Using Undergraduate Researchers to Build Vector and West Nile Virus Surveillance Capacity
Abstract
:1. Introduction
2. Experimental Section
2.1. Student Recruitment and Training
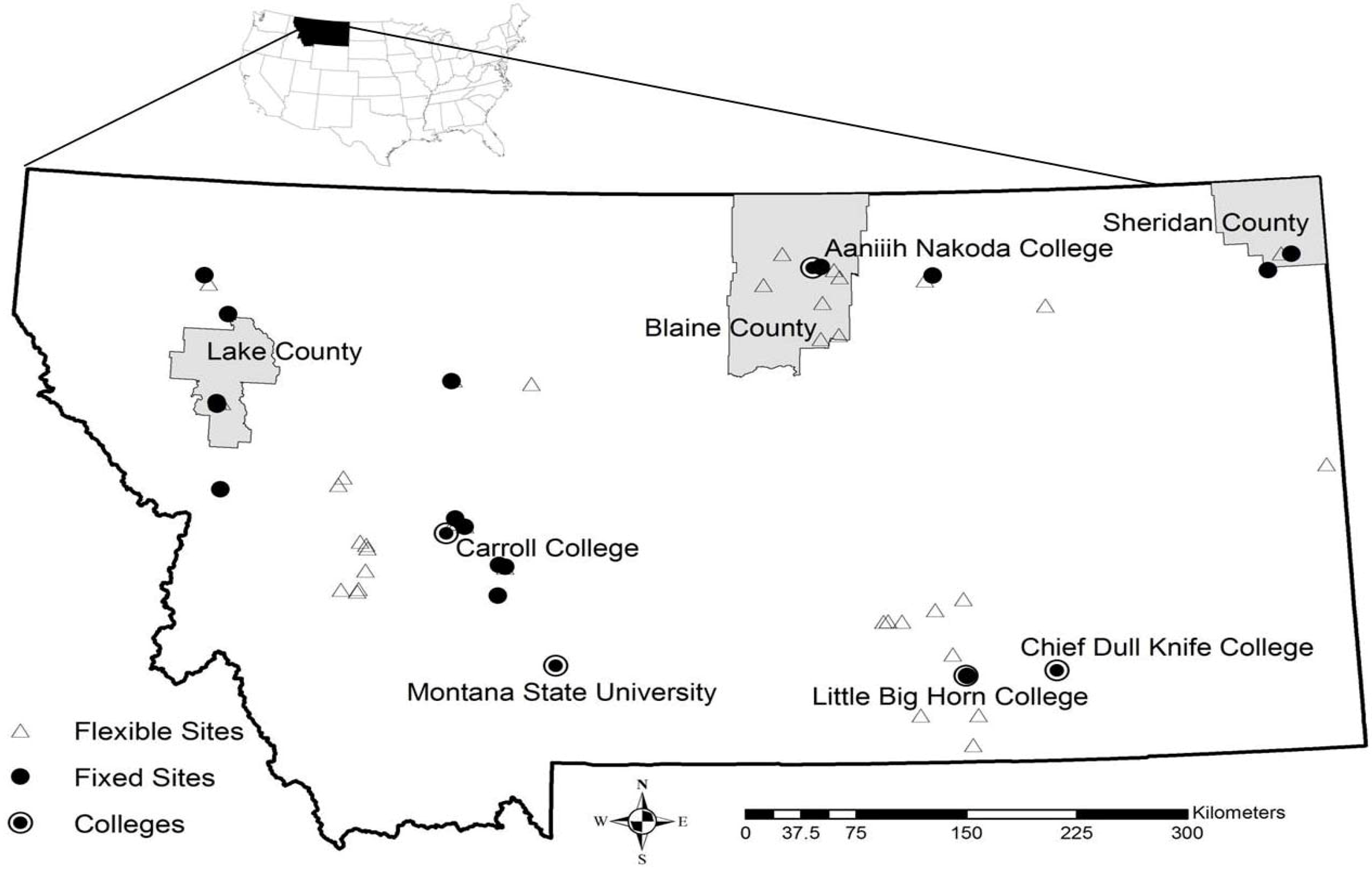
2.2. Sampling Protocol
2.3. Detection Protocol
2.4. Data Analysis and Dissemination
2.5. Assessing Student Outcomes
3. Results and Discussion
3.1. Surveillance Results
| Year | No. | Cx. tarsalis | No. Cx. tarsalis | No. | MIR | Human |
|---|---|---|---|---|---|---|
| trap nights | mean LTI | pools tested | positive | cases | ||
| 2003 | 180 | 50 | 389 | 134 | 14.9 | 228 |
| 2004 | 380 | 38 | 323 | 6 | 1.3 | 7 |
| 2005 | 289 | 67 | 557 | 15 | 0.8 | 25 |
| 2006 | 261 | 50 | 326 | 29 | 2.2 | 34 |
| 2007 | 222 | 95 | 458 | 78 | 3.7 | 202 |
| 2008 | 94 | 46 | 118 | 3 | 0.7 | 5 |
| 2009 | 204 | 35 | 145 | 5 | 0.7 | 5 |
| 2010 | 201 | 23 | 92 | 1 | 0.2 | 0 |
| 2011 | 230 | 181 | 832 | 0 | 0.0 | 1 |
| 2012 | 175 | 38 | 133 | 11 | 1.7 | 6 |
| Year | Sheridan County | Blaine County | Lake County | |||
|---|---|---|---|---|---|---|
| LTI | MIR | LTI | MIR | LTI | MIR | |
| 2005 | 210.4 | 1.4 | 86.0 | 0.0 | 39.8 | 0.0 |
| 2006 | 131.0 | 1.3 | 245.0 | 3.6 | 27.3 | 0.0 |
| 2007 | 334.0 | 6.0 | 185.0 | 7.6 | 44.7 | 0.0 |
| 2008 | 62.6 | 3.2 | 113.3 | 1.1 | 50.1 | 0.0 |
| 2009 | 197.5 | 1.0 | 152.7 | 0.7 | 25.8 | 0.0 |
| 2010 | 380.0 | 1.1 | 75.1 | 0.0 | 37.0 | 0.0 |
| 2011 | 1,690.0 | 0.0 | 204.6 | 0.0 | 46.9 | 0.0 |
| 2012 | 21.6 | 5.8 | 80.8 | 6.2 | 21.6 | 0.0 |
3.2. Patterns in Infection Rate
3.3. Student Outcomes
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Gubler, D.J. The global emergence/resurgence of arboviral diseases as public health problems. Arch. Med. Res. 2002, 33, 330–342. [Google Scholar] [CrossRef]
