Active Sensor for Microwave Tissue Imaging with Bias-Switched Arrays
Abstract
1. Introduction
2. Antenna Array Design
2.1. Carbon–Rubber Tissue Phantoms
2.2. Antenna Array Element
2.3. Antenna Array
2.4. Comparison Between a TEM Horn Antenna and the Proposed Antenna Element
3. Mixer and LNA Parameter Measurements and their Integrations with the Antenna
3.1. Low-Noise Amplifier
3.2. Mixer
3.3. Integrating the LNA and the Mixer with the Antenna
4. Results
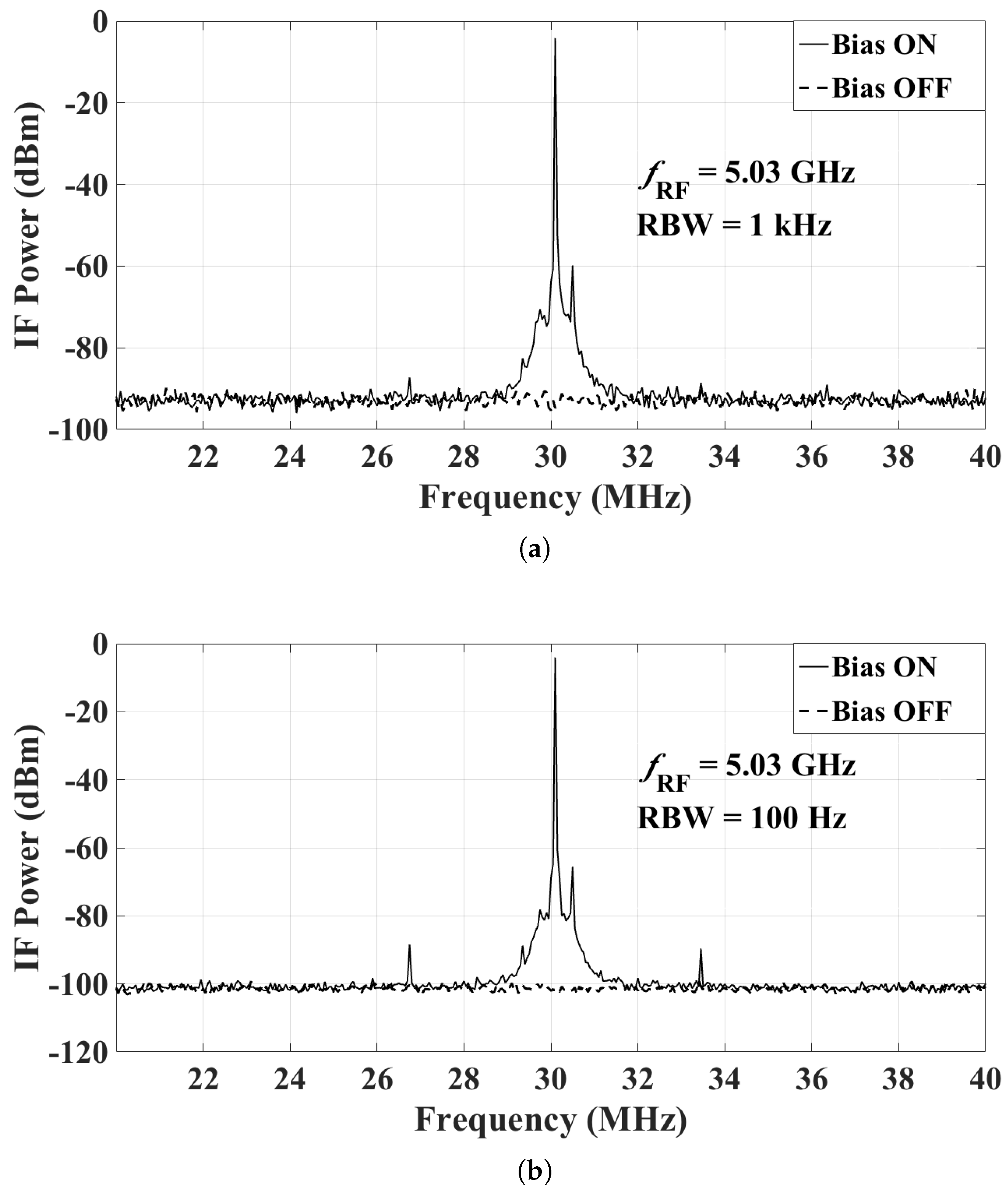
4.1. Dynamic Range of the Proposed Active Sensor
- RF frequency at GHz, GHz, and GHz;
- LO frequency at 3 GHz, 5 GHz, and GHz, respectively (so that the IF is at 30 MHz); and
- LO power at 0 dBm (as per the recommendation in the mixer datasheet).
