1. Introduction
Lower leg muscle activity is important for controlling the body. Relationships between lower leg muscle activity and the risk of falls among the elderly have already been reported [
1,
2]; delayed plantar flexion increases the risk of such falls. In addition, there is a certain amount of people in the young group (20–45 years) that experience falling [
3]. The ability to recover balance after a fall is related to lower leg muscle activity [
4]; further the time and volume of muscle activities are important for recovering balance. The relationship between the lower leg muscle activity and certain anomalies such as the freezing of gait in Parkinson’s disease, has also been studied [
5]. Training the lower leg muscles contributes to improvements in terms of gait, such as velocity, cadence, step time and so on [
6]. Thus, monitoring lower leg muscle activity when walking is beneficial for understanding the body conditions and preventing accidents. Currently, muscle activity analysis is conducted in specific facilities, such as hospitals and universities owing to the requirement of specific knowledge of kinematics and anatomy.
Electromyography (EMG) is used widely for monitoring muscle activity. Biopotential signals that activating muscle fibers propagate from the neuromuscular junction to the tendon along the muscle fibers. An electrical field is created by the stimulation from the neuron activating the muscle fiber’s chemical receptors [
7]. By placing electrodes on the surface of the skin, biopotential signals are measured as EMG signals. An EMG signal-based system has several applications [
8,
9,
10]. The simplification of EMG signal measurement is considered important for further popularization; however, it is necessary to select the muscle site to be measured, and to determine the position where the electrode is to be placed, based on kinematic and anatomical knowledge. It is difficult for users who have no kinematic and anatomical knowledge to place electrodes on specific muscles. With garment-type measurement systems, electrodes placements are included in the action of wearing socks. Therefore, garment-type measurement systems can be applied for daily use. There have been studies on conductive fabric-type EMG measurement materials for garment-type EMG measurement systems [
11,
12]. Myers et al. presented a silver nanowire (AgNW)-based dry electrode for electrophysiological wearable monitoring [
12]. They compared EMG signals measured using AgNW dry electrodes and conventional Ag/AgCl wet electrodes, and they confirmed that there were a few differences. Nippon Telegraph and Telephone Corporation and Toray developed “hitoe
®”, which is made of electro-conductive polymer nanofibers [
11]. Their technology can be applied to users who have metal allergies because it is not made of metal. Some other types of conductive fabric have also become commercially available [
13].
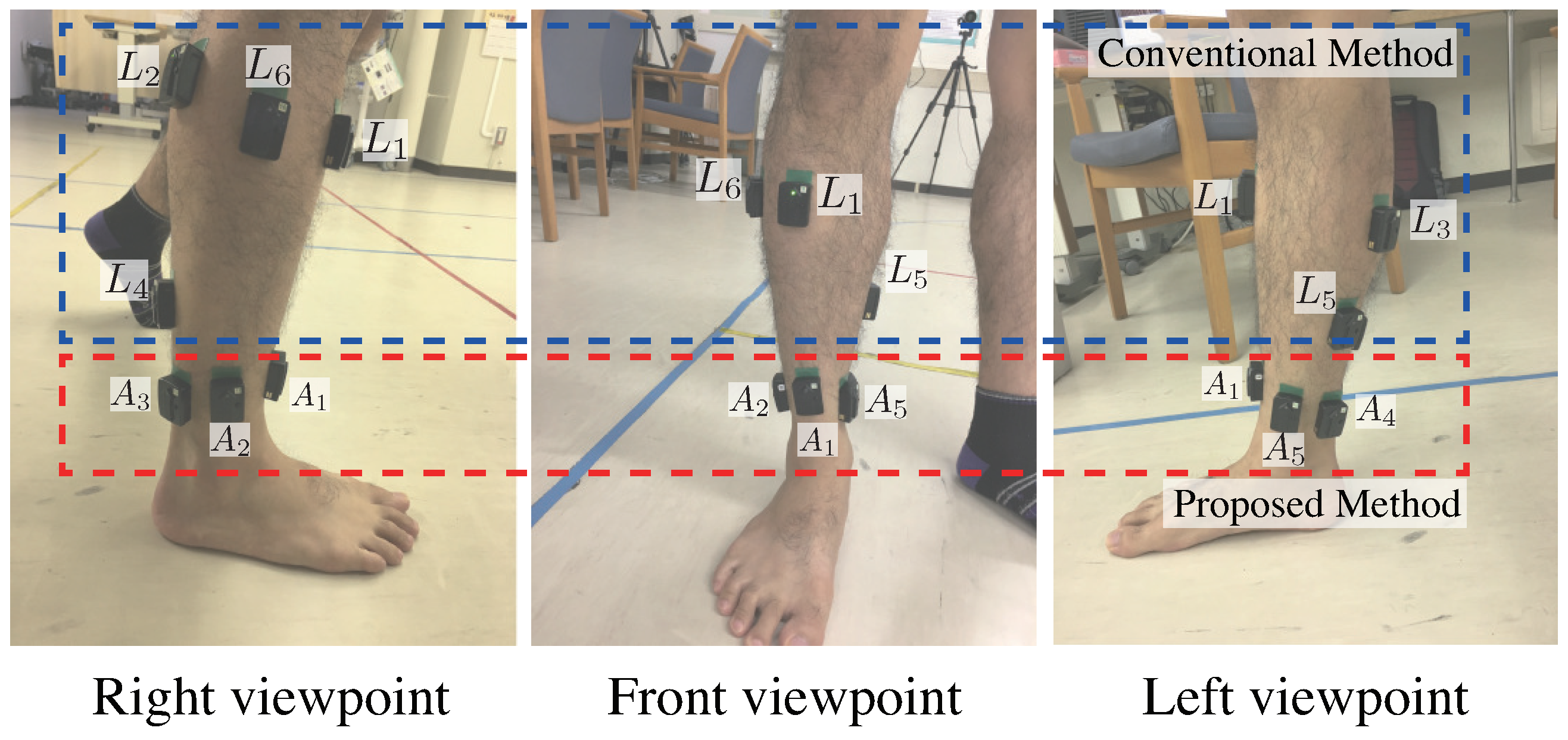
