1. Introduction
Wide health benefits resulting from maintaining the optimal concentration of vitamin D have been confirmed by many scientific publications over the last several years.
Vitamin D deficiency was associated with or coincided with many civilization diseases, such as cancer [
1,
2,
3,
4,
5], autoimmune diseases [
6,
7,
8], diabetes type 1 and 2 [
9,
10,
11], cardiovascular disease [
12,
13,
14], hypertension [
15,
16,
17] and depression, schizophrenia [
18,
19] or Alzheimer’s disease [
20]. Maintaining proper serum 25-hydroxyvitain D, i.e., 25(OH)D, became a serious problem and appeared to be an important aspect for human health [
21,
22,
23,
24,
25,
26]. Earlier, the criterion of vitamin D deficiency associated with the risk of deficiency rickets or osteomalacia was considered when a 25(OH)D concentration value below 10 ng/mL was noted in a given patient [
27]. However, in the last 10 years, many world scientific and medical societies have published guidelines on the recommended values of 25(OH)D-a metabolite recognized as an indicator of a vitamin D deficiency. The Institute of Medicine (IOM; Washington, DC, USA) recognized the value of 20 ng/mL as the minimum concentration of 25(OH)D necessary to obtain health benefits [
28]. The Endocrine Society (ES; Washington, DC, USA) recommended a minimum concentration of 25(OH)D > 30 ng/mL, and indicated the optimal range as being 40–60 ng/mL [
29]. In addition, the International Osteoporosis Foundation recommended a value of 30 ng/mL pointing to an important correlation with optimal parathormone (PTH) concentration values [
30].
Many other societies around the world have also developed their own guidelines. Comparing these data, it is possible to notice differences in the proposed target values. This is due to differences of opinion as to the perception of the effects of vitamin D by different groups of experts. The IOM recommendations focused on benefits limited to bone tissue and calcium–phosphate metabolism. The ES proposal pointed to the systemic effect of vitamin D. The ES defined values <30 ng/mL and <20 ng/mL as a “vitamin D insufficiency” or a “deficiency”, respectively.
The recommendations for Europe as well as for the Scandinavian countries, Germany, Austria and Switzerland, adopted a sufficient concentration of 25(OH)D as being at least 20 ng/mL [
31,
32,
33,
34].
In 2013, the guidelines for Central Europe were published for a healthy population and for the groups at risk of deficits. The recommended daily doses of vitamin D, the diagnostic criteria for 25(OH)D and the recommendations regarding maximum safe doses were published [
35]. In this document, a minimum target concentration of 25(OH)D of 30–50 ng/mL was proposed for both the calcemic and pleiotropic effects of vitamin D [
35]. In 2018 [
36] and 2023, [
37] updates were introduced with national, i.e., Polish, arrangements and guidelines. The detailed rules for vitamin D supplementation and treatment were described, the global and local recommendations on supplementation were compared and safety issues were discussed.
The symptoms of the toxic effect of vitamin D were shown to be extremely rarely observed, and accompanying 25(OH)D concentrations of up to 100 ng/mL had been established as safe for children and adults [
37,
38].
The problem of potential toxicity concerns only people with congenital hypersensitivity to vitamin D, including a group of genetically predisposed people, in whom the supply of even the recommended doses of vitamin D, considered as safe, may introduce health problems due to hypercalcemia and hypercalciuria [
39]. The risk of vitamin D intoxication, especially in unrecognized newborns, infants and toddlers with idiopathic infantile hypercalcemia (IIH) or with the Williams–Beuren syndrome, granulomatous diseases and lymphomas should be considered before the start of vitamin D supplementation [
40,
41,
42,
43,
44,
45,
46,
47,
48].
This study follows and continues the article “25(OH)D Concentration in Neonates, Infants, and Toddlers from Poland–Evaluation of Trends During Years 1981–2011” [
49], where we have retrospectively assessed the parameters of the Ca-P metabolism and 25(OH)D concentration among a population aged 0–18 years, but investigated in the previous period of time. Again, the 25(OH)D concentration data as well as the calcium–phosphate metabolism markers assayed in the years 2014–2019 were analyzed to investigate the scope of potential changes.
