Genome-Wide Identification and Functional Characterization of CCHC-Type Zinc Finger Genes in Ustilaginoidea virens
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fungal Strains and Growth Conditions
2.2. Transactivation Activity Assay in Yeast
2.3. DNA/RNA Manipulation and RT-qPCR
2.4. Gene Deletion and Complementation
2.5. Vegetative Growth and Conidiation
2.6. Stress Adaptation Assays and Sugar Utilization Assays
2.7. Pathogenicity and Plant Infection Assays
2.8. Generation of the GFP-UvCCHC Fusion Construct
2.9. RNA-seq and Data Analysis
2.10. Accession Numbers
2.11. Statistical Analysis
3. Results
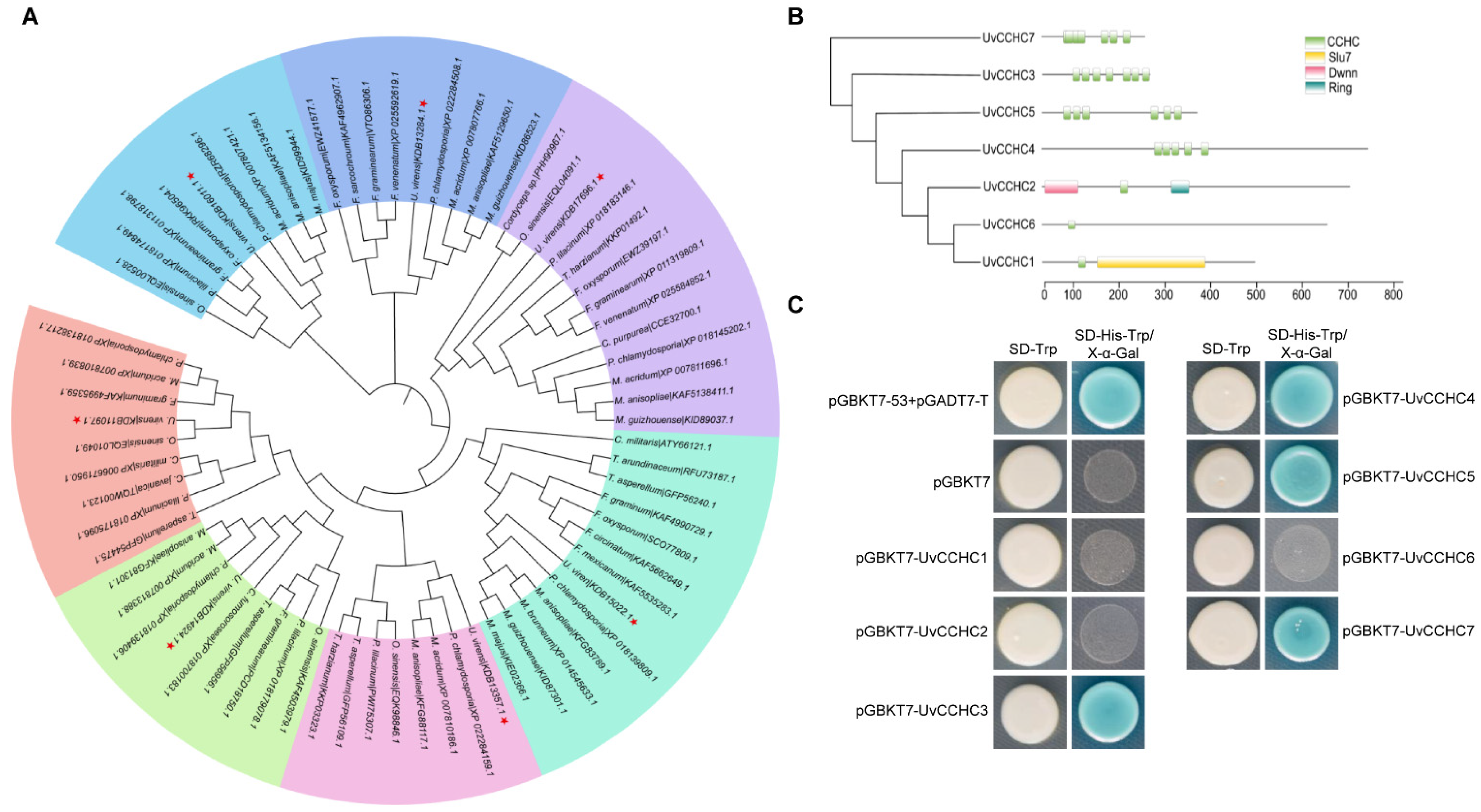
3.1. Identification of CCHC-Type Zinc-Finger Proteins in U. virens
3.2. Expression Patterns of CCHC-Type Zinc-Finger Genes in U. virens
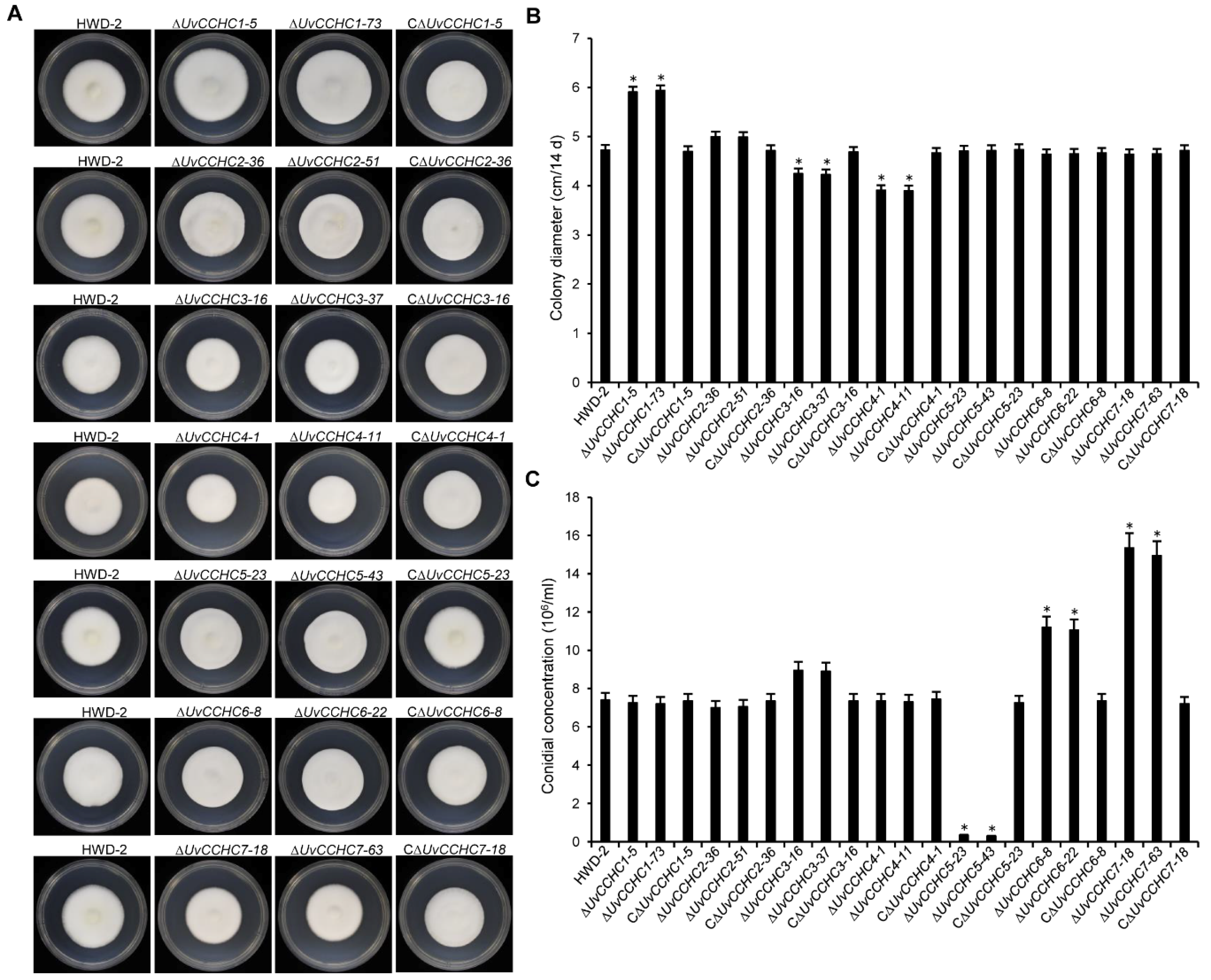
3.3. UvCCHC Genes Are Important for the Mycelial Growth and Conidiation of U. virens
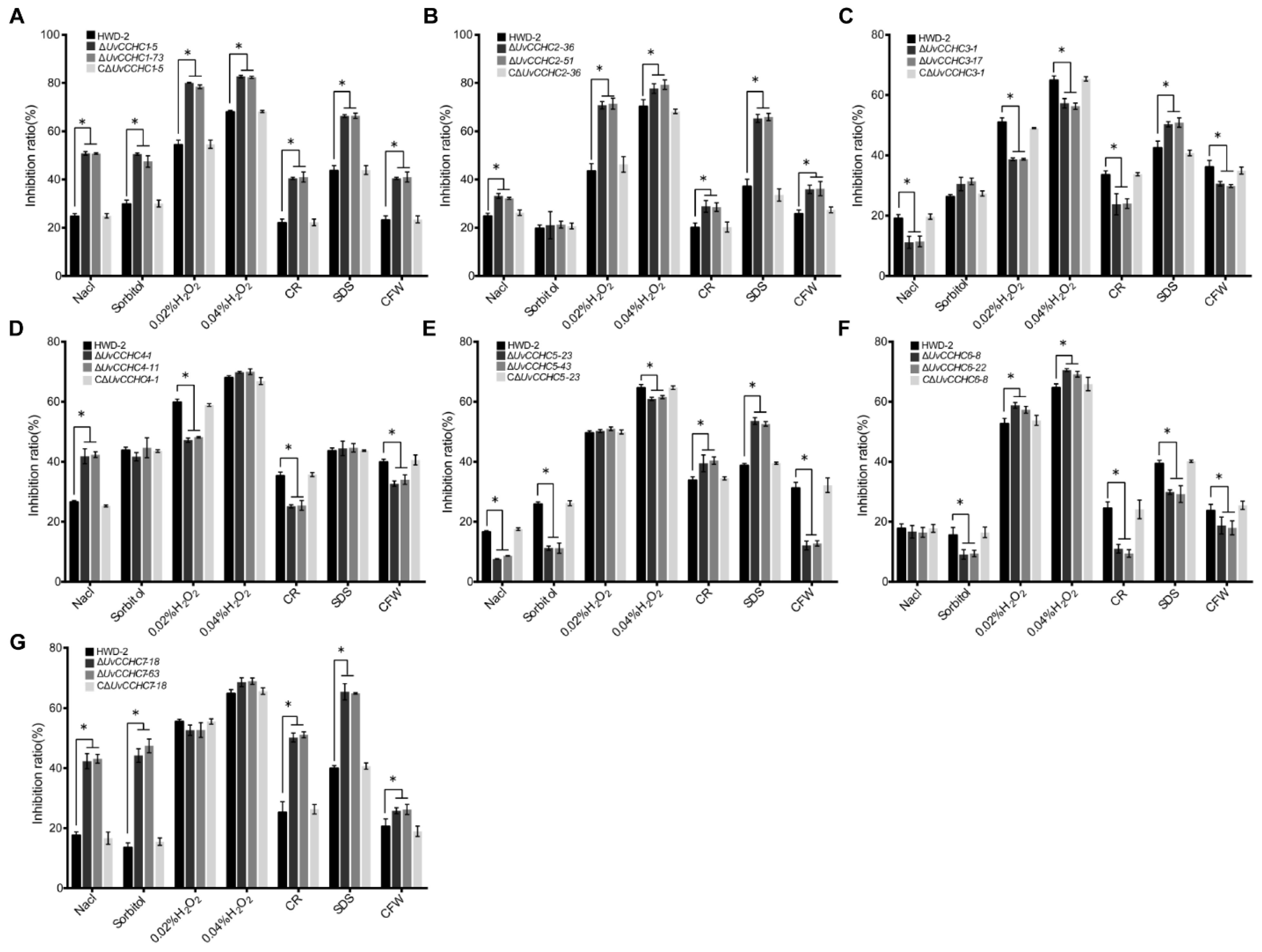
3.4. UvCCHC Genes Play Key Roles in the Responses of U. virens to Environmental Stress
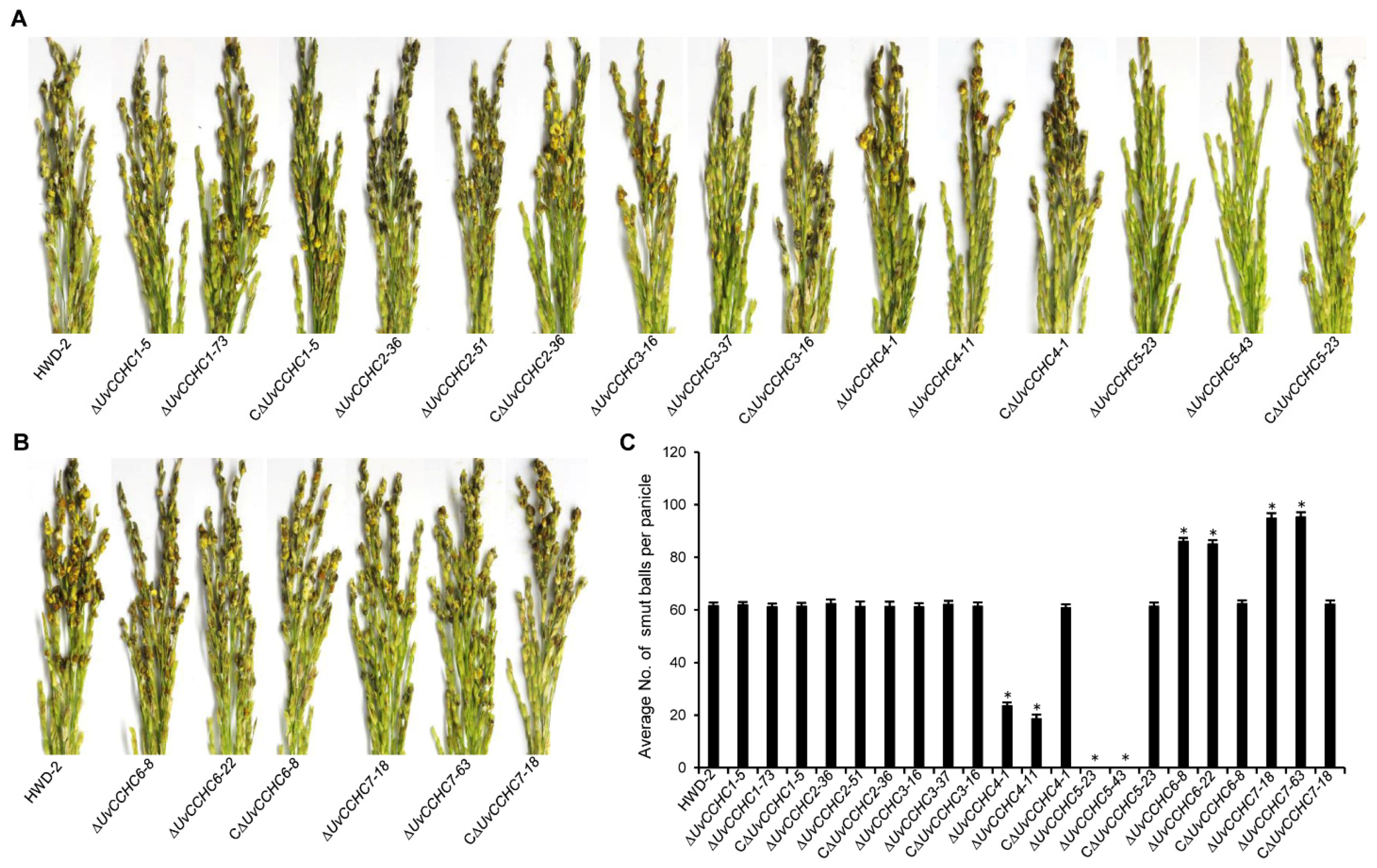
3.5. UvCCHC Genes Play Crucial Roles in the Virulence of U. virens
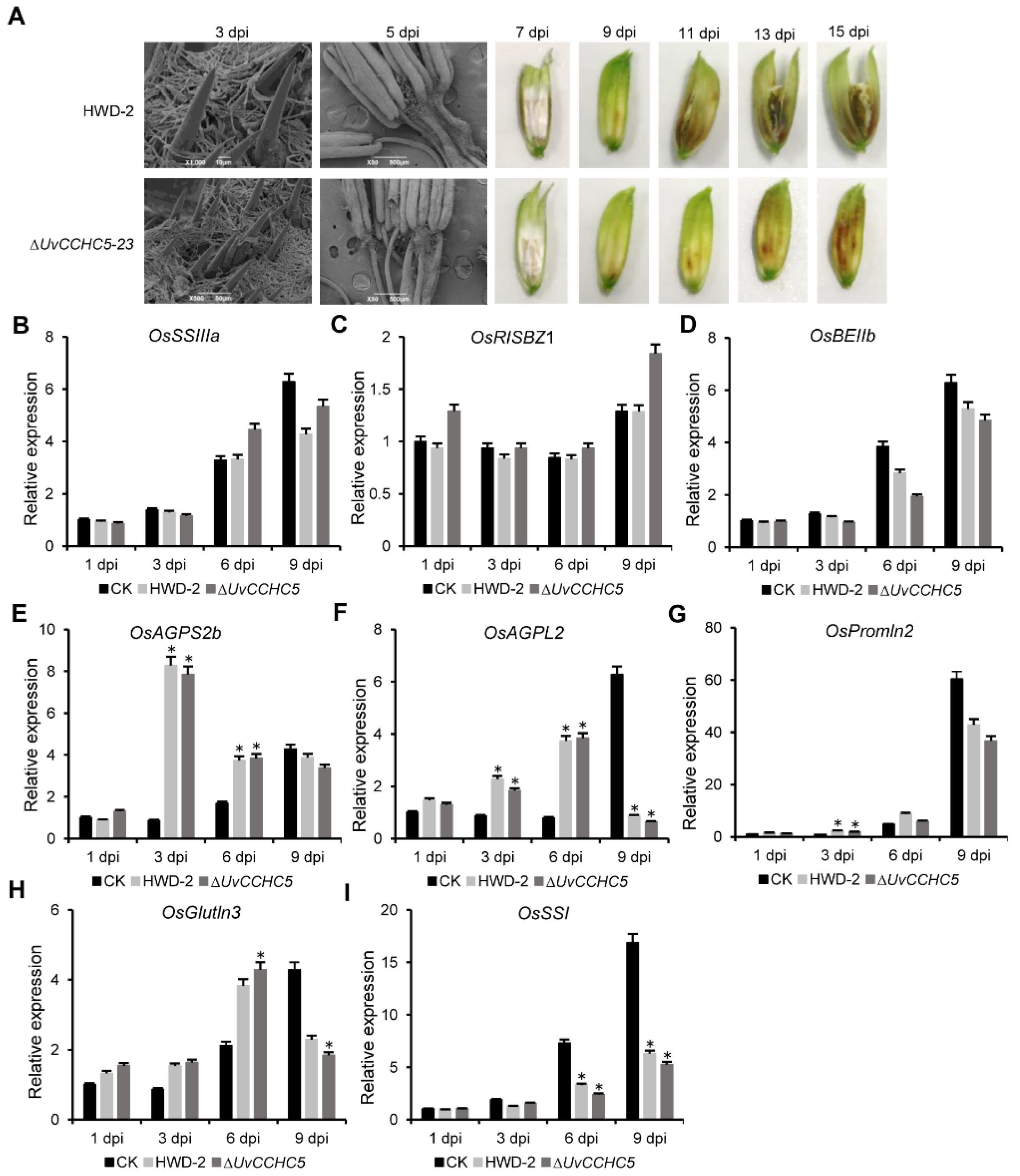
3.6. UvCCHC5 Regulates the Formation of Smut Balls
3.7. Deletion of UvCCHC5 Does Not Affect the Expression of Rice Genes Associated with Grain Filling
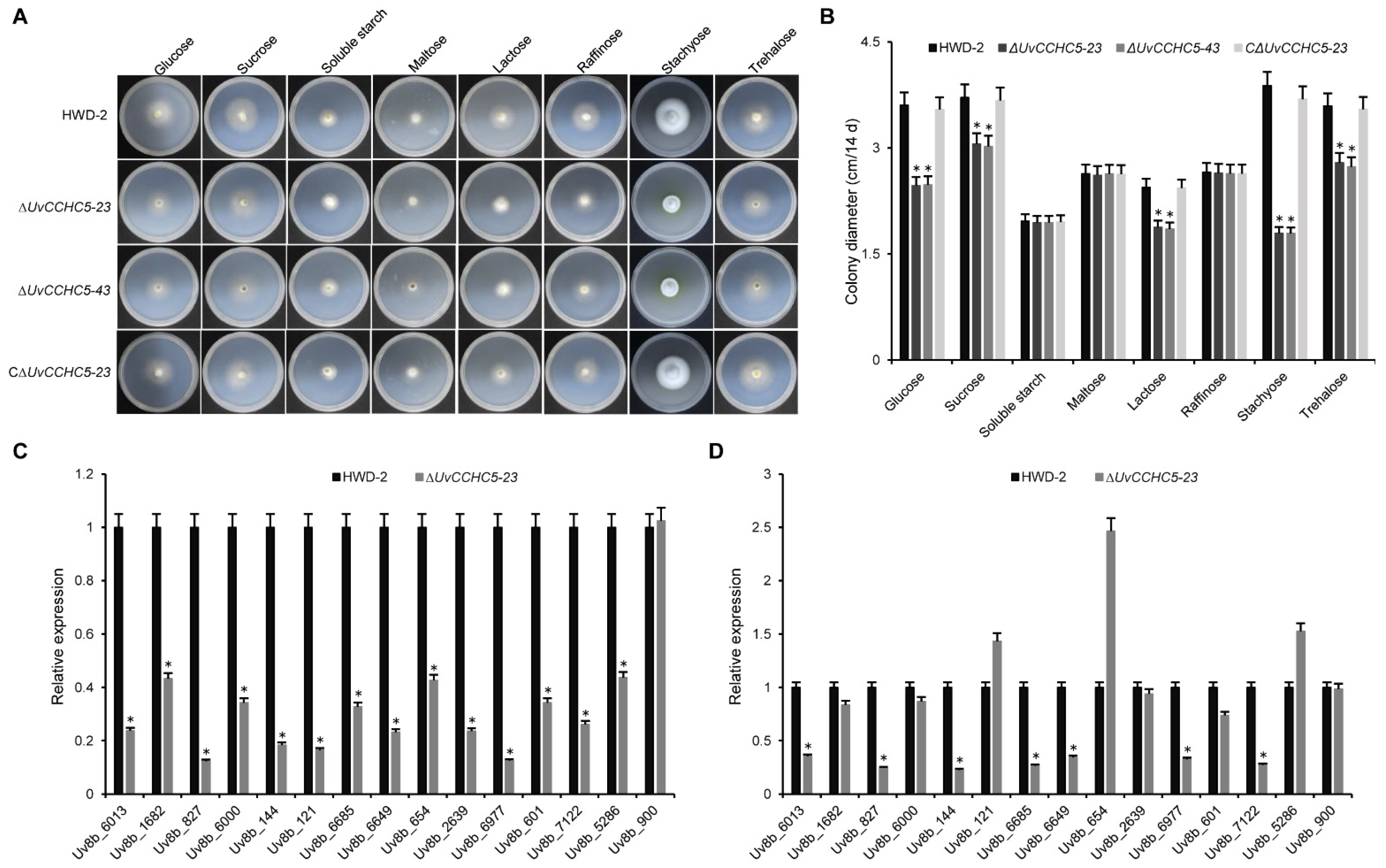
3.8. UvCCHC5 Plays Important Roles in Sugar Utilization, Carbohydrate Transport, and Transmembrane Transport
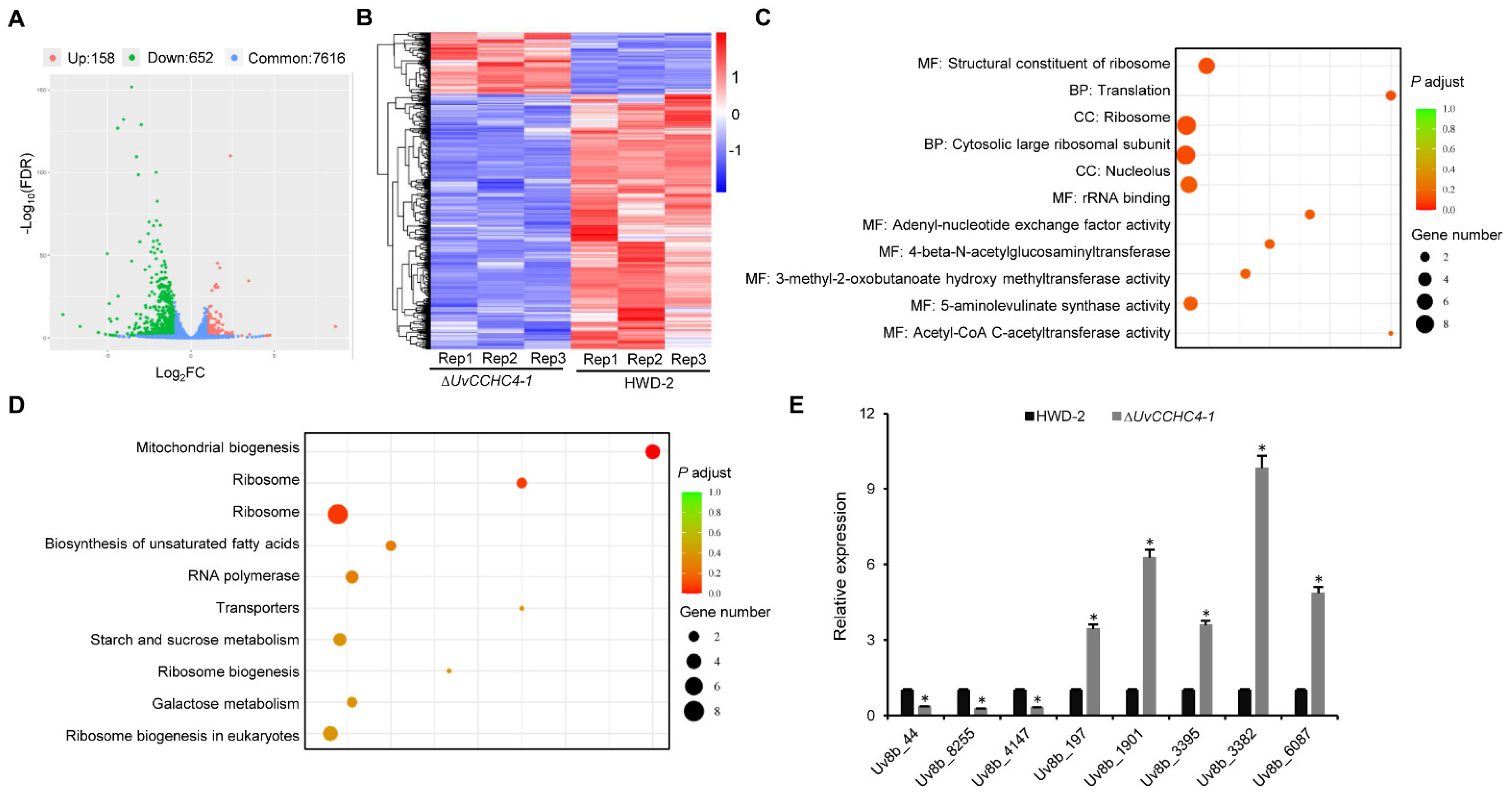
3.9. Genome-Wide Identification of Genes Regulated by UvCCHC4
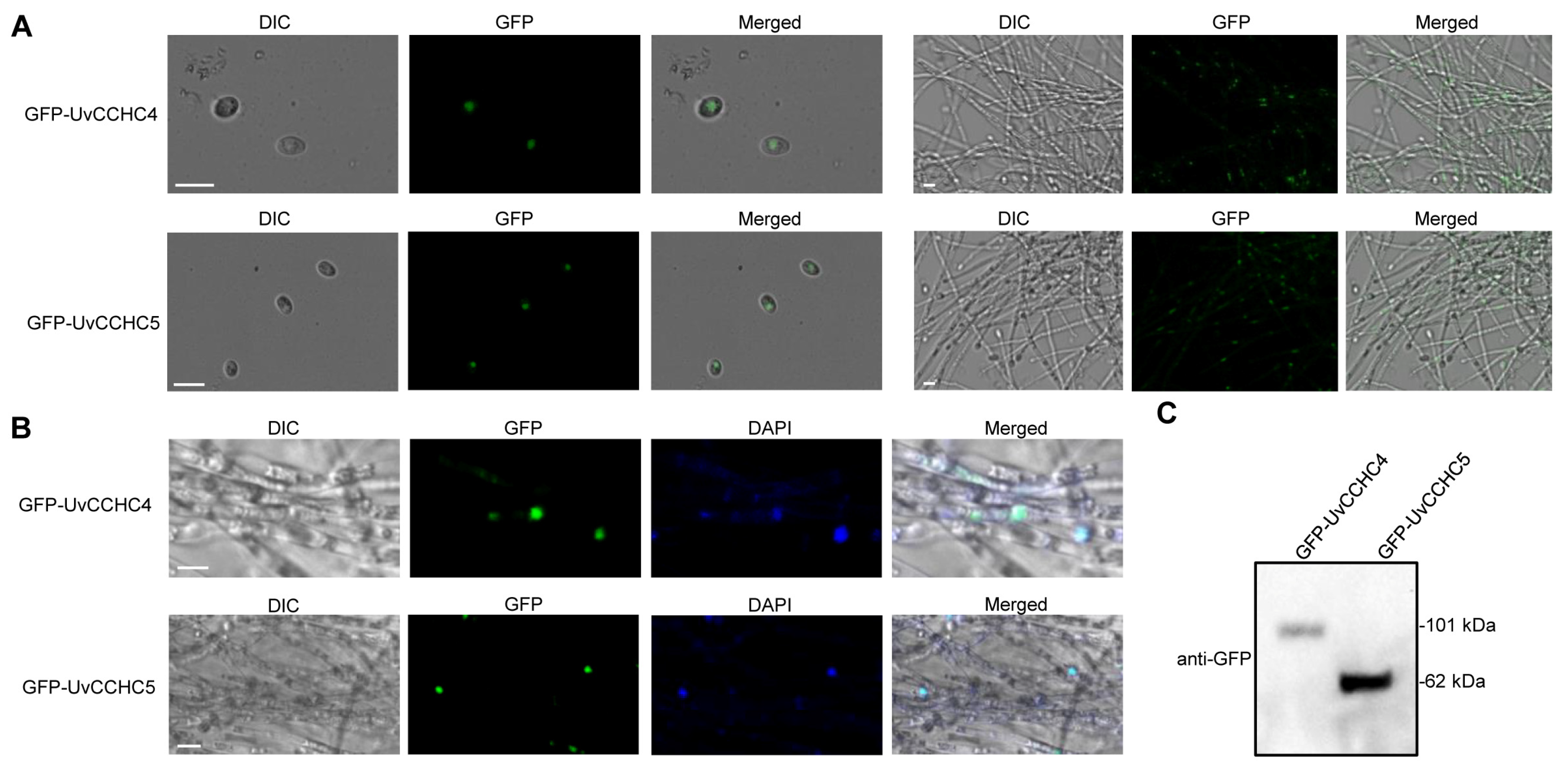
3.10. Subcellular Localization of UvCCHC4 and UvCCHC5 in U. virens
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Coradetti, S.T.; Craig, J.P.; Xiong, Y.; Shock, T.; Tian, C.; Glass, N.L. Conserved and essential transcription factors for cellulase gene expression in ascomycete fungi. Proc. Natl. Acad. Sci. USA 2012, 109, 7397–7402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- John, E.; Singh, K.B.; Oliver, R.P.; Tan, K.C. Transcription factor control of virulence in phytopathogenic fungi. Mol. Plant Pathol. 2021, 22, 858–881. [Google Scholar] [CrossRef]
- Laity, J.H.; Lee, B.M.; Wright, P.E. Zinc finger proteins: New insights into structural and functional diversity. Curr. Opin. Struc. Biol. 2001, 11, 39–46. [Google Scholar] [CrossRef]
