Toxic Metal Concentrations in Cigarettes Obtained from U.S. Smokers in 2009: Results from the International Tobacco Control (ITC) United States Survey Cohort
Abstract
:1. Introduction
2. Experimental Section
3. Results and Discussion
| Metals | Mean (μg·g−1) | Std. Deviation | Range |
|---|---|---|---|
| As | 0.17 | 0.06 | LOD–0.40 |
| Cd | 0.86 | 0.23 | LOD–3.10 |
| Cr (N = 310) | 2.35 | 0.86 | 0.60–7.50 |
| Ni | 2.21 | 0.54 | 0.60–4.40 |
| Pb (N = 310) | 0.44 | 0.24 | LOD–2.40 |
| Pb | Cd | As | Ni | Cr | ||
| Pb | Pearson Correlation | 1 | 0.187 ** | 0.075 | 0.107 | 0.046 |
| Cd | Pearson Correlation | 0.187 ** | 1 | 0.088 | 0.136 * | 0.081 |
| As | Pearson Correlation | 0.075 | 0.088 | 1 | −0.114 * | 0.021 |
| Ni | Pearson Correlation | 0.107 | 0.136 * | −0.114 * | 1 | 0.597 ** |
| Cr | Pearson Correlation | 0.046 | 0.081 | 0.021 | 0.597 ** | 1 |
| Philip Morris | ||||||
| Pb | Pearson Correlation | 1 | 0.108 | 0.225 * | 0.130 | 0.095 |
| Cd | Pearson Correlation | 0.108 | 1 | −0.045 | 0.135 | −0.177 |
| As | Pearson Correlation | 0.225 * | −0.045 | 1 | −0.095 | −0.004 |
| Ni | Pearson Correlation | 0.130 | 0.135 | −0.095 | 1 | 0.522 ** |
| Cr | Pearson Correlation | 0.095 | −0.177 | −0.004 | 0.522 ** | 1 |
| R.J. Reynolds | ||||||
| Pb | Pearson Correlation | 1 | −0.017 | 0.090 | 0.190 * | −0.060 |
| Cd | Pearson Correlation | −0.017 | 1 | −0.037 | −0.217 * | −0.103 |
| As | Pearson Correlation | 0.090 | −0.037 | 1 | 0.060 | 0.095 |
| Ni | Pearson Correlation | 0.190 * | −0.217 * | 0.060 | 1 | 0.546 ** |
| Cr | Pearson Correlation | −0.060 | −0.103 | 0.095 | 0.546 ** | 1 |
| Other | ||||||
| Pb | Pearson Correlation | 1 | 0.275 ** | −0.043 | 0.115 | 0.112 |
