1. Introduction
Adult female mosquitoes are vectors of numerous pathogens that cause diseases of significance to global health. Notably, hundreds of millions of people are infected with mosquito-borne malaria and dengue fever each year, leading to over 500,000 deaths [
1,
2]. Moreover, chikungunya fever and Zika virus, which were historically limited to parts of Africa and Asia, have recently emerged into global threats with unprecedented autochthonous transmission by mosquitoes in the Western hemisphere [
3,
4]. Effective, low-cost therapeutics and/or vaccines for treating and preventing these diseases have either not been developed or are not widely-available. Thus, the primary—and only universal—means to limit the spread of mosquito-borne diseases is to implement control of the mosquito vector, especially when a previously neglected arbovirus is emerging (e.g., Zika).
Chemical control of mosquitoes with insecticides remains a cornerstone to integrated vector management, especially in regions of the world where biological and genetic approaches are cost prohibitive or not logistically feasible. Most insecticides used for mosquito control target the nervous system, such as pyrethroids, carbamates, organophosphates, and organochlorides. Although these neurotoxins are highly effective at killing mosquitoes, the overuse of a limited number of active compounds has exerted a strong, selective pressure for traits that make mosquitoes resistant to conventional insecticides (e.g., knockdown resistance mutations, elevated detoxification mechanisms) [
5], analogous to the evolution of antibiotic resistance in pathogenic bacteria. Thus, the number of effective chemical tools available for mosquito control is diminishing, presenting an emerging challenge to the control of mosquito-borne diseases. In order to replenish our chemical “tool box”, mitigate insecticide resistance, and enhance our vector control capabilities, it is necessary to develop insecticides with novel mechanisms of action [
6,
7].
A physiological system in mosquitoes (and other insect pests/vectors) that has not been exploited to date for insecticide development is the renal excretory system. In the adult female mosquito, the only life stage that feeds on the blood of—and transmits pathogens to—vertebrate hosts, the Malpighian tubules play especially important roles in excreting (1) the excess water and ions that are absorbed into the extracellular fluid (hemolymph) after engorging on blood, and (2) the metabolic wastes that enter the hemolymph during the digestion and metabolism of blood (e.g., nitrogenous wastes). Thus, the Malpighian tubules offer a potential physiological target to exploit for developing insecticides that not only have novel mechanisms of action, but may also be highly selective to hematophagous adult females. The following pages highlight recent efforts by our group to understand the basic molecular mechanisms of Malpighian tubule function in adult female mosquitoes with the ultimate aim of developing novel mosquitocides.
2. Mosquito Malpighian Tubules
The morphology and physiology of mosquito Malpighian tubules have been the focus of recent comprehensive reviews [
8,
9,
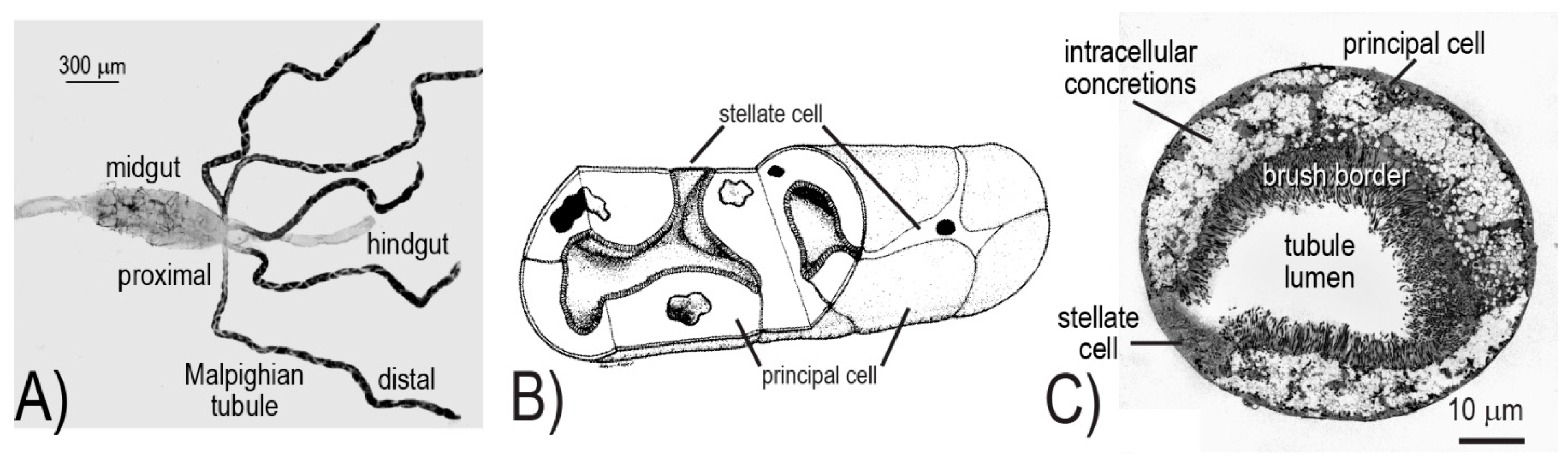
10]. In brief, the five Malpighian tubules together with the hindgut (i.e., ileum and rectum) form the renal excretory system of mosquitoes (
Figure 1A). The Malpighian tubules produce urine via a “two-step” process of transepithelial fluid secretion. That is, ions (primarily Na
+, K
+ and Cl
−) are actively transported by the tubule epithelial cells from the hemolymph to the tubule lumen, thereby generating an osmotic gradient for water to follow. The resulting urine is isosmotic to the hemolymph. The hindgut receives and modifies the urine (e.g., solute and/or water reabsorption), before excreting it from the mosquito via muscular contractions.
The cellular architecture of the Malpighian tubule epithelium is rather simple, consisting of only two different cell types: principal and stellate (
Figure 1B,C). The principal cells are large, thick, mitochondrion-rich cells with an elaborate apical (luminal) brush border; they are fusiform in shape and fold upon themselves to form the tubule lumen (
Figure 1B,C). On the other hand, stellate cells are small, thin, mitochondrion-poor cells with a highly in-folded basolateral (serosal) membrane; they possess a nucleated cell body with three to four arm-like projections that intercalate between principal cells, resulting in a “star-like” appearance (
Figure 1B,C).