- GOA (United States Government Accounting Office), Emerging Infectious Diseases: Review of State and Federal Disease Surveillance Efforts. GOA: Washington, DC, USA, 2004; Vol. GAO-04-877.
- Colwell, D.D.; Dantas-Torres, F.; Otranto, D. Vector-borne parasitic zoonoses: Emerging scenarios and new perspectives. Vet. Parasitol. 2011, 182, 14–21. [Google Scholar] [CrossRef]
- Pavlin, J.A.; Mostashari, F.; Kortepeter, M.G.; Hynes, N.A.; Chotani, R.A.; Mikol, Y.B.; Ryan, M.A.K.; Neville, J.S.; Gantz, D.T.; Writer, J.V.; et al. Innovative surveillance methods for rapid detection of disease outbreaks and bioterrorism: Results of an interagency workshop on health indicator surveillance. Am. J. Public Health 2003, 93, 1230–1235. [Google Scholar] [CrossRef]
- LaBeaud, A.D.; Aksoy, S. Neglected funding for vector-borne diseases: A near miss this time, a possible disaster the next time. PloS Neg. Trop. Dis. 2010, 4. [Google Scholar] [CrossRef]
- Kwan, J.L.; Park, B.K.; Carpenter, T.E.; Ngo, V.; Civen, R.; Reisen, W.K. Comparison of enzootic risk measures for predicting West Nile disease, Los Angeles, California, USA, 2004–2010. Emerg. Infect. Dis. 2012, 18, 1298–1306. [Google Scholar]
- Brownstein, J.S.; Holford, T.R.; Fish, D. Enhancing West Nile virus surveillance, United States. Emerg. Infect. Dis. 2004, 10, 1129–1133. [Google Scholar]
- Vazquez-Prokopec, G.M.; Chaves, L.F.; Ritchie, S.A.; Davis, J.; Kitron, U. Unforeseen costs of cutting mosquito surveillance budgets. PLoS Negl. Trop. Dis. 2010, 4. [Google Scholar] [CrossRef]
- Monath, T.P. Yellow fever: An update. Lancet Infect. Dis. 2001, 1, 11–20. [Google Scholar] [CrossRef]
- CDC. Outbreak of West Nile-like viral encephalitis—New York, 1999. MMWR 1999, 48, 845–849.