| Cd | Pearson Correlation | 0.275 ** | 1 | 0.268 * | 0.236 * | 0.291 ** |
| As | Pearson Correlation | −0.043 | 0.268 * | 1 | −0.172 | 0.080 |
| Ni | Pearson Correlation | 0.115 | 0.236 * | −0.172 | 1 | 0.565 ** |
| Cr | Pearson Correlation | 0.112 | 0.291 ** | 0.080 | 0.565 ** | 1 |

| Pack Number | Pack Name |
|---|---|
| 12 | Doral Ultra Light |
| 27 | Doral Menthol 100 |
| 42 | Misty Menthol Green 120 |
| 54 | Natural American Spirit Light |
| 61 | Basic Light |
| 69 | Marlboro Light |
| 71 | Marlboro Ultra Light 100 |
| 74 | Benson and Hedges Menthol 100 |
| 81 | Marlboro Light 100 |
| 82 | Basic Light |
| 85 | Marlboro Light |
| 100 | Marlboro Light 100 |
| 106 | Basic Light |
| 112 | Marlboro Ultra Light |
| 115 | Basic Ultra Light 100 |
| 118 | Marlboro Light 100 |
| 129 | Montclair Ultra Light |
| 144 | Poker Light 100 |
| 170 | Virginia Slims Luxury Ultra Light 120 |
| 184 | Main Street Menthol Light 100 |
| 214 | Marlboro Ultra Light 100 |
| 216 | Marlboro |
| 222 | Marlboro 100 |
| 246 | Eve Ultra Light Slim 120 |
| 255 | Ace Menthol Light 100 |
| 267 | Merit Ultra Light |
| 269 | Marlboro King |
| 273 | disCOUNT Full Flavor 100 |
| 320 | USA Gold FF 100 |
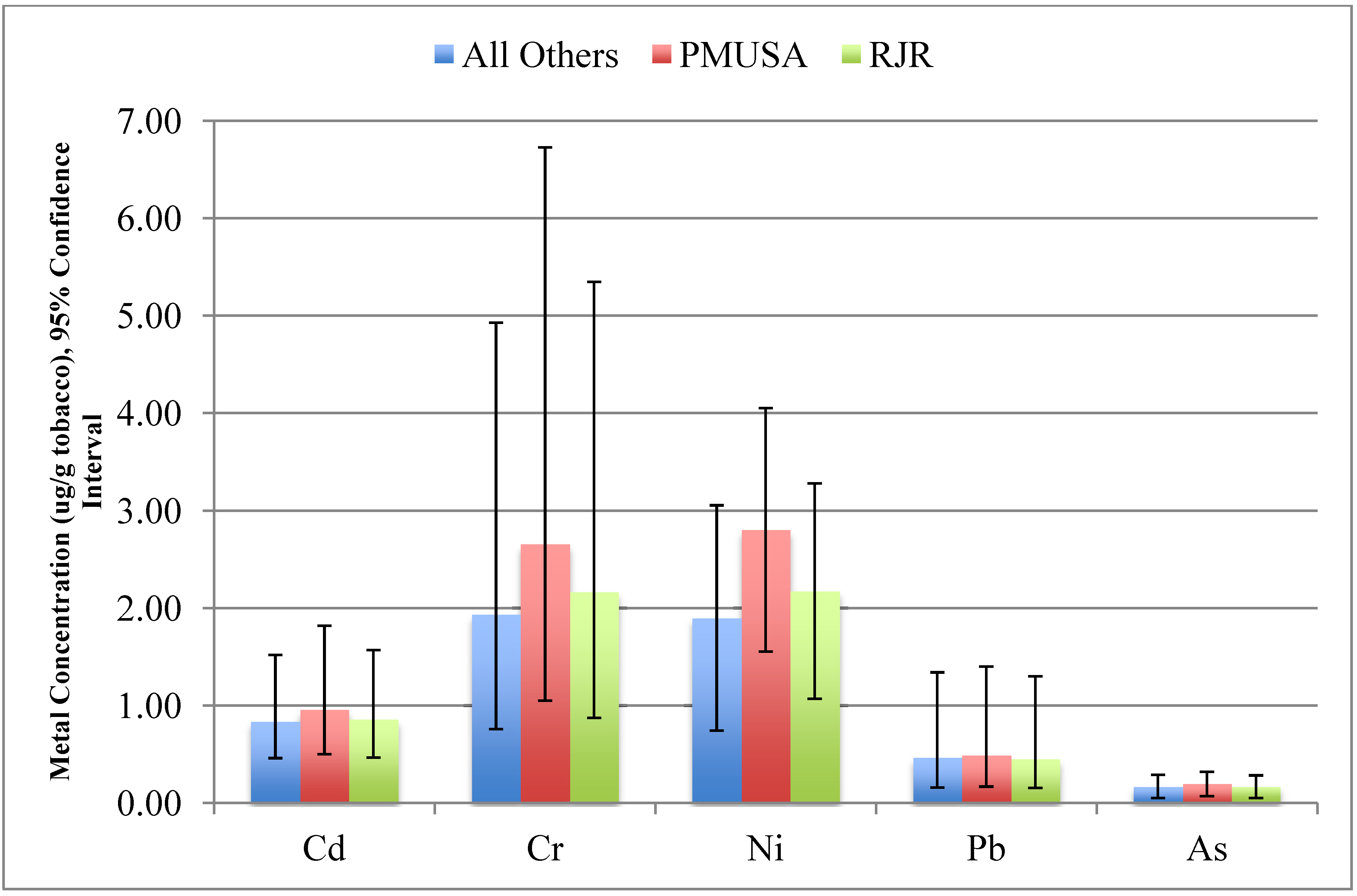
| As | Cd | Cr | Ni | Pb | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| B | SE | p | B | SE | p | B | SE | p | B | SE | p | B | SE | p | |
| (Intercept) | 0.347 | 0.0601 | <0.001 | 0.243 | 0.216 | 0.261 | −0.237 | 0.4311 | 0.583 | 1.92 | 0.5234 | <0.001 | −0.504 | 0.3838 | 0.189 |
| Other Manufacturer | 0.003 | 0.0097 | 0.791 | −0.022 | 0.04 | 0.586 | −0.112 | 0.0571 | 0.05 | −0.277 | 0.0961 | 0.004 | 0.026 | 0.0736 | 0.720 |
