Better than Membranes at the Origin of Life?
Abstract
:1. Introduction
2. Location of Membraneless Organelles at the Origins of Life?
3. Membraneless Organelles
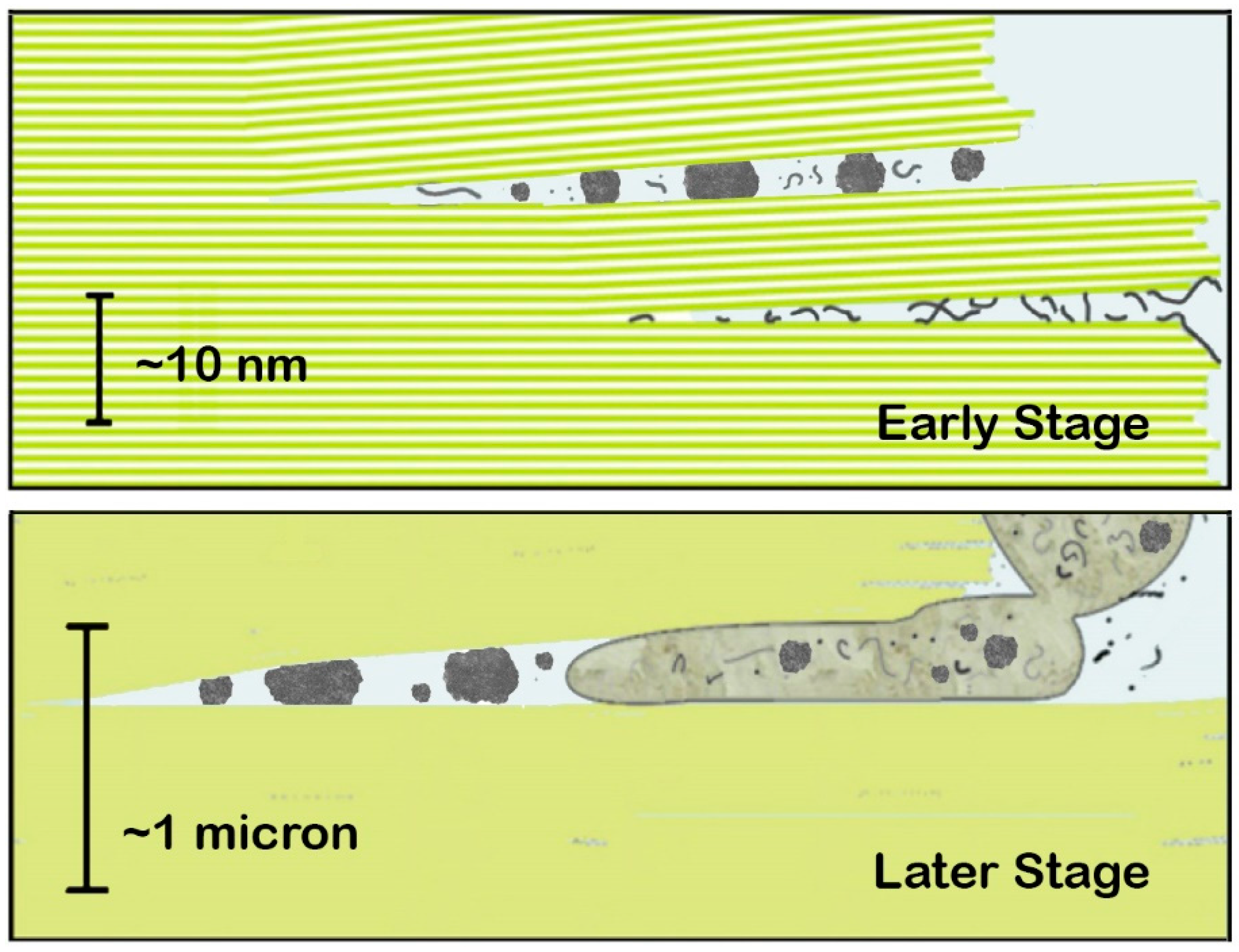
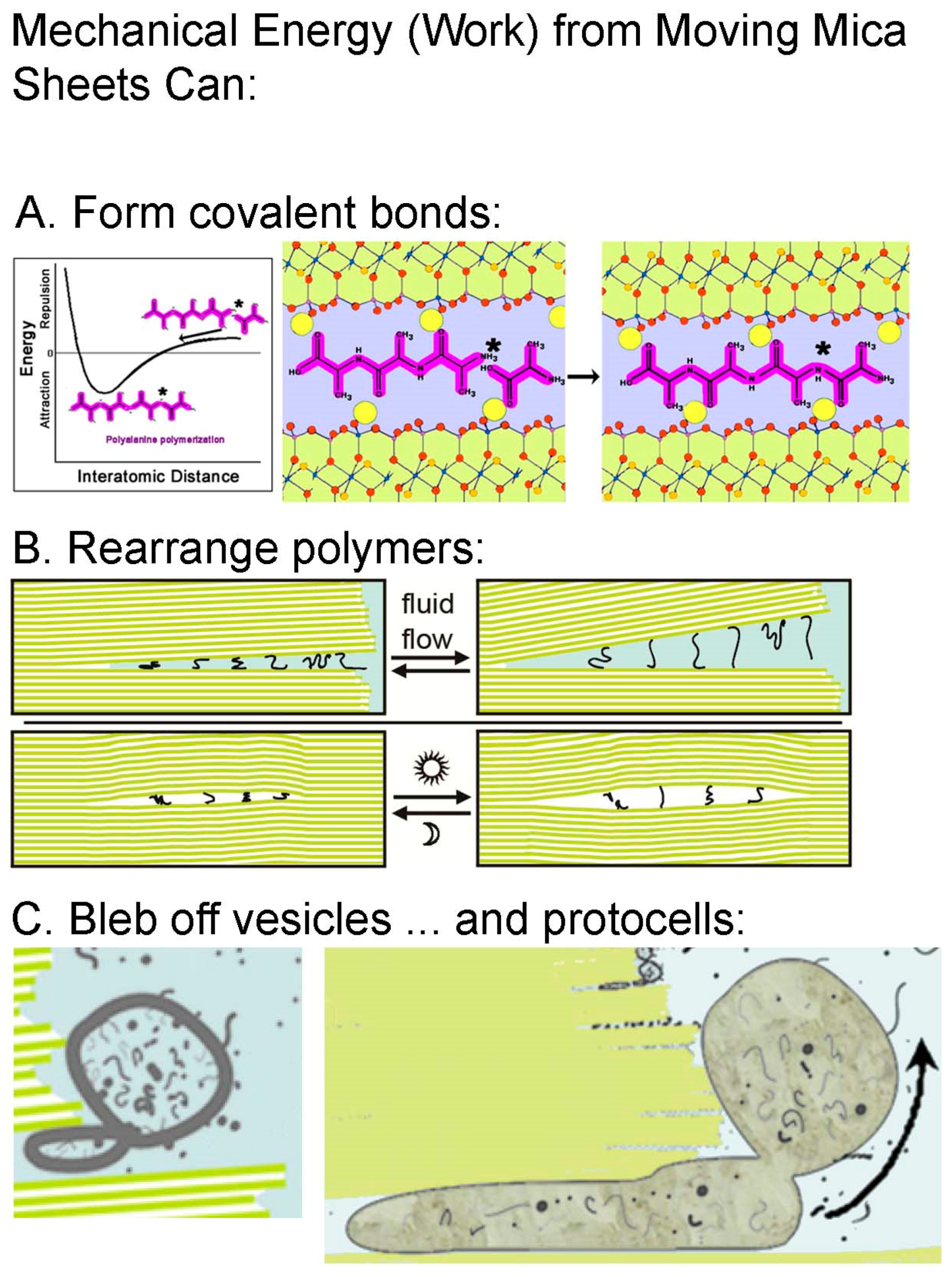
4. An Example of Chemical Emergence
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hansma, H.G. The Immobilization Antigen of Paramecium aurelia is a Single Polypeptide Chain. J. Protozool. 1975, 22, 257–259. [Google Scholar] [CrossRef] [PubMed]
- Woese, C.R.; Kandler, O.; Wheelis, M.L. Towards a natural system of organisms: Proposal for the domains Archaea, Bacteria, and Eucarya. Proc. Natl. Acad. Sci. USA 1990, 87, 4576–4579. [Google Scholar] [CrossRef] [PubMed]
- Matsumi, R.; Atomi, H.; Driessen, A.J.; van der Oost, J. Isoprenoid biosynthesis in Archaea–biochemical and evolutionary implications. Res. Microbiol. 2011, 162, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Szostak, J.W.; Bartel, D.P.; Luisi, P.L. Synthesizing life. Nature 2001, 409, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Cech, T.R. Crawling out of the RNA world. Cell 2009, 136, 599–602. [Google Scholar] [CrossRef] [PubMed]
- Hansma, H.G. Possible origin of life between mica sheets. J. Theor. Biol. 2010, 266, 175–188. [Google Scholar] [CrossRef] [PubMed]
- Hansma, H.G. Possible Origin of Life between Mica Sheets: How Life Imitates Mica. J. Biol. Struct. Dyn. 2013, 31, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Hansma, H.G. The Power of Crowding for the Origins of Life. Orig. Life Evol. Biosph. 