- Berg, J.M.; Shi, Y. The galvanization of biology: A growing appreciation for the roles of zinc. Science 1996, 271, 1081–1085. [Google Scholar] [CrossRef]
- Aceituno-Valenzuela, U.; Micol-Ponce, R.; Ponce, M.R. Genome-wide analysis of CCHC-type zinc finger (ZCCHC) proteins in yeast, Arabidopsis, and humans. Cell. Mol. Life Sci. 2020, 77, 3991–4014. [Google Scholar] [CrossRef]
- Espinosa, J.M.; Portal, D.; Lobo, G.S.; Pereira, C.A.; Alonso, G.D.; Gómez, E.B.; Lan, G.H.; Pomar, R.V.R.; Flawiá, M.M.; Torres, H.N. Trypanosoma cruzi poly-zinc finger protein: A novel DNA/RNA-binding CCHC-zinc finger protein. Mol. Biochem. Parasit. 2003, 131, 35–44. [Google Scholar] [CrossRef]
- Tan, Y.W.; Hong, W.; Liu, D.X. Binding of the 5′-untranslated region of coronavirus RNA to zinc finger CCHC-type and RNA-binding motif 1 enhances viral replication and transcription. Nucleic Acids Res. 2012, 40, 5065–5077. [Google Scholar] [CrossRef]
- Luo, X.; Wang, B.; Gao, S.; Zhang, F.; Terzaghi, W.; Dai, M. Genome-wide association study dissects the genetic bases of salt tolerance in maize seedlings. J. Integr. Plant Biol. 2019, 61, 658–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Q.; Wang, M.; Shen, J.; Chen, D.; Zheng, Y.; Zhang, W. ZmOST1 mediates abscisic acid regulation of guard cell ion channels and drought stress responses. J. Integr. Plant Biol. 2019, 61, 478–491. [Google Scholar] [CrossRef]
- Sun, Y.; Li, J.Q.; Yan, J.Y.; Yuan, J.J.; Li, G.X.; Wu, Y.R.; Xu, J.M.; Huang, R.F.; Harberd, N.P.; Ding, Z.J. Ethylene promotes seed iron storage during Arabidopsis seed maturation via ERF95 transcription factor. J. Integr. Plant Biol. 2020, 62, 1193–1212. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhang, T.Z. Functional diversifications of GhERF1 duplicate genes after the formation of allotetraploid cotton. J. Integr. Plant Biol. 2019, 61, 60–74. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Wei, X.; Jiao, G.; Chen, W.; Wu, Y.; Sheng, Z.; Hu, S.; Xie, L.; Wang, J.; Tang, S.; et al. GBSS-BINDING PROTEIN, encoding a CBM48 domain-containing protein, affects rice quality and yield. J. Integr. Plant Biol. 2020, 62, 948–966. [Google Scholar] [CrossRef] [Green Version]
