Preventing the Increase in Lysophosphatidic Acids: A New Therapeutic Target in Pulmonary Hypertension?
Abstract
:1. Introduction
2. Results
2.1. Angiotensin-II Induced Hypertension (Ang-II HTN)
2.2. Chronic Heart Failure (CHF)
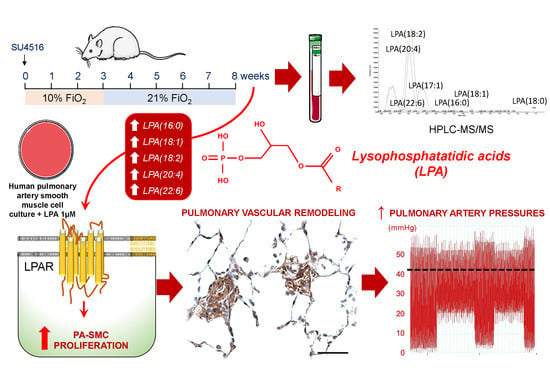
2.3. Pulmonary Hypertension (PH) Induced by Sugen/Hypoxia (SuHx)
2.4. Impact of LPA on Pulmonary Artery Smooth Muscle Cell (PA-SMC) Proliferation
2.5. Relationship between LPA Levels and Index of PH in Patients with Cardiac Dysfunction
3. Discussion
3.1. LPL, LPA and MAG Quantitation
3.2. CVD Models
3.3. LPA Signaling
4. Materials and Methods
4.1. Experimentation
4.1.1. Chemicals
4.1.2. Animals
4.2. LPL, LPA and MAG Quantitation
4.2.1. Blood Sampling and Processing
4.2.2. High Performance Liquid Chromatography (HPLC)
4.2.3. Tandem Mass Spectrometry (MS2)
4.3. Murine Models of Cardiovascular Diseases (CVD)
4.3.1. Angiotensin-II Induced Hypertension (HTN)
4.3.2. Chronic Heart Failure (CHF) Induced by Coronary Artery Ligation
4.3.3. Pulmonary Hypertension (PH) Induced by Sugen Hypoxia (SuHx)
4.4. Human PA-SMC Proliferation
4.5. Chronic Heart Failure Patients
4.6. Data and Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef] [PubMed]
- Soppert, J.; Lehrke, M.; Marx, N.; Jankowski, J.; Noels, H. Lipoproteins and lipids in cardiovascular disease: From mechanistic insights to therapeutic targeting. Adv. Drug Deliv. Rev. 2020, 159, 4–33. [Google Scholar] [CrossRef]
- Eilat-Tsanani, S.; Mor, E.; Schonmann, Y. Statin Use Over 65 Years of Age and All-Cause Mortality: A 10-Year Follow-Up of 19 518 People. J. Am. Geriatr. Soc. 2019, 67, 2038–2044. [Google Scholar] [CrossRef]
- McKenney, J.M. Optimizing LDL-C Lowering With Statins. Am. J. Ther. 2004, 11, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, R.; Garg, J.; Shah, N.; Sumner, A. PCSK9 inhibitors: A new era of lipid lowering therapy. World J. Cardiol. 2017, 9, 76–91. [Google Scholar] [CrossRef] [PubMed]
- Yeang, C.; Wilkinson, M.J.; Tsimikas, S. Lipoprotein(a) and oxidized phospholipids in calcific aortic valve stenosis. Curr. Opin. Cardiol. 2016, 31, 440–450. [Google Scholar] [CrossRef] [Green Version]
- Kotani, K.; Yamada, S.; Yamada, T.; Taniguchi, N.; Sakurabayashi, I. The relationship between oxidized lipoprotein(a) and carotid atherosclerosis in asymptomatic subjects: A comparison with native lipoprotein(a). Lipids Health Dis. 2011, 10, 174. [Google Scholar] [CrossRef] [Green Version]
- Parthasarathy, S.; Raghavamenon, A.; Garelnabi, M.O.; Santanam, N. Oxidized Low-Density Lipoprotein. Methods Mol Biol. 2009, 610, 403–417. [Google Scholar] [CrossRef] [Green Version]