To increase the opportunities for monitoring lower leg muscle activity, we focus on garment-type measurement systems using conductive fabric. Users do not need to place electrodes themselves, because conductive fabric electrodes are already implemented in garment-type measurement systems. However, garment-type measurement systems need high compression around the electrodes for preventing electrode displacement. If electrodes are distributed on a large area in a system, the area that requires high compression also expands, which makes it difficult for users to wear the electrodes. Considering the applications for daily use among a wide variety of users, including the elderly, it is important that the measurement system be easy to wear.
Accurate and repeatable EMG signals can be used for the analysis of signal amplitude, spectral variables and muscle fiber conduction velocity. Rainoldi et al. explored optimal electrode positions for obtaining accurate and repeatable EMG signals [
14]. It is common to measure EMG signals by placing a sensor electrode at the center of each target muscle. Such signals propagate to and from neighboring muscles [
15]. Therefore, it should be possible to measure EMG signals by placing electrodes at the distal positions close to the tendons of target muscles. We define biosignals not measured on the muscle bellies as
distal EMG signal. Using distal EMG signals, electrode positions can be designated for each situation based on propagation characteristics. Some studies used distal EMG signals to estimate facial expressions [
16,
17]. They revealed that distal EMG signals can be used for discriminating between smiles, frowns, and neutral facial expressions. Independent component analysis and artificial neural networks were used for separating signals and recognizing facial expressions, respectively. However, studies on the distal EMG-based analysis of other muscles, such as the lower leg, are lacking.
Our objective is to develop a sock-type wearable sensor consisting of electrodes that are implemented around the ankle for estimating lower leg muscle activity.
Figure 1 shows the concept for the proposed system. The proposed system requires high compression in the red area because electrodes are placed around the ankle, while high compression is required in the blue area if electrodes are positioned on each muscle belly. From a wearing pressure point of view, the proposed system is minimally restraining and easy to wear. The signals obtained from around the ankle are the distal EMG signals; which are the propagated biopotential signals of target muscles. The time and volume of muscle activity is a major target of muscle activity analysis [
18,
19]. We construct the EMG signal estimation system based on the distal EMG signals for the temporal-spatial muscle activity analysis.
Amplitude and spectral variables are the main features obtained from EMG signals [
20]. Amplitude features contain information on muscle force ratio, muscle activation (ON/OFF) state, and the timing of muscle contraction and relaxation. There are several gait analysis applications based on the amplitude features of EMG signals [
2,
21]. It was reported that the mean absolute values represent amplitude features more than other amplitude feature calculations [
22]. Therefore, mean absolute values are adopted as a muscle activity in this study.
