Diet and Genetics Influence Beef Cattle Performance and Meat Quality Characteristics
Abstract
:1. Introduction
2. Tropical Northern Australian Pastures and Beef Production
2.1. Beef Cattle Responses to Under-Nutrition
2.1.1. Decrease in Liveweight
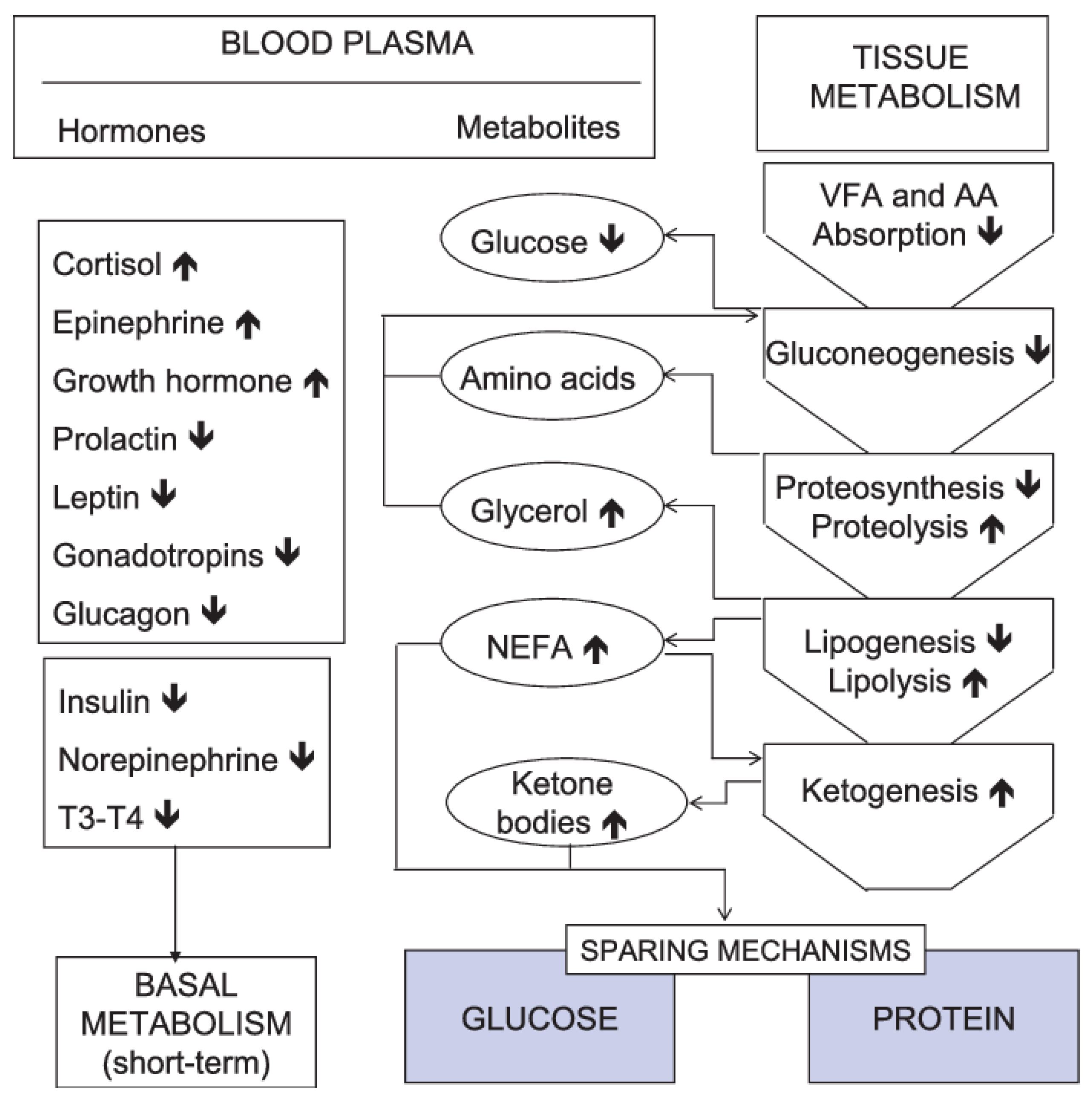
2.1.2. Metabolic and Body Composition Changes
2.2. Nutritional Supplementation to Improve Beef Cattle Performance on Low Quality Pastures
2.2.1. Feed Supplements During Grazing
2.2.2. Augmenting Pastures with Legumes
2.2.3. Use of Legumes in Northern Australia
2.3. Feedlot Finishing of Tropical Pasture-Backgrounded Cattle
3. Meat characteristics
3.1. Effect of Intramuscular Fat on Beef-Eating Characteristics
3.1.1. Tenderness
3.1.2. Flavour
3.1.3. Juiciness
3.2. Factors Influencing Beef Intramuscular Fat Content and Fatty Acid Composition
3.2.1. Pasture Versus Concentrate Diets
3.2.2. Oil Supplements
3.2.3. Micronutrients
3.2.4. Cattle Breed
3.3. Genes that Influence Carcass Fat Content and Fatty Acid Profiles
3.3.1. Stearoyl-CoA Desaturase (SCD)
3.3.2. Fatty Acid Synthase (FASN)
3.3.3. Fatty Acid Binding Protein 4 (FABP4)
4. Conclusions and Future Research
- However, only limited peer-reviewed published literature is available on the effect of Desmanthus on beef cattle growth and performance. These studies were either conducted indoors or in small sized paddocks (except one in 250 ha paddock) which may not be replicated in normal commercial farm settings. Hence, there is need to conduct more studies under commercial farm settings to determine the suitability of grass–Desmanthus pastures in northern Australian beef cattle production system.
- Tannin-containing pastures at 20–40 g/kg DM are reported to increase polyunsaturated fatty acids in meat by reducing rumen biohydrogenation of unsaturated fatty acids. There is need to study the effect of Desmanthus, a tannin-containing legume, on performance and meat characteristics of grazing cattle.
- Several genes such as SCD, FASN and FABP4 are reported to influence carcass fat traits in Korean and Japanese cattle as well as Australian temperate breeds such as Angus and Limousin. There is need to investigate the effect of these genes in northern Australian composite breeds.
- In addition, studies are required to determine finishing performance and carcass traits of northern Australian beef composite breeds backgrounded on newly introduced legume pastures, such as Desmanthus, to enable industry players to exploit them for greater economic gains.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- OECD Meat Consumption (Indicator). Available online: https://data.oecd.org/agroutput/meat-consumption.htm (accessed on 15 May 2019).
- OECD-FAO Agricultural Outlook 2018–2027. Available online: https://www.oecd-ilibrary.org/agriculture-and-food/oecd-fao-agricultural-outlook-2018-2027_agr_outlook-2018-en (accessed on 15 May 2019).
- Troy, D.J.; Tiwari, B.K.; Joo, S. Health implications of beef intramuscular fat consumption. Korean J. Food Sci. Anim. Resour. 2016, 36, 577–582. [Google Scholar] [CrossRef] [Green Version]
- Asp, M.L.; Richardson, J.R.; Collene, A.L.; Droll, K.R.; Belury, M.A. Dietary protein and beef consumption predict for markers of muscle mass and nutrition status in older adults. J. Nutr. Health Aging 2012, 16, 784–790. [Google Scholar] [CrossRef]
- Williams, P. Nutritional composition of red meat. J. Nutr. Diet. 2007, 64, S113–S119. [Google Scholar] [CrossRef] [Green Version]
- FAO Overview of Global Meat Market Developments in 2018. Available online: http://www.fao.org/3/ca3880en/ca3880en.pdf (accessed on 10 June 2019).
- Australian Bureau of Statistics Value of Agricultural Commodities Produced in Australia, 2017–2018. Available online: https://www.abs.gov.au/ausstats/[email protected]/0/58529ACD49B5ECE0CA2577A000154456?Opendocument (accessed on 12 August 2019).
- Meat and Livestock Australia State of the Industry Report 2018: The Australian Red Meat and Livestock Industry. Available online: http://rmac.com.au/wp-content/uploads/2018/09/SOTI18.pdf (accessed on 29 April 2019).
- Department of Agriculture and Fisheries The Queensland Beef Supply Chain. Available online: https://publications.qld.gov.au/dataset/23f4f979.../2-qld-beef-supply-chain.pdf%0A%0A (accessed on 29 April 2019).