- Oei, H.-H.S.; van der Meer, I.M.; Hofman, A.; Koudstaal, P.J.; Stijnen, T.; Breteler, M.M.; Witteman, J.C. Lipoprotein-Associated Phospholipase A2 Activity Is Associated with Risk of Coronary Heart Disease and Ischemic Stroke: The Rotterdam Study. Circulation 2005, 111, 570–575. [Google Scholar] [CrossRef] [Green Version]
- Colley, K.J.; Wolfert, R.L.; Cobble, M.E. Lipoprotein associated phospholipase A2: Role in atherosclerosis and utility as a biomarker for cardiovascular risk. EPMA J. 2011, 2, 27–38. [Google Scholar] [CrossRef] [Green Version]
- Gianazza, E.; Brioschi, M.; Fernandez, A.E.M.; Banfi, C. Lipoxidation in cardiovascular diseases. Redox Biol. 2019, 23, 101119. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Little, P.J.; Ta, H.T.; Xu, S.; Kamato, D. Lysophosphatidic acid and its receptors: Pharmacology and therapeutic potential in atherosclerosis and vascular disease. Pharmacol. Ther. 2019, 204, 107404. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Latif, A.; Heron, P.M.; Morris, A.J.; Smyth, S.S. Lysophospholipids in coronary artery and chronic ischemic heart disease. Curr. Opin. Lipidol. 2015, 26, 432–437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kleger, A.; Liebau, S.; Lin, Q.; von Wichert, G.; Seufferlein, T. The Impact of Bioactive Lipids on Cardiovascular Development. Stem Cells Int. 2011, 2011, 916180. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.; Goetzl, E.J.; An, S. Lysophosphatidic acid and sphingosine 1-phosphate stimulate endothelial cell wound healing. Am. J. Physiol. Cell Physiol. 2000, 278, C612–C618. [Google Scholar] [CrossRef] [PubMed]
- Moolenaar, W.H. Lysophosphatidic Acid, a Multifunctional Phospholipid Messenger. J. Biol. Chem. 1995, 270, 12949–12952. [Google Scholar] [CrossRef] [Green Version]
- Smyth, S.S.; Cheng, H.-Y.; Miriyala, S.; Panchatcharam, M.; Morris, A.J. Roles of lysophosphatidic acid in cardiovascular physiology and disease. Biochim. Biophys. Acta 2008, 1781, 563–570. [Google Scholar] [CrossRef] [Green Version]
- Yang, F.; Chen, G.-X. Production of extracellular lysophosphatidic acid in the regulation of adipocyte functions and liver fibrosis. World J. Gastroenterol. 2018, 24, 4132–4151. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Roberts, L.D.; Souza, A.L.; Gerszten, R.E.; Clish, C.B. Targeted Metabolomics. Curr. Protoc. Mol. Biol. 2012, 98, 30.2.1–30.2.24. [Google Scholar] [CrossRef]
- Triebl, A.; Trötzmüller, M.; Eberl, A.; Hanel, P.; Hartler, J.; Köfeler, H. Quantitation of phosphatidic acid and lysophosphatidic acid molecular species using hydrophilic interaction liquid chromatography coupled to electrospray ionization high resolution mass spectrometry. J. Chromatogr. A 2014, 1347, 104–110. [Google Scholar] [CrossRef]
- Colín-Santana, C.C.; Avendaño-Vázquez, S.E.; Alcántara-Hernández, R.; García-Sáinz, J.A. EGF and angiotensin II modulate lysophosphatidic acid LPA1 receptor function and phosphorylation state. Biochim. Biophys. Acta 2011, 1810, 1170–1177. [Google Scholar] [CrossRef] [PubMed]