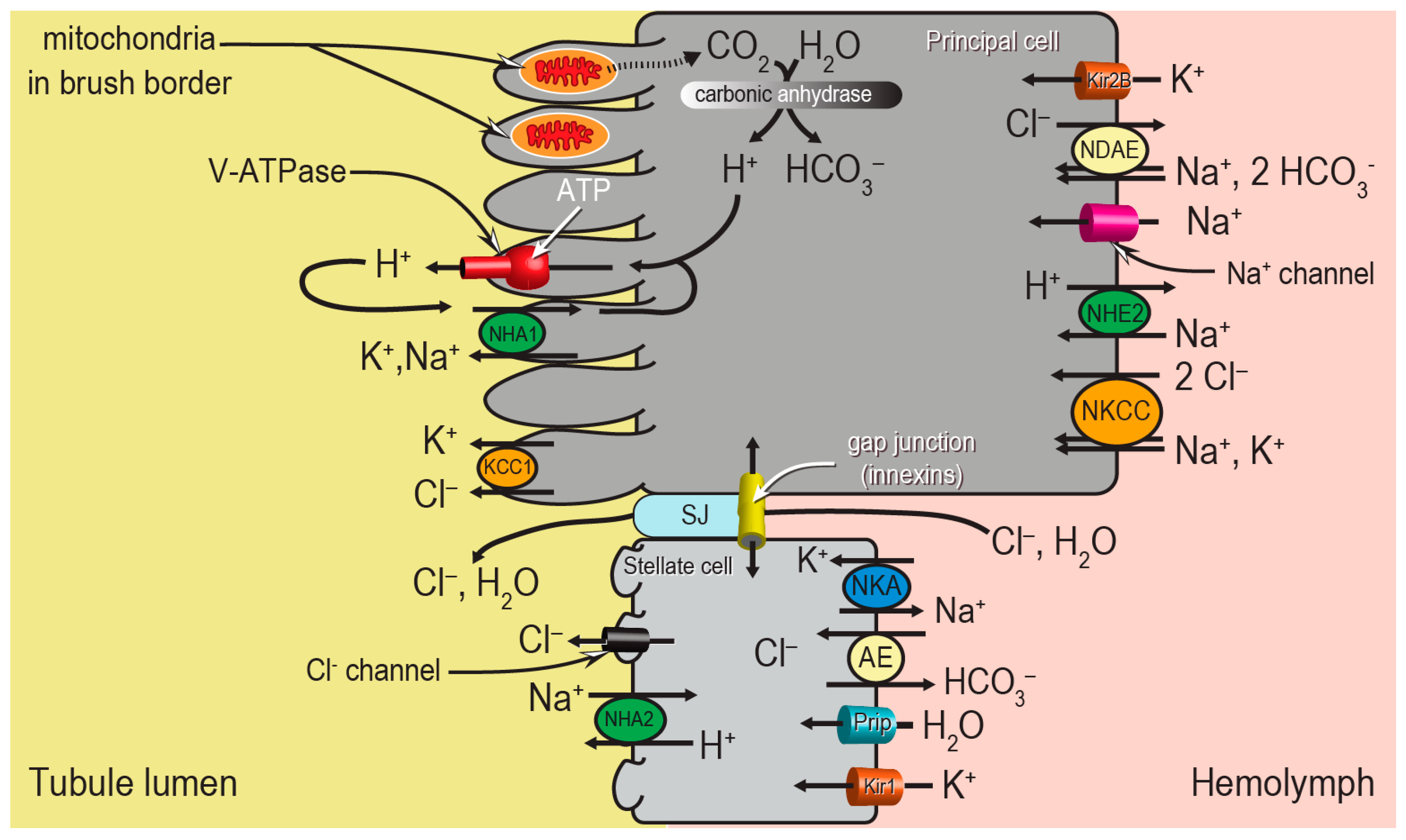
Despite the apparent morphological simplicity of the Malpighian tubule epithelium, the molecular mechanisms behind the “two-step” process of fluid secretion are complex, requiring the coordination of several ion transport mechanisms between the two cell types (
Figure 2). Principal cells generate the primary electrochemical gradient that drives the secondary transport of ions and water from the hemolymph to the tubule lumen. In particular, a V-type H
+-ATPase (V-ATPase) in the brush border pumps protons from the cytosol into the tubule lumen, thereby establishing an inside-negative voltage across both the apical and basolateral membranes of the epithelium. A Na
+-K
+-ATPase (NKA) on the basolateral membrane of stellate cells may play an accessory role to the V-ATPase. The negative membrane voltages drive ion uptake from the hemolymph across the basolateral membrane through (1) ion channels (e.g., Kir1, Kir2B, Na
+ channel), and (2) potentially electrogenic transporters in the apical membranes (e.g., NHA1, NHA2). In addition to the conductive pathways, several electroneutral mechanisms for ion transport driven by chemical gradients are present in the basolateral and apical membranes, such as cation chloride cotransporters (e.g., Na
+, K
+, Cl
− cotransporter (NKCC), K
+, Cl
− cotransporter (KCC1)), Na
+/H
+ exchangers (e.g., NHE2), and anion exchangers (e.g., Cl
−/HCO
3− anion exchanger (AE), Na
+-driven anion exchanger (NDAE)). The combined actions of the conductive and electroneutral ion transport mechanisms contribute to the movements of NaCl and KCl from the hemolymph into the tubule lumen, which provides the osmotic gradient for water to follow. This most likely occurs through stellate cells via aquaporin water channels (e.g., Prip) and/or the paracellular pathway via septate junctions (SJ) (
Figure 2).
In theory, any of the above molecular mechanisms represent potential targets to exploit for disrupting urine production and thereby osmotic/ionic homeostasis in mosquitoes. For adult female mosquitoes, this disruption could be especially debilitating after engorging on blood, when they ingest over the equivalent of their body mass. In response to this engorgement, the Malpighian tubules mediate a pronounced diuresis that eliminates ~40% of the water and Na
+, ~145% of the K
+, and ~60% of the Cl
− from the ingested blood plasma within 2 h [
13]. Moreover, limiting the capacity of Malpighian tubules to produce urine after a blood meal could dampen their capacity to excrete metabolic wastes associated with blood digestion (e.g., heme, nitrogenous wastes), which may lead to hemolymph poisoning and/or impaired egg production. It is with this vision in mind that motivated a collaborative effort by the Piermarini (The Ohio State University), Beyenbach (Cornell University), and Denton (Vanderbilt University) laboratories to identify K
+ channels expressed in the Malpighian tubules of adult female mosquitoes and develop small-molecule inhibitors of these channels to disrupt the capacity of Malpighian tubules to produce urine, with the ultimate aim of disrupting K
+ and fluid homeostasis in mosquitoes.