- Tang, K.; Zhao, L.; Ren, Y.; Yang, S.; Zhu, J.K.; Zhao, C. The transcription factor ICE1 functions in cold stress response by binding to the promoters of CBF and COR genes. J. Integr. Plant Biol. 2020, 62, 258–263. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Gao, J.; Zhang, Z.; Liu, W.; Cheng, P.; Mu, W.; Su, T.; Chen, S.; Chen, F.; Jiang, J. Overexpression of CmSOS1 confers waterlogging tolerance in Chrysanthemum. J. Integr. Plant Biol. 2020, 62, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Radkova, M.; Revalska, M.; Kertikova, D.; Iantcheva, A. Zinc finger CCHC-type protein related with seed size in model legume species Medicago truncatula. Biotechnol. Biotech. Equip. 2019, 33, 278–285. [Google Scholar] [CrossRef] [Green Version]
- Lu, X.; Zhou, Y.; Fan, F.; Peng, J.; Zhang, J. Coordination of light, circadian clock with temperature: The potential mechanisms regulating chilling tolerance in rice. J. Integr. Plant Biol. 2020, 62, 737–760. [Google Scholar] [CrossRef]
- Zhao, M.; Wang, L.; Wang, J.; Jin, J.; Zhang, N.; Lei, L.; Gao, T.; Jing, T.; Zhang, S.; Wu, Y. Induction of priming by cold stress via inducible volatile cues in neighboring tea plants. J. Integr. Plant Biol. 2020, 62, 1461–1468. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Lei, Y.; Lu, C.; Wang, L.; Wu, J. MYC2, MYC3, and MYC4 function additively in wounding-induced jasmonic acid biosynthesis and catabolism. J. Integr. Plant Biol. 2020, 62, 1159–1175. [Google Scholar] [CrossRef]
- Liu, M.; Zhu, J.; Dong, Z. Immediate transcriptional responses of Arabidopsis leaves to heat shock. J. Integr. Plant Biol. 2020, 63, 468–483. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, Y.; Pang, Y.; Yu, H.; Zhang, W.; Zhao, X.; Yu, J. The distinct roles of zinc finger CCHC-type (ZCCHC) superfamily proteins in the regulation of RNA metabolism. RNA Biol. 2021, 1–20. [Google Scholar] [CrossRef]
- de Paula, Q.A.; Mangrum, J.B.; Farrell, N.P. Zinc finger proteins as templates for metal ion exchange: Substitution effects on the C-finger of HIV nucleocapsid NCp7 using M (chelate) species (M=Pt, Pd, Au). J. Inorg. Biochem. 2009, 103, 1347–1354. [Google Scholar] [CrossRef]
- Shimizu, K.; Chen, W.; Ashique, A.M.; Moroi, R.; Li, Y.P. Molecular cloning, developmental expression, promoter analysis and functional characterization of the mouse CNBP gene. Gene 2003, 307, 51–62. [Google Scholar] [CrossRef]
- Berglund, J.A.; Chua, K.; Abovich, N.; Reed, R.; Rosbash, M. The splicing factor BBP interacts specifically with the pre-mRNA branchpoint sequence UACUAA C. Cell 1997, 89, 781–787. [Google Scholar] [CrossRef] [Green Version]
- Jones, M.H.; Frank, D.N.; Guthrie, C. Characterization and functional ordering of Slu7p and Prp17p during the second step of pre-mRNA splicing in yeast. Proc. Natl. Acad. Sci. USA 1995, 92, 9687–9691. [Google Scholar] [CrossRef] [Green Version]