- De Souza, A.M.; de Carvalho, T.L.G.; Lara, L.D.S.; Gomes-Quintana, E.; Gil Lopes, A.; Caruso-Neves, C. The stimulatory effect of angiotensin II on Na+-ATPase activity involves sequential activation of phospholipases and sustained PKC activity. Biochim. Biophys. Acta 2010, 1798, 354–359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Booz, G.W.; Taher, M.M.; Baker, K.M.; Singer, H.A. Angiotensin II induces phosphatidic acid formation in neonatal rat cardiac fibroblasts: Evaluation of the roles of phospholipases C and D. Mol. Cell. Biochem. 1994, 141, 135–143. [Google Scholar] [CrossRef]
- Wolf, G.; Wenzel, U.O. Angiotensin II and Cell Cycle Regulation. Hypertension 2004, 43, 693–698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsutsui, H.; Kinugawa, S.; Matsushima, S. Oxidative stress and heart failure. Am. J. Physiol. Heart Circ. Physiol. 2011, 301, H2181–H2190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Idkowiak-Baldys, J.; Apraiz, A.; Li, L.; Rahmaniyan, M.; Clarke, C.J.; Kraveka, J.M.; Asumendi, A.; Hannun, Y.A. Dihydroceramide desaturase activity is modulated by oxidative stress. Biochem. J. 2010, 427, 265–274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Briggs, M.A.; Petersen, K.S.; Kris-Etherton, P.M. Saturated Fatty Acids and Cardiovascular Disease: Replacements for Saturated Fat to Reduce Cardiovascular Risk. Healthcare 2017, 5, 29. [Google Scholar] [CrossRef] [Green Version]
- Mensink, R.P.; World Health Organization. Effects of Saturated Fatty Acids on Serum Lipids and Lipoproteins: A Systematic Review and Regression Analysis; World Health Organization: Geneva, Switzerland, 2016; Available online: https://apps.who.int/iris/handle/10665/246104 (accessed on 25 August 2021).
- Beam, J.; Botta, A.; Barendregt, R.; Ghosh, S. Dietary Fatty Acids, Redox Signaling, and the Heart. Syst. Biol. Free Radic. Antioxid. 2014, 1497–1522. [Google Scholar] [CrossRef]
- Gouaref, I.; Bouazza, A.; Abderrhmane, S.A.; Koceir, E.-A. Lipid Profile Modulates Cardiometabolic Risk Biomarkers Including Hypertension in People with Type-2 Diabetes: A Focus on Unbalanced Ratio of Plasma Polyunsaturated/Saturated Fatty Acids. Molecules 2020, 25, 4315. [Google Scholar] [CrossRef]
- Michalczyk, A.; Budkowska, M.; Dołęgowska, B.; Chlubek, D.; Safranow, K. Lysophosphatidic acid plasma concentrations in healthy subjects: Circadian rhythm and associations with demographic, anthropometric and biochemical parameters. Lipids Health Dis. 2017, 16, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Eddahibi, S.; Humbert, M.; Fadel, E.; Raffestin, B.; Darmon, M.; Capron, F.; Simonneau, G.; Dartevelle, P.; Hamon, M.; Adnot, S. Serotonin transporter overexpression is responsible for pulmonary artery smooth muscle hyperplasia in primary pulmonary hypertension. J. Clin. Investig. 2001, 108, 1141–1150. [Google Scholar] [CrossRef]