- Greenwood, P.L.; Gardner, G.E.; Ferguson, D.M. Current situation and future prospects for the Australian beef industry—A review. Asian Australas. J. Anim. Sci. 2018, 31, 992–1006. [Google Scholar] [CrossRef]
- Johnston, M.G.; Jeyaruban, D.J. Estimated additive and non-additive breed effects and genetic parameters for ultrasound scanned traits of a multi-breed beef population in tropical Australia. In Proceedings of the 10th World Congress of Genetics Applied to Livestock Production, Vancouver, BC, Canada, 17–22 August 2014. [Google Scholar]
- Bell, A.W.; Charmley, E.; Hunter, R.A.; Archer, J.A. The Australasian beef industries—Challenges and opportunities in the 21st century. Anim. Front. 2011, 1, 10–19. [Google Scholar] [CrossRef] [Green Version]
- Wolcott, M.L.; Johnston, D.J.; Barwick, S.A.; Iker, C.L.; Thompson, J.M.; Burrow, H.M. Genetics of meat quality and carcass traits and the impact of tenderstretching in two tropical beef genotypes. Anim. Prod. Sci. 2009, 49, 383–398. [Google Scholar] [CrossRef]
- Mannen, H. Identification and utilization of genes associated with beef qualities. Anim. Sci. J. 2011, 82, 1–7. [Google Scholar] [CrossRef]
- Mapiye, C.; Vahmani, P.; Mlambo, V.; Muchenje, V.; Dzama, K.; Hoffman, L.C.; Dugan, M.E.R. The trans-octadecenoic fatty acid profile of beef: Implications for global food and nutrition security. Food Res. Int. 2015, 76, 992–1000. [Google Scholar] [CrossRef]
- Zietemann, V.; KrÖger, J.; Enzenbach, C.; Jansen, E.; Fritsche, A.; Weikert, C.; Boeing, H.; Schulze, M.B. Genetic variation of the FADS1 FADS2 gene cluster and n-6 PUFA composition in erythrocyte membranes. Br. J. Nutr. 2010, 104, 1748–1759. [Google Scholar] [CrossRef] [Green Version]
- Ordovas, J.M. Identification of a functional polymorphism at the adipose fatty acid binding protein gene (FABP4) and demonstration of its association with cardiovascular disease: A path to follow. Nutr. Rev. 2007, 65, 130–134. [Google Scholar] [CrossRef]
- Michal, J.J.; Zhang, Z.W.; Gaskins, C.T.; Jiang, Z. The bovine fatty acid binding protein 4 gene is significantly associated with marbling and subcutaneous fat depth in Wagyu x Limousin F2 crosses. Anim. Genet. 2006, 37, 400–402. [Google Scholar] [CrossRef]
- Barendse, W.; Bunch, R.J.; Thomas, M.B.; Harrison, B.E. A splice site single nucleotide polymorphism of the fatty acid binding protein 4 gene appears to be associated with intramuscular fat deposition in longissimus muscle in Australian cattle. Anim. Genet. 2009, 40, 770–773. [Google Scholar] [CrossRef]
- Cho, S.A.; Park, T.S.; Yoon, D.H.; Cheong, H.S.; Namgoong, S.; Park, B.L.; Lee, H.W.; Han, C.S.; Kim, E.M.; Cheong, I.C.; et al. Identification of genetic polymorphisms in FABP3 and FABP4 and putative association with back fat thickness in Korean native cattle. BMB Rep. 2008, 41, 29–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.H.; van der Werf, J.H.J.; Lee, S.H.; Park, E.W.; Oh, S.J.; Gibson, J.P.; Thompson, J.M. Genetic polymorphisms of the bovine fatty acid binding protein 4 gene are significantly associated with marbling and carcass weight in Hanwoo (Korean Cattle ). Anim. Genet. 2010, 41, 442–444. [Google Scholar] [PubMed]
- Hoashi, S.; Hinenoya, T.; Tanaka, A.; Ohsaki, H.; Sasazaki, S.; Taniguchi, M.; Oyama, K.; Mukai, F.; Mannen, H. Association between fatty acid compositions and genotypes of FABP4 and LXR-alpha in Japanese Black cattle. BMC Genet. 2008, 9, 84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bezerra, R.L.; Ferreira, R.R.; Edvan, L.R.; Neto, G.S.; da Silva, L.A.; de Araújo, J.M. Protein supplementation is vital for beef cattle fed with tropical pasture. In Grasses as Food and Feed; Tadele, Z., Ed.; IntechOpen: London, UK, 2018; pp. 97–107. [Google Scholar]
- Hattersley, P.W. The distribution of C3 and C4 grasses in Australia in relation to climate. Oecologia 1983, 57, 113–128. [Google Scholar] [CrossRef]
- Hunt, L.P.; Mcivor, J.G.; Grice, A.C.; Bray, S.G. Principles and guidelines for managing cattle grazing in the grazing lands of northern Australia: Stocking rates, pasture resting, prescribed fire, paddock size and water points—A review. Rangel. J. 2014, 36, 105–119. [Google Scholar] [CrossRef]
- Tothill, J.; Gillies, C. The Pasture Lands of Northern Australia: Their Condition, Productivity and Sustainability; Occasional; Tropical Grasslands Society of Australia: St Lucia, QLD, Australia, 1992; ISBN 0959094849. [Google Scholar]
- Cameron, D.F.; Edye, L.A.; Chakraborty, S.; Manners, J.M.; Liu, C.J.; Date, R.A.; Boland, R.M. An integrated program to improve anthracnose resistance in Stylosanthes—A review. In Proceedings of the Proc 8th Australian Agronomy Conference, Toowoomba, Australia, 30 January–2 February 1996; pp. 112–115. [Google Scholar]
- Pengelly, B.C.; Conway, M.J. Pastures on cropping soils: Which tropical pasture legume to use? Trop. Grassl. 2000, 34, 162–168. [Google Scholar]
- Robertson, F.A.; Myers, R.J.K.; Saffigna, P.G. Nitrogen cycling in brigalow clay soils under pasture and cropping. Soil Res. 1997, 35, 1323. [Google Scholar] [CrossRef]
- Soil Science Australia State Soils. Available online: https://www.soilscienceaustralia.org.au/about/about-soil/state-soil (accessed on 15 May 2019).
- Gleeson, T.; Martin, P.; Misfud, C. Northern Australian Beef Industry: Assessment of Risks and Opportunities. Available online: https://docplayer.net/25202349-Northern-australian-beef-industry-assessment-of-risks-and-opportunities.html (accessed on 29 April 2019).
- Ivory, D.; Whiteman, P. Effect of Temperature on Growth of Five Subtropical Grasses. II. Effect of Low Night Temperature. Funct. Plant Biol. 2006, 5, 149. [Google Scholar] [CrossRef]
- McMeniman, N.P.; Beale, I.F.; Murphy, G.M. Nutritional evaluation of south-west Queensland pastures. I. The botanical and nutrient content of diets selected by sheep grazing on Mitchell grass and mulga/grassland associations. Aust. J. Agric. Res. 1986, 37, 289–302. [Google Scholar] [CrossRef]
- Wilson, J.R.; Mannetje, L. Senescence, digestibility and carbohydrate content of Buffel grass and Green Panic leaves in swards. Aust. J. Agric. Res. 1979, 29, 503–516. [Google Scholar] [CrossRef]
- Charmley, E.; Stephens, M.L.; Kennedy, P.M. Predicting livestock productivity and methane emissions in northern Australia: Development of a bio-economic modelling approach. Aust. J. Exp. Agric. 2008, 48, 109–113. [Google Scholar] [CrossRef]
- McCown, R.L. The climatic potential for beef cattle production in tropical Australia: Part I-Simulating the annual cycle of liveweight change. Agric. Syst. 1981, 6, 303–317. [Google Scholar] [CrossRef]