4.2. Comparison with Conventional RF-Switch System
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nikolova, N.K. Microwave imaging for breast cancer. IEEE Microw. Mag. 2011, 12, 78–94. [Google Scholar] [CrossRef]
- Ley, S.; Sachs, J.; Helbig, M. MNP enhanced microwave breast cancer imaging based on ultra-wideband pseudo-noise sensing. In Proceedings of the 11th European Conference on Antennas and Propagation (EUCAP), Paris, France, 19–24 March 2017; pp. 2754–2757. [Google Scholar]
- Conceição, R.C.; Mohr, J.J.; O’Halloran, M. An Introduction to Microwave Imaging for Breast Cancer Detection; Springer: Berlin, Germany, 2016. [Google Scholar]
- Kwon, S.; Lee, S. Recent advances in microwave imaging for breast cancer detection. Int. J. Biomed. Imaging 2016. [Google Scholar] [CrossRef] [PubMed]
- Bucci, O.M.; Bellizzi, G.; Borgia, A.; Costanzo, S.; Crocco, L.; Massa, G.D.; Scapaticci, R. Experimental framework for magnetic nanoparticles enhanced breast cancer microwave imaging. IEEE Access 2017, 5, 16332–16340. [Google Scholar] [CrossRef]
- Cheng, G.G.; Zhu, Y.; Grzesik, J. Microwave imaging of biological bone tissue. In Proceedings of the USNC-URSI Radio Science Meeting (Joint with AP-S Symposium), Memphis, TN, USA, 6–11 July 2014. [Google Scholar]
- Meaney, P.M.; Goodwin, D.; Golnabi, A.; Pallone, M.; Geimer, S.; Paulsen, K.D. 3D microwave bone imaging. In Proceedings of the 6th European Conference on Antennas and Propagation (EUCAP), Prague, Czech Republic, 26–30 March 2012; pp. 1770–1771. [Google Scholar]
- Ireland, D.; Bialkowski, K.; Abbosh, A. Microwave imaging for brain stroke detection using Born iterative method. IET Microw. Antennas Propag. 2013, 7, 909–915. [Google Scholar] [CrossRef]
- Tournier, P.H.; Bonazzoli, M.; Dolean, V.; Rapetti, F.; Hecht, F.; Nataf, F.; Aliferis, I.; El-Kanfoud, I.; Migliaccio, C.; de Buhan, M.; Darbas, M.; Semenov, S.; Pichot, C. Numerical modeling and high-speed parallel computing: new perspectives on tomographic microwave imaging for brain stroke detection and monitoring. IEEE Antennas Propag. Mag. 2017, 59, 98–110. [Google Scholar] [CrossRef]
- Dilman, İ.; Yıldırım, U.; Coşğun, S.; Doğu, S.; Çayören, M.; Akduman, I. Feasibility of brain stroke imaging with microwaves. In Proceedings of the IEEE Asia-Pacific Conference on Applied Electromagnetics (APACE), Langkawi, Malaysia, 11–13 December 2016; pp. 334–338. [Google Scholar]
- Zamani, A.; Rezaeieh, S.A.; Abbosh, A.M. Lung cancer detection using frequency-domain microwave imaging. Electron. Lett. 2015, 51, 740–741. [Google Scholar] [CrossRef]
- Babarinde, O.J.; Jamlos, M.F.; Soh, P.J.; Schreurs, D.M.M.P.; Beyer, A. Microwave imaging technique for lung tumour detection. In Proceedings of the German Microwave Conference (GeMiC), Bochum, Germany, 14–16 March 2016; pp. 100–103. [Google Scholar]