- Bordenave, J.; Thuillet, R.; Tu, L.; Phan, C.; Cumont, A.; Marsol, C.; Huertas, A.; Savale, L.; Hibert, M.; Galzi, J.-L.; et al. Neutralization of CXCL12 attenuates established pulmonary hypertension in rats. Cardiovasc. Res. 2019, 116, 686–697. [Google Scholar] [CrossRef] [PubMed]
- Morrell, N.W.; Adnot, S.; Archer, S.L.; Dupuis, J.; Lloyd Jones, P.; MacLean, M.R.; McMurtry, I.F.; Stenmark, K.R.; Thistlethwaite, P.A.; Weissmann, N.; et al. Cellular and Molecular Basis of Pulmonary Arterial Hypertension. J. Am. Coll. Cardiol. 2009, 54 (Suppl. 1), S20–S31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Y.; Bei, Y.; Shen, S.; Zhang, J.; Lu, Y.; Xiao, J.; Li, X. MicroRNA-222 Promotes the Proliferation of Pulmonary Arterial Smooth Muscle Cells by Targeting P27 and TIMP3. Cell. Physiol. Biochem. 2017, 43, 282–292. [Google Scholar] [CrossRef]
- Shlyonsky, V.; Naeije, R.; Mies, F. Possible Role of Lysophosphatidic Acid in Rat Model of Hypoxic Pulmonary Vascular Remodeling. Pulm. Circ. 2014, 4, 471–481. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.-J.; No, Y.R.; Dang, D.T.; Dang, L.H.; Yang, V.W.; Shim, H.; Yun, C.C. Regulation of Hypoxia-inducible Factor 1α (HIF-1α) by Lysophosphatidic Acid Is Dependent on Interplay between p53 and Krüppel-like Factor 5. J. Biol. Chem. 2013, 288, 25244–25253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ziello, J.E.; Jovin, I.S.; Huang, Y. Hypoxia-Inducible Factor (HIF)-1 regulatory pathway and its potential for therapeutic interven-tion in malignancy and ischemia. Yale J. Biol. Med. 2007, 80, 51–60. [Google Scholar]
- Cheng, H.-Y.; Dong, A.; Panchatcharam, M.; Mueller, P.; Yang, F.; Li, Z.; Mills, G.; Chun, J.; Morris, A.J.; Smyth, S.S. Lysophosphatidic Acid Signaling Protects Pulmonary Vasculature From Hypoxia-Induced Remodeling. Arter. Thromb. Vasc. Biol. 2012, 32, 24–32. [Google Scholar] [CrossRef] [Green Version]
- He, D.; Su, Y.; Usatyuk, P.V.; Spannhake, E.W.; Kogut, P.; Solway, J.; Natarajan, V.; Zhao, Y. Lysophosphatidic Acid Enhances Pulmonary Epithelial Barrier Integrity and Protects Endotoxin-induced Epithelial Barrier Disruption and Lung Injury. J. Biol. Chem. 2009, 284, 24123–24132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, C.; Liu, P.-P.; Tang, D.-D.; Song, R.; Zhang, Y.-Q.; Lei, S.; Wu, S.