- Brandão, R.K.C.; de Carvalho, G.; Silva, R.; Dias, D.; Mendes, F.; Lins, T.; Pereira, M.; Guimarães, J.; Tosto, M.; Rufino, L.; et al. Correlation between production performance and feeding behavior of steers on pasture during the rainy-dry transition period. Trop. Anim. Health Prod. 2018, 50, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Leng, R.A. Factors affecting the utilization of ‘poor-quality’ forages by ruminants particularly under tropical conditions. Nutr. Res. Rev. 1990, 3, 277–303. [Google Scholar] [CrossRef]
- Kanani, J.; Lukefahr, S.D.; Stanko, R.L. Evaluation of tropical forage legumes (Medicago sativa, Dolichos lablab, Leucaena leucocephala and Desmanthus bicornutus) for growing goats. Small Rumin. Res. 2006, 65, 1–7. [Google Scholar] [CrossRef]
- Johnson, D.E.; Johnson, K.A. Methane emissions from cattle. J. Anim. Sci. 1995, 73, 2483–2492. [Google Scholar] [CrossRef]
- Buxton, D.R.; Mertens, D.R.; Moore, K.J. Forage quality for ruminants: Plant and animal considerations. Prof. Anim. Sci. 1995, 11, 121–131. [Google Scholar] [CrossRef]
- Bowman, J.G.P.; Sowell, B.F.; Paterson, J.A. Liquid supplementation for ruminants fed low-quality forage diets: A review. Anim. Feed Sci. Technol. 1995, 55, 105–138. [Google Scholar] [CrossRef]
- Chilliard, Y.; Bocquier, F.; Doreau, M. Digestive and metabolic adaptations of ruminants to undernutrition, and consequences on reproduction. Reprod. Nutr. Dev. 1998, 38, 131–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baldwin, R.L. Digestion and metabolism of ruminants. Bioscience 1984, 34, 244–249. [Google Scholar] [CrossRef]
- Burrin, D.G.; Ferrell, C.L.; Britton, R.A.; Bauer, M. Level of nutrition and visceral organ size and metabolic activity in sheep. Br. J. Nutr. 1990, 64, 439–448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ortigues, I.; Doreau, M. Responses of the splanchnic tissues of ruminants to changes in intake: Absorption of digestion end products, tissue mass, metabolic activity and implications to whole animal energy metabolism. Ann. Zootech. 1995, 44, 321–346. [Google Scholar] [CrossRef] [Green Version]
- Yang, A.; Larsen, T.W.; Smith, S.B.; Tume, R.K. Δ9 Desaturase activity in bovine subcutaneous adipose tissue of different fatty acid composition. Lipids 1999, 34, 971–978. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.C.; Cross, H.R.; Carpenter, Z.L.; Murphy, C.E.; Savell, J.W.; Abraham, H.C.; Davis, G.W. Relationship of USDA maturity groups to palatability of cooked beef. J. Food Sci. 1982, 47, 1100–1107. [Google Scholar] [CrossRef]
- Preston, T.R.; Leng, R.A. Matching Ruminant Production Systems with Available Resources in the Tropics and Sub-Tropics; Penambul Books: Armidale, Australia, 1987. [Google Scholar]
- Panjaitan, T.; Quigley, S.P.; McLennan, S.R.; Poppi, D.P. Effect of the concentration of Spirulina (Spirulina platensis) algae in the drinking water on water intake by cattle and the proportion of algae bypassing the rumen. Anim. Prod. Sci. 2010, 50, 405–409. [Google Scholar] [CrossRef]
- Valente, E.E.L.; Paulino, M.F.; Detmann, E.; Valadares Filho, S.D.C.; Cardenas, J.E.G.; Dias, I.F.T. Requirement of energy and protein of beef cattle on tropical pasture. Acta Sci. Anim. Sci. 2013, 35, 417–424. [Google Scholar] [CrossRef] [Green Version]
- Batista, E.D.; Detmann, E.; Titgemeyer, E.C.; Valadares Filho, S.C.; Valadares, R.F.D.; Prates, L.L.; Rennó, L.N.; Paulino, M.F. Effects of varying ruminally undegradable protein supplementation on forage digestion, nitrogen metabolism, and urea kinetics in Nellore cattle fed low-quality tropical forage. J. Anim. Sci. 2016, 94, 201–216. [Google Scholar] [CrossRef] [Green Version]
- Lazzarini, I.; Detmann, E.; Sampaio, C.B.; Paulino, M.F.; Valadares Filho, S.D.; Souza, M.A.; Oliveira, F.A. Intake and digestibility in cattle fed low-quality tropical forage and supplemented with nitrogenous compounds. Rev. Bras. Zootec. 2009, 38, 2021–2030. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, R.M.; de Almeida, C.M.; Carvalho, B.C.; Neto, J.A.; Mota, V.A.; de Resende, F.D.; Siqueira, G.R. Effect of supplementation of beef cattle with different protein levels and degradation rates during transition from the dry to rainy season. Trop. Anim. Health Prod. 2016, 48, 95–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neves, D.S.; Silva, R.R.; da Silva, F.F.; Santos, L.V.; Abreu Filho, G.; de Souza, S.O.; Santos, M.D.; Rocha, W.J.; da Silva, A.P.; de Melo Lisboa, M.; et al. Increasing levels of supplementation for crossbred steers on pasture during the dry period of the year. Trop. Anim. Health Prod. 2018, 50, 1411–1416. [Google Scholar] [CrossRef] [PubMed]
- Poppi, D.P.; McLennan, S.R. Nutritional research to meet future challenges. Anim. Prod. Sci. 2010, 50, 329. [Google Scholar] [CrossRef]
- Department of Agriculture and Fisheries Supplementation Feeding Considerations. Available online: https://www.daf.qld.gov.au/business-priorities/agriculture/disaster-recovery/drought/managing/supplementation-feeding-considerations (accessed on 24 August 2019).
- Krebs, G.; Leng, R.A.; Nolan, J.V. Effect on bacterial kinetics in the rumen of eliminating rumen protozoa or supplementing with soyabean meal or urea in sheep on a low protein fibrous feed. In The Roles of Protozoa and Fungi in Ruminant Digestion; Nolan, J.V., Leng, R.A., Demeyer, D.I., Eds.; Penambul Books: Armidale, Australia, 1989; pp. 199–210. [Google Scholar]
- Hill, J.O.; Coates, D.B.; Whitbread, A.M.; Clem, R.L.; Robertson, M.J.; Pengelly, B.C. Seasonal changes in pasture quality and diet selection and their relationship with liveweight gain of steers grazing tropical grass and grass legume pastures in northern Australia. Anim. Prod. Sci. 2009, 49, 983–993. [Google Scholar] [CrossRef]
- Murphy, A.M.; Colucci, P.E. A tropical forage solution to poor quality ruminant diets: A review of Lablab purpureus. Livest. Res. Rural Dev. 1999, 11, 1999. [Google Scholar]
- Osuji, P.O.; Sibanda, S.; Nsahlai, I.V. Supplementation of maize stover for Ethiopian Menz sheep: Effects of cottonseed, noug (Guizotia abyssinica) or sunflower cake with or without maize on the intake, growth, apparent digestibility, nitrogen balance and excretion of purine derivatives. Anim. Sci. 1993, 57, 429–436. [Google Scholar] [CrossRef]
- Gillard, P. Improvement of native pasture with Townsville stylo in the dry tropics of sub-coastal northern Queensland [stylosanthes]. Aust. J. Exp. Agric. 1979, 19, 325–336. [Google Scholar] [CrossRef]
- Rochon, J.J.; Doyle, C.J.; Greef, J.M.; Hopkins, A.; Molle, G.; Sitzia, M.; Scholefield, D.; Smith, C.J. Grazing legumes in Europe: A review of their status, management, benefits, research needs and future prospects. Grass Forage Sci. 2004, 59, 197–214. [Google Scholar] [CrossRef]
