 b2 | 11.30 ± 0.10 b12 | 3.44 ± 0.10 b2 | |
| 40 | 241.84 ± 3.85 c2 | 11.48 ± 0.14 b12 | 3.58 ± 0.08 bc12 | |
| 60 | 245.65 ± 8.81 c2 | 11.90 ± 0.39 b1 | 3.74 ± 0.30 c2 | |
| 80 | 286.04 ± 14.66 d2 | 11.28 ± 0.27 b2 | 3.82 ± 0.12 c2 | |
| C 7.5 ppm | 0 | 119.23 ± 0.06 a2 | 11.04 ± 0.27 a23 | 3.27 ± 0.01 a3 |
| 20 | 187.73 ± 4.58 b23 | 11.51 ± 0.04 ab2 | 3.63 ± 0.05 b23 | |
| 40 | 253.29 ± 7.41 c2 | 11.91 ± 0.17 b23 | 3.90 ± 0.11 c2 | |
| 60 | 280.90 ± 3.13 d3 | 12.66 ± 0.47 c2 | 4.27 ± 0.09 d3 | |
| 80 | 309.68 ± 5.44 e3 | 11.75 ± 0.14 b23 | 4.27 ± 0.03 d3 | |
| C 10 ppm | 0 | 137.22 ± 7.41 a3 | 11.44 ± 0.17 a3 | 3.59 ± 0.12 a4 |
| 20 | 197.20 ± 6.00 b3 | 12.22 ± 0.41 b3 | 3.89 ± 0.24 b3 | |
| 40 | 267.24 ± 1.02 c3 | 12.41 ± 0.20 b3 | 4.55 ± 0.10 c3 | |
| 60 | 307.91 ± 4.41 d4 | 13.73 ± 0.64 c3 | 4.73 ± 0.10 c4 | |
| 80 | 318.34 ± 8.03 d3 | 12.30 ± 0.54 b3 | 4.64 ± 0.11 c4 |
3.3. Color in Manchego-Style Cheeses
| Cheese Samples | Ripening Time (days) | L* | a* | b* |
|---|---|---|---|---|
| B 10 ppm | 0 | 78.27 ± 0.85 a | − 2.27 ± 0.15 a1 | 6.00 ± 0.75 a1 |
| 20 | 77.67 ± 0.45 a | 0.23 ± 0.49 b1 | 9.93 ± 0.90 b1 | |
| 40 | 77.90 ± 0.70 a1 | 0.67 ± 0.12 b1 | 9.73 ± 1.65 b1 | |
| 60 | 77.13 ± 1.51 ab1 | 0.57 ± 0.67 b1 | 11.57 ± 1.66 c1 | |
| 80 | 75.33 ± 1.27 b12 | 0.43 ± 0.29 b1 | 11.70 ± 2.29 c1 | |
| C 5 ppm | 0 | 76.57 ± 1.50 a | − 3.17 ± 0.15 a1 | 10.53 ± 0.98 a2 |
| 20 | 76.43 ± 1.50 a | − 3.20 ± 0.69 a2 | 13.57 ± 0.99 b2 | |
| 40 | 74.80 ± 0.96 ab2 | − 1.47 ± 0.83 b2 | 18.90 ± 0.82 c3 | |
| 60 | 75.03 ± 0.49 ab12 | − 0.90 ± 0.78 bc23 | 18.07 ± 2.12 c2 | |
| 80 | 72.97 ± 0.35 b3 | − 0.27 ± 0.50 c1 | 17.70 ± 2.21 c2 | |
| C 7.5 ppm | 0 | 76.57 ± 0.25 ab | − 4.93 ± 0.15 a2 | 12.37 ± 1.05 a23 |
| 20 | 78.27 ± 1.31 a | − 4.87 ± 1.12 a3 | 15.20 ± 1.11 b23 | |
| 40 | 74.80 ± 1.23 bc2 | − 1.40 ± 0.72 b2 | 19.00 ± 1.84 c3 | |
| 60 | 74.03 ± 1.04 c2 | − 0.73 ± 0.76 b2 | 18.80 ± 1.77 c2 | |
| 80 | 74.77 ± 1.40 bc23 | − 0.43 ± 0.55 b1 | 22.60 ± 2.51 d3 | |
| C 10 ppm | 0 | 77.63 ± 0.78 | − 5.17 ± 0.21 a2 | 14.17 ± 0.75 a3 |
| 20 | 76.73 ± 2.12 | − 5.80 ± 0.46 a3 | 18.43 ± 1.95 b3 | |
| 40 | 77.37 ± 1.02 1 | − 2.13 ± 0.47 b2 | 13.80 ± 0.95 a2 | |
| 60 | 76.50 ± 0.36 1 | − 1.90 ± 0.78 b3 | 20.30 ± 0.90 b2 | |
| 80 | 77.27 ± 1.12 1 | − 2.10 ± 0.70 b2 | 24.10 ± 3.73 c3 |
3.4. Fatty Acid Composition of Cheeses
| Fatty Acid | Cheese Samples | |||
|---|---|---|---|---|
| B 10 ppm | C 5 ppm | C 7.5 ppm | C 10 ppm | |
| 4:0 | 5.49 ± 2.08 a | 11.90 ± 2.02 b | 8.14 ± 2.81 ab | 6.63 ± 2.66 ab |
| 6:0 | 3.94 ± 1.67 a | 4.84 ± 0.51 a | 5.74 ± 1.12 a | 5.11 ± 1.56 a |
| 8:0 | 2.97 ± 0.53 a | 2.98 ± 0.28 a | 4.67 ± 1.48 a | 4.41 ± 1.19 a |
| 10:0 | 7.07 ± 0.68 a | 6.72 ± 0.75 a | 6.34 ± 0.43 a | 7.43 ± 0.68 a |
| 12:0 | 4.59 ± 0.30 a | 4.13 ± 0.46 ab | 3.98 ± 0.01 b | 4.68 ± 0.27 a |
| 14:0 | 10.44 ± 0.50 a | 9.27 ± 0.57 b | 9.59 ± 0.17 b | 10.75 ± 0.65 a |
