Breathomics in Asthmatic Children Treated with Inhaled Corticosteroids
Abstract
:1. Introduction
2. Results
2.1. Study Population
2.2. Symptoms Control and Lung Function
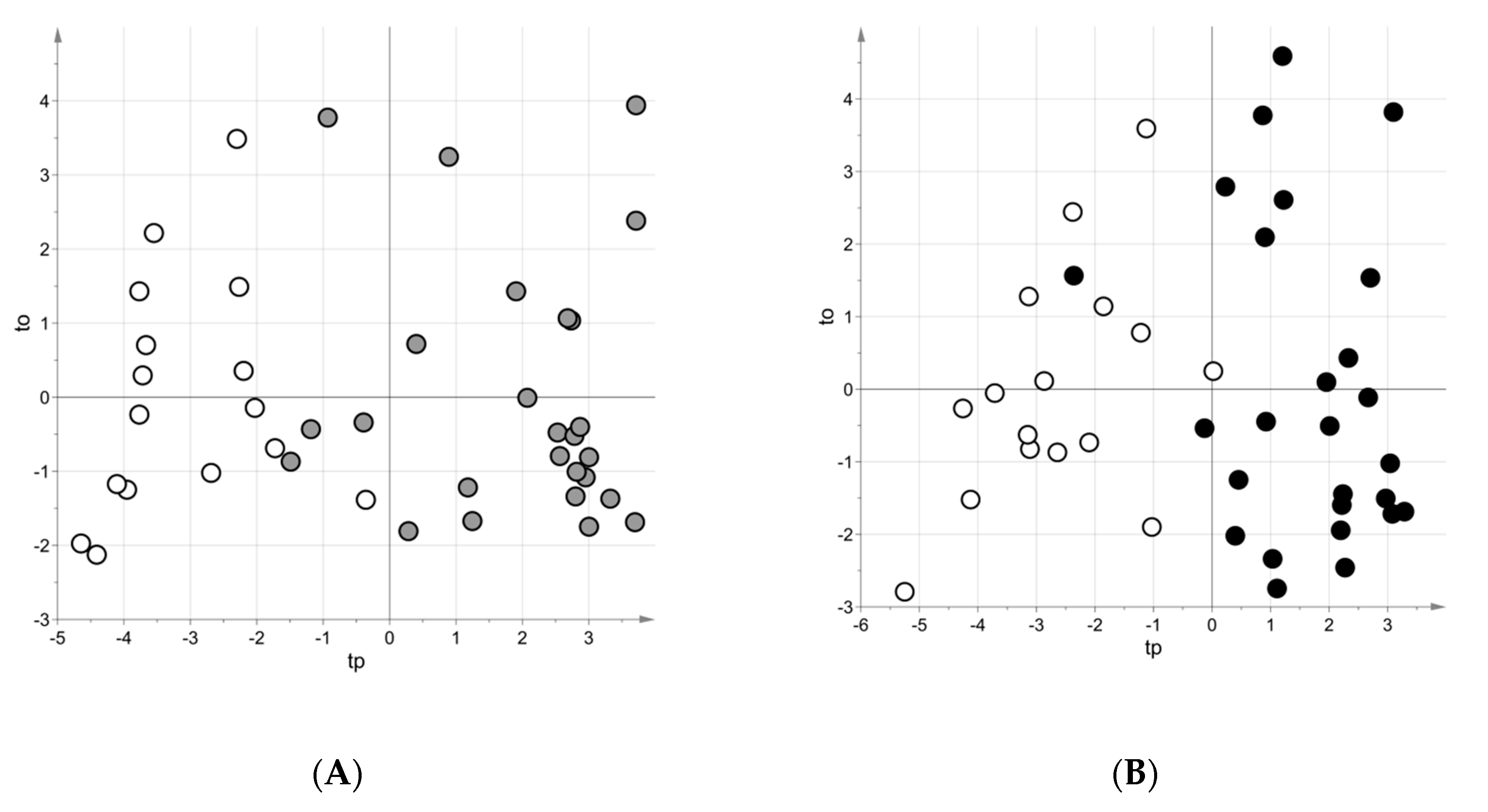
2.3. Metabolomics Analysis
2.3.1. Asthmatic Group at Recruitment (T0) vs. Controls
2.3.2. Asthmatic Group after Three Weeks of Treatment (T1) vs. Controls
2.3.3. Asthmatic Group at Recruitment (T0) vs. Asthmatic Group after Three Weeks of Treatment (T1)
2.3.4. Putative Markers Annotation
2.3.5. Target Metabolomics Analysis: Urinary Steroids
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Study Design
4.3. Metabolomic Analysis
4.3.1. EBC and Urine Collection
4.3.2. UPLC-MS (Ultra-Performance Liquid Chromatography-Mass Spectrometer) Analysis of EBC
4.3.3. Analysis of Urinary Steroids and BDP
4.3.4. Data Preprocessing
4.3.5. Statistical Data Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
UPLC-MS Analysis of EBC
| Time (min) | Flow (mL/min) | % A | % B |
|---|---|---|---|
| initial | 0.2 | 90 | 10 |
| 1.00 | 0.2 | 90 | 10 |
| 3.50 | 0.2 | 70 | 30 |
| 6.50 | 0.2 | 5 | 95 |
| 8.00 | 0.2 | 5 | 95 |
| 11.00 | 0.2 | 90 | 10 |
| Time (min) | Flow mL/min | % A | % B |
|---|---|---|---|
| Initial | 0.500 | 98 | 2 |
| 0.50 | 0.500 | 98 | 2 |
| 1.50 | 0.500 | 0.0 | 100 |
| 2.00 | 0.500 | 0.0 | 100 |
| 3.00 | 0.500 | 0.0 | 100 |
| 5.00 | 0.500 | 98 | 2 |
| 10.00 | 0.200 | 98 | 2 |
| 10.01 | 0.200 | 98 | 2 |
| 11.00 | 0.500 | 98 | 2 |
Appendix B
Analysis of Urinary Steroids and BDP
References
- Mokhallati, N.; Guilbert, T.W. Moving towards precision care for childhood asthma. Curr. Opin. Pediatr. 2016, 28, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Pajor, N.M.; Guilbert, T.W. Personalized Medicine and Pediatric Asthma. Immunol. Allergy Clin. N. Am. 2019, 39, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Licari, A.; Manti, S.; Castagnoli, R.; Marseglia, A.; Foiadelli, T.; Brambilla, I.; Marseglia, G.L. Immunomodulation in Pediatric Asthma. Front. Pediatr. 2019, 7, 289. [Google Scholar] [CrossRef] [PubMed]
- Grunwell, J.R.; Stephenson, S.T.; Tirouvanziam, R.; Brown, L.A.S.; Brown, M.R.; Fitzpatrick, A.M. Children with Neutrophil-Predominant Severe Asthma Have Proinflammatory Neutrophils with Enhanced Survival and Impaired Clearance. J. Allergy Clin. Immunol. Pract. 2019, 7, 516–525.e6. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, C.; Kucuksezer, U.C.; Akdis, M.; Akdis, C.A. The concepts of asthma endotypes and phenotypes to guide current and novel treatment strategies. Expert Rev. Respir. Med. 2018, 12, 733–743. [Google Scholar] [CrossRef]