- Vo, L.T.; Minet, M.; Schmitter, J.M.; Lacroute, F.; Wyers, F. Mpe1, a zinc knuckle protein, is an essential component of yeast cleavage and polyadenylation factor required for the cleavage and polyadenylation of mRNA. Mol. Cell. Biol. 2001, 21, 8346–8356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sammons, M.A.; Samir, P.; Link, A.J. Saccharomyces cerevisiae Gis2 interacts with the translation machinery and is orthogonal to myotonic dystrophy type 2 protein ZNF9. Biochem. Biophys. Res. Commun. 2011, 406, 13–19. [Google Scholar] [CrossRef]
- Miller, R.K.; D’Silva, S.; Moore, J.K.; Goodson, H.V. The CLIP-170 orthologue Bik1p and positioning the mitotic spindle in yeast. Curr. Top. Dev. Biol. 2006, 76, 49–87. [Google Scholar]
- Sun, W.X.; Fan, J.; Fang, A.F.; Li, Y.J.; Tariqjaveed, M.; Li, D.Y.; Hu, D.W.; Wang, W.M. Ustilaginoidea virens: Insights into an emerging rice pathogen. Annu. Rev. Phytopathol. 2020, 58, 363–385. [Google Scholar] [CrossRef]
- Koiso, Y.; Li, Y.; Iwasaki, S.; Kenji, H.; Tomowo, K.; Ryoichi, S.; Yoshikai, F.; Hiroshi, Y.; Zenji, S. Ustiloxins, antimitotic cyclic peptides from smut balls on rice panicles caused by Ustilaginoidea virens. J. Antibiot. 1994, 47, 765–773. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miyazaki, S.; Matsumoto, Y.; Uchihara, T.; Morimoto, K. High-performance liquid chromatographic determination of ustiloxin A in forage rice silage. J. Vet. Med. Sci. 2009, 71, 239–241. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Zhang, K.; Fang, A.F.; Han, Y.Q.; Yang, J.; Xue, M.; Bao, J.; Hu, D.; Zhou, B.; Sun, X.; et al. Specific adaptation of Ustilaginoidea virens in occupying host florets revealed by comparative and functional genomics. Nat. Commun. 2014, 5, 3849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.Y.; Liu, H.; Chen, X.L.; Huang, J.B.; Hsiang, T.; Zheng, L. ATAC-seq data for genome-wide profiling of transcription factor binding sites in the rice false smut fungus Ustilaginoidea virens. Mol. Plant Microbe Interact. 2021, 34, 830–834. [Google Scholar] [CrossRef]