| 14:1 n-9 | 0.21 ± 0.01 a | 0.20 ± 0.01 a | 0.21 ± 0.01 a | 0.22 ± 0.02 a |
| 14:1 n-7 | 0.22 ± 0.02 a | 0.19 ± 0.02 a | 0.19 ± 0.02 a | 0.22 ± 0.01 a |
| 14:1 n-5 | 0.40 ± 0.02 a | 0.36 ± 0.01 b | 0.36 ± 0.01 b | 0.42 ± 0.03 a |
| 15:0 | 0.75 ± 0.07 ab | 0.65 ± 0.05 a | 0.71 ± 0.02 a | 0.79 ± 0.04 b |
| 15:1 n-7 | 0.22 ± 0.01 a | 0.22 ± 0.01 a | 0.23 ± 0.01 a | 0.24 ± 0.01 a |
| 16:0 | 25.48 ± 1.68 a | 23.22 ± 1.01 a | 22.80 ± 2.56 a | 23.36 ± 2.79 a |
| 16:1 n-9 | 0.45 ± 0.02 a | 0.40 ± 0.03 a | 0.39 ± 0.06 a | 0.41 ± 0.07 a |
| 16:1 n-7 | 0.52 ± 0.14 a | 0.70 ± 0.15 a | 0.41 ± 0.14 a | 0.46 ± 0.15 a |
| 16:1 n-3 | 0.32 ± 0.01 a | 0.32 ± 0.01 a | 0.29 ± 0.04 a | 0.30 ± 0.06 a |
| 17:0 | 0.49 ± 0.03 a | 0.45 ± 0.03 a | 0.44 ± 0.05 a | 0.45 ± 0.09 a |
| 17:1 n-8 | 0.22 ± 0.01 a | 0.22 ± 0.01 a | 0.23 ± 0.02 a | 0.28 ± 0.07 a |
| 18:0 | 9.97 ± 0.70 a | 9.54 ± 0.31 a | 8.84 ± 0.80 a | 9.20 ± 0.91 a |
| 18:1 n-9 | 23.03 ± 1.81 a | 21.25 ± 0.66 a | 20.61 ± 2.78 a | 21.42 ± 2.86 a |
| 18:2 n-6 | 2.22 ± 0.22 a | 2.01 ± 0.10 a | 2.05 ± 0.26 a | 2.05 ± 0.25 a |
| 18:3 n-3 | 0.45 ± 0.05 a | 0.35 ± 0.05 ab | 0.32 ± 0.11 ab | 0.29 ± 0.06 b |
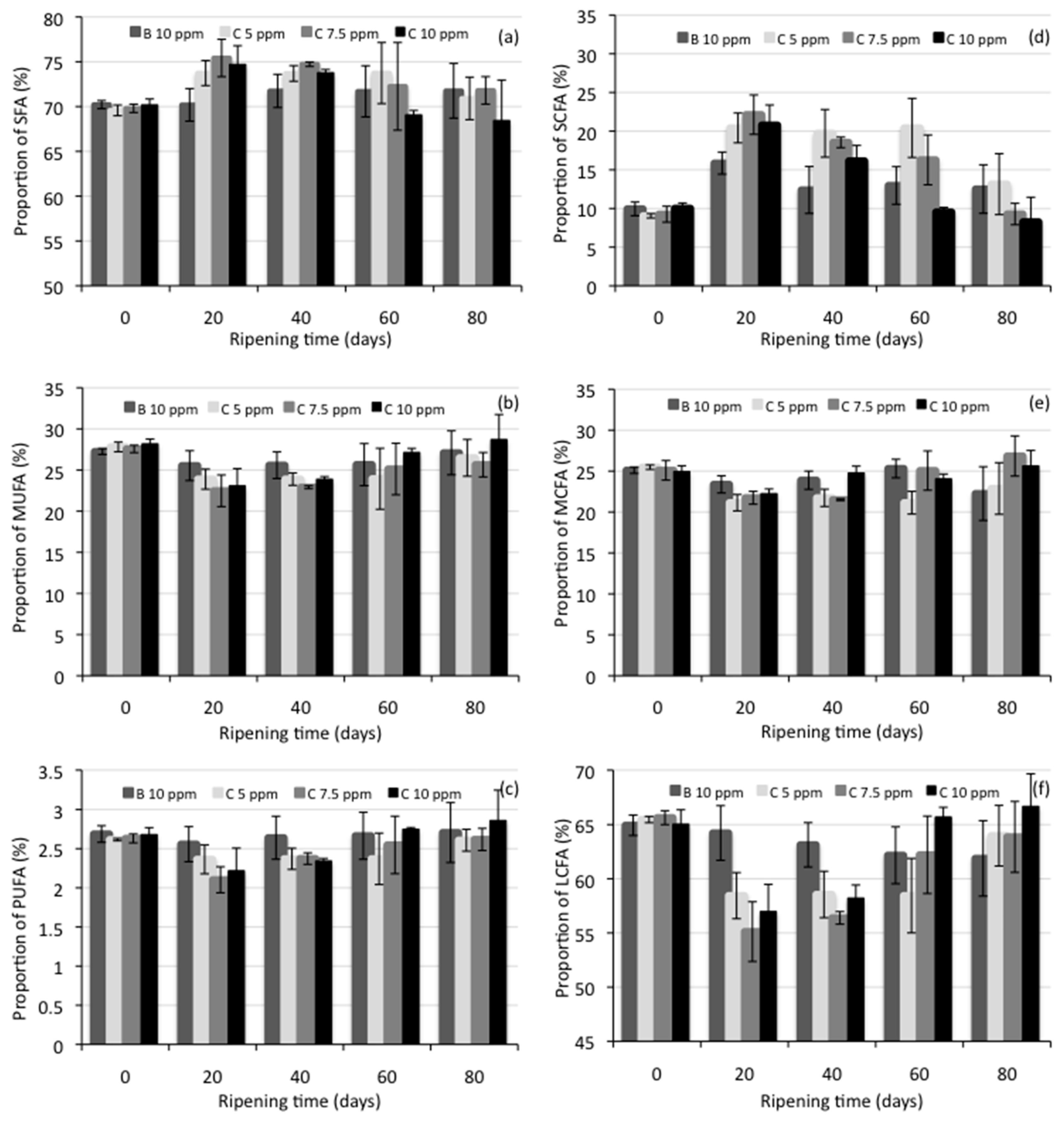
3.5. Lipolysis

3.6. Proteolysis

3.7. Principal Component Analysis

4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Granato, D.; Barba, F.J.; Bursać-Kovačević, D.; Lorenzo, J.M.; Cruz, A.G.; Putnik, P. Functional foods: Product development, technological trends, efficacy testing, and safety. Annu. Rev. Food Sci. 2020, 11, 93–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cencic, A.; Chingwaru, W. The role of functional foods, nutraceuticals, and food supplements in intestinal health. Nutrients 2010, 2, 611–625. [Google Scholar] [CrossRef] [PubMed]
- Câmara, J.S.; Albuquerque, B.R.; Aguiar, J.; Corrêa, R.C.; Gonçalves, J.L.; Granato, D.; Pereira, J.A.; Barros, L.; Ferreira, I.C. Food Bioactive Compounds and Emerging Techniques for Their Extraction: Polyphenols as a Case Study. Foods 2021, 10, 37. [Google Scholar] [CrossRef] [PubMed]
- Samtiya, M.; Aluko, R.E.; Dhewa, T.; Moreno-Rojas, J.M. Potential Health Benefits of Plant Food-Derived Bioactive Components: An Overview. Foods 2021, 10, 839. [Google Scholar] [CrossRef] [PubMed]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxidative Med. Cell Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koch, W. Dietary polyphenols—important non-nutrients in the prevention of chronic noncommunicable diseases. A systematic review. Nutrients 2019, 11, 1039. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arts, I.C.; Hollman, P.C. Polyphenols and disease risk in epidemiologic studies. Am. J. Clin. Nutr. 2005, 81, 317–325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raduly, F.M.; Raditoiu, V.; Raditoiu, A.; Purcar, V. Curcumin: Modern Applications for a Versatile Additive. Coatings 2021, 11, 519. [Google Scholar] [CrossRef]