2. Patients and Methods
2.1. The Study Group
The study group consisted, in total, of 49,424 pediatric cases aged 0–18 years with a mean age of 7.3 ± 6.0 years. A total of 17,636 patients from this group had results for a 25(OH)D concentration value, 26,331 had results for a calcium (Ca) in serum concentration value and 2839 had results for a parathormone (PTH) value. The study group represented the entire pediatric population of Poland, healthy or with congenital and acquired disorders of Ca-P homeostasis. The medical database analyzed in this study consisted of results assayed in our laboratory in the years 2014–2019. The inclusion criterion was access to the 25(OH)D measurement value, and/or the Ca and PTH values of a patient’s sample. All lab assays were conducted at the same time.
2.2. Methods
Biochemical parameters were assessed using the following methods:
- (1)
Total calcium (Ca) was assayed with the use of spectrophotometric assay (Alinity Abott);
- (2)
25-hydroxyvitamin D (25(OH)D) and 1,25-dihydroxyvitamin D (1,25(OH)2D) concentration values were assessed using chemiluminescence immunoassay CLIA (IDS-ISYS);
- (3)
Parathormone (PTH) activity measurements were conducted by immunoradiometric assay IRMA (Cisbio Bioassays).
The above-mentioned methods did not changed during the years that were included in this study, i.e., 2014–2019. The authors had access to The Children’s Memorial Health Institute’s medical database for the laboratory assay results that were evaluated.
2.3. Statistical Analyses
The medical database was investigated in terms of patient’s age, year of visit, quarter of the year of assay and 25(OH)D concentration value and/or Ca value or PTH value or 1,25(OH)2D value. The mean ± standard deviation values of 25(OH)D, Ca and PTH concentrations were calculated as general characteristics of the studied group. The distribution of the concentration values of 25(OH)D, Ca, PTH in relation to “subjective” reference values was also calculated. In cases with evident hypercalcemia, i.e., Ca ≥ 2.75 mmol/L, both 25(OH)D and 1,25(OH)2D as well as the PTH distribution of values were investigated. Finally, 25(OH)D concentration values were evaluated according to patient’s age, year of the visit and quarter of the year, and correlations between a measure of vitamin D status and patient’s age or Ca concentrations were estimated; mean ± standard deviation values and, if statistically significant, r values were provided. All the statistical analyses were carried out with the use of Statistica v. 10 (StatSoft Inc., Tulsa, OK, USA).
4. Discussion
There are many reports providing vitamin D recommended intakes, reference ranges for 25(OH)D and potential toxicity risks [
35,
36,
37,
50,
51]. Other reports confirmed that vitamin D deficiency is a serious healthcare problem [