3. Targeting Potassium Channels in Mosquito Malpighian Tubules
Several lines of evidence strongly support the hypothesis that inward rectifier K
+ (Kir) channels are major routes of K
+ uptake across the basolateral membrane of mosquito Malpighian tubules, making them potential molecular targets for disrupting urine production. In isolated Malpighian tubules of the yellow fever mosquito
Aedes aegypti, the Beyenbach laboratory has shown that barium, a canonical blocker of Kir channels, inhibits (1) over 50% of the transepithelial secretion of K
+ and fluid, and (2) ~60% of the basolateral membrane conductance [
14,
15]. Moreover, the basolateral membrane of principal cells exhibits a cation permeability sequence (Tl
+ >> K
+ > Rb
+ >> NH
4+) consistent with the presence of Kir channels [
16].
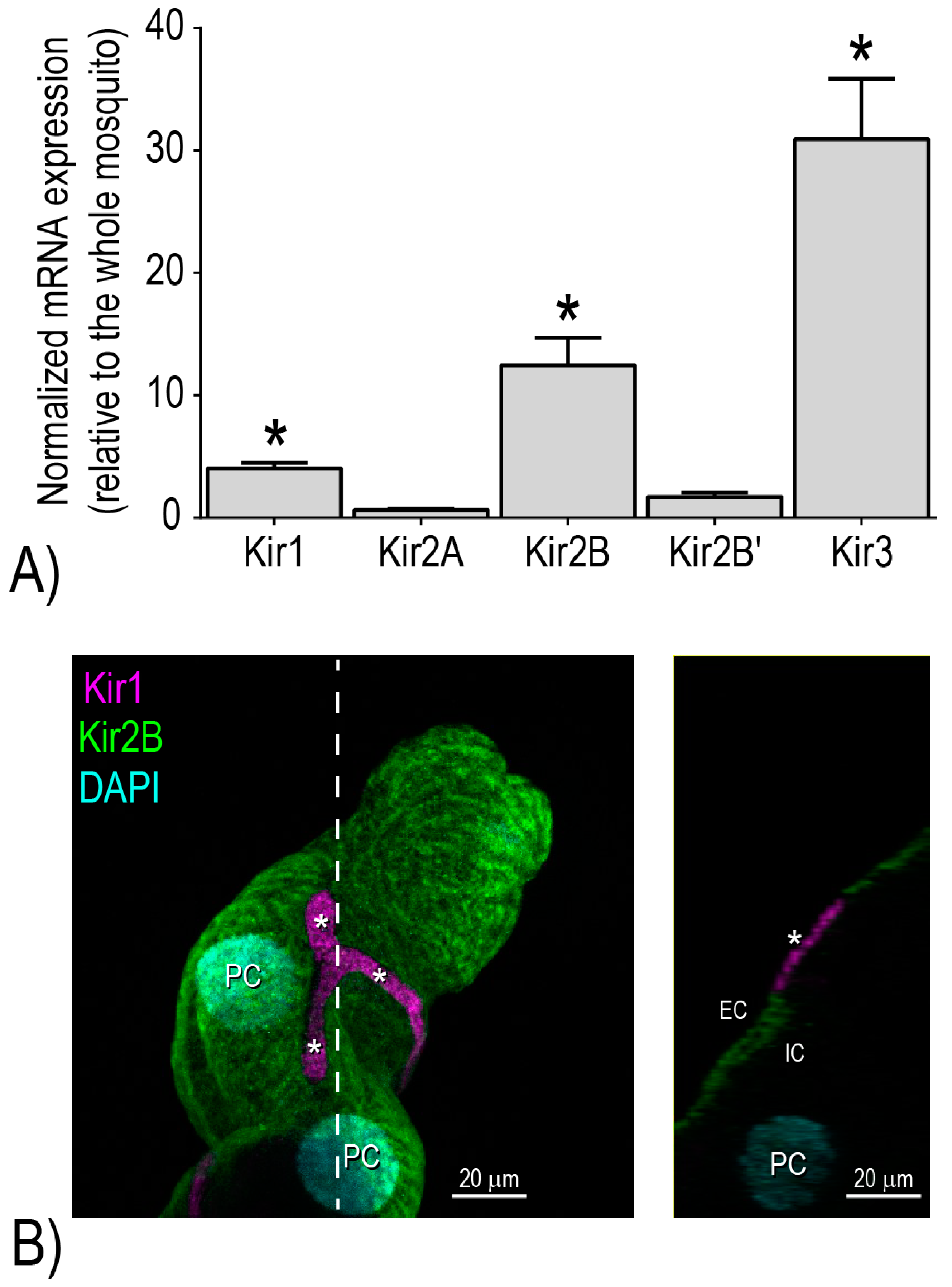
Notably, we have shown that the Malpighian tubules of adult female
Ae. aegypti are characterized by the enriched expression of three distinct mRNAs encoding Kir channel subunits:
AeKir1,
AeKir2B, and
AeKir3 [
16] (
Figure 3A). When expressed heterologously in
Xenopus oocytes,
AeKir1 and
AeKir2B each form functional channels mediating barium-inhibitable, inward-rectifying K
+ currents [
16]. Moreover, immunolabeling studies of Malpighian tubules localized
AeKir1 and
AeKir2B to the basolateral membranes of stellate and principal cells, respectively [
17] (
Figure 3B). On the other hand,
AeKir3 does not form functional K
+ channels in the plasma membrane when heterologously-expressed in
Xenopus oocytes [
16] or HEK-293 cells (Denton, personal observation), and its immunoreactivity localizes to intracellular compartments of principal and stellate cells in Malpighian tubules [
17]. Thus,
AeKir1 and
AeKir2B appear to be the best candidates for mediating the barium-sensitive transepithelial transport of K
+ and fluid in mosquito Malpighian tubules.