| RJR | 0.028 | 0.0151 | 0.06 | 0.111 | 0.0461 | 0.017 | 0.206 | 0.0519 | <0.001 | 0.628 | 0.1609 | <0.001 | 0.075 | 0.102 | 0.463 |
| PMUSA | REF | REF | REF | REF | REF | ||||||||||
| Menthol | −0.005 | 0.01 | 0.612 | 0.008 | 0.0335 | 0.818 | −0.051 | 0.0506 | 0.318 | 0.001 | 0.0991 | 0.99 | 0.197 | 0.1184 | 0.096 |
| Nonmenthol | REF | REF | REF | REF | REF | ||||||||||
| Light/Mild descriptor | 0.006 | 0.0098 | 0.528 | 0.041 | 0.037 | 0.264 | −0.039 | 0.0416 | 0.347 | −0.402 | 0.1723 | 0.02 | −0.003 | 0.073 | 0.966 |
| No descriptor | REF | REF | REF | REF | REF | ||||||||||
| Per cigarette dry weight (g) | 0.014 | 0.0636 | 0.822 | −0.2 | 0.2835 | 0.48 | −0.249 | 0.2655 | 0.348 | −0.638 | 0.4666 | 0.171 | 0.564 | 0.6339 | 0.374 |
| % moisture | −0.013 | 0.0036 | <0.001 | −0.02 | 0.0189 | 0.29 | 0.081 | 0.0279 | 0.004 | 0.057 | 0.0339 | 0.093 | −0.049 | 0.0336 | 0.142 |
| (Scale) | 0.005 | 0.056 | 0.662 | 0.271 | 0.059 | ||||||||||
| % Transfer Range | Median Potential Daily Exposure (IQR) | Range | Cal OEHHA (μg/day, inhalation NSRL a) | |
|---|---|---|---|---|
| As | 0–7% | 0.06 μg (0.05) | 0–0.24 | 0.06 μg/day |
| Cd | 7%–22% | 1.36 μg (0.77) | 0.46–6.48 | 0.05 μg/day |
| Cr | 0.43%–1.74% | 0.26 μg (0.18) | 0.06–1.11 | 0.001 μg/day Cr(VI) |
| Ni | 0.1%–2.4% | 0.30 μg (0.15) | 0.07–1.19 | – |
| Pb | 0.16%–6.3% | 0.14 μg (0.10) | 0–1.58 | 0.5 μg/day (MADL) |
4. Conclusions
Acknowledgements
Conflicts of Interest
References
- Family Smoking Prevention and Tobacco Control Act. Public Law 111–31. 22 June 2009. Available online: http://www.gpo.gov/fdsys/pkg/PLAW-111publ31/pdf/PLAW-111publ31.pdf (accessed on 2 December 2013).