- Radrizzani, A.; Nasca, J.A. The effect of Leucaena leucocephala on beef production and its toxicity in the Chaco Region of Argentina. Trop. Grassl. Forrajes Trop. 2014, 2, 127–129. [Google Scholar] [CrossRef] [Green Version]
- Jackson, F.S.; Barry, T.N.; Lascano, C.; Palmer, B. The extractable and bound condensed tannin content of leaves from tropical tree, shrub and forage legumes. J. Sci. Food Agric. 1996, 71, 103–110. [Google Scholar] [CrossRef]
- Piluzza, G.; Sulas, L.; Bullitta, S. Tannins in forage plants and their role in animal husbandry and environmental sustainability: A review. Grass Forage Sci. 2014, 69, 32–48. [Google Scholar] [CrossRef]
- Thi, M.N.; Van Binh, D.; Ørskov, E.R. Effect of foliages containing condensed tannins and on gastrointestinal parasites. Anim. Feed Sci. Technol. 2005, 121, 77–87. [Google Scholar]
- McSweeney, C.S.; Palmer, B.; McNeill, D.M.; Krause, D.O. Microbial interactions with tannins: Nutritional consequences for ruminants. Anim. Feed Sci. Technol. 2001, 91, 83–93. [Google Scholar] [CrossRef]
- Puchala, R.; Min, B.R.; Goetsch, A.L.; Sahlu, T. The effect of a condensed tannin-containing forage on methane emission by goats. J. Anim. Sci. 2005, 83, 182–186. [Google Scholar] [CrossRef] [PubMed]
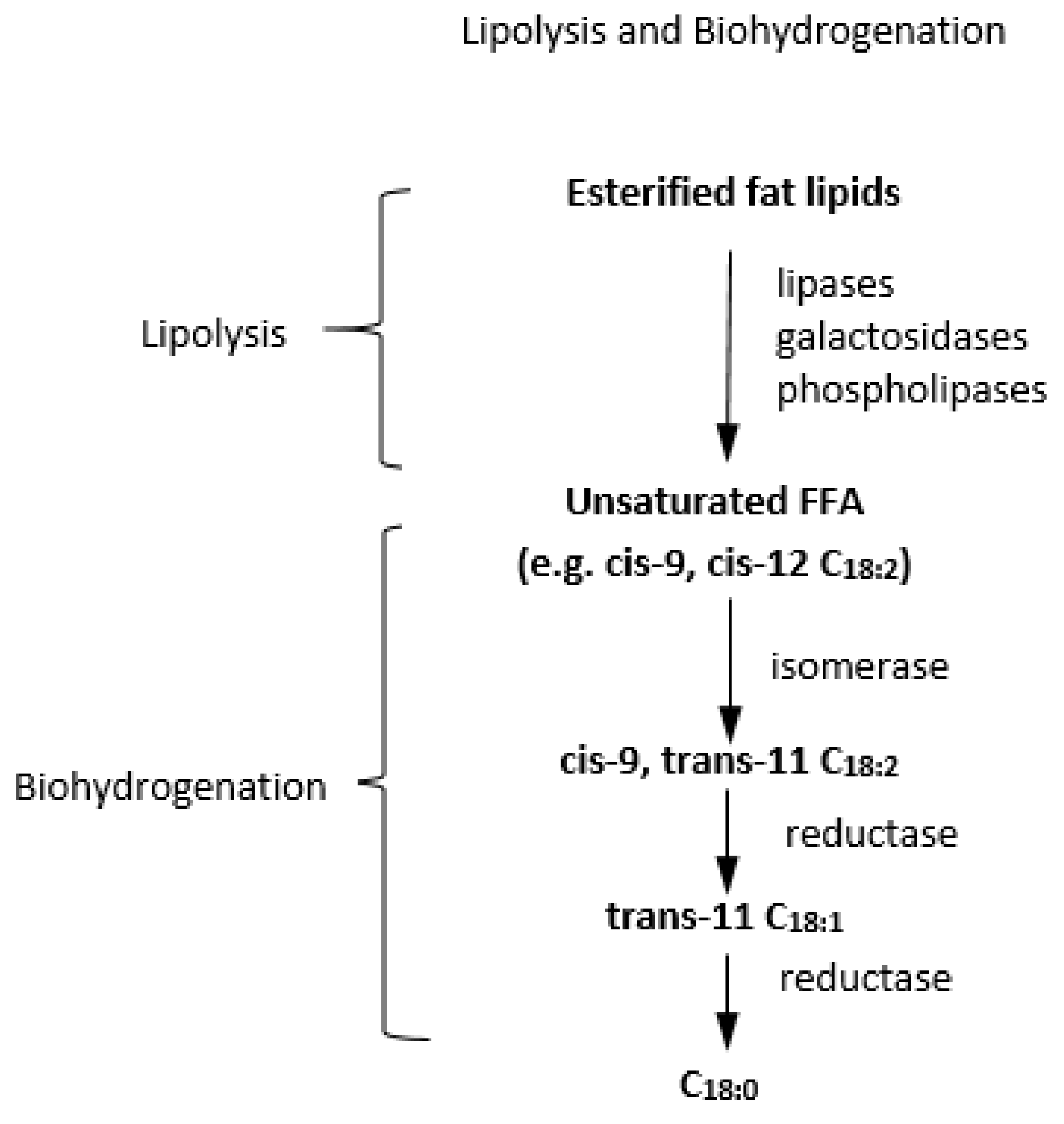
- Toral, P.G.; Monahan, F.J.; Hervas, G.; Frutos, P.; Moloney, A.P. Review: Modulating ruminal lipid metabolism to improve the fatty acid composition of meat and milk. challenges and opportunities. Animal 2018, 12, s272–s281. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, T.C. Lipid Metabolism in the Rumen. J. Dairy Sci. 1993, 76, 3851–3863. [Google Scholar] [CrossRef]
- Garton, G.A.; Lough, A.K.; Vioque, E. Glyceride Hydrolysis and Glycerol Fermentation by Sheep Rumen Contents. J. Gen. Microbiol. 1961, 25, 215–225. [Google Scholar] [CrossRef] [Green Version]
- Kronberg, S.L.; Scholljegerdes, E.J.; Barceló-Coblijn, G.; Murphy, E.J. Flaxseed treatments to reduce biohydrogenation of α-linolenic acid by rumen microbes in cattle. Lipids 2007, 42, 1105–1111. [Google Scholar] [CrossRef]
- Khiaosa-Ard, R.; Bryner, S.F.; Scheeder, M.R.L.; Wettstein, H.R.; Leiber, F.; Kreuzer, M.; Soliva, C.R. Evidence for the inhibition of the terminal step of ruminal α-linolenic acid biohydrogenation by condensed tannins. J. Dairy Sci. 2009, 92, 177–188. [Google Scholar] [CrossRef] [Green Version]
- Vasta, V.; Makkar, H.P.S.; Mele, M.; Priolo, A. Ruminal biohydrogenation as affected by tannins in vitro. Br. J. Nutr. 2009, 102, 82–92. [Google Scholar] [CrossRef] [Green Version]
- Alves, S.P.; Francisco, A.; Costa, M.; Santos-Silva, J.; Bessa, R.J.B. Biohydrogenation patterns in digestive contents and plasma of lambs fed increasing levels of a tanniferous bush (Cistus ladanifer L.) and vegetable oils. Anim. Feed Sci. Technol. 2017, 225, 157–172. [Google Scholar] [CrossRef]
- Campidonico, L.; Toral, P.G.; Priolo, A.; Luciano, G.; Valenti, B.; Hervás, G.; Frutos, P.; Copani, G.; Ginane, C.; Niderkorn, V. Fatty acid composition of ruminal digesta and longissimus muscle from lambs fed silage mixtures including red clover, sainfoin, and timothy. J. Anim. Sci. 2016, 94, 1550–1560. [Google Scholar] [CrossRef] [PubMed]
- Scislowski, V.; Bauchart, D.; Gruffat, D.; Laplaud, P.M.; Durand, D. Effects of dietary n-6 or n-3 polyunsaturated fatty acids protected or not against ruminal hydrogenation on plasma lipids and their susceptibility to peroxidation in fattening steers. J. Anim. Sci. 2005, 83, 2162–2174. [Google Scholar] [CrossRef] [PubMed]
- Niezen, J.H.; Waghorn, T.S.; Charleston, W.A.G.; Waghorn, G.C. Growth and gastrointestinal nematode parasitism in lambs grazing either lucerne ( Medicago sativa ) or sulla ( Hedysarum coronarium ) which contains condensed tannins. J. Agric. Sci. 1995, 125, 281–289. [Google Scholar] [CrossRef]
- Min, B.; Hart, S. Tannins for suppression of internal parasites. J. Anim. Sci. 2003, 81, E102–E109. [Google Scholar]
- Molan, A.L.; Waghorn, G.C.; Mcnabb, W.C. Condensed tannins and gastro-intestinal parasites in sheep. 57 Proc. N. Z. Grassl. Assoc. 1999, 61, 57–61. [Google Scholar]
- Max, R.A.; Buttery, P.J.; Wakelin, D.; Kimambo, A.E.; Kassuku, A.A.; Mtenga, L.A. The potential of controlling gastrointestinal parasitic infections in tropical small ruminants using plants high in tannins or extracts from them. In The Contribution of Small Ruminants in Alleviating Poverty: Communicating Messages from Research; Smith, T., Godfrey, S.H., Buttery, P.J., Owen, E., Eds.; Natural Resources International: England, UK, 2004; pp. 115–125. [Google Scholar]
- Marley, C.L.; Cook, R.; Keatinge, R.; Barrett, J.; Lampkin, N.H. The effect of birdsfoot trefoil (Lotus corniculatus) and chicory (Cichorium intybus) on parasite intensities and performance of lambs naturally infected with helminth parasites. Vet. Parasitol. 2003, 112, 147–155. [Google Scholar] [CrossRef]
- Hoskin, S.O.; Wilson, P.R.; Barry, T.N.; Charleston, W.A.G.; Waghorn, G.C. Effect of forage legumes containing condensed tannins on lungworm (Dictyocaulus sp.) and gastrointestinal parasitism in young red deer (Cervus elaphus). Res. Vet. Sci. 2000, 68, 223–230. [Google Scholar] [CrossRef]
- Mannetje, L. Harry Stobbs Memorial Lecture, 1994: Potential and prospects of legume-based pastures in the tropics. Trop. Grassl. 1997, 31, 81–94. [Google Scholar]
- Meat and Livestock Australia State of the Industry Report: The Australian Red Meat and Livestock Industry. Available online: https://www.mla.com.au/globalassets/mla-corporate/research-and-development/documents/industry-issues/state-of-the-industry-v-1.2-final.pdf (accessed on 29 April 2019).
- Coates, D.B.; Miller, C.P.; Hendricksen, R.E.; Jones, R.J. Stability and productivity of Stylosanthes pastures in Australia. II. Animal production from Stylosanthes pastures. Trop. Grassl. 1997, 31, 494–502. [Google Scholar]
- Meat and Livestock Australia Legumes for Clay Soils. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&ved=2ahUKEwjp6eOtsN7iAhUNfysKHVxNC4cQFjAAegQIAxAC&url=https%3A%2F%2Fwww.mla.com.au%2Fdownload%2Ffinalreports%3FitemId%3D874&usg=AOvVaw3MfMlkc2BzojTHeSR7NdVZ (accessed on 29 April 2019).
- Schlink, A.C.; Burt, R.L. Assessment of the chemical composition of selected tropical legume seeds as animal feed. Trop. Agric. 1993, 70, 169–173. [Google Scholar]
- Cook, B.G.; Pengelly, B.C.; Brown, S.D.; Donnelly, J.L.; Eagles, D.A.; Franco, M.A.; Hanson, J.; Mullen, B.F.; Partridge, I.J.; Peters, M.; et al. Tropical Forages: An Interactive Selection Tool. Available online: http://www.tropicalforages.info/key/forages/Media/Html/entities/index.htm (accessed on 10 June 2019).