- Kunnumakkara, A.B.; Harsha, C.; Banik, K.; Vikkurthi, R.; Sailo, B.L.; Bordoloi, D.; Gupta, S.C.; Aggarwal, B.B. Is curcumin bioavailabili-ty a problem in humans: Lessons from clinical trials. Expert Opin. Drug Met. 2019, 15, 705–733. [Google Scholar] [CrossRef]
- Wu, C.; Li, L.; Zhong, Q.; Cai, R.; Wang, P.; Xu, X.; Zhou, G.; Han, M.; Liu, Q.; Hu, T.; et al. Myofibrillar protein–curcumin nanocomplexes prepared at different ionic strengths to improve oxidative stability of marinated chicken meat products. LWT 2019, 99, 69–76. [Google Scholar] [CrossRef]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Aditya, N.P.; Espinosa, Y.G.; Norton, I.T. Encapsulation systems for the delivery of hydrophilic nutraceuticals: Food application. Biotechnol. Adv. 2017, 35, 450–457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ochoa-Flores, A.A.; Hernández-Becerra, J.A.; Cavazos-Garduño, A.; Soto-Rodríguez, I.; Sanchez-Otero, M.G.; Vernon-Carter, E.J.; García, H.S. Enhanced bioavailability of curcumin nanoemulsions stabilized with phosphatidylcholine modified with medium chain fatty acids. Curr. Drug Deliv. 2017, 14, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Salazar-Montoya, J.A.; González-Cuello, R.; Flores-Girón, E.; Ramos-Ramírez, E.G. Effect of free and microencapsulated Lactococcus lactis on composition and rheological properties of Manchego-type cheeses during ripening. Food Res. Int. 2018, 105, 59–64. [Google Scholar] [CrossRef]
- González-Córdova, A.F.; Yescas, C.; Ortiz-Estrada, Á.M.; Hernández-Mendoza, A.; Vallejo-Cordoba, B. Invited review: Artisanal Mexican cheeses. J. Dairy Sci. 2016, 99, 3250–3262. [Google Scholar] [CrossRef] [Green Version]
- González-Viñas, M.A.; Poveda, J.; Ruiz, A.G.; Cabezas, L. Changes in chemical, sensory and rheological characteristics of Manchego cheeses during ripening. J. Sens. Stud. 2001, 16, 361–371. [Google Scholar] [CrossRef]
- Balthazar, C.F.; Pimentel, T.C.; Ferrão, L.L.; Almada, C.N.; Santillo, A.; Albenzio, M.; Mollakhalili, N.; Mortazavian, A.M.; Nascimento, J.S.; Silva, M.C. Sheep milk: Physicochemical characteristics and relevance for functional food development. Compr. Rev. Food Sci. Food Saf. 2017, 16, 247–262. [Google Scholar] [CrossRef] [PubMed]
- Mezo-Solís, J.A.; Moo-Huchin, V.M.; Sánchez-Zarate, A.; Gonzalez-Ronquillo, M.; Estrada-León, R.J.; Ibáñez, R.; Toro-Mujica, P.; Chay-Canul, A.J.; Vargas-Bello-Pérez, E. Physico-chemical, sensory and texture properties of an aged Mexican Manchego-style cheese produced from hair sheep milk. Foods 2020, 9, 1666. [Google Scholar] [CrossRef] [PubMed]
- Ochoa-Flores, A.A.; Hernández-Becerra, J.A.; Velázquez-Martínez, J.R.; Piña-Gutiérrez, J.M.; Hernández-Castellano, L.E.; Toro-Mujica, P.; Chay-Canul, A.J.; Vargas-Bello-Pérez, E. Chemical and fatty acid composition of Manchego type and Panela cheeses manufactured from either hair sheep milk or cow milk. J. Dairy Sci. 2021, 104, 7457–7465. [Google Scholar] [CrossRef]