- U.S. Department of Health and Human Services. How Tobacco Smoke Causes Disease: The Biology and Behavioral Basis for Smoking-Attributable Disease: A Report of the Surgeon General; U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health: Atlanta, GA, USA, 2010. [Google Scholar]
- Talhout, R.; Schulz, T.; Florek, E.; Benthem, J.V.; Wester, P.; Opperhuizen, A. Hazardous compounds in tobacco smoke. Int. J. Environ. Res. Public Health 2011, 8, 613–628. [Google Scholar] [CrossRef]
- Rodgman, A.; Perfetti, T.A. The Chemical Components of Tobacco and Tobacco Smoke; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Tobacco Smoke and Involuntary Smoking. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; World Health Organization, International Agency for Research on Cancer: Lyon, France, 2004; Volume 83. [Google Scholar]
- Dorne, J.L.; Kass, G.E.; Bordajandi, L.R.; Amzal, B.; Bertelsen, U.; Castoldi, A.F.; Heppner, C.; Eskola, M.; Fabiansson, S.; Ferrari, P.; et al. Human risk assessment of heavy metals: Principles and applications. Met Ions Life Sci. 2011, 8, 27–60. [Google Scholar]
- Gidlow, D.A. Lead toxicity. Occup. Med. (Lond). 2004, 54, 76–81. [Google Scholar] [CrossRef]
- Fresquez, M.R.; Pappas, R.S.; Watson, C.H. Establishment of Toxic Metal Reference Range in Tobacco from U.S. Cigarettes. J. Anal. Toxicol. 2013, 37, 298–304. [Google Scholar] [CrossRef]
- Chiba, M.; Masironi, R. Toxic and Trace-Elements in Tobacco and Tobacco-Smoke. Bull. WHO 1992, 70, 269–275. [Google Scholar]
- European Commission. Ambient Air Pollution by AS, CD and NI compounds: Position Paper—Final Verision; LUA NRW: Essen, Germany, 2001. [Google Scholar]
- Lugon-Moulin, N.; Martin, F.; Krauss, M.R.; Ramey, P.B.; Rossi, L. Arsenic Concentration in Tobacco Leaves: A Study on Three Commercially Important Tobacco (Nicotiana tabacum L.) Types. Water Air Soil Pollut. 2008, 192, 315–319. [Google Scholar] [CrossRef]
- Pappas, R.S. Toxic elements in tobacco and in cigarette smoke: Inflammation and sensitization. Metallomics 2011, 3, 1181–1198. [Google Scholar] [CrossRef]
- Stojanović, D.; Nikić, D.; Lazarević, K. The level of nickel in smoker's blood and urine. Cent. Eur. J. Public Health. 2004, 12, 187–189. [Google Scholar]
- Galazyn-Sidorczuk, M.; Brzóska, M.M.; Moniuszko-Jakoniuk, J. stimation of Polish cigarettes contamination with cadmium and lead, and exposure to these metals via smoking. Environ. Monit. Assess. 2008, 137, 481–493. [Google Scholar] [CrossRef]
- Richter, P.A.; Bishop, E.E.; Wang, J.; Swahn, M.H. Tobacco smoke exposure and levels of urinary metals in the U.S. youth and adult population: the National Health and Nutrition Examination Survey (NHANES) 1999–2004. Int. J. Environ. Res. Public Health. 2009, 6, 1930–1946. [Google Scholar] [CrossRef]
- Tellez-Plaza, M.; Navas-Acien, A.; Caldwell, K.L.; Menke, A.; Muntner, P.; Guallar, E. Reduction in cadmium exposure in the United States population, 1988–2008: The contribution of declining smoking rates. Environ. Health Perspect. 2012, 120, 204–209. [Google Scholar]
- Marano, K.M.; Naufal, Z.S.; Kathman, S.J.; Bodnar, J.A.; Borgerding, M.F.; Garner, C.D.; Wilson, C.L. Cadmium exposure and tobacco consumption: Biomarkers and risk assessment. Regul. Toxicol. Pharmacol. 2012, 64, 243–52. [Google Scholar] [CrossRef]
- Serdar, M.A.; Akin, B.S.; Razi, C.; Akin, O.; Tokgoz, S.; Kenar, L.; Aykut, O. The correlation between smoking status of family members and concentrations of toxic trace elements in the hair of children. Biol. Trace Elem. Res. 2012, 148, 11–17. [Google Scholar] [CrossRef]
- Marano, K.M.; Naufal, Z.S.; Kathman, S.J.; Bodnar, J.A.; Borgerding, M.F.; Wilson, C.L. Arsenic exposure and tobacco consumption: Biomarkers and risk assessment. Regul. Toxicol. Pharmacol. 2012, 64, 225–232. [Google Scholar] [CrossRef]
- Liu, C.; Wright, C.G.; McAdam, K.G.; Taebunpakul, S.; Heroult, J.; Braybrook, J.; Goenaga-Infante, H. Arsenic Speciation in Tobacco and Cigarette Smoke. Beiträge zur Tabakforschung International (Contrib. Tob. Res.) 2012, 25, 375–380. [Google Scholar]