- Clem, R.L. Animal production from legume-based ley pastures in southeastern Queensland. In Tropical Legumes for Sustainable Farming Systems in Southern Africa and Australia; Whitbread, A.M., Pengelly, B.C., Eds.; Australian Centre for International Agricultural Research: Canberra, Australia, 2004; pp. 136–144. ISBN 1863204199 (print)r1863204202. [Google Scholar]
- Vandermeulen, S.; Singh, S.; Ramírez-Restrepo, C.A.; Kinley, R.D.; Gardiner, C.P.; Holtum, J.A.M.; Hannah, I.; Bindelle, J. In vitro assessment of ruminal fermentation, digestibility and methane production of three species of Desmanthus for application in northern Australian grazing systems. Crop Pasture Sci. 2018, 69, 797–807. [Google Scholar] [CrossRef]
- Gardiner, C.P.; Swan, S.J. Abandoned pasture legumes offer potential economic and environmental benefits in semiarid clay soil rangelands. In Proceedings of the Australian Rangeland Society 15th Biennial Conference Proceedings, Charters Towers, QLD, Australia, 28 September–2 October 2008; p. 93. [Google Scholar]
- Gonzalez-V, E.A.; Hussey, M.A.; Ortega-S, J.A. Nutritive value of Desmanthus associated with Kleingrass during the establishment year. Rangel. Ecol. Manag. 2005, 58, 308–314. [Google Scholar] [CrossRef]
- Isbell, R.F. The Australian Soil Classification, 2nd ed.; CSIRO: Melbourne, Australia, 2016; ISBN 9781486304639. [Google Scholar]
- Gardiner, C.; Bielig, L.; Schlink, A.; Coventry, R.; Waycott, M. Desmanthus—A new pasture legume for the dry tropics. In Proceedings of the 4th International Crop Science Congress, Brisbane, Australia, 26 September–1 October 2004; pp. 1–6. [Google Scholar]
- Gardiner, C.; Kempe, C.; Kempe, N.; Campbell, G.; Fleury, H.; Malau-Aduli, A.; Walker, G.; Suybeng, B.; Mwangi, F. Progardes Desmanthus—An update. In Proceedings of the Northern Beef Research Update Conference, Brisbane, Australia, 19–22 August 2019; p. 33. [Google Scholar]
- Gardiner, C.; Parker, A. Steer liveweight gains on ProgardesTM Desmanthus/Buffel pastures in Queensland. In Proceedings of the 2nd Australian and New Zealand Societies of Animal Production Joint Conference, England, New Zealand, 2–5 July 2012. [Google Scholar]
- Aoetpah, A.; Gardiner, C.; Gummow, B.; Walker, G. Growth and eye muscle area of cross-bred Boer goats fed Desmanthus cultivar JCU 1 hay. In Proceedings of the 32nd Biennial Conference of the Australian Society of Animal Production, Wagga Wagga, Australia, 2–4 July 2018; p. 2563. [Google Scholar]
- Rusiyantono, Y.; Syukur, S.H. The effect of supplementation of different legume leaves on feed intake, digestion and growth of Kacang goats given Mulato grass. J. Agric. Sci. Technol. 2017, 7, 117–122. [Google Scholar]
- Pacheco, P.S.; Pascoal, L.L.; Restle, J.; Vaz, F.N.; Arboitte, M.Z.; Vaz, R.Z.; Santos, J.P.A.; de Oliveira, T.M.L. Risk assessment of finishing beef cattle in feedlot: Slaughter weights and correlation amongst input variables. Rev. Bras. Zootec. 2014, 43, 92–99. [Google Scholar] [CrossRef] [Green Version]
- Meat and Livestock Australia Lotfeeding and Intensive Finishing. Available online: https://www.mla.com.au/research-and-development/feeding-finishing-nutrition/Lotfeeding-intensive-finishing/# (accessed on 29 April 2019).
- Drouillard, J.S.; Kuhl, G.L. Effects of previous grazing nutrition and management on feedlot performance of cattle. J. Anim. Sci. 1999, 77, 136–146. [Google Scholar] [CrossRef]
- Coffey, K.P.; Lomas, L.W.; Moyer, J.L. Grazing and subsequent feedlot performance by steers that grazed different types of fescue pasture. J. Prod. Agric. 2013, 3, 415–420. [Google Scholar] [CrossRef]
- Burton, R.O.J.; Berends, P.T.; Moyer, J.L.; Coffey, K.P.; Lomas, L.W. Economic analysis of grazing and subsequent feeding of steers from three fescue pasture alternatives. J. Prod. Agric. 1994, 7, 482–489. [Google Scholar] [CrossRef]
- Johnson, H.E.; DiCostanzo, A. A meta-analysis on the effects of backgrounding strategy on feedlot and carcass performance. J. Anim. Sci. 2017, 95, 49. [Google Scholar] [CrossRef] [Green Version]
- Maltin, C.; Balcerzak, D.; Tilley, R.; Delday, M. Determinants of meat quality: Tenderness. Proc. Nutr. Soc. 2003, 62, 337–347. [Google Scholar] [CrossRef]
- Wood, J.D.; Enser, M.; Fisher, A.V.; Nute, G.R.; Richardson, R.I.; Sheard, P.R. Manipulating meat quality and composition. Proc. Nutr. Soc. 1999, 58, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.D. Consequences for meat quality of reducing carcass fatness. In Reducing Fat in Meat Animals; Wood, J.D., Fisher, A.V., Eds.; Elsevier Applied Science: Barking, UK, 1990; pp. 344–397. ISBN 1851664556. [Google Scholar]
- Webb, E.C. Manipulating beef quality through feeding. S. Afr.J. Food Sci. Nutr. 2006, 7, 5–15. [Google Scholar]
- Webb, E.C.; O’Neill, H.A. The animal fat paradox and meat quality. Meat Sci. 2008, 80, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Dujková, R.; Ranganathan, Y.; Dufek, A.; Macák, J.; Bezdíček, J. Polymorphic effects of FABP4 and SCD genes on intramuscular fatty acid profiles in longissimus muscle from two cattle breeds. Acta Vet. BRNO 2015, 84, 327–336. [Google Scholar] [CrossRef] [Green Version]
- Scollan, N.; Hocquette, J.F.; Nuernberg, K.; Dannenberger, D.; Richardson, I.; Moloney, A. Innovations in beef production systems that enhance the nutritional and health value of beef lipids and their relationship with meat quality. Meat Sci. 2006, 74, 17–33. [Google Scholar] [CrossRef]
- French, P.; Stanton, C.; Lawless, F.; O’Riordan, E.G.; Monahan, F.J.; Caffrey, P.J.; Moloney, A.P. Fatty acid composition, including conjugated linoleic acid, of intramuscular fat from steers offered grazed grass, grass silage, or concentrate-based diets. J. Anim. Sci. 2000, 78, 2849–2855. [Google Scholar] [CrossRef]
- Dehghan, M.; Mente, A.; Zhang, X.; Swaminathan, S.; Li, W.; Mohan, V.; Iqbal, R.; Kumar, R.; Wentzel-Viljoen, E.; Rosengren, A.; et al. Associations of fats and carbohydrate intake with cardiovascular disease and mortality in 18 countries from five continents (PURE): A prospective cohort study. Lancet 2017, 390, 2050–2062. [Google Scholar] [CrossRef] [Green Version]
- Astrup, A.; Bertram, H.C.S.; Bonjour, J.P.; De Groot, L.C.P.; De Oliveira Otto, M.C.; Feeney, E.L.; Garg, M.L.; Givens, I.; Kok, F.J.; Krauss, R.M.; et al. WHO draft guidelines on dietary saturated and trans fatty acids: Time for a new approach? BMJ 2019, 366, 14137. [Google Scholar] [CrossRef] [Green Version]
- Guasch-Ferré, M.; Zong, G.; Willett, W.C.; Zock, P.L.; Wanders, A.J.; Hu, F.B.; Sun, Q. Associations of monounsaturated fatty acids from plant and animal sources with total and cause-specific mortality in two us prospective cohort studies. Circ. Res. 2019, 124, 1266–1275. [Google Scholar] [CrossRef]
- Scollan, N.D.; Choi, N.J.; Kurt, E.; Fisher, A.V.; Enser, M.; Wood, J.D. Manipulating the fatty acid composition of muscle and adipose tissue in beef cattle. Br. J. Nutr. 2001, 85, 115–124. [Google Scholar] [CrossRef] [Green Version]
- McAfee, A.J.; McSorley, E.M.; Cuskelly, G.J.; Moss, B.W.; Wallace, J.M.W.; Bonham, M.P.; Fearon, A.M. Red meat consumption: An overview of the risks and benefits. Meat Sci. 2010, 84, 1–13. [Google Scholar] [CrossRef]
- FAO Fats and Fatty Acids in Human Nutrition Report of an Expert Consultation. Available online: http://www.fao.org/3/a-i1953e.pdf (accessed on 15 May 2019).
- WHO Diet, Nutrition and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation, 28 January–1 February 2002. Available online: https://www.who.int/dietphysicalactivity/publications/trs916/en/ (accessed on 15 May 2019).