- Nikolova, N.K. Microwave biomedical imaging. In Wiley Encyclopedia of Electrical and Electronics Engineering; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014. [Google Scholar]
- Lazebnik, M.; McCartney, L.; Popovic, D.; Watkins, C.B.; Lindstrom, M.J.; Harter, J.; Sewall, S.; Magliocco, A.; Booske, J.H.; Okoniewski, M.; Hagness, S.C. A large-scale study of the ultrawideband microwave dielectric properties of normal breast tissue obtained from reduction surgeries. Phys. Med. Biol. 2007, 52, 2637. [Google Scholar] [CrossRef] [PubMed]
- Lazebnik, M.; Popovic, D.; McCartney, L.; Watkins, C.B.; Lindstrom, M.J.; Harter, J.; Sewall, S.; Ogilvie, T.; Magliocco, A.; Breslin, T.M.; et al. A large-scale study of the ultrawideband microwave dielectric properties of normal, benign and malignant breast tissues obtained from cancer surgeries. Phys. Med. Biol. 2007, 52, 6093. [Google Scholar] [CrossRef] [PubMed]
- Halter, R.J.; Zhou, T.; Meaney, P.M.; Hartov, A.; Barth Jr, R.J.; Rosenkranz, K.M.; Wells, W.A.; Kogel, C.A.; Borsic, A.; Rizzo, E.J.; et al. The correlation of in vivo and ex vivo tissue dielectric properties to validate electromagnetic breast imaging: initial clinical experience. Physiol. Meas. 2009, 30, S121. [Google Scholar] [CrossRef] [PubMed]
- Sugitani, T.; Kubota, S.i.; Kuroki, S.i.; Sogo, K.; Arihiro, K.; Okada, M.; Kadoya, T.; Hide, M.; Oda, M.; Kikkawa, T. Complex permittivities of breast tumor tissues obtained from cancer surgeries. Appl. Phys. Lett. 2014, 104, 253702. [Google Scholar] [CrossRef]
- Klemm, M.; Gibbins, D.; Leendertz, J.; Horseman, T.; Preece, A.; Benjamin, R.; Craddock, I. Development and testing of a 60-element UWB conformal array for breast cancer imaging. In Proceedings of the 5th European Conference on Antennas and Propagation (EUCAP), Rome, Italy, 11–15 April 2011; pp. 3077–3079. [Google Scholar]
- Song, H.; Sasada, S.; Kadoya, T.; Okada, M.; Arihiro, K.; Xiao, X.; Kikkawa, T. Detectability of breast tumor by a hand-held impulse-radar detector: performance evaluation and pilot clinical study. Sci. Rep. 2017, 7, 16353. [Google Scholar] [CrossRef] [PubMed]
- Amineh, R.K.; Ravan, M.; Khalatpour, A.; Nikolova, N.K. Three-dimensional near-field microwave holography using reflected and transmitted signals. IEEE Trans. Antennas Propag. 2011, 59, 4777–4789. [Google Scholar] [CrossRef]
- Amineh, R.K.; McCombe, J.J.; Khalatpour, A.; Nikolova, N.K. Microwave holography using point-spread functions measured with calibration objects. IEEE Trans. Instrum. Meas. 2015, 64, 403–417. [Google Scholar] [CrossRef]