- Sardiñas-Valdés, M.; Hernández-Becerra, J.A.; García-Galindo, H.S.; Chay-Canul, A.J.; Velázquez-Martínez, J.R.; Ochoa-Flores, A.A. Physicochemical and sensory properties of Manchego-type cheese fortified with nano-emulsified curcumin. Int. Food Res. J. 2021, 28, 326–336. [Google Scholar]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis, 18th ed.; AOAC: Washington DC, USA, 2005. [Google Scholar]
- Rashidinejad, A.; Birch, E.J.; Sun-Waterhouse, D.; Everett, D.W. Effects of catechin on the phenolic content and antioxidant properties of low-fat cheese. Int. J. Food Sci. Technol. 2013, 48, 2448–2455. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Method Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a measure of ‘‘antioxidant power’’: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carvalho, A.P.; Malcata, F.X. Preparation of fatty acid methyl esters for gas-chromatographic analysis of marine lipids: Insight studies. J. Agric. Food Chem. 2005, 53, 5049–5059. [Google Scholar] [CrossRef]
- Deeth, H.C.; Fitz-Gerald, C.H. Lipolysis in dairy products: A review. Aust. J. Dairy Technol. 1976, 31, 53–64. [Google Scholar] [CrossRef]
- Gripon, J.C.; Desmazeaud, M.J.; Le Bars, D.; Bergere, J.L. Etude du rôle des micro-organismes et des enzymes au cours de la maturation des fromages. II.-Influence de la présure commerciale. Le Lait 1975, 55, 502–516. [Google Scholar] [CrossRef] [Green Version]
- Cabezas, L.; Sánchez, I.; Poveda, J.M.; Seseña, S.; Palop, M.L. Comparison of microflora, chemical and sensory characteristics of artisanal Manchego cheeses from two dairies. Food Control. 2007, 18, 11–17. [Google Scholar] [CrossRef]
- Lobato-Calleros, C.; Velázquez-Varela, J.; Sánchez-García, J.; Vernon-Carter, E.J. Dynamic rheology of Mexican Manchego cheese-like products containing canola oil and emulsifier blends. Food Res. Int. 2003, 36, 81–90. [Google Scholar] [CrossRef]
- Giroux, H.J.; De Grandpré, G.; Fustier, P.; Champagne, C.P.; St-Gelais, D.; Lacroix, M.; Britten, M. Production and characterization of Cheddar-type cheese enriched with green tea extract. Dairy Sci. Technol. 2013, 93, 241–254. [Google Scholar] [CrossRef]
- Han, J.; Britten, M.; St-Gelais, D.; Champagne, C.P.; Fustier, P.; Salmieri, S.; Lacroix, M. Polyphenolic compounds as functional ingredients in cheese. Food Chem. 2011, 124, 1589–1594. [Google Scholar] [CrossRef]
- Lešić, T.; Pleadin, J.; Krešić, G.; Vahčić, N.; Markov, K.; Vrdoljak, M.; Frece, J. Chemical and fatty acid composition of cow and sheep milk cheeses in a lamb skin sack. J. Food Compos. Anal. 2016, 46, 70–77. [Google Scholar] [CrossRef]