- Ramadan, A.A.; Gaffin, J.M.; Israel, E.; Phipatanakul, W. Asthma and Corticosteroid Responses in Childhood and Adult Asthma. Clin. Chest Med. 2019, 40, 163–177. [Google Scholar] [CrossRef]
- Du, W.; Zhou, L.; Ni, Y.; Yu, Y.; Wu, F.; Shi, G. Inhaled corticosteroids improve lung function, airway hyper-responsiveness and airway inflammation but not symptom control in patients with mild intermittent asthma: A meta-analysis. Exp. Ther. Med. 2017, 14, 1594–1608. [Google Scholar] [CrossRef] [Green Version]
- Cowan, D.C.; Taylor, D.R.; Peterson, L.E.; Cowan, J.O.; Palmay, R.; Williamson, A.; Hammel, J.; Erzurum, S.C.; Hazen, S.L.; Comhair, S.A. Biomarker-based asthma phenotypes of corticosteroid response. J. Allergy Clin. Immunol. 2015, 135, 877–883.e1. [Google Scholar] [CrossRef] [Green Version]
- Potaczek, D.P.; Miethe, S.; Schindler, V.; Alhamdan, F.; Garn, H. Role of airway epithelial cells in the development of different asthma phenotypes. Cell. Signal. 2020, 69, 109523. [Google Scholar] [CrossRef]
- Tyler, S.R.; Bunyavanich, S. Leveraging-omics for asthma endotyping. J. Allergy Clin. Immunol. 2019, 144, 13–23. [Google Scholar] [CrossRef]
- Carraro, S.; Giordano, G.; Reniero, F.; Perilongo, G.; Baraldi, E. Metabolomics: A new frontier for research in pediatrics. J. Pediatr. 2009, 154, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Patti, G.J.; Yanes, O.; Siuzdak, G. Innovation: Metabolomics: The apogee of the omics trilogy. Nat. Rev. Mol. Cell Biol. 2012, 13, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Bardanzellu, F.; Fanos, V. How could metabolomics change pediatric health? Ital. J. Pediatr. 2020, 46, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelly, R.S.; Dahlin, A.; McGeachie, M.J.; Qiu, W.; Sordillo, J.; Wan, E.S.; Wu, A.C.; Lasky-Su, J. Asthma Metabolomics and the Potential for Integrative Omics in Research and the Clinic. Chest 2017, 151, 262–277. [Google Scholar] [CrossRef] [Green Version]
- Golebski, K.; Kabesch, M.; Melén, E.; Potočnik, U.; Van Drunen, C.M.; Reinarts, S.; Der Zee, A.H.M.-V.; Vijverberg, S.J. Childhood asthma in the new omics era: Challenges and perspectives. Curr. Opin. Allergy Clin. Immunol. 2020, 20, 155–161. [Google Scholar] [CrossRef]
- Zhu, Z.; Camargo, C.A.; Hasegawa, K. Metabolomics in the prevention and management of asthma. Expert Rev. Respir. Med. 2019, 13, 1135–1138. [Google Scholar] [CrossRef] [Green Version]