2014, 44, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Ferris, J.P.; Hill, A.R., Jr.; Liu, R.; Orgel, L.E. Synthesis of long prebiotic oligomers on mineral surfaces. Nature 1996, 381, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Hansma, H.G. Could Life Originate between Mica Sheets?: Mechanochemical Biomolecular Synthesis and the Origins of Life. In Probing Mechanics at Nanoscale Dimensions; Tamura, N., Minor, A., Murray, C., Frontmatter, L.F., Eds.; Materials Research Society: Warrendale, PA, USA, 2009. [Google Scholar]
- Christof, J.; Gebhardt, M.; Rief, M. Force signaling in biology. Science 2009, 324, 1278–1280. [Google Scholar] [CrossRef] [PubMed]
- Ingber, D.E. The origin of cellular life. Bioessays 2000, 22, 1160–1170. [Google Scholar] [CrossRef]
- Bustamante, C.; Chemla, Y.R.; Forde, N.R.; Izhaky, D. Mechanical processes in biochemistry. Annu. Rev. Biochem. 2004, 73, 705–748. [Google Scholar] [CrossRef] [PubMed]
- Keller, D.; Bustamante, C. The mechanochemistry of molecular motors. Biophys. J. 2000, 78, 541–556. [Google Scholar] [CrossRef]
- Radmacher, M.; Fritz, M.; Hansma, H.G.; Hansma, P.K. Direct observation of enzyme activity with the atomic force microscope. Science 1994, 265, 1577–1579. [Google Scholar] [CrossRef] [PubMed]
- Hammes-Schiffer, S.; Benkovic, S.J. Relating protein motion to catalysis. Annu. Rev. Biochem. 2006, 75, 519–541. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.-W. Mechanochemical organic synthesis. Chem. Soc. Rev. 2013, 42, 7668–7700. [Google Scholar] [CrossRef] [PubMed]
- Brangwynne, C.P. Phase transitions and size scaling of membraneless organelles. J. Cell Biol. 2013, 203, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Hyman, A.A.; Weber, C.A.; Jülicher, F. Liquid-liquid phase separation in biology. Annu. Rev. Cell Dev. Biol. 2014, 30, 39–58. [Google Scholar] [CrossRef] [PubMed]