- Department of Health, London (United Kingdom). Nutritional aspects of cardiovascular disease. Report of the cardiovascular review group committee on medical aspects of food policy. Nutr. Asp. Cardiovasc. Dis. 1994, 46, 1–186. [Google Scholar]
- Lorenzen, C.L.; Golden, J.W.; Martz, F.A.; Grün, I.U.; Ellersieck, M.R.; Gerrish, J.R.; Moore, K.C. Conjugated linoleic acid content of beef differs by feeding regime and muscle. Meat Sci. 2007, 75, 159–167. [Google Scholar] [CrossRef]
- Muir, P.D.; Deaker, J.M.; Bown, M.D. Effects of forage- and grain-based feeding systems on beef quality: A review. N. Z. J. Agric. Res. 1998, 41, 623–635. [Google Scholar] [CrossRef]
- Hocquette, J.F.; Gondret, F.; Baza, E.; Mdale, F.; Jurie, C.; Pethick, D.W. Intramuscular fat content in meat-producing animals: Development, genetic and nutritional control, and identification of putative markers. Animal 2010, 4, 303–319. [Google Scholar] [CrossRef] [Green Version]
- Jeremiah, L.E. The influence of subcutaneous fat thickness and marbling on beef: Palatability and consumer acceptability. Food Res. Int. 1996, 29, 513–520. [Google Scholar] [CrossRef]
- Steen, R.W.J.; Lavery, N.P.; Kilpatrick, D.J.; Porter, M.G. Effects of pasture and high-concentrate diets on the performance of beef cattle, carcass composition at equal growth rates, and the fatty acid composition of beef. N. Z. J. Agric. Res. 2003, 46, 69–81. [Google Scholar] [CrossRef] [Green Version]
- De Smet, S.; Raes, K.; Demeyer, D. Meat fatty acid composition as affected by fatness and genetic factors: A review. Anim. Res. 2004, 53, 81–98. [Google Scholar] [CrossRef]
- Insausti, K.; Beriain, M.; Alzueta, M.; Carr, T.; Purroy, A. Lipid composition of the intramuscular fat of beef from Spanish cattle breeds stored under modified atmosphere. Meat Sci. 2004, 66, 639–646. [Google Scholar] [CrossRef]
- Špehar, M.; Vincek, D.; Žgur, S. Beef Quality: Factors affecting tenderness and marbling. Stočarstvo 2008, 62, 463–478. [Google Scholar]
- Hwang, I.H.; Thompson, J.M. Effects of pH early postmortem on meat quality in beef longissimus. Asian Australas. J. Anim. Sci. 2003, 16, 1218–1223. [Google Scholar] [CrossRef]
- Smith, G.C.; Dutson, T.R.; Hostetler, R.L.; Carpenter, Z.L. Fatness, rate of chilling and tenderness of lamb. J. Food Sci. 1976, 41, 748–756. [Google Scholar] [CrossRef]
- Sañudo, C.; Nute, G.R.; Campo, M.M.; María, G.; Baker, A.; Sierra, I.; Enser, M.E.; Wood, J.D. Assessment of commercial lamb meat quality by British and Spanish taste panels. Meat Sci. 1998, 48, 91–100. [Google Scholar] [CrossRef]
- Juárez, M.; Aldai, N.; López-Campos, Ó.; Dugan, M.; Uttaro, B.; Aalhus, J. Beef texture and juiciness. In Handbook of Meat and Meat Processing; Hui, Y.H., Ed.; CRC Press: Boca Raton, FL, USA, 2012; pp. 177–206. [Google Scholar]
- Arshad, M.S.; Sohaib, M.; Ahmad, R.S.; Nadeem, M.T.; Imran, A.; Arshad, M.U.; Kwon, J.H.; Amjad, Z. Ruminant meat flavor influenced by different factors with special reference to fatty acids. Lipids Health Dis. 2018, 17, 223. [Google Scholar] [CrossRef] [Green Version]
- Mottram, D.S.; Salter, L.J. Flavor formation in meat-related maillard systems containing phospholipids. In Thermal Generation of Aromas; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 1989; pp. 442–451. [Google Scholar]
- Mottram, D. Meat flavour. In Understanding Natural Flavors; Piggott, J.R., Paterson, A., Eds.; Springer: Boston, MA, USA, 1994; pp. 140–163. [Google Scholar]
- Melton, S.L.; Black, J.M.; Davis, G.W.; Backus, W.R. Flavor and Selected Chemical Components of Ground Beef from Steers Backgrounded on Pasture and Fed Corn up to 140 Days. J. Food Sci. 1982, 47, 699–704. [Google Scholar] [CrossRef]
- Lee, J.Y.; Oh, D.Y.; Kim, H.J.; Jang, G.S.; Lee, S.U. Detection of superior genotype of fatty acid synthase in Korean native cattle by an environment-adjusted statistical model. Asian Australas. J. Anim. Sci. 2017, 30, 765–772. [Google Scholar] [CrossRef] [Green Version]
- McMillin, K.W.; Hoffman, L.C. Improving the quality of meat from ratites. In Improving the Sensory and Nutritional Quality of Fresh Meat; Kerry, J., Ed.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 418–446. ISBN 9781845693435. [Google Scholar]
- Wood, J.D. Fat deposition and the quality of fat tissue in meat animals. In Fats in Animal Nutrition; Wiseman, J., Ed.; Elsevier: Amsterdam, The Netherlands, 1984; pp. 407–435. ISBN 9781483100357. [Google Scholar]
- Mapiye, C.; Aalhus, J.L.; Turner, T.D.; Rolland, D.C.; Basarab, J.A.; Baron, V.S.; McAllister, T.A.; Block, H.C.; Uttaro, B.; Lopez-Campos, O.; et al. Effects of feeding flaxseed or sunflower-seed in high-forage diets on beef production, quality and fatty acid composition. Meat Sci. 2013, 95, 98–109. [Google Scholar] [CrossRef]
- Whetsell, M.S.; Rayburn, E.B.; Lozier, J.D. Human Health Effects of Fatty Acids in Beef. Available online: https://pdfs.semanticscholar.org/d2b7/462e78b4e9c11c17a99a5aabed57d4e2fb49.pdf (accessed on 14 August 2019).
- Smith, S.B.; Gill, C.A.; Lunt, D.K.; Brooks, M.A. Regulation of fat and fatty acid composition in beef cattle. Asian-Australas. J. Anim. Sci. 2009, 22, 1225–1233. [Google Scholar] [CrossRef]
- Nürnberg, K.; Ender, B.; Papstein, H.J.; Wegner, J.; Ender, K.; Nürnberg, G. Effects of growth and breed on the fatty acid composition of the muscle lipids in cattle. Z. Lebensm. Forsch. A 1999, 208, 332–335. [Google Scholar] [CrossRef]
- Malau-Aduli, A.E.O.; Siebert, B.D.; Bottema, C.D.K.; Pitchford, W.S. A comparison of the fatty acid composition of triacylglycerols in adipose tissue from Limousin and Jersey cattle. Aust. J. Agric. Res. 1997, 48, 715–722. [Google Scholar] [CrossRef] [Green Version]
- Malau-Aduli, A.E.O.; Siebert, B.D.; Bottema, C.D.K.; Pitchford, W.S. Breed comparison of the fatty acid composition of muscle phospholipids in Jersey and Limousin cattle. J. Anim. Sci. 1998, 76, 766–773. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abe, T.; Saburi, J.; Hasebe, H.; Nakagawa, T.; Misumi, S.; Nade, T.; Nakajima, H.; Shoji, N.; Kobayashi, M.; Kobayashi, E. Novel mutations of the FASN gene and their effect on fatty acid composition in Japanese black beef. Biochem. Genet. 2009, 47, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Savage, D.B. Nutritional management of heifers in northern Australia. In Recent Advances in Animal Nutrition in Australia; Cronj, P.B., Richards, N., Eds.; Animal Science, University of New England: Armidale, Australia, 2005; Volume 15, pp. 205–214. ISBN 1863899278. [Google Scholar]
- Drouillard, J.S. Current situation and future trends for beef production in the United States of America—A review. Asian Australas. J. Anim. Sci. 2018, 31, 1007–1016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Priolo, A.; Bella, M.; Lanza, M.; Galofaro, V.; Biondi, L.; Barbagallo, D.; Ben Salem, H.; Pennisi, P. Carcass and meat quality of lambs fed fresh sulla (Hedysarum coronarium L.) with or without polyethylene glycol or concentrate. Small Rumin. Res. 2005, 59, 281–288. [Google Scholar] [CrossRef]
- Dierking, R.M.; Kallenbach, R.L.; Grün, I.U. Effect of forage species on fatty acid content and performance of pasture-finished steers. Meat Sci. 2010, 85, 597–605. [Google Scholar] [CrossRef]
- Kook, K.; Choi, B.H.; Sun, S.S.; Garcia, F.; Myung, K.H. Effect of fish oil supplement on growth performance, ruminal metabolism and fatty acid composition of longissimus muscle in Korean cattle. Asian Australas. J. Anim. Sci. 2002, 15, 66–71. [Google Scholar] [CrossRef]