- Lv, B.; Zheng, L.; Liu, H.; Tang, J.T.; Hsiang, T.; Huang, J.B. Use of random T-DNA mutagenesis in identification of gene UvPRO1, a regulator of conidiation, stress response, and virulence in Ustilaginoidea virens. Front. Microbiol. 2016, 7, 2086. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.J.; Yu, M.N.; Song, T.Q.; Cao, H.J.; Yong, M.L.; Qi, Z.Q.; Du, Y.; Zhang, R.S.; Yin, X.L.; Liu, Y.F. A homeobox transcription factor UvHOX2 regulates chlamydospore formation, conidiogenesis and pathogenicity in Ustilaginoidea virens. Front. Microbiol. 2019, 10, 1071. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.Y.; Hai, D.; Tang, J.T.; Liu, H.; Huang, J.B.; Luo, C.X.; Hsiang, T.; Zheng, L. UvCom1 is an important regulator required for development and infection in the rice false smut fungus Ustilaginoidea virens. Phytopathology 2020, 110, 483–493. [Google Scholar] [CrossRef]
- Song, T.Q.; Zhang, X.; Zhang, Y.; Liang, D.; Yan, J.; Yu, J.; Yu, M.; Cao, H.; Yong, M.; Pan, X.; et al. Genome-Wide Identification of Zn (2)-Cys (6) Class Fungal-Specific Transcription Factors (ZnFTFs) and Functional Analysis of UvZnFTF1 in Ustilaginoidea virens. Rice Sci. 2021, 28, 567–578. [Google Scholar]
- Xu, Y.; Wu, S.; Yu, Z.; Moeketsi, E.K.; Yang, Z.; Zhang, Z.; Zhang, H. Transcription factor UvMsn2 is important for vegetative growth, conidiogenesis, stress response, mitochondrial morphology and pathogenicity in the rice false smut fungus Ustilaginoidea virens. Phytopathol. Res. 2021, 3, 16. [Google Scholar] [CrossRef]
- Chen, X.; Li, P.; Liu, H.; Chen, X.; Huang, J.; Luo, C.; Li, G.; Hsiang, T.; Collinge, D.; Zheng, L. A novel transcription factor UvCGBP1 regulates development and virulence of rice false smut fungus Ustilaginoidea virens. Virulence 2021, 12, 1563–1579. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Y.; Tang, J.T.; Pei, Z.X.; Liu, H.; Huang, J.B.; Luo, C.X.; Tom, H.; Zheng, L. The “pears and lemons” protein UvPal1 regulates development and virulence of Ustilaginoidea virens. Environ. Microbiol. 2020, 22, 5414–5432. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Y.; Xu, Q.T.; Duan, Y.H.; Liu, H.; Chen, X.L.; Huang, J.B.; Luo, C.X.; Zhou, D.X.; Zheng, L. Ustilaginoidea virens modulates lysine 2-hydroxyisobutyrylation in rice flowers during infection. J. Integr. Plant Biol. 2021, 63, 1801–1814. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef] [Green Version]
- Xie, C.; Mao, X.; Huang, J.; Ding, Y.; Wu, J.; Dong, S. KOBAS 2.0: A web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res. 2011, 39, 316–322. [Google Scholar] [CrossRef] [Green Version]
- Fan, J.; Guo, X.Y.; Li, L.; Huang, F.; Sun, W.X.; Li, Y.; Huang, Y.Y.; Xu, Y.J.; Shi, J.; Lei, Y.; et al. Infection of Ustilaginoidea virens intercepts rice seed formation but activates grain-filling-related genes. J. Integr. Plant. Biol. 2015, 57, 577–590. [Google Scholar] [CrossRef] [Green Version]