- Marko, J.F. The liquid drop nature of nucleoli. Nucleus 2012, 3, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Brangwynne, C.P.; Tompa, P.; Pappu, R.V. Polymer physics of intracellular phase transitions. Nat. Phys. 2015, 11, 899–904. [Google Scholar] [CrossRef]
- Ellis, J.C.; Brown, D.D.; Brown, J.W. The small nucleolar ribonucleoprotein (snoRNP) database. RNA 2010, 16, 664–666. [Google Scholar] [CrossRef] [PubMed]
- Weber, S.C.; Brangwynne, C.P. Getting RNA and protein in phase. Cell 2012, 149, 1188–1191. [Google Scholar] [CrossRef] [PubMed]
- Shorter, J. Membraneless organelles: Phasing in and out. Nat. Chem. 2016, 8, 528–530. [Google Scholar] [CrossRef] [PubMed]
- Nott, T.J.; Craggs, T.D.; Baldwin, A.J. Membraneless organelles can melt nucleic acid duplexes and act as biomolecular filters. Nat. Chem. 2016, 8, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Gesteland, R.F.; Cech, T.R.; Atkins, J.F. (Eds.) The RNA World: The Nature of Modern RNA Suggests a Prebiotic RNA, 3rd ed.; Cold Spring Harbor Monograph Series; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2006. [Google Scholar]
- Fox, G.; (University of Houston, Houston TX, USA). Personal Communication, 2015.
- Lin, Y.; Protter, D.S.; Rosen, M.K.; Parker, R. Formation and maturation of phase-separated liquid droplets by RNA-binding proteins. Mol. Cell 2015, 60, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Shorter, J. It’s raining liquids: RNA tunes viscoelasticity and dynamics of membraneless organelles. Mol. Cell 2015, 60, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Deamer, D. First Life: Discovering the Connections between Stars, Cells, and How Life Began; University of California Press: Oakland, CA, USA, 2011. [Google Scholar]