- Brinkman, P.; Ahmed, W.M.; Gómez, C.; Knobel, H.H.; Weda, H.; Vink, T.J.; Nijsen, T.M.; Wheelock, C.E.; Dahlen, S.-E.; Montuschi, P.; et al. Exhaled volatile organic compounds as markers for medication use in asthma. Eur. Respir. J. 2020, 55, 1900544. [Google Scholar] [CrossRef]
- Bannier, M.A.G.E.; Rosias, P.P.R.; Jöbsis, Q.; Dompeling, E. Exhaled Breath Condensate in Childhood Asthma: A Review and Current Perspective. Front. Pediatr. 2019, 7, 150. [Google Scholar] [CrossRef] [Green Version]
- Carraro, S.; Rezzi, S.; Reniero, F.; Héberger, K.; Giordano, G.; Zanconato, S.; Guillou, C.; Baraldi, E. Metabolomics applied to exhaled breath condensate in childhood asthma. Am. J. Respir. Crit. Care Med. 2007, 175, 986–990. [Google Scholar] [CrossRef]
- Stocchero, M. Relevant and irrelevant predictors in PLS2. J. Chemom. 2020. [Google Scholar] [CrossRef]
- Esther, C.R.; Boysen, G.; Olsen, B.M.; Collins, L.B.; Ghio, A.J.; Swenberg, J.W.; Boucher, R.C. Mass spectrometric analysis of biomarkers and dilution markers in exhaled breath condensate reveals elevated purines in asthma and cystic fibrosis. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2009, 296, L987–L993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montuschi, P. LC/MS/MS analysis of leukotriene B4 and other eicosanoids in exhaled breath condensate for assessing lung inflammation. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2009, 877, 1272–1280. [Google Scholar] [CrossRef] [PubMed]
- Carraro, S.; Giordano, G.; Reniero, F.; Stocchero, M.; Sterk, P.; Baraldi, E.; Carpi, D. Asthma severity in childhood and metabolomic profiling of breath condensate. Allergy 2013, 68, 110–117. [Google Scholar] [CrossRef]
- Claar, D.; Hartert, T.V.; Peebles, R.S. The role of prostaglandins in allergic lung inflammation and asthma. Expert Rev. Respir. Med. 2015, 9, 55–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, L.M.; Belvisi, M.G.; Bode, K.A.; Bauer, J.; Schmidt, C.; Suchy, M.-T.; Tsikas, D.; Scheuerer, J.; Lasitschka, F.; Gröne, H.-J.; et al. Bronchial epithelial cell-derived prostaglandin E2 dampens the reactivity of dendritic cells. J. Immunol. 2011, 186, 2095–2105. [Google Scholar] [CrossRef] [Green Version]
- Pavord, I.D.; Tattersfield, A.E. Bronchoprotective role for endogenous prostaglandin E2. Lancet Lond. Engl. 1995, 345, 436–438. [Google Scholar] [CrossRef]
- Belvisi, M.G. Regulation of inflammatory cell function by corticosteroids. Proc. Am. Thorac. Soc. 2004, 1, 207–214. [Google Scholar] [CrossRef] [Green Version]