- Viana, G.F.; Garcia, K.S.; Menezes-Filho, J.A. Assessment of carcinogenic heavy metals levels in Brazilian cigarettes. Environ. Monit.Assess. 2001, 181, 255–265. [Google Scholar]
- O’Connor, R.J.; Li, Q.; Stephens, W.E.; Hammond, D.; Elton-Marshall, T.; Cummings, K.M.; Giovino, G.A.; Fong, G.T. Cigarettes sold in China: Design, emissions and metals. Tob. Control. 2010, 19, i47–i53. [Google Scholar] [CrossRef]
- Stephens, W.E.; Calder, A.; Newton, J. Source and health implications of high toxic metal concentrations in illicit tobacco products. Environ. Sci. Technol. 2005, 39, 479–488. [Google Scholar] [CrossRef]
- Pappas, R.S.; Polzin, G.M.; Watson, C.H.; Ashley, D.L. Cadmium, lead, and thallium in smoke particulate from counterfeit cigarettes compared to authentic US brands. Food Chem. Toxicol. 2007, 45, 202–209. [Google Scholar] [CrossRef]
- Wertz, M.S.; Kyriss, T.; Paranjape, S.; Glantz, S.A. The toxic effects of cigarette additives. Philip Morris’ project mix reconsidered: an analysis of documents released through litigation. PLoS Med. 2011, 8. [Google Scholar] [CrossRef]
- Deyton, L.; Sharfstein, J.; Hamburg, M. Tobacco product regulation—A public health approach. N. Engl. J. Med. 2010, 362, 1753–1756. [Google Scholar] [CrossRef]
- Deyton, L.R. FDA tobacco product regulations: A powerful tool for tobacco control. Public Health Reports 2011, 126, 167–169. [Google Scholar]
- Food and Drug Administration. Harmful and Potentially Harmful Constituents in Tobacco Products and Tobacco Smoke; Established List; FDA–2012–N–0143; FDA: Rockville, MD, USA, 2012. [Google Scholar]
- Capehart, T. Tobacco Industry Downsizing, Restructuring. USDA Economic Research Service. Agricultural Outlook. January–February 2002. Available online: http://www.ers.usda.gov/publications/agoutlook/jan2002/ao288c.pdf (accessed on 2 December 2013).
- Fix, B.V.; O’Connor, R.J.; Fond, G.T.; Borland, R.; Cummings, K.M.; Hyland, A. Smokers’ Reactions to FDA regulation of tobacco products: Findings from the 2009 ITC United States survey. BMC Public Health 2011, 11. [Google Scholar] [CrossRef]
- Fong, G.T.; Cummings, K.M.; Borland, R.; Hastings, G.; Hyland, A.; Giovino, G.A.; Hammond, D.; Thompson, M.E. The conceptual framework of the International Tobacco Control (ITC) Policy Evaluation Project. Tob. Control 2006, 15, iii3–iii11. [Google Scholar]
- Thompson, M.E.; Fong, G.T.; Hammond, D.; Boudreau, C.; Driezen, P.; Hyland, A.; Borland, R.; Cummings, K.M.; Hastings, G.B.; Siahpush, M.; et al. Methods of the International Tobacco Control (ITC) Four Country Survey. Tob. Control. 2006, 15, iii12–iii18. [Google Scholar]
- O’Connor, R.J.; Caruso, R.V.; Borland, R.; Cummings, K.M.; Bansal-Travers, M.; Fix, B.V.; King, B.; Hammond, D.; Fong, G.T. Relationship of cigarette-related perceptions to cigarette design features: Findings from the 2009 ITC United States Survey. Nicotine Tob. Res. 2013, in press. [Google Scholar]
- O’Connor, R.J.; Hammond, D.; McNeill, A.; King, B.; Kozlowski, L.T.; Giovino, G.A.; Cummings, K.M. How do different cigarette design features influence the standard tar yields of popular cigarette brands sold in different countries? Tob. Control. 2008, 17 (Suppl 1), i1–i5. [Google Scholar]
- O’Connor, R.J.; Wilkins, K.J.; Caruso, R.V.; Cummings, K.M.; Kozlowski, L.T. Cigarette characteristic and emission variations across high-, middle- and low-income countries. Public Health. 2010, 124, 667–674. [Google Scholar] [CrossRef]
- Caruso, R.V.; O’Connor, R.J. Cigarette design features in low-, middle-, and high-income countries. J. Environ. Public Health 2012. [Google Scholar] [CrossRef]
- Stephens, W.E.; Calder, A. Analysis of non-organic elements in plant foliage using polarised X-ray fluorescence spectrometry. Anal. Chim. Acta 2004, 527, 89–96. [Google Scholar] [CrossRef]
- Proposition 65 No Significant Risk Level (NSRLs) for Carcinogens and Maximum Allowable Dose Levels (MADLs) for Chemicals Causing Reproductive Toxicity. Available online: http://oehha.ca.gov/prop65/pdf/safeharbor070113.pdf (accessed on 28 November 2013).