To further explore this hypothesis, the Denton laboratory developed a thallium (Tl
+) flux assay for
AeKir1 (expressed heterologously in HEK-293 cells) to allow for the high-throughput discovery of novel small-molecule inhibitors of mosquito Kir channels [
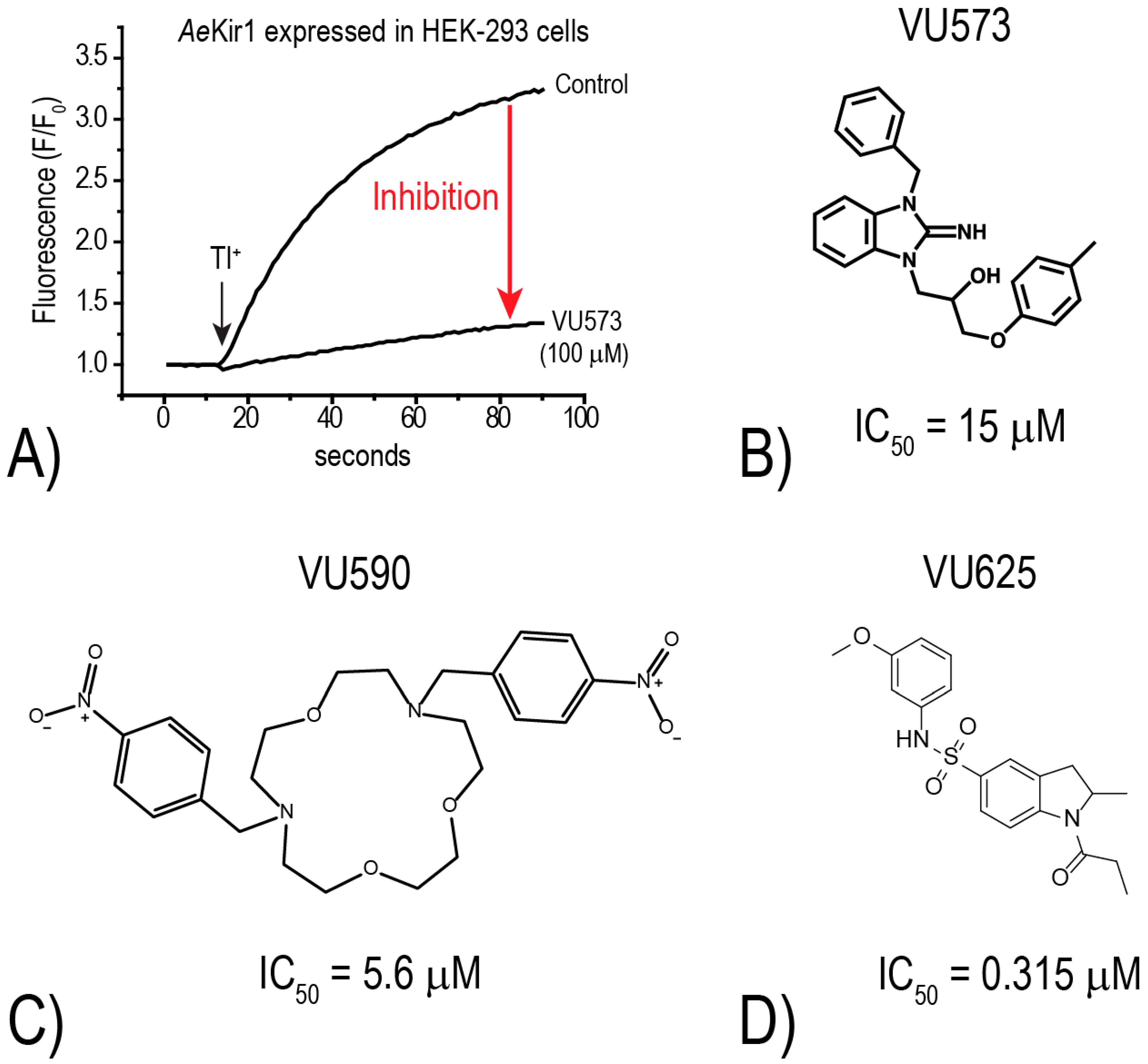
18]. The assay utilizes an intracellular, Tl
+-sensitive fluorescent dye and follows the principle that Kir channels mediate the uptake of Tl
+ into cells. Thus, upon the addition of Tl
+ to the extracellular bath of cells expressing functional Kir channels, intracellular fluorescence increases as Tl
+ moves through the Kir channel pore (e.g., “Control” in
Figure 4A). In the presence of a small molecule inhibitor, the rate of fluorescence increase is reduced (e.g., “VU573” in
Figure 4A). Using this Tl
+-flux assay, we have screened over 75,000 small molecules from the Vanderbilt Institute of Chemical Biology library against mosquito Kir1 and identified over 300 potential inhibitors. To date, we have focused most of our attention on three small molecule inhibitors of
AeKir1: VU573, VU590, and VU625 (
Figure 4B–D). Intriguingly, each of these
AeKir1 inhibitors has a unique effect on
AeKir2B (expressed heterologously in
Xenopus oocytes). That is, the K
+ currents mediated by
AeKir2B are (1) stimulated by VU573, (2) not affected by VU590, and (3) are inhibited by VU625 [
19,
20].
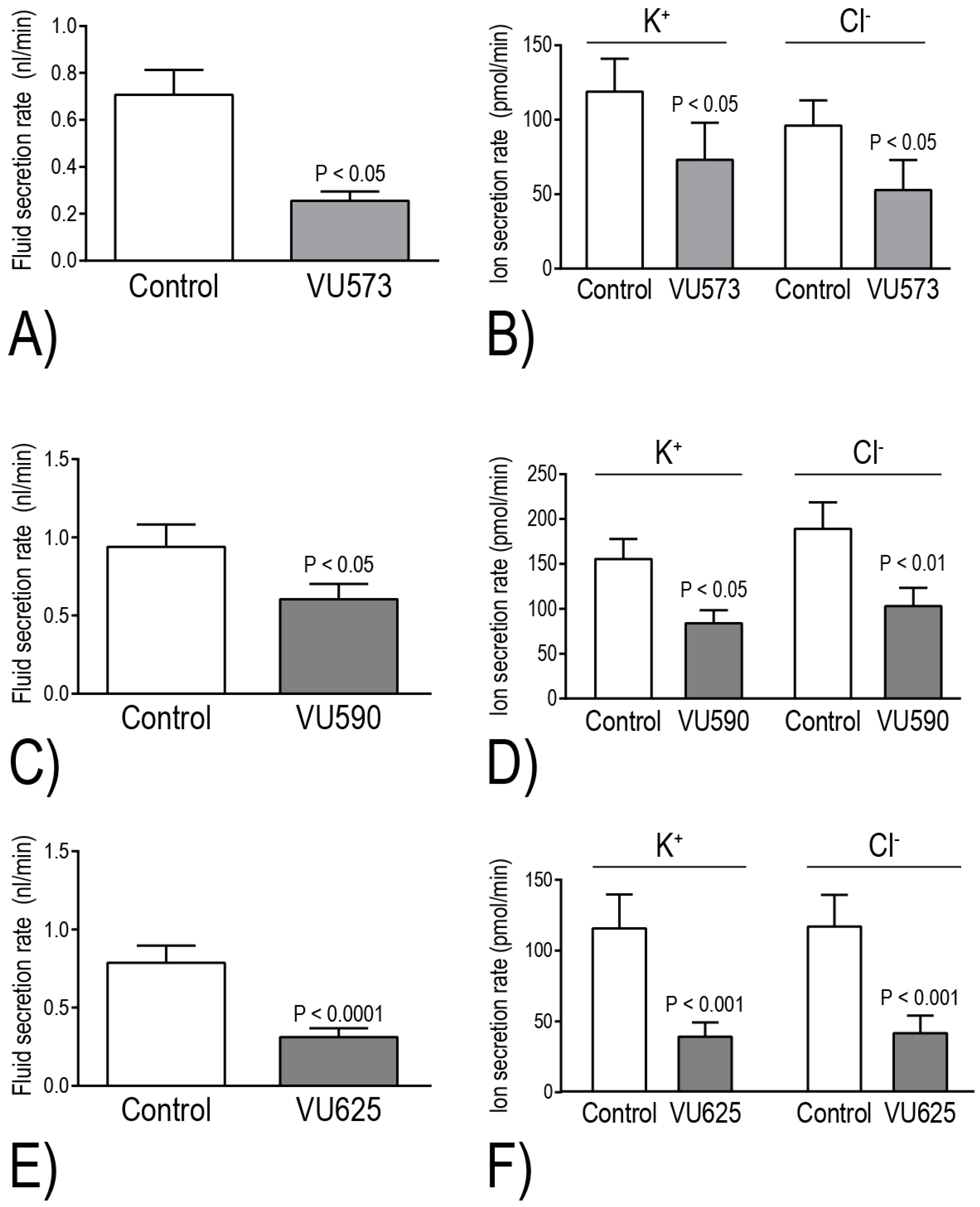
The Beyenbach laboratory has evaluated the effects of VU573, VU590, and VU625 on isolated Malpighian tubule function in vitro via the Ramsay assay of fluid secretion. Within 2 h of adding one of these molecules to the peritubular bath, the secretion of fluid and KCl is reduced by ~40%–60%, consistent with the inhibition of basolateral Kir channels [
17,
18,
21] (
Figure 5). Taking into consideration the aforementioned differential pharmacological effects of the small molecules on
AeKir1 vs.