- Reinke, S.N.; Gallart-Ayala, H.; Gómez, C.; Checa, A.; Fauland, A.; Naz, S.; Kamleh, M.A.; Djukanovic, R.; Hinks, T.S.; Wheelock, C.E. Metabolomics analysis identifies different metabotypes of asthma severity. Eur. Respir. J. 2017, 49, 1601740. [Google Scholar] [CrossRef] [Green Version]
- Wendell, S.G.; Baffi, C.; Holguin, F. Fatty acids, inflammation, and asthma. J. Allergy Clin. Immunol. 2014, 133, 1255–1264. [Google Scholar] [CrossRef] [Green Version]
- Harb, H.; Irvine, J.; Amarasekera, M.; Hii, C.S.; Kesper, D.A.; Ma, Y.F.; D’Vaz, N.; Renz, H.; Potaczek, D.P.; Prescott, S.L.; et al. The role of PKCζ in cord blood T-cell maturation towards Th1 cytokine profile and its epigenetic regulation by fish oil. Biosci. Rep. 2017, 37, BSR20160485. [Google Scholar] [CrossRef] [Green Version]
- Acevedo, N.; Frumento, P.; Harb, H.; Alashkar Alhamwe, B.; Johansson, C.; Eick, L.; Alm, J.; Renz, H.; Scheynius, A.; Potaczek, D.P. Histone Acetylation of Immune Regulatory Genes in Human Placenta in Association with Maternal Intake of Olive Oil and Fish Consumption. Int. J. Mol. Sci. 2019, 20, 1060. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ho, W.E.; Xu, Y.-J.; Cheng, C.; Peh, H.Y.; Tannenbaum, S.R.; Wong, W.F.; Ong, C.N. Metabolomics Reveals Inflammatory-Linked Pulmonary Metabolic Alterations in a Murine Model of House Dust Mite-Induced Allergic Asthma. J. Proteome Res. 2014, 13, 3771–3782. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Cui, F.-X.; Jia, H.-M.; Zhou, C.; Yang, Y.; Zhang, H.-W.; Ding, G.; Zou, Z.-M. Aberrant purine metabolism in allergic asthma revealed by plasma metabolomics. J. Pharm. Biomed. Anal. 2016, 120, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Yoder, M.; Zhuge, Y.; Yuan, Y.; Holian, O.; Kuo, S.; Van Breemen, R.B.; Thomas, L.L.; Lum, H. Bioactive lysophosphatidylcholine 16:0 and 18:0 are elevated in lungs of asthmatic subjects. Allergy Asthma Immunol. Res. 2014, 6, 61–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quinn, K.; Schedel, M.; Nkrumah-Elie, Y.; Joetham, A.; Armstrong, M.; Cruickshank-Quinn, C.I.; Reisdorph, N.; Gelfand, E.W. Dysregulation of metabolic pathways in a mouse model of allergic asthma. Allergy 2017, 72, 1327–1337. [Google Scholar] [CrossRef]
- Leigh, R.; Mostafa, M.M.; King, E.M.; Rider, C.F.; Shah, S.; Dumonceaux, C.; Traves, S.L.S.L.; McWhae, A.; Kolisnik, T.; Kooi, C.; et al. An inhaled dose of budesonide induces genes involved in transcription and signaling in the human airways: Enhancement of anti- and proinflammatory effector genes. Pharmacol. Res. Perspect. 2016, 4, e00243. [Google Scholar] [CrossRef]