-J. Targeting the RhoA-ROCK pathway to regulate T-cell homeostasis in hypoxia-induced pulmonary arterial hypertension. Pulm. Pharmacol. Ther. 2018, 50, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Tager, A.M.; LaCamera, P.; Shea, B.S.; Campanella, G.S.; Selman, M.; Zhao, Z.; Polosukhin, V.; Wain, J.; Karimi-Shah, B.A.; Kim, N.D.; et al. The lysophosphatidic acid receptor LPA1 links pulmonary fibrosis to lung injury by mediating fibroblast recruitment and vascular leak. Nat. Med. 2007, 14, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.; Zhao, Y.; Feng, R.; Liu, Y.; Wang, S.; Wei, W.; Ding, Q.; An, M.; Wen, J.; Li, L. Lysophosphatidic acid accelerates lung fibrosis by inducing differentiation of mesenchymal stem cells into myofibroblasts. J. Cell. Mol. Med. 2013, 18, 156–169. [Google Scholar] [CrossRef]
- Kritikou, E.; Van Puijvelde, G.H.M.; Van Der Heijden, T.; Van Santbrink, P.J.; Swart, M.; Schaftenaar, F.H.; Kröner, M.J.; Kuiper, J.; Bot, I. Inhibition of lysophosphatidic acid receptors 1 and 3 attenuates atherosclerosis development in LDL-receptor deficient mice. Sci. Rep. 2016, 6, 37585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cui, M.-Z. Lysophosphatidic acid effects on atherosclerosis and thrombosis. Clin. Lipidol. 2011, 6, 413–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kano, K.; Matsumoto, H.; Inoue, A.; Yukiura, H.; Kanai, M.; Chun, J.; Ishii, S.; Shimizu, T.; Aoki, J. Molecular mechanism of lysophosphatidic acid-induced hypertensive response. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Bandoh, K.; Aoki, J.; Taira, A.; Tsujimoto, M.; Arai, H.; Inoue, K. Lysophosphatidic acid (LPA) receptors of the EDG family are differentially activated by LPA species. Structure-activity relationship of cloned LPA receptors. FEBS Lett. 2000, 478, 159–165. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; He, D.; Su, Y.; Berdyshev, E.; Chun, J.; Natarajan, V.; Zhao, Y. Lysophosphatidic acid receptor 1 modulates lipopolysaccharide-induced inflammation in alveolar epithelial cells and murine lungs. Am. J. Physiol. Lung Cell Mol. Physiol. 2011, 301, L547–L556. [Google Scholar] [CrossRef]
- Huang, L.S.; Fu, P.; Patel, P.; Harijith, A.; Sun, T.; Zhao, Y.; Garcia, J.G.N.; Chun, J.; Natarajan, V. Lysophosphatidic Acid Receptor–2 Deficiency Confers Protection against Bleomycin-Induced Lung Injury and Fibrosis in Mice. Am. J. Respir. Cell Mol. Biol. 2013, 49, 912–922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Merabet, N.; Bellien, J.; Glevarec, E.; Nicol, L.; Lucas, D.; Remy-Jouet, I.; Bounoure, F.; Dreano, Y.; Wecker, D.; Thuillez, C.; et al. Soluble epoxide hydrolase inhibition improves myocardial perfusion and function in experimental heart failure. J. Mol. Cell. Cardiol. 2012, 52, 660–666. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 25 August 2021).
- Signorell, A.; Aho, A.; Alfons, A.; Anderegg, N.; Aragon, T.; Arachchige, C.; Arppe, A.; Baddeley, A.; Barton, K.; Bolker, B.; et al. DescTools: Tools for Descriptive Statistics. R Package Version 0.99.42. 2021. Available online: https://cran.r-project.org/package=DescTools (accessed on 25 August 2021).