25,
26,
27,
28,
29,
30,
52].
In our earlier study titled “25(OH)D Concentration in Neonates, Infants, and Toddlers from Poland–Evaluation of Trends During Years 1981–2011” [
49] we showed a significantly higher average 25(OH)D concentration (37.5 ng/mL) in the group of newborns and infants (0–18 month) compared to children and adolescents (18 month–18 years of life) [
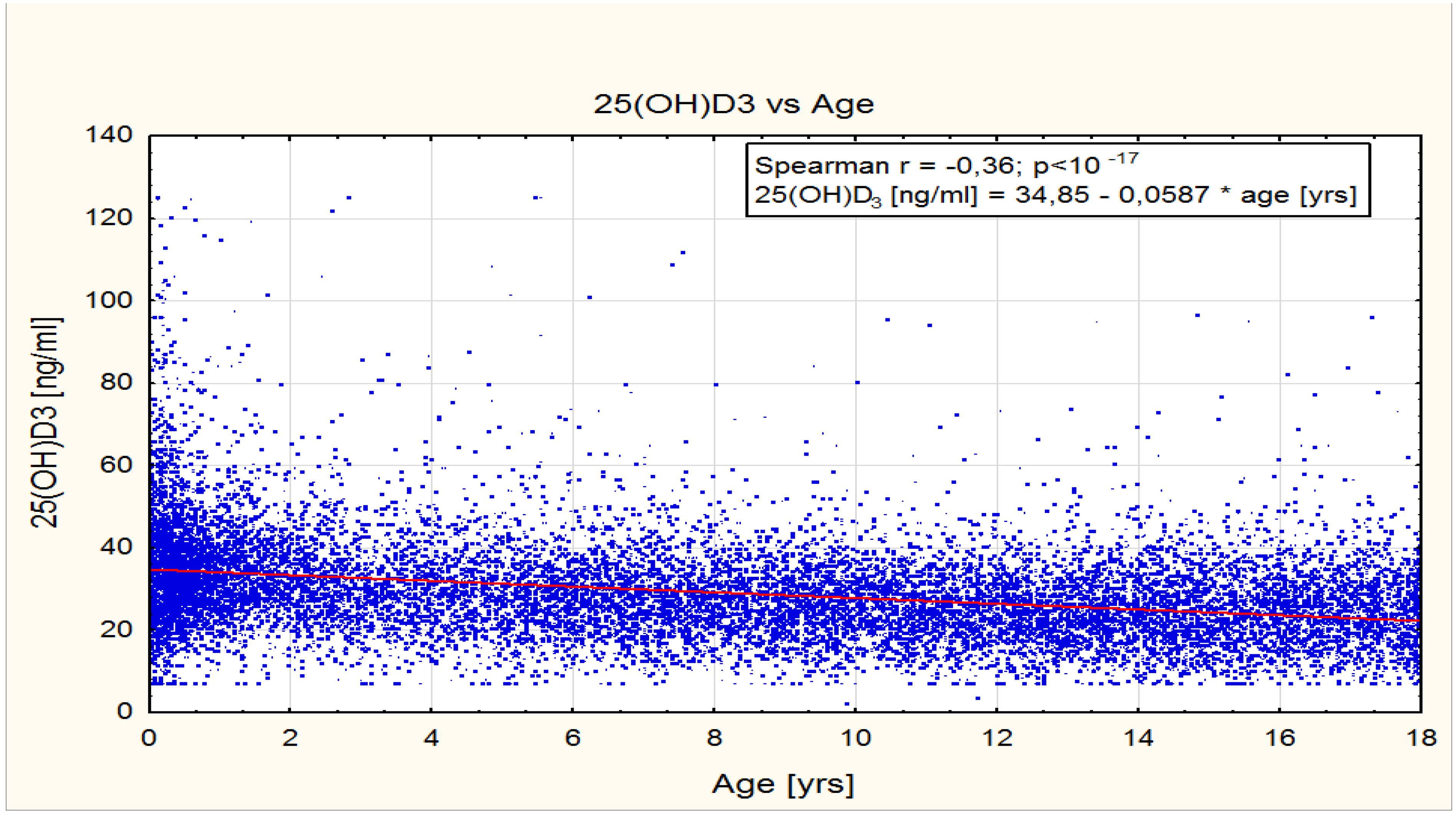
49]. A clear negative trend showing decreasing 25(OH)D concentrations with age was revealed, especially after the age of 3 [
49]. In this paper, we decided to present another time period, i.e., the evaluation of 25(OH)D concentration values in the years 2014–2019. Our study was based on a representative group of 49,424 pediatric patients aged 0–18 years admitted to the consultation clinic or hospitalized to the Children’s Memorial Health Institute over the course of more than 5 years. From this group we separated patients using the available disposable results of 25(OH)D, Ca, PTH (in serum) concentrations.
It was again a wide and diverse population, which included patients admitted for the first time as well as those who were on a subsequent visit to the clinic or undergoing a repeat hospitalization.
The reference point for the statistical evaluation was the available value of 25(OH)D concentration noted during a visit to the consultation clinic.
The Children’s Memorial Health Institute uniquely takes care of pediatric patients suffering from almost every disorder or disease, therefore, the types of suspected or diagnosed diseases or disorders were at that time diverse. It was assumed that the patients under assessment were supplemented with cholecalciferol according to the current recommendations [
35,
36]. In addition, it was assumed that the vitamin D supplementation guidelines [
35,
36], which were recently updated [
37], are the mostly followed by parents or caregivers of neonates and infants up to 18 months of age.
The comparison of the mean concentrations of 25(OH)D in the recent study group showed a fairly clear negative correlation in the age groups 0–1 year and 1–3 years. The average values of 25(OH)D concentration were 36.3 ng/mL and 33.6 ng/mL, respectively. Among these age groups, 25(OH)D concentrations remained, in general, within the optimal concentration ranges of 30–50 ng/mL [
35,
36,
37]. There was a significant negative correlation between 25(OH)D concentration and the overall age of patients in the range 0–18 years. However, in the groups of toddlers and younger children aged 3–10 years, as well as in older children and teenagers aged 10–18 years, the average 25(OH)D concentrations did not reach the optimal values of 30–50 ng/mL and were 28.7 ng/mL and 25.4 ng/mL, respectively.