- Pavord, I.D.; Beasley, R.; Agusti, A.; Anderson, G.P.; Bel, E.; Brusselle, G.; Cullinan, P.; Custovic, A.; Ducharme, F.M.; Fahy, J.V.; et al. After asthma: Redefining airways diseases. Lancet 2018, 391, 350–400. [Google Scholar] [CrossRef]
- Yeo, S.-H.; Aggarwal, B.; Shantakumar, S.; Mulgirigama, A.; Daley-Yates, P. Efficacy and safety of inhaled corticosteroids relative to fluticasone propionate: A systematic review of randomized controlled trials in asthma. Expert Rev. Respir. Med. 2017, 11, 763–778. [Google Scholar] [CrossRef]
- Amar, N.J.; Moss, M.H.; Kerwin, E.M.; Li, J.; Small, C.J. Safety and efficacy of beclomethasone dipropionate delivered by breath-actuated or metered-dose inhaler for persistent asthma. Allergy Asthma Proc. 2016, 37, 359–369. [Google Scholar] [CrossRef]
- Nave, R.; Fisher, R.; McCracken, N. In vitro metabolism of beclomethasone dipropionate, budesonide, ciclesonide, and fluticasone propionate in human lung precision-cut tissue slices. Respir. Res. 2007, 8, 65. [Google Scholar] [CrossRef] [Green Version]
- (*NEW) 2018 GINA Report: Global Strategy for Asthma Management and Prevention. Global Initiative for Asthma-GINA. Available online: https://ginasthma.org/2018-gina-report-global-strategy-for-asthma-management-and-prevention/ (accessed on 20 June 2018).
- Stanojevic, S. Standardisation of lung function test interpretation: Global Lung Function Initiative. Lancet Respir. Med. 2018, 6, 10–12. [Google Scholar] [CrossRef]
- Cooper, B.G.; Stocks, J.; Hall, G.L.; Culver, B.; Steenbruggen, I.; Carter, K.W.; Thompson, B.R.; Graham, B.L.; Miller, M.R.; Ruppel, G.; et al. The Global Lung Function Initiative (GLI) Network: Bringing the world’s respiratory reference values together. Breathe 2017, 13, e56–e64. [Google Scholar] [CrossRef] [PubMed]
- Horváth, I.; Barnes, P.J.; Loukides, S.; Sterk, P.J.; Högman, M.; Olin, A.-C.; Amann, A.; Antus, B.; Baraldi, E.; Bikov, A.; et al. A European Respiratory Society technical standard: Exhaled biomarkers in lung disease. Eur. Respir. J. 2017, 49, 1600965. [Google Scholar] [CrossRef] [Green Version]
- Benjamini, Y. Discovering the False Discovery Rate. J. R. Stat. Soc. Ser. B Stat. Methodol. 2010, 72, 405–416. Available online: https://rss.onlinelibrary.wiley.com/doi/full/10.1111/j.1467-9868.2010.00746.x (accessed on 24 April 2020).