AeKir2B, it was possible to dissect the relative contributions of each Kir channel to transepithelial K
+ secretion in isolated Malpighian tubules. Notably, VU590 and VU625 decrease transepithelial K
+ secretion by 46% and 66%, respectively [
17] (
Figure 5D,F). These data indicate that
AeKir1 in stellate cells and
AeKir2B in principal cells respectively contribute to ~50% and 20% of the transepithelial K
+ transport. Thus, the inhibition of at least
AeKir1 can potentially have major impacts on mosquito urinary K
+ excretion and thereby hemolymph K
+ homeostasis.
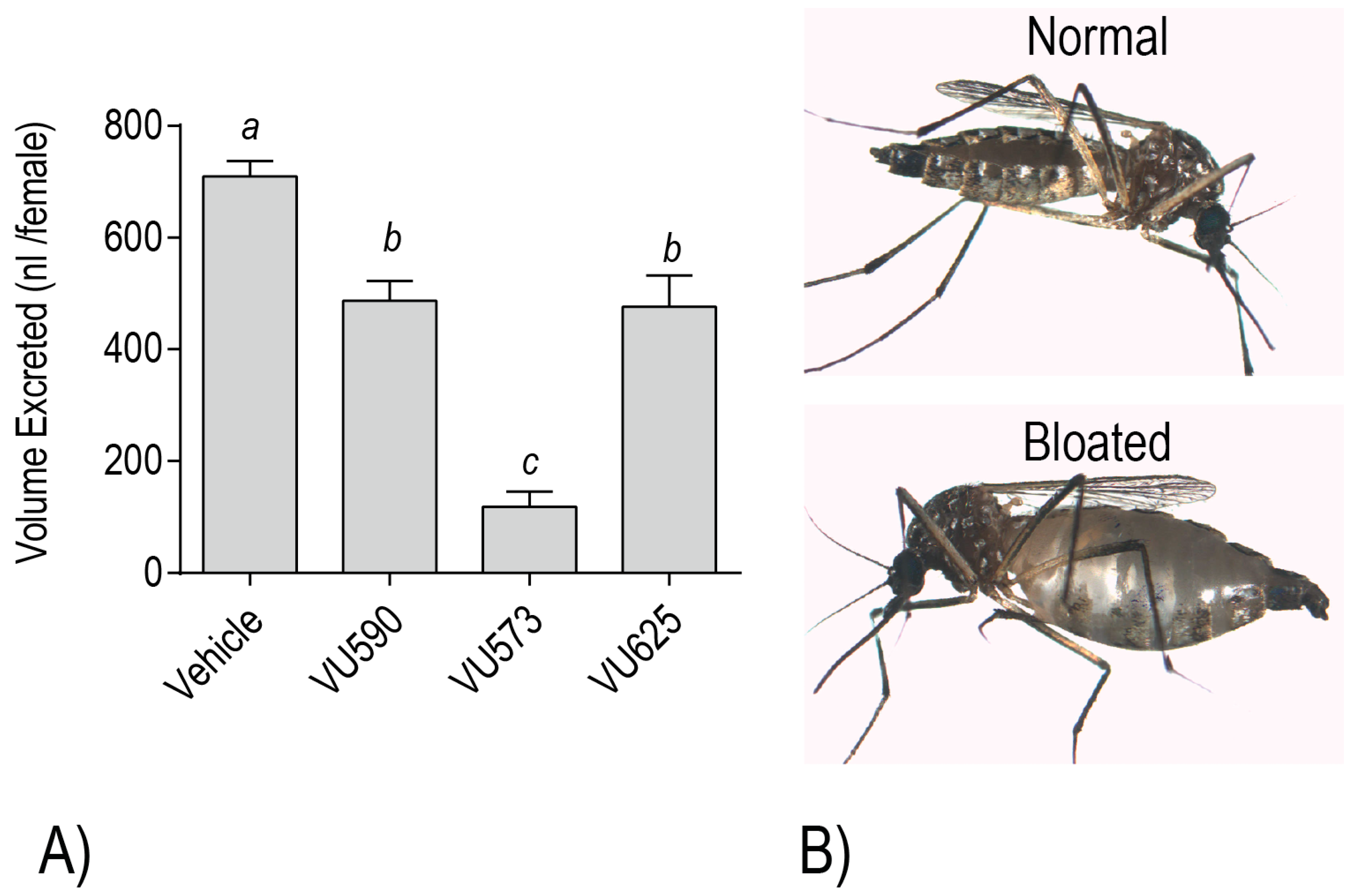
To further test this possibility, we evaluated the effects of VU573, VU590, and VU625 on mosquito behavior/survival and excretory function. Notably, injection of each small molecule into the hemolymph of adult female
Ae. aegypti elicits dose-dependent toxic effects within 24 h, manifested as death or a loss of flight [
18,
19,
20]. Each of the molecules also reduces the whole mosquito capacity for diuresis, consistent with an inhibitory effect on the renal excretory system [
18,
19,
20,
21] (
Figure 6A). Moreover, mosquitoes treated with VU573 are more susceptible to a hemolymph load of K
+, which is likely a consequence of a decreased capacity for renal K
+ excretion [
18,
21]. Among the more dramatic renal effects that manifest in adult female mosquitoes after hemolymph injection of VU573 or VU590 is extreme abdominal swelling, presumably due to extracellular fluid retention associated with impaired renal function [
18,
20] (
Figure 6B). In some cases, we have observed that the swelling causes a physical rupturing of the abdominal wall (Piermarini, unpublished observations).