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. Available online: https://ggplot2.tidyverse.org (accessed on 25 August 2021).
- Xiao, N. Ggsci: Scientific Journal and Sci-Fi Themed Color Palettes for ‘ggplot2′. R Package Version 2.9. 2018. Available online: https://CRAN.R-project.org/package=ggsci (accessed on 25 August 2021).
- Kassambara, A. Ggpubr: ’Ggplot2′ Based Publication Ready Plots. R Package Version 0.3.0. 2020. Available online: https://CRAN.R-project.org/package=ggpubr (accessed on 25 August 2021).







| Analyte | Control Group (N = 8) | HTN Group (N = 8) | p-Value | Adjusted p-Value a |
|---|---|---|---|---|
| LPA(16:0) | 0.11 [0.11–0.12] | 0.15 [0.13–0.22] | 0.0229 | 0.241 |
| LPA(18:0) b | 0.09 [0.08–0.10] | 0.11 [0.10–0.14] | 0.0832 | 0.341 |
| LPA(18:1) | 0.09 [0.09–0.10] | 0.13 [0.10–0.14] | 0.0438 | 0.241 |
| LPA(18:2) b | 1.78 [1.57–1.97] | 2.20 [1.56–2.46] | 0.163 | 0.398 |
| LPA(20:4) b | 1.91 [1.50–2.13] | 2.56 [2.14–2.92] | 0.0308 | 0.241 |
| LPA(22:6) b | 0.10 [0.08–0.11] | 0.14 [0.11–0.17] | 0.0931 | 0.341 |
| LPC(16:0) | 16.7 [16.2–17.4] | 17.3 [15.0–18.0] | 0.568 | 0.781 |
| LPC(16:1) b | 1.24 [1.10–1.40] | 1.58 [1.40–1.77] | 0.0438 | 0.241 |
| LPC(18:0) b | 29.7 [27.3–32.9] | 30.4 [28.7–31.7] | 0.914 | 0.914 |
| LPC(18:1) | 7.24 [6.36–7.53] | 7.22 [6.83–8.14] | 0.343 | 0.629 |
| LPC(18:2) b | 31.5 [28.5–32.6] | 30.9 [28.2–32.0] | 0.749 | 0.867 |
| LPC(20:4) b | 11.1 [10.3–12.1] | 12.6 [11.6–13.7] | 0.148 | 0.398 |
| LPC(22:6) b | 0.34 [0.31–0.43] | 0.43 [0.38–0.46] | 0.269 | 0.538 |
| LPE(16:0) | 0.54 [0.47–0.61] | 0.62 [0.58–0.68] | 0.118 | 0.371 |
| LPE(18:0) b | 0.80 [0.76–0.93] | 0.95 [0.89–1.00] | 0.205 | 0.451 |
| LPE(18:1) b | 0.22 [0.21–0.27] | 0.25 [0.22–0.27] | 0.376 | 0.636 |
| LPE(18:2) b | 0.45 [0.44–0.52] | 0.46 [0.41–0.49] | 0.439 | 0.690 |
| LPE(20:4) b | 0.16 [0.15–0.17] | 0.17 [0.16–0.20] | 0.836 | 0.876 |
| LPE(22:6) b | 0.11 [0.09–0.12] | 0.12 [0.10–0.15] | 0.612 | 0.792 |
| MAG(18:1) | 0.13 [0.10–0.16] | 0.12 [0.11–0.15] | 0.805 | 0.876 |
| MAG(18:2) b | 0.67 [0.57–1.00] | 0.64 [0.58–0.79] | 0.673 | 0.823 |
| MAG(20:4) b | 0.02 [0.02–0.03] | 0.02 [0.01–0.02] | 0.508 | 0.745 |
| Analyte | Control Group (N = 8) | CHF Group (N = 9) | p-Value | Adjusted p-Value a |
|---|---|---|---|---|
| LPA(16:0) | 0.058 [0.046–0.060] | 0.082 [0.056–0.103] | 0.025 | 0.186 |
| LPA(18:0) b | 0.050 [0.048–0.057] | 0.070 [0.065–0.080] | 0.336 | 0.615 |
| LPA(18:1) | 0.069 [0.063–0.076] | 0.067 [0.054–0.075] | 0.757 | 0.779 |
| LPA(18:2) b | 0.87 [0.77–0.95] | 1.00 [0.88–1.06] | 0.193 | 0.499 |
| LPA(20:4) b | 1.24 [1.04–1.31] | 1.21 [1.14–1.56] | 0.204 | 0.499 |
| LPA(22:6) b | 0.066 [0.062–0.070] | 0.068 [0.061–0.084] | 0.447 | 0.615 |