- Hammond, D.; O’Connor, R.J. Constituents in tobacco and smoke emissions from Canadian cigarettes. Tob. Control. 2008, 17 (Suppl 1), i24–i31. [Google Scholar]
- Hecht, S.S. Research opportunities related to establishing standards for tobacco products under the Family Smoking Prevention and Tobacco Control Act. Nicotine Tob. Res. 2012, 14, 18–28. [Google Scholar] [CrossRef]
- Hecht, S.S. Lung carcinogenesis by tobacco smoke. Int. J. Cancer. 2012, 131, 2724–2732. [Google Scholar] [CrossRef]
- Kuo, C.Y.; Wong, R.H.; Lin, J.Y.; Lai, J.C.; Lee, H. Accumulation of chromium and nickel metals in lung tumors from lung cancer patients in Taiwan. J. Toxicol. Environ. Health A. 2006, 69, 1337–1344. [Google Scholar] [CrossRef]
- Khlifi, R.; Hamza-Chaffai, A. Head and neck cancer due to heavy metal exposure via tobacco smoking and professional exposure: A review. Toxicol. Appl. Pharmacol. 2010, 248, 71–88. [Google Scholar] [CrossRef]
- Adams, S.V.; Passarelli, M.N.; Newcomb, P.A. Cadmium exposure and cancer mortality in the Third National Health and Nutrition Examination Survey cohort. Occup. Environ. Med. 2012, 69, 153–156. [Google Scholar] [CrossRef]
- Catalani, S.; De Palma, G.; Mangili, A.; Apostoli, P. Metallic elements in lung tissues: Results of a meta-analysis. Acta. Biomed. 2008, 79 (Suppl 1), 52–63. [Google Scholar]
- Khlifi, R.; Olmedo, P.; Gil, F.; Hammami, B.; Chakroun, A.; Rebai, A.; Hamza-Chaffai, A. Arsenic, cadmium, chromium and nickel in cancerous and healthy tissues from patients with head and neck cancer. Sci. Total Environ. 2013. [Google Scholar] [CrossRef]
- Jones, M.R.; Apelberg, B.J.; Tellez-Plaza, M.; Samet, J.M.; Navas-Acien, A. Menthol cigarettes, race/ethnicity, and biomarkers of tobacco use in U.S. adults: the 1999–2010 National Health and Nutrition Examination Survey (NHANES). Cancer Epidemiol. Biomarkers Prev. 2013, 22, 224–232. [Google Scholar]
- WHO Study Group on Tobacco Product Regulation. Report on the Scientific Basis of Tobacco Product Regulation; WHO Technical Report Series; World Health Organization: Geneva, Switzerland, 2012; pp. 25–28. [Google Scholar]
- Kozlowski, L.T. The proposed tobacco regulation: the triumph of hope over experience? Tob. Control 2008, 17, 74–75. [Google Scholar] [CrossRef]
- Givel, M. In search of the less hazardous cigarette. Int. J. Health Serv. 2011, 41, 77–94. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Caruso, R.V.; O'Connor, R.J.; Stephens, W.E.; Cummings, K.M.; Fong, G.T. Toxic Metal Concentrations in Cigarettes Obtained from U.S. Smokers in 2009: Results from the International Tobacco Control (ITC) United States Survey Cohort. Int. J. Environ. Res. Public Health 2014, 11, 202-217. https://doi.org/10.3390/ijerph110100202
Caruso RV, O'Connor RJ, Stephens WE, Cummings KM, Fong GT. Toxic Metal Concentrations in Cigarettes Obtained from U.S. Smokers in 2009: Results from the International Tobacco Control (ITC) United States Survey Cohort. International Journal of Environmental Research and Public Health. 2014; 11(1):202-217. https://doi.org/10.3390/ijerph110100202
Chicago/Turabian StyleCaruso, Rosalie V., Richard J. O'Connor, W. Edryd Stephens, K. Michael Cummings, and Geoffrey T. Fong. 2014. "Toxic Metal Concentrations in Cigarettes Obtained from U.S. Smokers in 2009: Results from the International Tobacco Control (ITC) United States Survey Cohort" International Journal of Environmental Research and Public Health 11, no. 1: 202-217. https://doi.org/10.3390/ijerph110100202