- Atasoy, A.F.; Türkoğlu, H. Changes of composition and free fatty acid contents of Urfa cheeses (a white-brined Turkish cheese) during ripening: Effects of heat treatments and starter cultures. Food Chem. 2008, 110, 598–604. [Google Scholar] [CrossRef]
- Virto, M.; Chávarri, F.; Bustamante, M.A.; Barron, L.J.R.; Aramburu, M.; Vicente, M.S.; Pérez-Elortondo, F.J.; Albisu, M.; De Renobales, M. Lamb rennet paste in ovine cheese manufacture. Lipolysis and flavour. Int. Dairy J. 2003, 13, 391–399. [Google Scholar] [CrossRef]
- Amalraj, A.; Pius, A.; Gopi, S.; Gopi, S. Biological activities of curcuminoids, other biomolecules from turmeric and their derivatives—A review. J. Tradit. Complement. Med. 2017, 7, 205–233. [Google Scholar] [CrossRef] [Green Version]
- Rashidinejad, A.; Birch, E.J.; Everett, D.W. Effects of (+)-catechin on the composition, phenolic content and antioxidant activity of full-fat cheese during ripening and recovery of (+)-catechin after simulated in vitro digestion. Antioxidants 2016, 5, 29. [Google Scholar] [CrossRef] [Green Version]
- Batool, M.; Nadeem, M.; Imran, M.; Khan, I.T.; Bhatti, J.A.; Ayaz, M. Lipolysis and antioxidant properties of cow and buffalo cheddar cheese in accelerated ripening. Lipids Health Dis. 2018, 17, 228. [Google Scholar] [CrossRef]
- Khan, I.T.; Nadeem, M.; Imran, M.; Ajmal, M.; Ali, S. Antioxidant activity, fatty acids characterization and oxidative stability of Gouda cheese fortified with mango (Mangifera indica L.) kernel fat. J. Food Sci. Technol. 2018, 55, 992–1002. [Google Scholar] [CrossRef]
- Perna, A.; Intaglietta, I.; Simonetti, A.; Gambacorta, E. Short communication: Effect of genetic type on antioxidant activity of Caciocavallo cheese during ripening. J. Dairy Sci. 2015, 98, 3690–3694. [Google Scholar] [CrossRef] [Green Version]
- González-Martín, I.; Hernández-Hierro, J.M.; Vivar-Quintana, A.; Revilla, I.; González-Pérez, C. The application of near infrared spectroscopy technology and a remote reflectance fibre-optic probe for the determination of peptides in cheeses (cow’s, ewe’s and goat’s) with different ripening times. Food Chem. 2009, 114, 1564–1569. [Google Scholar] [CrossRef]
- Gupta, A.; Mann, B.; Kumar, R.; Sangwan, R.B. Antioxidant activity of Cheddar cheeses at different stages of ripening. Int. J. Dairy Technol. 2009, 62, 339–347. [Google Scholar] [CrossRef]
- Carvalho, G.P.; Santos, R.; Fino, A.; Ferreira, P.; Rodrigues, F.M.; Dias, J. Evolution during three ripening stages of Évora cheese. Foods 2020, 9, 1140. [Google Scholar] [CrossRef]
- Tarakci, Z.; Temiz, H.; Aykut, U.; Turhan, S. Influence of wild garlic on color, free fatty acids, and chemical and sensory properties of Herby Pickled cheese. Int. J. Food Prop. 2011, 14, 287–299. [Google Scholar] [CrossRef] [Green Version]
- Pinho, O.; Mendes, E.; Alves, M.M.; Ferreira, I.M.P.L.V.O. Chemical, physical, and sensorial characteristics of “Terrincho” ewe cheese: Changes during ripening and intravarietal comparison. J. Dairy Sci. 2004, 87, 249–257. [Google Scholar] [CrossRef] [Green Version]