| LPC(16:0) | 13.0 [11.3–14.2] | 12.3 [11.1–13.2] | 0.302 | 0.615 |
| LPC(16:1) b | 1.06 [0.98–1.37] | 0.93 [0.71–1.60] | 0.750 | 0.779 |
| LPC(18:0) b | 19.4 [15.7–20.5] | 18.7 [17.1–21.4] | 0.608 | 0.743 |
| LPC(18:1) | 5.02 [4.61–5.64] | 3.97 [3.3–4.48] | 0.016 | 0.186 |
| LPC(18:2) b | 19.7 [18.7–21.6] | 19.4 [17.1–20.5] | 0.430 | 0.615 |
| LPC(20:4) b | 8.97 [8.27–10.63] | 8.4 [8.16–8.81] | 0.350 | 0.615 |
| LPC(22:6) b | 0.386 [0.331–0.393] | 0.325 [0.274–0.362] | 0.196 | 0.499 |
| LPE(16:0) | 0.55 [0.52–0.58] | 0.70 [0.61–0.72] | 0.017 | 0.186 |
| LPE(18:0) b | 0.74 [0.63–0.79] | 0.82 [0.78–0.88] | 0.046 | 0.250 |
| LPE(18:1) b | 0.25 [0.23–0.27] | 0.27 [0.25–0.30] | 0.425 | 0.615 |
| LPE(18:2) b | 0.38 [0.37–0.42] | 0.44 [0.37–0.48] | 0.383 | 0.615 |
| LPE(20:4) b | 0.158 [0.143–0.169] | 0.168 [0.167–0.201] | 0.089 | 0.390 |
| LPE(22:6) b | 0.12 [0.11–0.14] | 0.14 [0.12–0.15] | 0.478 | 0.619 |
| MAG(18:1) | 0.07 [0.06–0.09] | 0.11 [0.10–0.12] | 0.126 | 0.462 |
| MAG(18:2) b | 0.18 [0.15–0.21] | 0.17 [0.11–0.26] | 0.756 | 0.779 |
| MAG(20:4) b | 0.0061 [0.0049–0.0084] | 0.0068 [0.0045–0.0094] | 0.779 | 0.779 |
| Analyte | Control Group (N = 10) | PH Group (N = 7) | p-Value | Adjusted p-Value a |
|---|---|---|---|---|
| LPA(16:0) | 0.091 [0.080–0.12] | 0.21 [0.19–0.26] | <0.001 | 0.003 |
| LPA(18:0) b | 0.13 [0.11–0.17] | 0.19 [0.17–0.25] | 0.080 | 0.219 |
| LPA(18:1) | 0.062 [0.056–0.085] | 0.156 [0.125–0.182] | <0.001 | 0.001 |
| LPA(18:2) b | 0.99 [0.80–1.29] | 2.31 [2.03–4.00] | <0.001 | 0.001 |
| LPA(20:4) b | 1.22 [1.05–1.51] | 2.18 [1.99–2.76] | <0.001 | 0.004 |
| LPA(22:6) b | 0.064 [0.057–0.082] | 0.137 [0.119–0.160] | <0.001 | <0.001 |
| LPC(16:0) | 16.8 [16.0–17.3] | 16.7 [14.9–17.2] | 0.357 | 0.604 |
| LPC(16:1) b | 1.96 [1.625–2.07] | 1.99 [1.585–2.86] | 0.348 | 0.604 |
| LPC(18:0) b | 32.8 [31.3–35.9] | 33.7 [30.7–36.0] | 0.749 | 0.827 |
| LPC(18:1) | 7.01 [6.66–7.32] | 6.80 [6.11–7.31] | 0.485 | 0.686 |
| LPC(18:2) b | 28.7 [26.3–30.2] | 28.1 [26.8–30.0] | 0.669 | 0.827 |
| LPC(20:4) b | 11.1 [10.3–11.8] | 9.3 [7.6–10.4] | 0.231 | 0.462 |
| LPC(22:6) b | 0.36 [0.32–0.39] | 0.36 [0.33–0.39] | 0.693 | 0.827 |
| LPE(16:0) | 0.78 [0.73–0.79] | 0.81 [0.80–1.03] | 0.015 | 0.056 |
| LPE(18:0) b | 1.14 [1.05–1.28] | 1.39 [1.23–1.48] | 0.053 | 0.168 |
| LPE(18:1) b | 0.29 [0.27–0.34] | 0.38 [0.335–0.42] | 0.149 | 0.328 |
| LPE(18:2) b | 0.53 [0.47–0.59] | 0.62 [0.53–0.66] | 0.457 | 0.686 |
| LPE(20:4) b | 0.23 [0.21–0.25] | 0.22 [0.18–0.24] | 0.131 | 0.320 |
| LPE(22:6) b | 0.158 [0.143–0.167] | 0.176 [0.146–0.179] | 0.752 | 0.827 |
| MAG(18:1) | 0.095 [0.066–0.156] | 0.089 [0.058–0.113] | 0.499 | 0.686 |
| MAG(18:2) b | 0.70 [0.40–1.10] | 0.60 [0.41–0.98] | 0.860 | 0.860 |
| MAG(20:4) b | 0.014 [0.011–0.018] | 0.015 [0.006–0.023] | 0.806 | 0.844 |