To confirm that the above effects on Malpighian tubules and mosquitoes were specific to the inhibition of
AeKir1, we developed so-called “inactive” analogs of each molecule that do not inhibit the activity of
AeKir1 in vitro. Importantly, the inactive analogs neither inhibit fluid/K
+ secretion in isolated Malpighian tubules nor exhibit toxic effects on adult female mosquitoes when injected into the hemolymph [
17,
18,
19,
20,
21]. Taken together, the above in vitro and in vivo results indicate that the inhibition of
AeKir1 disrupts K
+ and fluid secretion at the level of the Malpighian tubules, leading to disruptions of hemolymph K
+ and fluid homeostasis at the level of the whole mosquito. In other words, small molecule inhibitors of
AeKir1 elicit renal failure in mosquitoes, suggesting that they have potential for development into mosquitocides with novel mechanisms of action.
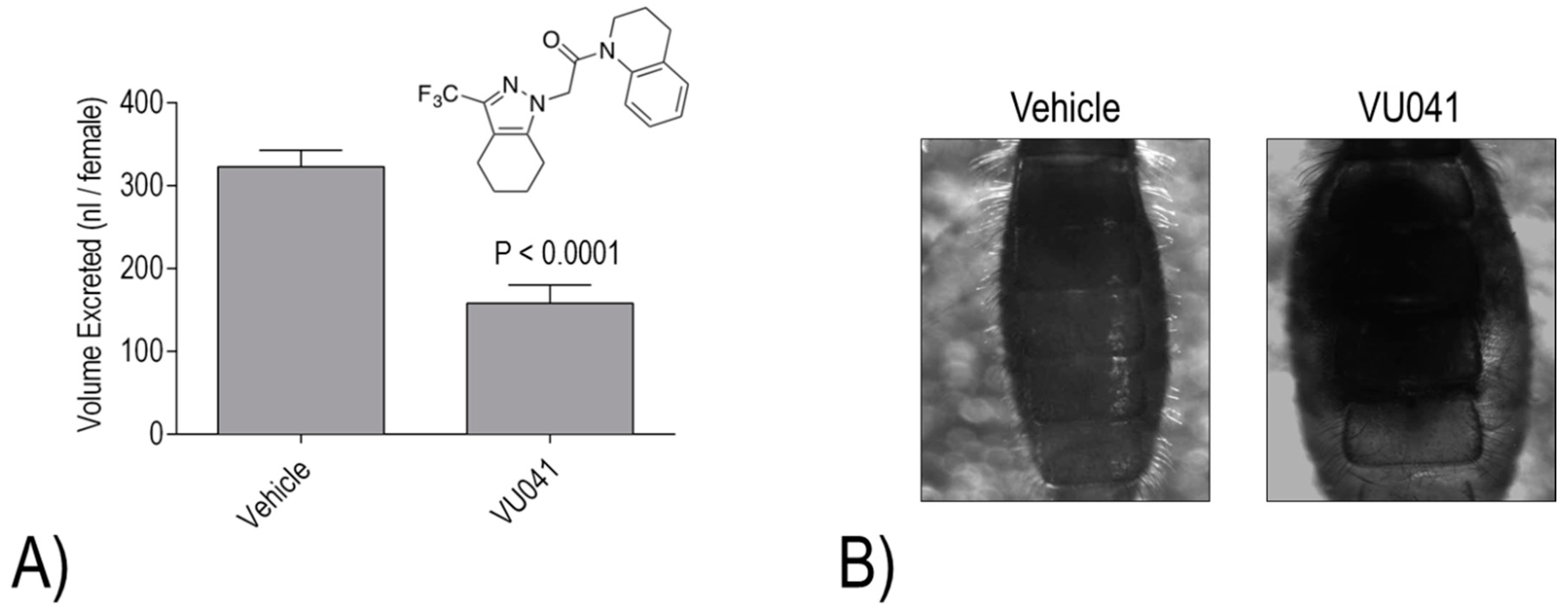
Despite the promising activities of VU573, VU590, and VU625, they each share one common limitation for mosquitocide development: they are unable to elicit toxic effects when applied topically to the cuticle. Thus, our most recent effort has focused on a small molecule inhibitor of
AeKir1 (i.e., VU041) with a high calculated permeability coefficient (cLogP), which makes a molecule more likely to penetrate the cuticle of insects [
23]. VU041 was discovered by the Denton laboratory in a high-throughput screen for inhibitors of the Kir1 ortholog of
Anopheles gambiae (
AnKir1) [
23]; it is equally effective against
AeKir1 [
23]. Notably, VU041 is toxic to adult female
Ae. aegypti and
An. gambiae within 24 h of topical application to the cuticle [
23]. Moreover, VU041 exhibits similar efficacy against insecticide-susceptible and -resistant strains of both mosquito species [
23]. We have confirmed that topical application of VU041 inhibits the diuretic capacity of
Ae. aegypti, consistent with an effect on the Malpighian tubules [
23] (
Figure 7A). Furthermore, in
An. gambiae, topical treatment with VU041 leads to apparent fluid retention in the abdomen 24 h after a blood meal [
23] (
Figure 7B). These effects on the renal system likely contribute to impaired blood meal processing/metabolism as indicated by the decreased fecundity of both species if treated with VU041 after engorgement [
23]. Thus, VU041 provides a promising chemical scaffold for developing a next-generation mosquitocide with a novel mechanism of action (i.e., renal failure) that can potentially be used to mitigate resistance of mosquitoes to conventional insecticides.
4. Finding New Targets: Transcriptomic Insights into Malpighian Tubule Function in Blood Fed Mosquitoes
As described above, the Malpighian tubules of mosquitoes play a key role in mediating the post-prandial diuresis, which alleviates the acute challenges to hemolymph salt and water balance after engorging on blood. However, once the post-prandial diuresis subsides (~1–2 h post-blood meal), the potential roles of the Malpighian tubules in detoxifying and excreting metabolites generated during blood meal digestion, such as heme and nitrogenous wastes (e.g., NH3), have not been widely investigated. Thus, we initiated transcriptomic studies on the Malpighian tubules of the Asian tiger mosquito, Aedes albopictus, before and after a blood meal to generate insights.