- Ostadrahimi, M.; Asefi, M.; LoVetri, J.; Bridges, G.E.; Shafai, L. An MST-based microwave tomography system using homodyne receiver. In Proceedings of the 2013 IEEE Antennas and Propagation Society International Symposium (APSURSI), Orlando, FL, USA, 7–13 July 2013; pp. 814–815. [Google Scholar]
- Crocker, D.A.; Donnell, K.M. Application of electrically invisible antennas to the modulated scatterer technique. IEEE Trans. Instrum. Meas. 2015, 64, 3526–3535. [Google Scholar] [CrossRef]
- Joisel, A.; Mallorqui, J.; Broquetas, A.; Geffrin, J.; Joachimowicz, N.; Lossera, M.; Joire, L.; Bolomey, J.C. Microwave imaging techniques for biomedical applications. In Proceedings of the 16th IEEE on Instrumentation and Measurement Technology Conference IMTC/99, Venice, Italy, 24–26 May 1999; Volume 3, pp. 1591–1596. [Google Scholar]
- Bolomey, J.C.; Gardiol, F.E. Engineering Applications of the Modulated Scatterer Technique; Artech House: Norwood, MA, USA, 2001. [Google Scholar]
- Abou-Khousa, M.A.; Ghasr, M.T.; Kharkovsky, S.; Pommerenke, D.; Zoughi, R. Modulated elliptical slot antenna for electric field mapping and microwave imaging. IEEE Trans. Antennas Propag. 2011, 59, 733–741. [Google Scholar] [CrossRef]
- Sturley, K.R. Radio Receiver Design; Chapman & Hall: London, UK, 1953. [Google Scholar]
- Agilent AN 1275, Automatic Frequency Settling Time Measurements Speeds Time-to-Market for RF Design: Application Note. Available online: http://literature.cdn.keysight.com/litweb/pdf/5964-4335E.pdf (accessed on 22 April 2018).
- Guan, X.; Hashemi, H.; Hajimiri, A. A fully integrated 24-GHz eight-element phased-array receiver in silicon. IEEE J. Solid-State Circuits 2004, 39, 2311–2320. [Google Scholar] [CrossRef]
- Tajik, D.; Foroutan, F.; Shumakov, D.S.; Pitcher, A.D.; Nikolova, N.K. Real-time microwave imaging of a compressed breast phantom with planar scanning. IEEE J. Electromagn. RF Microw. Med. Biol. 2017. under revision. [Google Scholar]
- Beaverstone, A.S.; Nikolova, N.K. Modeling and design of a switched transceiver array for tissue imaging. In Proceedings of the IEEE MTT-S International Conference on Numerical Electromagnetic and Multiphysics Modeling and Optimization (NEMO), Ottawa, ON, Canada, 11–14 August 2015; pp. 1–3. [Google Scholar]
- Beaverstone, A.S.; Nikolova, N.K. Switched sensor array for near-field microwave imaging of tissue. In Proceedings of the IEEE International Symposium on Antennas and Propagation & USNC/URSI National Radio Science Meeting, Vancouver, BC, Canada, 19–24 July 2015; pp. 354–355. [Google Scholar]
- ACR BI-RADS® ATLAS-MAMMOGRAPHY. Available online: https://www.acr.org/-/media/ACR/Files/RADS/BI-RADS/Mammography-Reporting.pdf (accessed on 1 January 2018).
- University of Wisconsin-Madison Numerical Breast Phantom Repository. Available online: https://uwcem.ece.wisc.edu/phantomRepository.html (accessed on 1 January 2018).