| Analyte | Mass Transition | MS Parameters | Adduct | Retention Time (min) | |||
|---|---|---|---|---|---|---|---|
| m/z (MS1) | m/z (MS2) | DP (V) | CE (eV) | CXP (V) | |||
| LPA(16:0) | 409.5 | 152.9 | −50 | −40 | −10 | [M–H]− | 6.8 |
| LPA(17:1) a | 421.1 | 152.9 | −50 | −40 | −10 | [M–H]− | 6.5 |
| LPA(18:0) b | 437.3 | 152.9 | −50 | −40 | −10 | [M–H]− | 7.7 |
| LPA(18:1) | 435.2 | 152.9 | −50 | −40 | −10 | [M–H]− | 7.0 |
| LPA(18:2) b | 433.2 | 152.9 | −50 | −40 | −10 | [M–H]− | 6.3 |
| LPA(20:4) b | 457.2 | 152.9 | −50 | −40 | −10 | [M–H]− | 6.3 |
| LPA(22:6) b | 481.2 | 152.9 | −50 | −40 | −10 | [M–H]− | 6.3 |
| LPC(16:0) | 480.6 | 255.4 | −80 | −40 | −10 | [M–CH3]− | 7.2 |
| LPC(16:1) b | 478.6 | 253.4 | −100 | −40 | −10 | [M–CH3]− | 6.3 |
| LPC(17:1) a | 492.3 | 267.3 | −80 | −40 | −10 | [M–CH3]− | 6.8 |
| LPC(18:0) b | 508.4 | 283.5 | −100 | −40 | −10 | [M–CH3]− | 8.1 |
| LPC(18:1) | 506.4 | 281.5 | −80 | −40 | −10 | [M–CH3]− | 7.4 |
| LPC(18:2) b | 504.4 | 279.5 | −100 | −40 | −10 | [M–CH3]− | 6.7 |
| LPC(20:4) b | 528.4 | 303.4 | −100 | −40 | −10 | [M–CH3]− | 6.7 |
| LPC(22:6) b | 552.4 | 327.4 | −100 | −40 | −10 | [M–CH3]− | 6.6 |
| LPE(16:0) | 452.4 | 196.0 | −50 | −40 | −10 | [M–H]− | 7.2 |
| LPE(17:1) | 464.2 | 196.0 | −80 | −40 | −10 | [M–H]− | 6.9 |
| LPE(18:0) b | 480.3 | 196.0 | −50 | −40 | −10 | [M–H]− | 8.1 |
| LPE(18:1) b | 478.3 | 196.0 | −50 | −40 | −10 | [M–H]− | 7.4 |
| LPE(18:2) b | 476.3 | 196.0 | −50 | −40 | −10 | [M–H]− | 6.8 |
| LPE(20:4) b | 500.6 | 196.0 | −50 | −40 | −10 | [M–H]− | 6.7 |
| LPE(22:6) b | 524.3 | 196.0 | −50 | −40 | −10 | [M–H]− | 6.7 |
| MAG(18:1) | 357.2 | 265.0 | 50 | 17 | 16 | [M+H]+ | 8.0 |
| MAG(18:2) b | 355.2 | 263.0 | 50 | 17 | 16 | [M+H]+ | 7.5 |
| MAG(20:4) b | 379.3 | 287.1 | 50 | 17 | 16 | [M+H]+ | 7.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duflot, T.; Tu, L.; Leuillier, M.; Messaoudi, H.; Groussard, D.; Feugray, G.; Azhar, S.; Thuillet, R.; Bauer, F.; Humbert, M.; et al. Preventing the Increase in Lysophosphatidic Acids: A New Therapeutic Target in Pulmonary Hypertension? Metabolites 2021, 11, 784. https://doi.org/10.3390/metabo11110784
Duflot T, Tu L, Leuillier M, Messaoudi H, Groussard D, Feugray G, Azhar S, Thuillet R, Bauer F, Humbert M, et al. Preventing the Increase in Lysophosphatidic Acids: A New Therapeutic Target in Pulmonary Hypertension? Metabolites. 2021; 11(11):784. https://doi.org/10.3390/metabo11110784
Chicago/Turabian StyleDuflot, Thomas, Ly Tu, Matthieu Leuillier, Hind Messaoudi, Déborah Groussard, Guillaume Feugray, Saïda Azhar, Raphaël Thuillet, Fabrice Bauer, Marc Humbert, and et al. 2021. "Preventing the Increase in Lysophosphatidic Acids: A New Therapeutic Target in Pulmonary Hypertension?" Metabolites 11, no. 11: 784. https://doi.org/10.3390/metabo11110784