- El-Nimr, A.A.; Eissa, H.A.; El-Abd, M.M.; Mehriz, A.A.; Abbas, H.M.; Bayoumi, H.M. Water Activity, color characteristics and sensory properties of egyptian Gouda cheese during ripening. J. Am. Sci. 2010, 6, 447–453. [Google Scholar] [CrossRef]
- Lee, J.S.; Choi, H.Y.; Kim, K.H.; Chun, S.S.; Baen, I. Physicochemical and sensory properties of Appenzeller cheese supplemented with shrimp powder. Korean J. Food Sci. Anim. Resour. 2015, 35, 232–239. [Google Scholar] [CrossRef] [Green Version]
- Ibáñez, R.A.; Waldron, D.S.; McSweeney, P.L.H. Effect of fat content and temperature on the translucency of Cheddar cheese. Int. Dairy J. 2016, 54, 33–42. [Google Scholar] [CrossRef]
- Juric, M.; Bertelsen, G.; Mortensen, G.; Petersen, M.A. Light-induced colour and aroma changes in sliced, modified atmosphere packaged semi-hard cheeses. Int. Dairy J. 2003, 13, 239–249. [Google Scholar] [CrossRef]
- Kristensen, D.; Orlien, V.; Mortensen, G.; Brockhoff, P.; Skibsted, L.H. Light-induced oxidation in sliced Havarti cheese packaged in modified atmosphere. Int. Dairy J. 2000, 10, 95–103. [Google Scholar] [CrossRef]
- Pillonel, L.; Badertscher, R.; Bütikofer, U.; Casey, M.; Dalla-Torre, M.; Lavanchy, P.; Meyer, J.; Tabacchi, R.; Bosset, J.O. Analytical methods for the determination of the geographic origin of Emmentaler cheese. Main framework of the project; chemical, biochemical, microbiological, colour and sensory analyses. Eur. Food Res. Technol. 2002, 215, 260–267. [Google Scholar] [CrossRef] [Green Version]
- Rohm, H.; Jaros, D. Colour of hard cheese. 2. Factors of influence and relation to compositional parameters. Z. Für Lebensm. Und Forsch. 1997, 204, 259–264. [Google Scholar] [CrossRef]
- Collins, Y.F.; McSweeney, P.L.H.; Wilkinsonc, M.G. Lipolysis and free fatty acid catabolism in cheese: A review of current knowledge. Int. Dairy J. 2003, 13, 841–866. [Google Scholar] [CrossRef]
- Prandini, A.; Sigolo, S.; Piva, G. A comparative study of fatty acid composition and CLA concentration in commercial cheeses. J. Food Compos. Anal. 2011, 24, 55–61. [Google Scholar] [CrossRef]
- Kurćubić, V.S.; Vujic, J.M.; Iliĉić, M.D.; Vranić, D.; Vesković-Moracanin, S.M.; Mašković, P.Z. Effect of plant extracts of Kitaibelia vitifolia on antioxidant activity, chemical characteristics, microbiological status and sensory properties of Pirotski Kachkaval cheese. Hem. Ind. 2015, 69, 85–93. [Google Scholar] [CrossRef]
- Khalifa, S.A.; Wahdan, K.M. Improving the quality characteristics of white soft cheese using cranberry (Vaccinium macrocarpon) fruit extract. Int. Food Res. J. 2015, 22, 2203–2211. [Google Scholar]
- Del Olmo, A.; López-Pérez, O.; Picon, A.; Gaya, P.; Nuñez, M. Cheese supplementation with five species of edible seaweeds: Effect on proteolysis, lipolysis and volatile compounds. Int. Dairy J. 2019, 90, 104–113. [Google Scholar] [CrossRef]