- Bradt, H. Madagascar: The Bradt Travel Guide; The Globe Pequot Press Inc.: Guilford, CT, USA, 2007. [Google Scholar]
- Sivarajasingham, S.; Alexander, L.T.; Cady, J.G.; Cline, M.G. Laterite. Adv. Agron. 1962, 14, 1–60. [Google Scholar]
- Damer, B.; Deamer, D. Coupled phases and combinatorial selection in fluctuating hydrothermal pools: A scenario to guide experimental approaches to the origin of cellular life. Life 2015, 5, 872–887. [Google Scholar] [CrossRef] [PubMed]
- Deamer, D. Membranes and the Origin of Life: A Century of Conjecture. J. Mol. Evol. 2016, 83, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Martin, W.; Baross, J.; Kelley, D.; Russell, M.J. Hydrothermal vents and the origin of life. Nat. Rev. Microbiol. 2008, 6, 805–814. [Google Scholar] [CrossRef] [PubMed]
- Attwater, J.; Wochner, A.; Holliger, P. In-ice evolution of RNA polymerase ribozyme activity. Nat. Chem. 2013, 5, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Wachtershauser, G. Before enzymes and templates: Theory of surface metabolism. Microbiol. Rev. 1988, 52, 452–484. [Google Scholar] [PubMed]
- Dyson, F.J. Origins of Life, Revised Edition; Cambridge University Press: Cambridge, UK; New York, NY, USA, 1999; p. 100. [Google Scholar]

© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hansma, H.G. Better than Membranes at the Origin of Life? Life 2017, 7, 28. https://doi.org/10.3390/life7020028
Hansma HG. Better than Membranes at the Origin of Life? Life. 2017; 7(2):28. https://doi.org/10.3390/life7020028
Chicago/Turabian StyleHansma, Helen Greenwood. 2017. "Better than Membranes at the Origin of Life?" Life 7, no. 2: 28. https://doi.org/10.3390/life7020028