- Stocchero, M.; Locci, E.; d’Aloja, E.; Nioi, M.; Baraldi, E.; Giordano, G. PLS2 in Metabolomics. Metabolites 2019, 9, 51. [Google Scholar] [CrossRef] [Green Version]
- Westerhuis, J.A.; van Velzen, E.J.J.; Hoefsloot, H.C.J.; Smilde, A.K. Multivariate paired data analysis: Multilevel PLSDA versus OPLSDA. Metabolomics 2010, 6, 119–128. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Heading | Asthmatic Children | Healthy Children | p Value | ||
|---|---|---|---|---|---|
| Age (year) | 9.1 (6.5, 13.4) | 10.2 (6.2,14.4) | 0.16 | ||
| Sex (male/female) | 20/6 | 11/5 | 0.72 | ||
| BMI | 17.0 (14.2, 21.7) | 17.9 (14.3, 25.0) | 0.18 | ||
| Allergic (yes/no) | 23/3 | 0/16 | <0.001 | ||
| T0 | T1 | ||||
| FEV1 | Z-score % predicted | −0.59 (−2.54, +1.38) 92.87 (69.19, 116.55) | −0.35 (−2.07, +1.37) 95.69 (75.73, 115.65) | −0.16 (−1.54, +1.22) 97.99 (81.67, 114.31) | 0.07 a 0.23 b 0.01 c |
| FEF 25–75 | Z-score % predicted | −0.22 (−2.06, +1.62) 95.71 (52.27, 139.15) | −0.10 (−2.08, +1.88) 98.52 (50.76, 146.28) | 0.33 (1.23, 1.89) 108.49 (70.75, 146.23) | 0.02 a 0.07 b 0.08 c |
| m/z | Retention Time (min) | Adduct | Annotation | Class |
|---|---|---|---|---|
| 172.1332 | 4.42 | M − H | 9-amino-nonanoic acid | omega-amino fatty acids |
| 198.1864 | 4.92 | M + H − H2O | 12-amino-dodecanoic acid | omega-amino fatty acids |
| 295.1543 | 5.53 | M − H | lactone of PGF-MUM | prostaglandins |
| 371.2280 | 2.84 | M + H − NH3, M + H | N-linoleoyl taurine | N-acyl amides |
| 415.2546 | 3.12 | M + H − NH3, M + H | 17-phenoxy trinor PGF2α ethyl amide | prostaglandins |
| 520.3342 | 3.58 | M + H − NH3, M + H | lysoPC (18:2(9Z,12Z)) | mono glycerophospholipids |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferraro, V.A.; Carraro, S.; Pirillo, P.; Gucciardi, A.; Poloniato, G.; Stocchero, M.; Giordano, G.; Zanconato, S.; Baraldi, E. Breathomics in Asthmatic Children Treated with Inhaled Corticosteroids. Metabolites 2020, 10, 390. https://doi.org/10.3390/metabo10100390
Ferraro VA, Carraro S, Pirillo P, Gucciardi A, Poloniato G, Stocchero M, Giordano G, Zanconato S, Baraldi E. Breathomics in Asthmatic Children Treated with Inhaled Corticosteroids. Metabolites. 2020; 10(10):390. https://doi.org/10.3390/metabo10100390
Chicago/Turabian StyleFerraro, Valentina Agnese, Silvia Carraro, Paola Pirillo, Antonina Gucciardi, Gabriele Poloniato, Matteo Stocchero, Giuseppe Giordano, Stefania Zanconato, and Eugenio Baraldi. 2020. "Breathomics in Asthmatic Children Treated with Inhaled Corticosteroids" Metabolites 10, no. 10: 390. https://doi.org/10.3390/metabo10100390