- Dhiman, T.R.; Zaman, S.; Olson, K.C.; Bingham, H.R.; Ure, A.L.; Pariza, M.W. Influence of feeding soybean oil on conjugated linoleic acid content in beef. J. Agric. Food Chem. 2005, 53, 684–689. [Google Scholar] [CrossRef]
- Nuernberg, K.; Dannenberger, D.; Nuernberg, G.; Ender, K.; Voigt, J.; Scollan, N.D.; Wood, J.D.; Nute, G.R.; Richardson, R.I. Effect of a grass-based and a concentrate feeding system on meat quality characteristics and fatty acid composition of longissimus muscle in different cattle breeds. Livest. Prod. Sci. 2005, 94, 137–147. [Google Scholar] [CrossRef]
- Tume, R.K. The effects of environmental factors on fatty acid composition and the assessment of marbling in beef cattle: A review. Aust. J. Exp. Agric. 2004, 44, 663–668. [Google Scholar] [CrossRef]
- Jacques, J.; Chouinard, Y.; Gariépy, C.; Cinq-Mars, D. Meat quality, organoleptic characteristics and fatty acid composition of Dorset lambs fed different forage to concentrate ratio or fresh grass. Can. J. Anim. Sci. 2016, 97, 290–301. [Google Scholar] [CrossRef]
- Poulson, C.S.; Dhiman, T.R.; Ure, A.L.; Cornforth, D.; Olson, K.C. Conjugated linoleic acid content of beef from cattle fed diets containing high grain, CLA, or raised on forages. Livest. Prod. Sci. 2004, 91, 117–128. [Google Scholar] [CrossRef]
- Duckett, S.K.; Neel, J.P.S.; Lewis, R.M.; Fontenot, J.P.; Clapham, W.M. Effects of forage species or concentrate finishing on animal performance, carcass and meat quality. J. Anim. Sci. 2013, 91, 1454–1467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Realini, C.E.; Duckett, S.K.; Brito, G.W.; Dalla Rizza, M.; De Mattos, D. Effect of pasture vs. concentrate feeding with or without antioxidants on carcass characteristics, fatty acid composition, and quality of Uruguayan beef. Meat Sci. 2004, 66, 567–577. [Google Scholar] [CrossRef]
- Park, S.J.; Beak, S.H.; Jung, D.J.S.; Kim, S.Y.; Jeong, I.H.; Piao, M.Y.; Kang, H.J.; Fassah, D.M.; Na, S.W.; Yoo, S.P.; et al. Genetic, management and nutritional factors affecting intramuscular fat deposition in beef cattle—A review. Asian Australas. J. Anim. Sci. 2018, 31, 1043–1061. [Google Scholar] [CrossRef] [Green Version]
- Duckett, S.K.; Wagner, D.G.; Yates, L.D.; Dolezal, H.G.; May, S.G. Effects of time on feed on beef nutrient composition. J. Anim. Sci. 1993, 71, 2079–2088. [Google Scholar] [CrossRef] [Green Version]
- Schönfeldt, H.C.; Naudé, R.T.; Boshoff, E. Effect of age and cut on the nutritional content of South African beef. Meat Sci. 2010, 86, 674–683. [Google Scholar] [CrossRef]
- Kelava Ugarković, N.; Ivanković, A.; Konjačić, M. Effect of breed and age on beef carcass quality, fatness and fatty acid composition. Arch. Anim. Breed. 2013, 56, 958–970. [Google Scholar] [CrossRef]
- Bednárová, A.; Mocák, J.; Gössler, W.; Velik, M.; Kaufmann, J.; Staruch, L. Effect of animal age and gender on fatty acid and elemental composition in Austrian beef applicable for authentication purposes. Chem. Pap. 2013, 67, 274–283. [Google Scholar] [CrossRef]
- Chail, A.; Legako, J.F.; Pitcher, L.R.; Griggs, T.C.; Ward, R.E.; Martini, S.; MacAdam, J.W. Legume finishing provides beef with positive human dietary fatty acid ratios and consumer preference comparable with grain-finished beef. J. Anim. Sci. 2016, 94, 2184–2197. [Google Scholar] [CrossRef] [Green Version]
- Ramírez-Retamal, J.; Morales, R.; Martínez, M.E.; Barra, R. de la Effect of the type of pasture on the meat characteristics of Chilote lambs. Food Nutr. Sci. 2014, 05, 635–644. [Google Scholar]
- Moloney, A.P.; Fievez, V.; Martin, B.; Nute, G.R.; Richardson, R.I. Botanically diverse forage-based rations for cattle: Implications for product composition, product quality and consumer health. In Proceedings of the 22nd General Meeting of the European Grassland Federation, Uppsala, Sweden, 9–12 June 2008; pp. 361–374. [Google Scholar]
- Lee, M.R.F.; Winters, A.L.; Scollan, N.D.; Dewhurst, R.J.; Theodorou, M.K.; Minchin, F.R. Plant-mediated lipolysis and proteolysis in red clover with different polyphenol oxidase activities. J. Sci. Food Agric. 2004, 84, 1639–1645. [Google Scholar] [CrossRef]
- Ashes, J.R.; Siebert, B.D.; Gulati, S.K.; Cuthbertson, A.Z.; Scott, T.W. Incorporation of n-3 fatty acids of fish oil into tissue and serum lipids of ruminants. Lipids 1992, 27, 629–631. [Google Scholar] [CrossRef] [PubMed]
- Richardson, R.I.; Hallett, K.G.; Ball, R.; Nute, G.R.; Wood, J.D.; Scollan, N.D. Effect of free and ruminally protected fish oils on fatty acid composition, sensory and oxidative characteristics of beef loin muscle. In Proceedings of the 50th International Congress Meat Science and Technology, Heksinki, Finland, 8–13 August 2004; Volume 2, p. 43. [Google Scholar]
- Kruk, Z.A.; Bottema, C.D.K.; Davis, J.J.; Siebert, B.D.; Harper, G.S.; Di, J.; Pitchford, W.S. Effects of vitamin A on growth performance and carcass quality in steers. Livest. Sci. 2008, 119, 12–21. [Google Scholar] [CrossRef]
- Oka, A.; Maruo, Y.; Miki, T.; Yamasaki, T.; Saito, T. Influence of vitamin A on the quality of beef from the Tajima strain of Japanese black cattle. Meat Sci. 1998, 48, 159–167. [Google Scholar] [CrossRef]
- Hida, Y.; Kawada, T.; Kayahashi, S.; Ishihara, T.; Fushiki, T. Counteraction of retinoic acid and 1,25-dihydroxyvitamin D3 on up-regulation of adipocyte differentiation with PPARγ ligand, an antidiabetic thiazolidinedione, in 3T3-L1 cells. Life Sci. 1998, 62, PL205–PL211. [Google Scholar] [CrossRef]
- Smith, S.B.; Kawachi, H.; Choi, C.B.; Choi, C.W.; Wu, G.; Sawyer, J.E. Cellular regulation of bovine intramuscular adipose tissue development and composition. J. Anim. Sci. 2009, 87, E72–E82. [Google Scholar] [CrossRef] [Green Version]
- McDowell, L.R. Vitamins in Animal Nutrition: Comparative Aspects to Human Nutrition; Academic Press: Gainesville, FL, USA, 1989; ISBN 9780124833722. [Google Scholar]
- Kawachi, H. Micronutrients affecting adipogenesis in beef cattle. Anim. Sci. J. 2006, 77, 463–471. [Google Scholar] [CrossRef]
- Ohashi, H. Effect of vitamin C on the quality of Wagyu beef. Res. Bull. Aichi Agric. Res. Cent. 2000, 32, 207–214. [Google Scholar]
- Kawada, T.; Aoki, N.; Kamei, Y.; Maeshige, K.; Nishiu, S.; Sugimoto, E. Comparative investigation of vitamins and their analogues on terminal differentiation, from preadipocytes to adipocytes, of 3T3-L1 cells. Comp. Biochem. Physiol. Part A Physiol. 1990, 96, 323–326. [Google Scholar] [CrossRef]
- Montgomery, J.L.; Blanton, J.R.; Horst, R.L.; Galyean, M.L.; Morrow, K.J.; Wester, D.B.; Miller, M.F. Effects of biological type of beef steers on vitamin D, calcium, and phosphorus status. J. Anim. Sci. 2004, 82, 2043–2049. [Google Scholar] [CrossRef]
- Lee, C.E.; Park, N.K.; Seong, P.N.; Jin, S.H.; Park, B.Y.; Kim, K.I. Effects of deletion of Ca Supplement (limestone) on growth and beef quality in Hanwoo finishing steers. J. Anim. Sci. Technol. 2003, 45, 455–462. [Google Scholar] [CrossRef] [Green Version]
- Vernon, R.G. Lipid metabolism in the adipose tissue of ruminant animals. In Lipid Metabolism in Ruminant Animals; Christie, W., Ed.; Elsevier: Ayr, UK, 1981; pp. 279–362. ISBN 9780080237893. [Google Scholar]
- Zembayashi, M.; Nishimura, K.; Lunt, D.K.; Smith, S.B. Effect of breed type and sex on the fatty acid composition of subcutaneous and intramuscular lipids of finishing steers and heifers. J. Anim. Sci. 1995, 73, 3325–3332. [Google Scholar] [CrossRef] [PubMed]