- Song, J.H.; Wei, W.; Lv, B.; Lin, Y.; Yin, W.X.; Peng, Y.L.; Schnabel, G.; Huang, J.B.; Jiang, D.H.; Luo, C.X. Rice false smut fungus hijacks the rice nutrients supply by blocking and mimicking the fertilization of rice ovary. Environ. Microbiol. 2016, 18, 3840–3849. [Google Scholar] [CrossRef]
- Liang, Y.; Han, Y.; Wang, C.; Jiang, C.; Xu, J.R. Targeted deletion of the USTA and UvSLT2 genes efficiently in Ustilaginoidea virens with the CRISPR-Cas9 system. Front. Plant Sci. 2018, 9, 699. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Y.; Pei, Z.X.; Li, P.P.; Li, X.B.; Duan, Y.H.; Liu, H.; Chen, X.L.; Zheng, L.; Luo, C.X.; Huang, J.B. Quantitative proteomics analysis reveals the function of the putative ester cyclase UvEC1 in the pathogenicity of the rice false smut fungus Ustilaginoidea virens. Int. J. Mol. Sci. 2021, 22, 4069. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Y.; Li, X.B.; Li, P.P.; Chen, X.L.; Liu, H.; Huang, J.B.; Luo, C.X.; Tom, H.; Zheng, L. Comprehensive identification of lysine 2-hydroxyisobutyrylated proteins in Ustilaginoidea virens reveals the involvement of lysine 2-hydroxyisobutyrylation in fungal virulence. J. Integr. Plant. Biol. 2021, 63, 409–425. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.T.; Ding, H.; Huang, L.; Wang, Y.H.; Yu, M.N.; Zheng, R.; Yu, J.J.; Liu, Y.F. Low-affinity iron transport protein Uvt3277 is important for pathogenesis in the rice false smut fungus Ustilaginoidea virens. Curr. Genet. 2017, 63, 131–144. [Google Scholar] [CrossRef]
- Fang, A.F.; Gao, H.; Zhang, N.; Zheng, X.H.; Qiu, S.S.; Li, Y.J.; Zhou, S.; Cui, F.H.; Sun, W.X. A novel effector gene SCRE2 contributes to full virulence of Ustilaginoidea virens to rice. Front. Microbiol. 2019, 10, 845. [Google Scholar] [CrossRef]
- Xie, S.L.; Wang, Y.F.; Wei, W.; Li, C.Y.; Liu, Y.; Qu, J.S.; Meng, Q.H.; Lin, Y.; Yin, W.X.; Yang, Y.N.; et al. The Bax inhibitor UvBI-1, a negative regulator of mycelial growth and conidiation, mediates stress response and is critical for pathogenicity of the rice false smut fungus Ustilaginoidea virens. Curr. Genet. 2019, 65, 1–13. [Google Scholar] [CrossRef]