This observation indirectly confirms the widespread use of anti-rickets prophylaxis by pediatricians in the group of the youngest children under the age of 3 years and is in agreement with our previous findings [
49]. Further, a strong negative correlation was observed between 25(OH)D concentrations and children of ages 6 and 12 months caused by rapid weight gain which was hypothesized as a reason to increase the supplementation dose of cholecalciferol in infants [
53].
The observed changes in the 25(OH)D concentration values within different time periods showed variable average 25(OH)D concentrations from the optimal concentration range of 31.2 ng/mL noted in 2014 to 29 ng/mL noted in 2015–2017, and again in the concentration of 32.1 ng/mL revealed in 2018–2019.
Recommendations for cholecalciferol supplementation in individual pediatric age groups were not changed within the period of our study, so the above-mentioned slight fluctuations in average 25(OH)D concentrations can be explained by the diversity of disease etiology and or by incomplete compliance with supplementation recommendations.
A comparison of the 25(OH)D concentrations in individual quarters of the year confirms the significant role of skin synthesis in the supply of vitamin D. The visible effect of skin synthesis was revealed especially during the summer months, and it also persisted in autumn months. However, it should be noted that the average concentrations of 25(OH)D in the second and third quarters of the year only slightly exceeded the lower limit of optimal concentrations, approximately 31.3 ng/mL and 30.6 ng/mL, respectively.
Of note, in our laboratory, 25(OH)D concentration measurements were carried out using an automatic IDS-ISYS system based on chemiluminescence immunoassay.
This method was compared to and validated with the Good Laboratory Practice requirements, and the high precision of 25(OH)D measurements was confirmed with a DEQAS international quality certificate.
The analyses of relations between calcemia and vitamin D status provided interesting observations: more than 72% of patients with 25(OH)D concentration <10 ng/mL (severe vitamin D deficiency) had a normal serum Ca concentration (2.25–2.65 mmol/L). In the subgroup with hypocalcemia (Ca < 2.25 mmol/L), 25% of patients had 25(OH)D concentration <20 ng/mL and 57% had 25(OH)D concentration <30 ng/mL. In the analyzed subgroup with hypercalcemia (Ca > 2.65 mmol/L), 25(OH)D concentrations were both very low and high and there was no correlation between the severity of hypercalcemia and 25(OH)D concentration. Despite hypercalcemia, more than 30% of patients did not achieve optimal 25(OH)D concentration values of at least 30 ng/mL. Moreover, a number of cases with hypercalcemia had 25(OH)D concentrations lower than 10 ng/mL or higher than 50 ng/mL. These results generally confirm a lack of clear correlation between 25(OH)D and Ca serum concentrations and indirectly showed that the causes of disturbances in the Ca-P metabolism may have various causes.
The evaluation of the active metabolite 1,25(OH)
2D showed that more than 54% of the estimated population had a concentration above 80 pg/mL; in clinical practice this is considered suitable for further detailed analysis [
45,
54,
55].
High concentrations of calcitriol can also indicate other disorders causing disturbances in Ca-P metabolism, but due to very limited number of cases the further speculations in this case were omitted by us.
The studies conducted in various sites of the world and in Poland have already uncovered vitamin D status in the general population, including pediatric patients, and confirmed the problem of low 25(OH)D concentrations in both healthy and diseased populations [
22,
23,
24,
25,
27,
29,
33,
56,
57].
Nonetheless, more research focused on vitamin D and its metabolites is still required, and our study results, due to its limitations, provided only the estimation of negative trends of 25(OH)D concentration with age. The evaluation of the pediatric population covering over 5 years can, in our opinion, provide information not only to the past and current state and trends in the vitamin D status but also to the relation between calcium and 25(OH)D concentrations, at least in 0–18 year olds residing in Poland. To conclude, vitamin D deficiency (<20 ng/mL) was noted in 18.9% subjects in 2014–2019. The effective prevention of vitamin D deficiency was observed in children aged 3 years and younger. A relationship between the concentrations of calcium and 25(OH)D was not observed.