- Trehan, A. Numerical and Physical Models for Microwave Breast Imaging. Master’s Thesis, McMaster University, Hamilton, ON, Canada, 2009. [Google Scholar]
- Moll, J.; Wcõrtge, D.; Krozer, V.; Santorelli, A.; Popović, M.; Bazrafshan, B.; Hübner, F.; Vogl, T.J.; Nikolova, N. Quality control of carbon-rubber tissue phantoms: Comparative MRI, CT, X-ray and UWB microwave measurements. In Proceedings of the 11th European Conference on Antennas and Propagation (EUCAP), Paris, France, 19–24 March 2017; pp. 2723–2727. [Google Scholar]
- Lazebnik, M.; Okoniewski, M.; Booske, J.H.; Hagness, S.C. Highly accurate Debye models for normal and malignant breast tissue dielectric properties at microwave frequencies. IEEE Microw. Wirel. Compon. Lett. 2007, 17, 822–824. [Google Scholar] [CrossRef]
- Baskharoun, Y.; Trehan, A.; Nikolova, N.K.; Noseworthy, M.D. Physical phantoms for microwave imaging of the breast. In Proceedings of the 2012 IEEE Topical Conference on Biomedical Wireless Technologies, Networks, and Sensing Systems (BioWireleSS), Santa Clara, CA, USA, 15–18 January 2012; pp. 73–76. [Google Scholar]
- Nikolova, N.K. Introduction to Microwave Imaging; Cambridge University Press: Cambridge, UK, 2017. [Google Scholar]
- Bucci, O.; Franceschetti, G. On the spatial bandwidth of scattered fields. IEEE Trans. Antennas Propag. 1987, 35, 1445–1455. [Google Scholar] [CrossRef]
- Bucci, O.; Isernia, T. Electromagnetic inverse scattering: retrievable information and measurement strategies. Radio Sci. 1997, 32, 2123–2137. [Google Scholar] [CrossRef]
- Bucci, O.M.; Crocco, L.; Scapaticci, R.; Bellizzi, G. On the design of phased arrays for medical applications. Proc. IEEE 2016, 104, 633–648. [Google Scholar] [CrossRef]
- Klemm, M.; Craddock, I.J.; Leendertz, J.A.; Preece, A.; Benjamin, R. Radar-based breast cancer detection using a hemispherical antenna array; experimental results. IEEE Trans. Antennas Propag. 2009, 57, 1692–1704. [Google Scholar] [CrossRef]
- Gibbins, D.; Klemm, M.; Craddock, I.J.; Leendertz, J.A.; Preece, A.; Benjamin, R. A comparison of a wide-slot and a stacked patch antenna for the purpose of breast cancer detection. IEEE Trans. Antennas Propag. 2010, 58, 665–674. [Google Scholar] [CrossRef]
- Amineh, R.K.; Trehan, A.; Nikolova, N.K. TEM horn antenna for ultra-wide band microwave breast imaging. Prog. Electromagn. Res. B 2009, 13, 59–74. [Google Scholar] [CrossRef]
- Kanj, H.; Popovic, M. Miniaturized microstrip-fed “Dark Eyes” antenna for near-field microwave sensing. IEEE Antennas Wirel. Propag. Lett. 2005, 4, 397–401. [Google Scholar] [CrossRef]
- Islam, M.A.; Kiourti, A.; Volakis, J.L. A novel body-worn RF sensor for deep tissue imaging. In Proceedings of the 2015 IEEE MTT-S 2015 International Microwave Workshop Series on RF and Wireless Technologies for Biomedical and Healthcare Applications (IMWS-BIO), Taipei, Taiwan, 21–23 September 2015; pp. 43–44. [Google Scholar]
- Aguilar, S.M.; Al-Joumayly, M.A.; Burfeindt, M.J.; Behdad, N.; Hagness, S.C. Multiband miniaturized patch antennas for a compact, shielded microwave breast imaging array. IEEE Trans. Antennas Propag. 2014, 62, 1221–1231. [Google Scholar] [CrossRef] [PubMed]
- Sugitani, T.; Kubota, S.; Toya, A.; Kikkawa, T. A compact 4X4 planar UWB antenna array for 3-D breast cancer detection. IEEE Antennas Propag. Soc. Int. Symp. 2012, 12, 733–736. [Google Scholar] [CrossRef]
- EM Software and Systems. Available online: https://altairhyperworks.com/product/FEKO (accessed on 3 May 2018).
- Dadash, S.; Moussakhani, K.; McCombe, J. Quad-Ridge Horn Antenna for Tissue Measurements; Computational Electromagnetics Res. Lab. Tech. Report; McMaster University: Hamilton, ON, Canada, 2014. [Google Scholar]
- TB-830A+ Evaluation Board. Available online: https://www.minicircuits.com/WebStore/dashboard.html?model=TB-830A%2B (accessed on 14 September 2017).