- Guo, W.W.; Gao, Y.X.; Yu, Z.M.; Xiao, Y.H.; Zhang, Z.G.; Zhang, H.F. The adenylate cyclase UvAc1 and phosphodiesterase UvPdeH control the intracellular cAMP level, development, and pathogenicity of the rice false smut fungus Ustilaginoidea virens. Fungal Genet. Biol. 2019, 129, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.T.; Bai, J.; Chen, X.Y.; Zheng, L.; Liu, H.; Huang, J.B. Two protein kinases UvPmk1 and UvCDC2 with significant functions in conidiation, stress response and pathogenicity of rice false smut fungus Ustilaginoidea virens. Curr. Genet. 2019, 66, 409–420. [Google Scholar] [CrossRef]
- Meng, S.; Xiong, M.; Jagernath, J.S.; Wang, C.; Qiu, J.; Shi, H.; Kou, Y. UvAtg8-mediated autophagy regulates fungal growth, stress responses, conidiation, and pathogenesis in Ustilaginoidea virens. Rice 2020, 13, 56. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Yang, J.; Fang, A.F.; Wang, J.Y.; Li, D.Y.; Li, Y.J.; Wang, S.Z.; Cui, F.H.; Yu, J.J.; Liu, Y.F.; et al. The essential effector SCRE1 in Ustilaginoidea virens suppresses rice immunity via a small peptide region. Mol. Plant Pathol. 2020, 21, 445–459. [Google Scholar] [CrossRef] [Green Version]
- Fan, J.; Du, N.; Li, L.; Li, G.; Wang, Y.; Zhou, Y.; Hu, X.; Liu, J.; Zhao, J.; Li, Y.; et al. A core effector UV_1261 promotes Ustilaginoidea virens infection via spatiotemporally suppressing plant defense. Phytopathol. Res. 2019, 1, 11. [Google Scholar] [CrossRef] [Green Version]
- Yan, N. Structural advances for the major facilitator superfamily (MFS) transporters. Trends Biochem. Sci. 2013, 38, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Yang, J.; Wang, Y.Q.; Li, G.B.; Li, Y.; Huang, F.; Wang, W.M. Current understanding on Villosiclava virens, a unique flower-infecting fungus causing rice false smut disease. Mol. Plant Pathol. 2016, 17, 1321–1330. [Google Scholar] [CrossRef]

| Gene ID | Function Description | Name |
|---|---|---|
| Uv8b_1580 | Pre-mRNA-splicing factor slu7 | UvCCHC1 |
| Uv8b_2941 | Retinoblastoma-binding protein | UvCCHC2 |
| Uv8b_4070 | Cellular nucleic acid-binding protein | UvCCHC3 |
| Uv8b_4168 | Zinc-knuckle domain-containing protein | UvCCHC4 |
| Uv8b_5736 | Putative zinc-knuckle transcription factor | UvCCHC5 |
| Uv8b_5874 | Zinc-finger domain-containing protein | UvCCHC6 |
| Uv8b_8085 | Zinc-knuckle domain-containing protein | UvCCHC7 |
| Strain | Description | Reference |
|---|---|---|
| HWD-2 | Wild-type strain | [35] |
| ΔUvCCHC1-5 | UvCCHC1 deletion mutant of HWD-2 | This study |
| ΔUvCCHC1-73 | UvCCHC1 deletion mutant of HWD-2 | This study |
| CΔUvCCHC1-5 | UvCCHC1 complementation strain from ΔUvCCHC1-5 | This study |