- Poveda, J.M.; Pérez-Coello, M.S.; Cabezas, L. Seasonal variations in the free fatty acid composition of Manchego cheese and changes during ripening. Eur. Food Res. Technol. 2000, 210, 314–317. [Google Scholar] [CrossRef]
- Partidario, A.M.; Barbosa, M.; Boas, L.V. Free fatty acids, triglycerides and volatile compounds in Serra da Estrela cheese—changes throughout ripening. Int. Dairy J. 1998, 8, 873–881. [Google Scholar] [CrossRef]
- Poveda, J.M.; Cabezas, L.; García, A. Changes in physicochemical properties and proteolysis in Manchego cheese preserved in olive oil. Milchwissenschaft 1999, 54, 252–255. [Google Scholar]
- Sieber, R.; Bütikofer, U.; Egger, C.; Portmann, R.; Walther, B.; Wechsler, D. ACE-inhibitory activity and ACE-inhibiting peptides in different cheese varieties. Dairy Sci. Technol. 2010, 90, 47–73. [Google Scholar] [CrossRef]
- Pinho, O.; Ferreira, I.M.; Mendes, E.; Oliveira, B.M.; Ferreira, M. Effect of temperature on evolution of free amino acid and biogenic amine contents during storage of Azeitão cheese. Food Chem. 2001, 75, 287–291. [Google Scholar] [CrossRef]
- Sousa, M.J.; Ardö, Y.; McSweeney, P.L.H. Advances in the study of proteolysis in cheese during ripening. Int. Dairy J. 2001, 11, 327–334. [Google Scholar] [CrossRef]
- Vivar-Quintana, A.M.; Blanco-López, M.A.; Revilla, I.; González-Martín, I.; Hernández-Hierro, J.M.; González-Pérez, C. Seasonal evolution of hydrophilic and hydrophobic peptide contents in cheeses made from ewe’s goat’s or cow’s milk. Czech. J. Food Sci. 2009, 27, S106–S108. [Google Scholar] [CrossRef] [Green Version]
- Bergamini, C.V.; Hynes, E.R.; Zalazar, C.A. Influence of probiotic bacteria on the proteolysis profile of a semi-hard cheese. Int. Dairy J. 2006, 16, 856–866. [Google Scholar] [CrossRef]
- Mallatou, H.; Pappa, E.C.; Boumba, V.A. Proteolysis in Teleme cheese made from ewes’, goats’ or a mixture of ewes’ and goats’ milk. Int. Dairy J. 2004, 14, 977–987. [Google Scholar] [CrossRef]
- Ardö, Y. Evaluating Proteolysis by Analysing the N Content of Cheese Fractions; Bulletin IDF 337; International Dairy Federation: Brussels, Belgium, 1999; pp. 4–9. [Google Scholar]
- Gardiner, G.; Ross, R.P.; Collins, J.K.; Fitzgerald, G.; Stanton, C. Development of a probiotic Cheddar cheese containing human-derived Lactobacillus paracasei strains. Appl. Environ. Microb. 1998, 64, 2192–2199. [Google Scholar] [CrossRef] [Green Version]
| Cheese Samples | Ripening Time (Days) | Moisture (%) | Fat (%) | Total Protein (%) | Ash (%) | aw |
|---|---|---|---|---|---|---|
| B 10 ppm | 0 | 46.66 ± 0.77 a | 27.20 ± 0.43 b | 22.09 ± 0.26 a | 3.40 ± 0.03 a | 0.973 ± 0.002 |
| 20 | 46.40 ± 3.31 ab1 | 26.72 ± 0.80 b | 22.32 ± 0.09 ab12 | 3.53 ± 0.02 bc | 0.972 ± 0.001 | |
| 40 | 45.41 ± 1.56 ab | 23.41 ± 1.45 a | 22.24 ± 0.15 ab | 3.50 ± 0.02 b1 | 0.972 ± 0.001 | |