In collaboration with Dr. Bryan Cassone (Brandon University), we used RNA-Seq to characterize and quantify global transcript expression in Malpighian tubules isolated from
Ae. albopictus at 3 h, 12 h, and 24 h post-blood meal (all compared to control Malpighian tubules from non-blood fed mosquitoes). Within 24 h post-blood meal, over 80% of the ingested protein is digested [
24]; thus, it is a period of intense metabolic activity. The changes to transcript expression in the Malpighian tubules after blood feeding were dramatic: over 3200 transcripts (i.e., ~40% of the transcriptome) were differentially-expressed at each time point relative to non-blood fed controls [
12,
25]. Moreover, the changes to transcript expression in the Malpighian tubules after blood feeding were dynamic: there were roughly equal numbers of up- and down-regulated transcripts at each time point [
12,
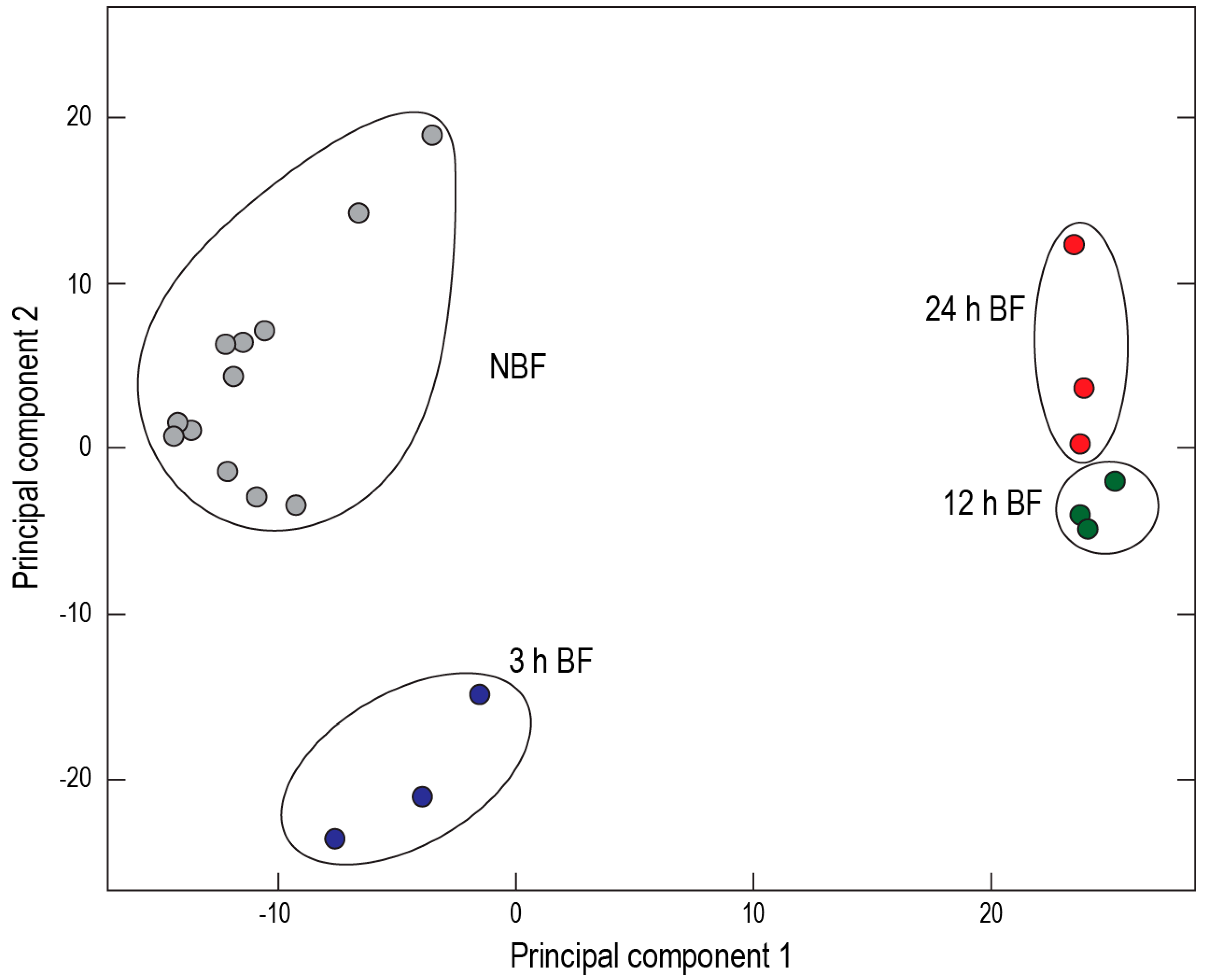
25]. Furthermore, a two-dimensional principal component analysis of transcript expression demonstrated spatial clustering of the sequenced cDNA libraries by treatment and time point (
Figure 8). These findings suggest that the Malpighian tubules of
Ae. albopictus are in the midst of a functional transition after a blood meal.