| ΔUvCCHC2-36 | UvCCHC2 deletion mutant of HWD-2 | This study |
| ΔUvCCHC2-51 | UvCCHC2 deletion mutant of HWD-2 | This study |
| CΔUvCCHC2-36 | UvCCHC3 complementation strain from ΔUvCCHC2-36 | This study |
| ΔUvCCHC3-16 | UvCCHC3 deletion mutant of HWD-2 | This study |
| ΔUvCCHC3-37 | UvCCHC3 deletion mutant of HWD-2 | This study |
| CΔUvCCHC3-16 | UvCCHC3 complementation strain from ΔUvCCHC3-16 | This study |
| ΔUvCCHC4-1 | UvCCHC4 deletion mutant of HWD-2 | This study |
| ΔUvCCHC4-11 | UvCCHC4 deletion mutant of HWD-2 | This study |
| CΔUvCCHC4-1 | UvCCHC4 complementation strain from ΔUvCCHC4-1 | This study |
| ΔUvCCHC5-23 | UvCCHC5 deletion mutant of HWD-2 | This study |
| ΔUvCCHC5-43 | UvCCHC5 deletion mutant of HWD-2 | This study |
| CΔUvCCHC5-23 | UvCCHC5 complementation strain from ΔUvCCHC5-23 | This study |
| ΔUvCCHC6-8 | UvCCHC6 deletion mutant of HWD-2 | This study |
| ΔUvCCHC6-22 | UvCCHC6 deletion mutant of HWD-2 | This study |
| CΔUvCCHC6-8 | UvCCHC6 complementation strain from ΔUvCCHC6-8 | This study |
| ΔUvCCHC7-18 | UvCCHC7 deletion mutant of HWD-2 | This study |
| ΔUvCCHC7-63 | UvCCHC7 deletion mutant of HWD-2 | This study |
| CΔUvCCHC7-18 | UvCCHC7 complementation strain from ΔUvCCHC7-18 | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Pei, Z.; Peng, L.; Qin, Q.; Duan, Y.; Liu, H.; Chen, X.; Zheng, L.; Luo, C.; Huang, J. Genome-Wide Identification and Functional Characterization of CCHC-Type Zinc Finger Genes in Ustilaginoidea virens. J. Fungi 2021, 7, 947. https://doi.org/10.3390/jof7110947
Chen X, Pei Z, Peng L, Qin Q, Duan Y, Liu H, Chen X, Zheng L, Luo C, Huang J. Genome-Wide Identification and Functional Characterization of CCHC-Type Zinc Finger Genes in Ustilaginoidea virens. Journal of Fungi. 2021; 7(11):947. https://doi.org/10.3390/jof7110947
Chicago/Turabian StyleChen, Xiaoyang, Zhangxin Pei, Lin Peng, Qin Qin, Yuhang Duan, Hao Liu, Xiaolin Chen, Lu Zheng, Chaoxi Luo, and Junbin Huang. 2021. "Genome-Wide Identification and Functional Characterization of CCHC-Type Zinc Finger Genes in Ustilaginoidea virens" Journal of Fungi 7, no. 11: 947. https://doi.org/10.3390/jof7110947
APA StyleChen, X., Pei, Z., Peng, L., Qin, Q., Duan, Y., Liu, H., Chen, X., Zheng, L., Luo, C., & Huang, J. (2021). Genome-Wide Identification and Functional Characterization of CCHC-Type Zinc Finger Genes in Ustilaginoidea virens. Journal of Fungi, 7(11), 947. https://doi.org/10.3390/jof7110947