- Wright, W.F.; Pritt, B.S. Update: The diagnosis and management of Dengue virus infection in North America. Diagn. Microbiol. Infect. Dis. 2012, 73, 215–220. [Google Scholar] [CrossRef]
- Lopatto, D. Undergraduate research experiences support science career decisions and active learning. CBE Life Sci. Educ. 2007, 6, 297–306. [Google Scholar] [CrossRef]
- Hunter, A.B.; Laursen, S.L.; Seymour, E. Becoming a scientist: The role of undergraduate research in students’ cognitive, personal, and professional development. Sci. Educ. 2007, 91, 36–74. [Google Scholar] [CrossRef]
- Russell, S.H.; Hancock, M.P.; McCullough, J. The pipeline—Benefits of undergraduate research experiences. Science 2007, 316, 548–549. [Google Scholar] [CrossRef]
- Jones, M.T.; Barlow, A.E.L.; Villarejo, M. Importance of undergraduate research for minority persistence and achievement in biology. J. Higher Educ. 2010, 81, 82–115. [Google Scholar] [CrossRef]
- Johnson, G.; Rolston, M.; Mason, K. Montana Mosquito and West Nile Virus Surveillance Program: 2003–2004; Technical report submitted to the Montana Department of Public Health and Human Services: Helena, MT, USA, 2005. [Google Scholar]
- CDC. Epidemic/Epizootic West Nile Virus in the United States: Guidelines for Surveillance, Prevention, and Control. Centers for Disease Control and Prevention: Fort Collins, CO, USA, 2003; pp. 1–78. Available online: http://www.cdc.gov/ncidod/dvbid/westnile/resources/wnv-guidelines-apr-2001.pdf (accessed on 17 November 2012).
- Lanciotti, R.S.; Kerst, A.J.; Nasci, R.S.; Godsey, M.S.; Mitchell, C.J.; Savage, H.M.; Komar, N.; Panella, N.A.; Allen, B.C.; Volpe, K.E.; et al. Rapid detection of West Nile virus from human clinical specimens, field-collected mosquitoes, and avian samples by a Taqman reverse transcriptase-PCR assay. J. Clin. Microbiol. 2000, 38, 4066–4071. [Google Scholar]
- Kauffman, E.B.; Jones, S.A.; Dupuis, A.P.; Ngo, K.A.; Bernard, K.A.; Kramer, L.D. Virus detection protocols for West Nile virus in vertebrate and mosquito specimens. J. Clin. Microbiol. 2003, 41, 3661–3667. [Google Scholar] [CrossRef]
- Silver, J.B. Mosquito Ecology: Field Sampling Methods, 3rd ed.; Springer: Berlin, Germany, 2008; p. 1498. [Google Scholar]
- Dye, C. The analysis of parasite transmission by bloodsucking insects. Annu. Rev. Entomol. 1992, 37, 1–19. [Google Scholar]
- Johnson, G.; Nemeth, N.; Hale, K.; Lindsey, N.; Panella, N.; Komar, N. Surveillance for West Nile virus in American white pelicans, Montana, USA, 2006–2007. Emerg. Infect. Dis. 2010, 16, 406–411. [Google Scholar] [CrossRef]
- Walter, S.D.; Hildreth, S.W.; Beaty, B.J. Estimation of infection-rates in populations of organisms using pools of variable size. Am. J. Epidemiol. 1980, 112, 124–128. [Google Scholar]
- DPHHS. West Nile Virus Homepage. Montana Department of Public Health and Human Services: Helena, MT, USA, 2010. Available online: http://www.dphhs.mt.gov/publichealth/cdepi/surveillance/westnilevirus.shtml (accessed on 27 January 2013).
- Barker, C.M.; Bolling, B.G.; Moore, C.G.; Eisen, L. Relationship between distance from major larval habitats and abundance of adult mosquitoes in semiarid plains landscapes in Colorado. J. Med. Entomol. 2009, 46, 1290–1298. [Google Scholar]
- Bolling, B.G.; Barker, C.M.; Moore, C.G.; Pape, W.J.; Eisen, L. Seasonal patterns for entomological measures of risk for exposure to Culex vectors and West Nile virus in relation to human disease cases in northeastern Colorado. J. Med. Entomol. 2009, 46, 1519–1531. [Google Scholar] [CrossRef]
- Theophilides, C.N.; Ahearn, S.C.; Grady, S.; Merlino, M. Identifying West Nile virus risk areas: The dynamic continuous-area space-time system. Am. J. Epidemiol. 2003, 157, 843–854. [Google Scholar] [CrossRef]
- Allan, B.F.; Langerhans, R.B.; Ryberg, W.A.; Landesman, W.J.; Griffin, N.W.; Katz, R.S.; Oberle, B.J.; Schutzenhofer, M.R.; Smyth, K.N.; de St Maurice, A.; et al. Ecological correlates of risk and incidence of West Nile virus in the United States. Oecologia 2009, 158, 699–708. [Google Scholar] [CrossRef]