The main contributions of this paper are:
To verify that distal EMG signals can be used for estimating lower leg muscle activity
To design low restrained electrodes positions for ease of wearing
To implement the signal processing of mean absolute values estimation based on the distal EMG signals
We outline our system’s configuration in
Section 2. Three analyses from the data of one experiment for evaluating the accuracy for estimating EMG signals based on distal EMG signals are described in
Section 3. We explain the experimental results in
Section 4, and describe the discussion for results in
Section 5. The conclusion is described in
Section 6.
5. Discussion
The goal of this study was to develop a sock-type system for estimating lower leg muscle activity based on distal EMG signals around the ankles. Multiple band-pass filter-based feature extraction was proposed for increasing the estimation accuracy. We conducted experiments involving five channels with conductive fabric for obtaining distal EMG signals, and we estimated the mean absolute value signals of target muscles (tibialis anterior, gastrocnemius lateral, gastrocnemius medial, soleus lateral, soleus medial, and peroneus ). In the experiment of the mean absolute value estimation accuracy, the estimation accuracy of the proposed method was compared with that of a baseline method that is based on a single bandpass filter. As shown in
Table 3 and
Table 4, the accuracies of the proposed method were higher than those of the baseline method. This result indicates that the proposed method can estimate mean absolute values at a high accuracy than the baseline method. In addition, this result indicates that the proposed method is applicable to all conducted tasks in this experiment at high accuracy. As shown in
Figure 9, the trend of the estimated signals from our system is close to that of the measured signals. The variance of the average measured signal and the average estimated signal are large, as shown in
Figure 9. We considered that this was caused from the individual variability of muscle activity in the gait. This result indicates that our system is user-dependent owing to the individual differences of muscle activity in the gait.
In the dependability analysis, the effect of body movements on estimation accuracy were evaluated by constructing two estimation models. As shown in
Table 5, there were no significant differences between the first model (assumed to be low impacts of body movements) and the second model (assumed to be high impacts of body movements). Therefore, the effects of body movement on the estimation accuracy can be considered to be small. This result is assumed to depend on the kinds of tasks, the body characteristics of subjects, the duration of tasks, and the size of socks. When the above conditions are determined, the dependability test is carried out. Most results from the temporal-spatial muscle activity analysis exceeded 80%, as shown in
Table 6 and
Table 7. Therefore, our system can be used to analyze information such as muscle-force ratio, muscle activation (ON/OFF) state, and the timing of muscle contraction and relaxation. In this experiment, the same tasks were used for calibration and estimation. Our system estimates EMG signals of target muscles based on distal EMG signals, which are biosignals propagated from several muscles. Therefore, distal EMG signals are defined by the contraction of each muscle. The results indicate that target muscle activity under the same muscle-contraction patterns as calibration can be estimated with high accuracy. Movement composed of the same muscle-contraction patterns as those of the target movement is assumed to be an appropriate calibration movement.
We applied multiple band-pass filters to the distal EMG signals. Since good results were obtained using our system, separating raw distal EMG signals into multiple frequency bands is considered beneficial. Raw distal EMG signals are assumed to contain several signals whose frequency distributions differ. One reason for this is that noise of a specific frequency distribution has been separated. Another is that the frequency distributions of each target muscle might originally be different. A third reason is that frequency distributions slightly change over the course of signal propagation, even if the frequency distributions of the original signals are the same. It is said that the frequency distributions of EMG signals are affected by propagation velocity [
37]. It is assumed that longer the propagation path of the EMG signals, the more susceptible they are to the effect of propagation velocity. Although computational cost is considered to be a concern, it is not supposed to be a major problem in the offline analysis as in this experiment.
The reason the data of the two subjects were noisy is attributed to the fact that the socks did not fit them.
Table 8 summarizes the length around the ankle [cm] for each subject. Two subjects whose data were noisy were subjects B and D, and their lengths around the ankle were shorter than others. The necessity to choose suitable socks could be a limitation of the proposed system. The impact of sweat also could be a limitation. It is assumed that the effect of sweat on these electrodes may cause an increase in the biopotential voltage measured from the surface of the skin with a decrease in skin impedance. The correction of biopotential voltage based on impedance analysis is considered to reduce the impact of sweat. The necessity of the calibration and recalibration is supposed to depend on whether the same signal characteristics as in the calibration are measured. Biopotential signals, which are measured from the surface of the skin, vary depending on muscle masses, skin impedance, and the coordination of muscle activity. It is assumed that they are different among individuals, and therefore, the estimation model should be calibrated for each user. There is also a possibility that the shifts or rotations of electrodes by re-wearing the socks. The impact of the shifts or rotations of electrodes should be investigated in further study.