To further explore this notion, we conducted a Database for Annotation, Visualization and Integrated Discovery (DAVID) functional cluster analysis [
26,
27] on the differentially-expressed transcripts and performed functional assays on mosquitoes and Malpighian tubules [
12,
25]. Among the transcripts that were down-regulated 3–24 h after a blood meal, we identified DAVID molecular pathways (e.g., oxidative phosphorylation, ATPase) and gene families (e.g., V-ATPase subunits, aquaporins) associated with active transepithelial fluid secretion [
12,
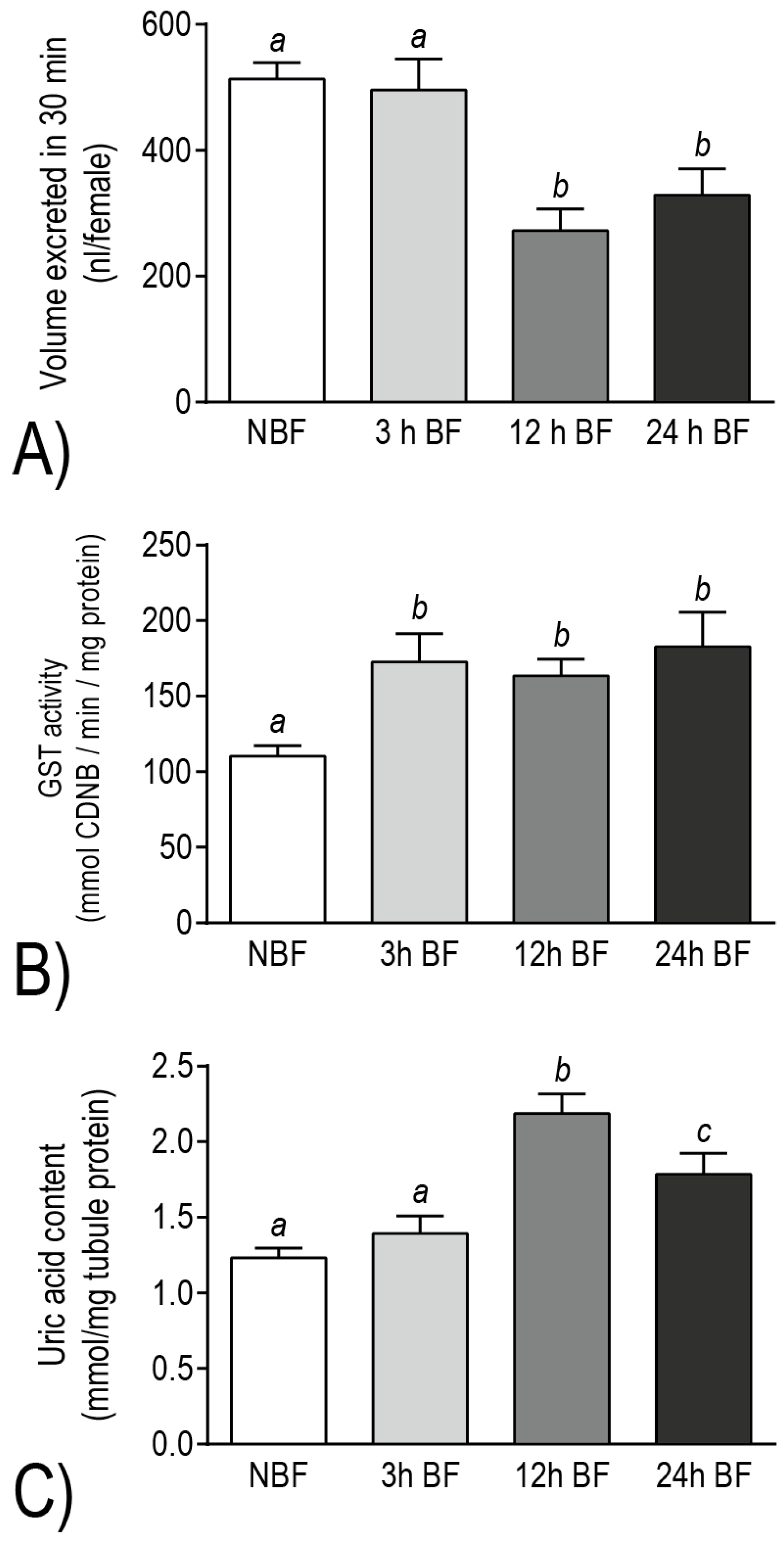
25]. Consistent with these transcript changes, adult female mosquitoes exhibited a decreased capacity for diuresis at 12 h and 24 h post-blood meal relative to non-blood fed females [
12] (
Figure 9A). On the other hand, among the transcripts that were up-regulated after a blood meal, we identified DAVID molecular pathways (e.g., thioredoxin, ATP-binding cassette transporter, cytochrome P450, tryptophan oxidation) and gene families (e.g., glutathione
S-transferases, glutathione peroxidases, glutaredoxins, xanthine dehydrogenases) associated with redox homeostasis and metabolite detoxification [
12,
25]. Consistent with these transcript changes, Malpighian tubules exhibited biochemical increases of (1) glutathione
S-transferase (GST) activity at 3–24 h post-blood meal and (2) uric acid at 12–24 h post-blood meal, relative to the Malpighian tubules of non-blood fed controls [
12] (
Figure 9B,C). Thus, the molecular and physiological/biochemical results both indicate that the Malpighian tubules of
Ae. albopictus undergo a functional transition after a blood meal, wherein molecular resources appear to be diverted away from mechanisms of diuresis and are reinvested into mechanisms associated with redox homeostasis and detoxification of blood-meal metabolites.
Although additional efforts are required to validate that the up-regulated metabolic pathways and transcripts in the Malpighian tubules are vital for mosquito survival and/or fecundity after a blood meal, they may offer unexploited molecular targets for developing insecticides with novel modes of action. For example, several transcripts that encode putative antioxidant enzymes are up-regulated in Malpighian tubules within 24 h post-blood meal (
Figure 10). Notably, in some cases, the transcript up-regulation is dramatic, such as two thioredoxin reductases at 3 h (log2 fold change = 3.7) and a thioredoxin peroxidase (AAEL002309-RA) at 24 h (log2 fold change = 2.6). In addition, a metabolic pathway associated with the production of xanthurenic acid, a chelator of heme and iron [
28], from tryptophan is up-regulated within 24 h (
Figure 11). In particular, transcripts predicted to encode tryptophan 2,3-dioxygenase and kynurenine 3-monoxygenase are highly up-regulated at 12 h and 24 h (log2 fold change = 2.8 to 5.4). Notably, in the Malpighian tubules of
An. gambiae, a transcript for kynurenine 3-monoxygenase was also highly enriched at 3 h post-blood meal [
29]. These findings suggest that the Malpighian tubules contribute to antioxidant production and heme detoxification in mosquitoes during blood meal processing. An alternate, but not mutually-exclusive, explanation is that the Malpighian tubules are highly-susceptible to heme-induced, oxidative tissue damage, and thereby require an up-regulation of antioxidant and heme-chelating mechanisms after a blood meal to maintain normal function. Thus, inhibiting one or more of the above mechanisms with small molecules may disrupt the renal contributions to hemolymph redox homeostasis and/or Malpighian tubule function in general after blood feeding, which may lead to premature death and/or reduced fecundity.
High-throughput assays for discovering small-molecule inhibitors of mammalian thioredoxin reductase, tryptophan 2,3-dioxygenase, and kynurenine 3-monoxygenase, which are human drug targets, have already been developed [
30,
31,
32]. Moreover, inhibitors of mosquito tryptophan 2,3-dioxygenase and 3-hydroxykynurenrine transaminase have already been discovered, some of which exhibit larvicidal activity [
33,
34]. Thus, pursuing a small-molecule approach, parallel to the one we have used for Kir channels, may be warranted for identifying potential mosquitocides that disrupt the antioxidant and xanthurenic acid-synthesis enzymes up-regulated in the Malpighian tubules after a blood meal.