- TB-771+ Evaluation Board. Available online: https://www.minicircuits.com/WebStore/dashboard.html?model=MDA4-752H%2B (accessed on 14 September 2017).
- R3969/R3970-Advantest. Available online: https://www.advantest.com/documents/11348/146687/pdf_mn_ER3969_OPERATING_MANUAL.pdf (accessed on 3 Janurary 2018).





















| Antenna Scanning | RF-Switched Array | MST Array | Bias-Switched Array | |
|---|---|---|---|---|
| Input/Output Frequency | RF/RF | RF/RF | RF/RF | RF/IF |
| Number of Multiplexed Ports | 9 [21] | 60 [18] | 1024 [24] | Hundreds/Thousands |
| Acquisition Time | Slow | Fast | Fast | Fast |
| Positioning Uncetianties | High | Low | Low | Low |
| Interconnect Complexity | Low | High | Low | Low |
| Interconnect Losses and Distortion | High | High | Very High | Low |
| The Largest Inter-Element Spacing (mm) | Bandwidth (GHz) | Back Shielding | Number of Elements | |
|---|---|---|---|---|
| Antenna Reported in [18] | Yes | 60 | ||
| Antenna Reported in [49] | No | 16 | ||
| The Proposed Antenna Array | 12 | Yes | 121 |
| RF | LO | IF | Mixer Gain (dB) | |
|---|---|---|---|---|
| Power (dBm) | −5 | 0 | 4.6 | 9.6 |
| Frequency (GHz) | 3.03 | 3 | 0.03 | |
| Power (dBm) | −5 | 0 | 1.6 | 6.6 |
| Frequency (GHz) | 5.03 | 5 | 0.03 | |
| Power (dBm) | −5 | 0 | −3.2 | 1.8 |
| Frequency (GHz) | 7.5 | 7.47 | 0.03 |
| RF | LO | IF | System Gain (dB) | |
|---|---|---|---|---|
| Power (dBm) | −30 | 0 | 0.2 | 30.2 |
| Frequency (GHz) | 3.03 | 3 | 0.03 | |
| Power (dBm) | −30 | 0 | −3.4 | 26.6 |
| Frequency (GHz) | 5.03 | 5 | 0.03 | |
| Frequency (GHz) | 7.5 | 7.47 | 0.03 |
| System 1 (dBm) | System 2 (dBm) | Difference (dB) | |
|---|---|---|---|
| = 3.03 GHz | −19 | −25 | 6 |
| = 5.03 GHz | −27 | −35 | 8 |
| = 7.5 GHz | −34 | −41 | 7 |
| Measured (dB) | From Users Manual [54] (dB) | |
|---|---|---|
| 3.03 GHz | 6 | 6 or less |
| 5.03 GHz | 8 | 7 or less |
| 7.5 GHz | 7 | 8.5 or less |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Foroutan, F.; Nikolova, N.K. Active Sensor for Microwave Tissue Imaging with Bias-Switched Arrays. Sensors 2018, 18, 1447. https://doi.org/10.3390/s18051447
Foroutan F, Nikolova NK. Active Sensor for Microwave Tissue Imaging with Bias-Switched Arrays. Sensors. 2018; 18(5):1447. https://doi.org/10.3390/s18051447
Chicago/Turabian StyleForoutan, Farzad, and Natalia K. Nikolova. 2018. "Active Sensor for Microwave Tissue Imaging with Bias-Switched Arrays" Sensors 18, no. 5: 1447. https://doi.org/10.3390/s18051447
APA StyleForoutan, F., & Nikolova, N. K. (2018). Active Sensor for Microwave Tissue Imaging with Bias-Switched Arrays. Sensors, 18(5), 1447. https://doi.org/10.3390/s18051447