| 60 | 44.89 ± 2.18 ab | 25.25 ± 0.79 ab1 | 22.47 ± 0.43 ab | 3.60 ± 0.03 cd1 | 0.970 ± 0.002 | |
| 80 | 43.48 ± 1.60 b | 26.82 ± 1.22 b1 | 22.91 ± 0.41 b | 3.64 ± 0.03 d1 | 0.970 ± 0.001 | |
| C 5 ppm | 0 | 46.58 ± 2.36 a | 27.37 ± 0.55 bc | 22.20 ± 0.18 | 3.38 ± 0.11 a | 0.975 ± 0.001 a |
| 20 | 46.61 ± 0.88 a1 | 24.68 ± 1.25 ab | 22.41 ± 0.38 12 | 3.58 ± 0.03 b | 0.973 ± 0.003 a | |
| 40 | 44.92 ± 2.18 ab | 24.29 ± 2.23 a | 22.54 ± 0.23 | 3.57 ± 0.05 b1 | 0.973 ± 0.001 a | |
| 60 | 42.73 ± 1.85 b | 27.49 ± 1.47 bc12 | 22.89 ± 0.41 | 3.62 ± 0.06 b1 | 0.972 ± 0.003 ab | |
| 80 | 42.57 ± 2.69 b | 28.91 ± 2.47 c12 | 22.73 ± 0.29 | 3.71 ± 0.01 c1 | 0.969 ± 0.002 b | |
| C 7.5 ppm | 0 | 45.70 ± 1.18 a | 26.89 ± 0.54 b | 22.17 ± 0.19 a | 3.45 ± 0.04 a | 0.974 ± 0.001 a |
| 20 | 45.13 ± 0.94 a12 | 26.01 ± 2.12 b | 21.89 ± 0.34 a1 | 3.58 ± 0.02 b | 0.973 ± 0.003 ab | |
| 40 | 43.68 ± 2.80 ab | 25.09 ± 2.65 a | 22.77 ± 0.53 ab | 3.61 ± 0.07 b12 | 0.973 ± 0.004 ab | |
| 60 | 42.69 ± 2.50 ab | 28.57 ± 0.61 c2 | 23.34 ± 0.12 b | 3.60 ± 0.02 b1 | 0.970 ± 0.001 b | |
| 80 | 41.13 ± 1.29 b | 27.63 ± 1.17 b12 | 23.44 ± 0.37 b | 3.66 ± 0.03 b1 | 0.971 ± 0.001 ab | |
| C 10 ppm | 0 | 45.34 ± 1.95 a | 28.12 ± 1.22 b | 22.40 ± 0.61 a | 3.47 ± 0.09 a | 0.973 ± 0.001 a |
| 20 | 42.42 ± 0.11 b2 | 27.58 ± 2.31 b | 23.13 ± 0.37 ab2 | 3.60 ± 0.03 b | 0.972 ± 0.000 a | |
| 40 | 42.44 ± 1.17 ab | 24.41 ± 0.72 a | 22.79 ± 0.63 ab | 3.72 ± 0.06 c2 | 0.972 ± 0.003 ab | |
| 60 | 42.51 ± 0.44 b | 29.46 ± 2.00 b2 | 23.42 ± 1.40 b | 3.71 ± 0.06 c2 | 0.971 ± 0.002 ab | |
| 80 | 41.72 ± 1.63 b | 30.08 ± 1.31 b2 | 23.55 ± 0.62 b | 3.85 ± 0.04 d2 | 0.968 ± 0.003 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sardiñas-Valdés, M.; García-Galindo, H.S.; Chay-Canul, A.J.; Velázquez-Martínez, J.R.; Hernández-Becerra, J.A.; Ochoa-Flores, A.A. Ripening Changes of the Chemical Composition, Proteolysis, and Lipolysis of a Hair Sheep Milk Mexican Manchego-Style Cheese: Effect of Nano-Emulsified Curcumin. Foods 2021, 10, 1579. https://doi.org/10.3390/foods10071579
Sardiñas-Valdés M, García-Galindo HS, Chay-Canul AJ, Velázquez-Martínez JR, Hernández-Becerra JA, Ochoa-Flores AA. Ripening Changes of the Chemical Composition, Proteolysis, and Lipolysis of a Hair Sheep Milk Mexican Manchego-Style Cheese: Effect of Nano-Emulsified Curcumin. Foods. 2021; 10(7):1579. https://doi.org/10.3390/foods10071579
Chicago/Turabian StyleSardiñas-Valdés, Mariam, Hugo Sergio García-Galindo, Alfonso Juventino Chay-Canul, José Rodolfo Velázquez-Martínez, Josafat Alberto Hernández-Becerra, and Angélica Alejandra Ochoa-Flores. 2021. "Ripening Changes of the Chemical Composition, Proteolysis, and Lipolysis of a Hair Sheep Milk Mexican Manchego-Style Cheese: Effect of Nano-Emulsified Curcumin" Foods 10, no. 7: 1579. https://doi.org/10.3390/foods10071579