- Reeves, W.C.; Milby, M.M.; Reisen, W.K. Development of a Statewide Arbovirus Surveillance Program and Models of Vector Populations and Virus Transmission. In Epidemiology and Control of Mosquito-Borne Arboviruses in California, 1943–1987; Reeves, W.C., Ed.; California Mosquito Vector Control Association: Sacramento, CA, USA, 1990; pp. 431–458. [Google Scholar]
- Diuk-Wasser, M.A.; Toure, M.B.; Dolo, G.; Bagayoko, M.; Sogoba, N.; Traore, S.F.; Manoukis, N.; Taylor, C.E. Vector abundance and malaria transmission in rice-growing villages in Mali. Am. J. Trop. Med. Hyg. 2005, 72, 725–731. [Google Scholar]
- Darbro, J.M.; Harrington, L.C. Avian defensive behavior and blood-feeding success of the West Nile vector mosquito, Culex pipiens. Behav. Ecol. 2007, 18, 750–757. [Google Scholar]
- Ezenwa, V.O.; Godsey, M.S.; King, R.J.; Guptill, S.C. Avian diversity and West Nile virus: Testing associations between biodiversity and infectious disease risk. Proc. Royal Soc. B Biol. Sci. 2006, 273, 109–117. [Google Scholar]
- Styer, L.M.; Meola, M.A.; Kramer, L.D. West Nile virus infection decreases fecundity of Culex tarsalis females. J. Med. Entomol. 2007, 44, 1074–1085. [Google Scholar] [CrossRef]
- Ciota, A.T.; Styer, L.M.; Meola, M.A.; Kramer, L.D. The costs of infection and resistance as determinants of West Nile virus susceptibility in Culex mosquitoes. BMC Ecol. 2011, 11. [Google Scholar] [CrossRef]
- Richards, S.L.; Lord, C.C.; Pesko, K.; Tabachnick, W.J. Environmental and biological factors influencing Culex pipiens quinquefasciatus Say (Diptera: Culicidae) vector competence for Saint Louis encephalitis virus. Am. J. Trop. Med. Hyg. 2009, 81, 264–272. [Google Scholar]
- Reisen, W.K.; Fang, Y.; Martinez, V.M. Effects of temperature on the transmission of West Nile virus by Culex tarsalis (Diptera : Culicidae). J. Med. Entomol. 2006, 43, 309–317. [Google Scholar] [CrossRef]
- Ruiz, M.O.; Chaves, L.F.; Hamer, G.L.; Sun, T.; Brown, W.M.; Walker, E.D.; Haramis, L.; Goldberg, T.L.; Kitron, U.D. Local impact of temperature and precipitation on West Nile virus infection in Culex species mosquitoes in northeast Illinois, USA. Parasit. Vec. 2010, 3. [Google Scholar] [CrossRef]
- Chen, C.C.; Epp, T.; Jenkins, E.; Waldner, C.; Curry, P.S.; Soos, C. Predicting weekly variation of Culex tarsalis (Diptera: Culicidae) West Nile virus infection in a newly endemic region, the Canadian prairies. J. Med. Entomol. 2012, 49, 1144–1153. [Google Scholar] [CrossRef]
- Lambrechts, L.; Paaijmans, K.P.; Fansiri, T.; Carrington, L.B.; Kramer, L.D.; Thomas, M.B.; Scott, T.W. Impact of daily temperature fluctuations on dengue virus transmission by Aedes aegypti. Proc. Natl. Acad. Sci. USA 2011, 108, 7460–7465. [Google Scholar] [CrossRef]
- Zohrabian, A.; Meltzer, M.I.; Ratard, R.; Billah, K.; Molinari, N.A.; Roy, K.; Scott, R.D.; Petersen, L.R. West Nile virus economic impact, Louisiana, 2002. Emerg. Infect. Dis. 2004, 10, 1736–1744. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hokit, G.; Alvey, S.; Geiger, J.M.O.; Johnson, G.D.; Rolston, M.G.; Kinsey, D.T.; Bear, N.T. Using Undergraduate Researchers to Build Vector and West Nile Virus Surveillance Capacity. Int. J. Environ. Res. Public Health 2013, 10, 3192-3202. https://doi.org/10.3390/ijerph10083192
Hokit G, Alvey S, Geiger JMO, Johnson GD, Rolston MG, Kinsey DT, Bear NT. Using Undergraduate Researchers to Build Vector and West Nile Virus Surveillance Capacity. International Journal of Environmental Research and Public Health. 2013; 10(8):3192-3202. https://doi.org/10.3390/ijerph10083192
Chicago/Turabian StyleHokit, Grant, Sam Alvey, Jennifer M. O. Geiger, Gregory D. Johnson, Marni G. Rolston, Daniel T. Kinsey, and Neva Tall Bear. 2013. "Using Undergraduate Researchers to Build Vector and West Nile Virus Surveillance Capacity" International Journal of Environmental Research and Public Health 10, no. 8: 3192-3202. https://doi.org/10.3390/ijerph10083192