- Dannenberger, D.; Nuernberg, K.; Nuernberg, G.; Scollan, N.; Steinhart, H.; Ender, K. Effect of pasture vs. concentrate diet on CLA isomer distribution in different tissue lipids of beef cattle. Lipids 2005, 40, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Laborde, F.L.; Mandell, I.B.; Tosh, J.J.; Buchanan-Smith, J.G.; Wilton, J.W. Effect of management strategy on growth performance, carcass characteristics, fatty acid composition, and palatability attributes in crossbred steers. Can. J. Anim. Sci. 2011, 82, 49–57. [Google Scholar] [CrossRef]
- Oka, A.; Iwaki, F.; Dohgo, T.; Ohtagaki, S.; Noda, M.; Shiozaki, T.; Endoh, O.; Ozaki, M. Genetic effects on fatty acid composition of carcass fat of Japanese Black Wagyu steers. J. Anim. Sci. 2002, 80, 1005–1011. [Google Scholar] [CrossRef]
- Pitchford, W.S.; Deland, M.P.B.; Siebert, B.D.; Malau-Aduli, A.E.O.; Bottema, C.D.K. Genetic variation in fatness and fatty acid composition of crossbred cattle. J. Anim. Sci. 2002, 80, 2825–2832. [Google Scholar] [CrossRef] [Green Version]
- Rempel, L.A.; Casas, E.; Shackelford, S.D.; Wheeler, T.L. Relationship of polymorphisms within metabolic genes and carcass traits in crossbred beef cattle. J. Anim. Sci. 2012, 90, 1311–1316. [Google Scholar] [CrossRef]
- Shin, S.C.; Chung, E.R. Association of SNP marker in the leptin gene with carcass and meat quality traits in Korean cattle. Asian Australas. J. Anim. Sci. 2006, 20, 1–6. [Google Scholar] [CrossRef]
- Barendse, W.; Bunch, R.J.; Harrison, B.E.; Thomas, M.B. The growth hormone 1 GH1:c.457C>G mutation is associated with intramuscular and rump fat distribution in a large sample of Australian feedlot cattle. Anim. Genet. 2006, 37, 211–214. [Google Scholar] [CrossRef]
- Bhuiyan, M.S.A.; Yu, S.L.; Jeon, J.T.; Yoon, D.; Cho, Y.M.; Park, E.W.; Kim, N.K.; Kim, K.S.; Lee, J.H. DNA polymorphisms in SREBF1 and FASN genes affect fatty acid composition in Korean cattle (Hanwoo). Asian Australas. J. Anim. Sci. 2009, 22, 765–773. [Google Scholar] [CrossRef]
- Zhang, S.; Knight, T.J.; Reecy, J.M.; Beitz, D.C. DNA polymorphisms in bovine fatty acid synthase are associated with beef fatty acid composition. Anim. Genet. 2008, 39, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Matsuhashi, T.; Maruyama, S.; Uemoto, Y.; Kobayashi, N.; Mannen, H.; Abe, T.; Sakaguchi, S.; Kobayashi, E. Effects of bovine fatty acid synthase, stearoyl-coenzyme A desaturase, sterol regulatory element-binding protein 1, and growth hormone gene polymorphisms on fatty acid composition and carcass traits in Japanese Black cattle. J. Anim. Sci. 2011, 89, 12–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaplanová, K.; Dufek, A.; Dračková, E.; Simeonovová, J.; Šubrt, J.; Vrtková, I.; Dvořák, J. The association of CAPN1, CAST, SCD, and FASN polymorphisms with beef quality traits in commercial crossbred cattle in the czech republic. Czech J. Anim. Sci. 2013, 58, 489–496. [Google Scholar] [CrossRef]
- Westerling, D.B.; Hedrick, H.B. Fatty acid composition of bovine lipids as influenced by diet, sex and anatomical location and relationship to sensory characteristics. J. Anim. Sci. 1979, 48, 1343–1348. [Google Scholar] [CrossRef]
- May, S.G.; Sturdivant, C.A.; Lunt, D.K.; Miller, R.K.; Smith, S.B. Comparison of sensory characteristics and fatty acid composition between Wagyu crossbred and Angus steers. Meat Sci. 1993, 35, 289–298. [Google Scholar] [CrossRef]
- Sturdivant, C.A.; Lunt, D.K.; Smith, G.C.; Smith, S.B. Fatty acid composition of subcutaneous and intramuscular adipose tissues and M. longissimus dorsi of Wagyu cattle. Meat Sci. 1992, 32, 449–458. [Google Scholar] [CrossRef]
- Archibeque, S.L.; Lunt, D.K.; Gilbert, C.D.; Tume, R.K.; Smith, S.B. Fatty acid indices of stearoyl-CoA desaturase do not reflect actual stearoyl-CoA desaturase enzyme activities in adipose tissues of beef steers finished with corn-, flaxseed-, or sorghum-based diets. J. Anim. Sci. 2005, 83, 1153–1166. [Google Scholar] [CrossRef]
- Lee, S.H.; Yoon, D.H.; Choi, N.J.; Hwang, S.H.; Cheong, E.Y.; Oh, S.J.; Cheong, I.C.; Lee, C.S. Developmental relationship of unsaturated fatty acid composition and stearoyl-CoA desaturase mRNA level in Hanwoo steers’ muscle. Asian Australas. J. Anim. Sci. 2005, 18, 562–566. [Google Scholar] [CrossRef]
- Taniguchi, M.; Utsugi, T.; Oyama, K.; Mannen, H.; Kobayashi, M.; Tanabe, Y.; Ogino, A.; Tsuji, S. Genotype of stearoyl-CoA desaturase is associated with fatty acid composition in Japanese Black cattle. Mamm. Genome 2004, 14, 142–148. [Google Scholar] [CrossRef]
- Roy, R.; Taourit, S.; Zaragoza, P.; Eggen, A.; Rodellar, C. Genomic structure and alternative transcript of bovine fatty acid synthase gene (FASN): Comparative analysis of the FASN gene between monogastric and ruminant species. Cytogenet. Genome Res. 2005, 111, 65–73. [Google Scholar] [CrossRef]
- Morris, C.A.; Cullen, N.G.; Glass, B.C.; Hyndman, D.L.; Manley, T.R.; Hickey, S.M.; McEwan, J.C.; Pitchford, W.S.; Bottema, C.D.K.; Lee, M.A.H. Fatty acid synthase effects on bovine adipose fat and milk fat. Mamm. Genome 2007, 18, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.; Lee, Y.; La, B.; Yeo, J.; Chung, E.; Kim, Y.; Lee, C. Fatty acid composition of beef is associated with exonic nucleotide variants of the gene encoding FASN. Mol. Biol. Rep. 2012, 39, 4083–4090. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.H.F.; Chang, I.; Hudak, C.S.S.; Hyun, S.; Kwan, H.Y.; Sul, H.S. A Role of DNA-PK for the Metabolic Gene Regulation in Response to Insulin. Cell 2009, 136, 1056–1072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, R.H.F.; Sul, H.S. DNA-PK: Relaying the insulin signal to USF in lipogenesis. Cell Cycle 2009, 8, 1977–1978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kulig, H.; Kowalewska-Łuczak, I.; Kmieć, M.; Wojdak-Maksymiec, K. ANXA9, SLC27A3, FABP3 and FABP4 single nucleotide polymorphisms in relation to milk production traits in Jersey cows. Czech J. Anim. Sci. 2010, 55, 463–467. [Google Scholar] [CrossRef] [Green Version]
| Pasture | Supplement | Outcome | Reference |
|---|---|---|---|
| Urochloa decumbens | Corn, Corn gluten, Soybean, Urea, | ADG up to 0.75 kg | [51] |
| Urochloa decumbens hay | Pure casein, urea and ammonia | Increase NDF digestion | [52] |
| Urochloa decumbens hay | Urea, ammonium sulphate and albumin | Increased DMI and | [53] |
| NDF digestion | |||
| Urochloa brizantha | Cottonseed meal, corn and urea | ADG of up to 0.3 kg | [54] |
| Urochloa brizantha | Soybean meal, urea and grain sorghum | ADG of up to 0.5 kg | [55] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mwangi, F.W.; Charmley, E.; Gardiner, C.P.; Malau-Aduli, B.S.; Kinobe, R.T.; Malau-Aduli, A.E.O. Diet and Genetics Influence Beef Cattle Performance and Meat Quality Characteristics. Foods 2019, 8, 648. https://doi.org/10.3390/foods8120648
Mwangi FW, Charmley E, Gardiner CP, Malau-Aduli BS, Kinobe RT, Malau-Aduli AEO. Diet and Genetics Influence Beef Cattle Performance and Meat Quality Characteristics. Foods. 2019; 8(12):648. https://doi.org/10.3390/foods8120648
Chicago/Turabian StyleMwangi, Felista W., Edward Charmley, Christopher P. Gardiner, Bunmi S. Malau-Aduli, Robert T. Kinobe, and Aduli E. O. Malau-Aduli. 2019. "Diet and Genetics Influence Beef Cattle Performance and Meat Quality Characteristics" Foods 8, no. 12: 648. https://doi.org/10.3390/foods8120648





