Application of Near-Infrared Spectroscopy to Investigate Some Endogenic Properties of Pleurotus ostreatus Cultivars
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Sample Preparation
2.2.2. Free Amino Acid Content (fAA)
2.2.3. Total Polyphenol Content (TPC)
2.2.4. Antioxidant Capacity (Ferric Reducing Ability of Plasma—FRAP)
2.2.5. Fourier-Transform Near Infrared Spectroscopy (FT-NIR)
2.3. Chemometrics
2.3.1. Kruskal–Wallis Test
2.3.2. Principal Component Analysis—PCA
2.3.3. Linear Discriminant Analysis—LDA
3. Results
3.1. Chemical Measurements
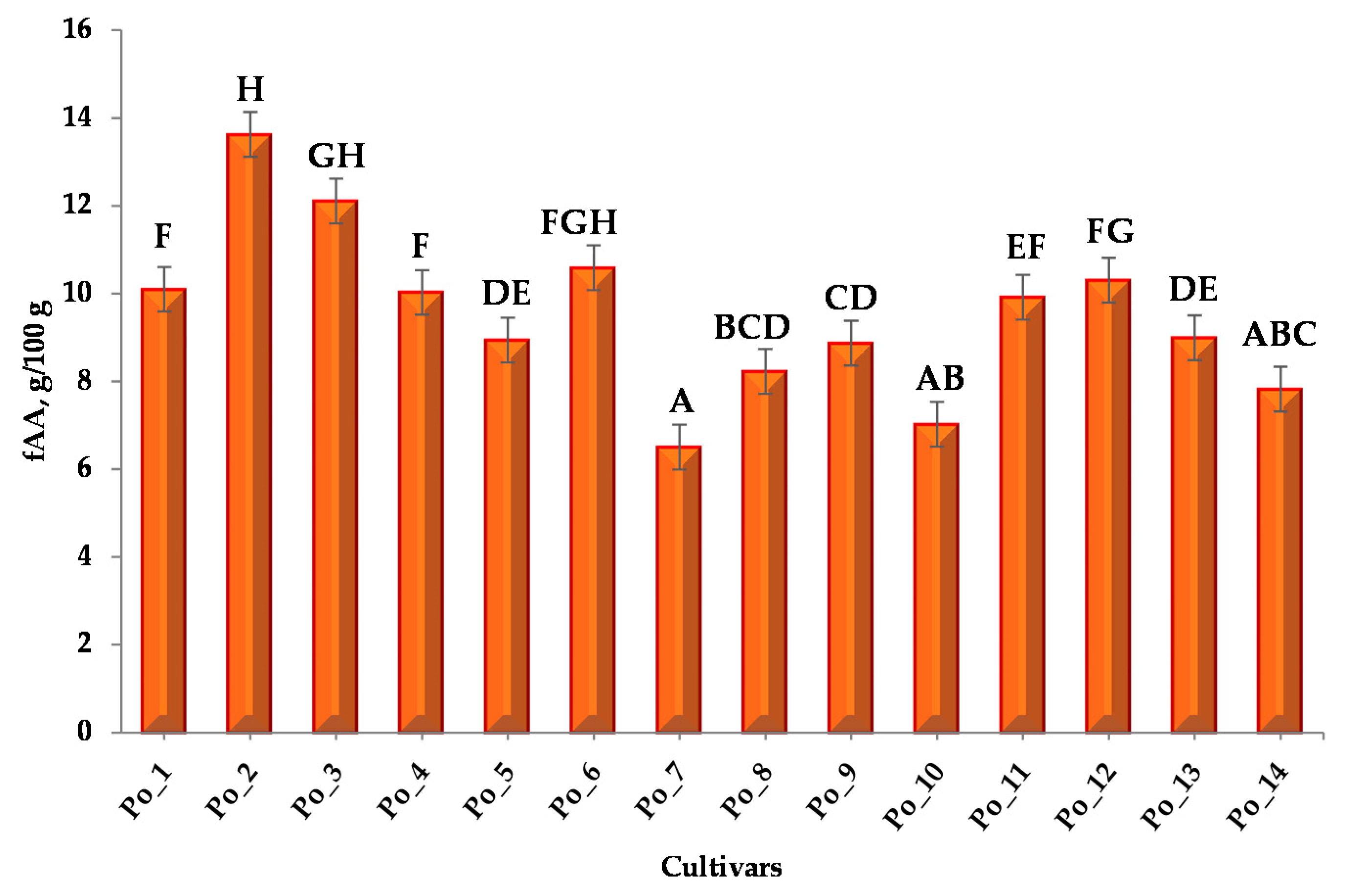
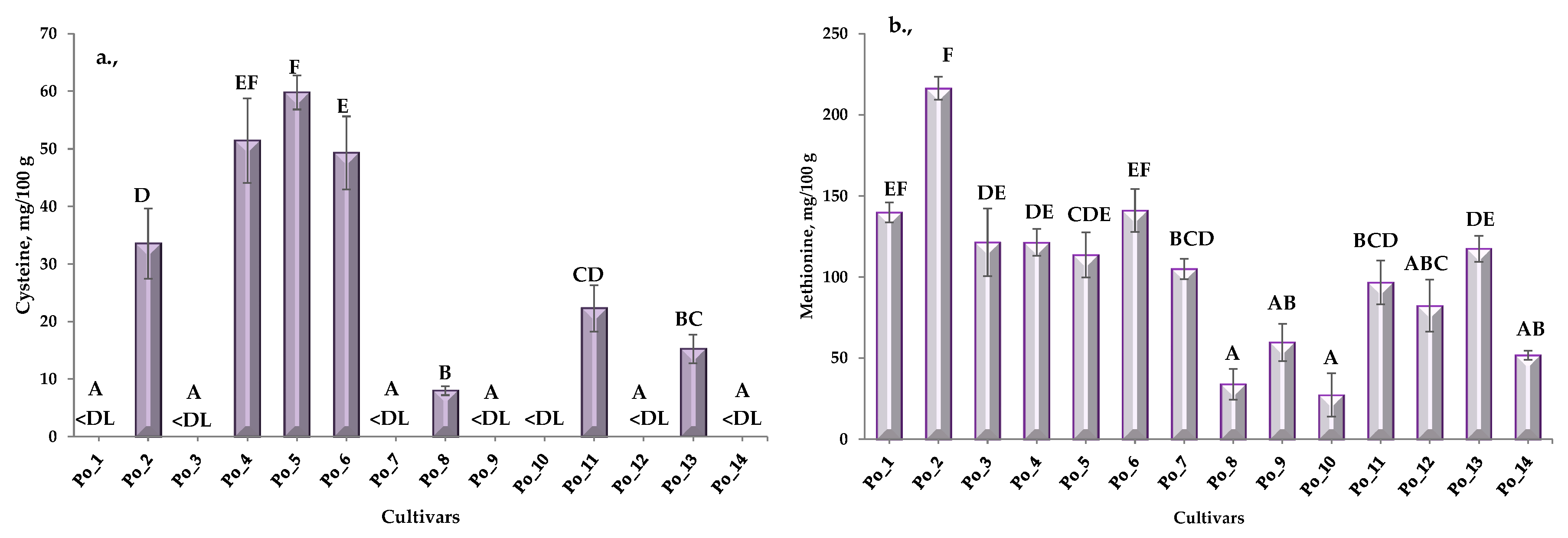
3.1.1. Free Amino Acid Content (fAA)
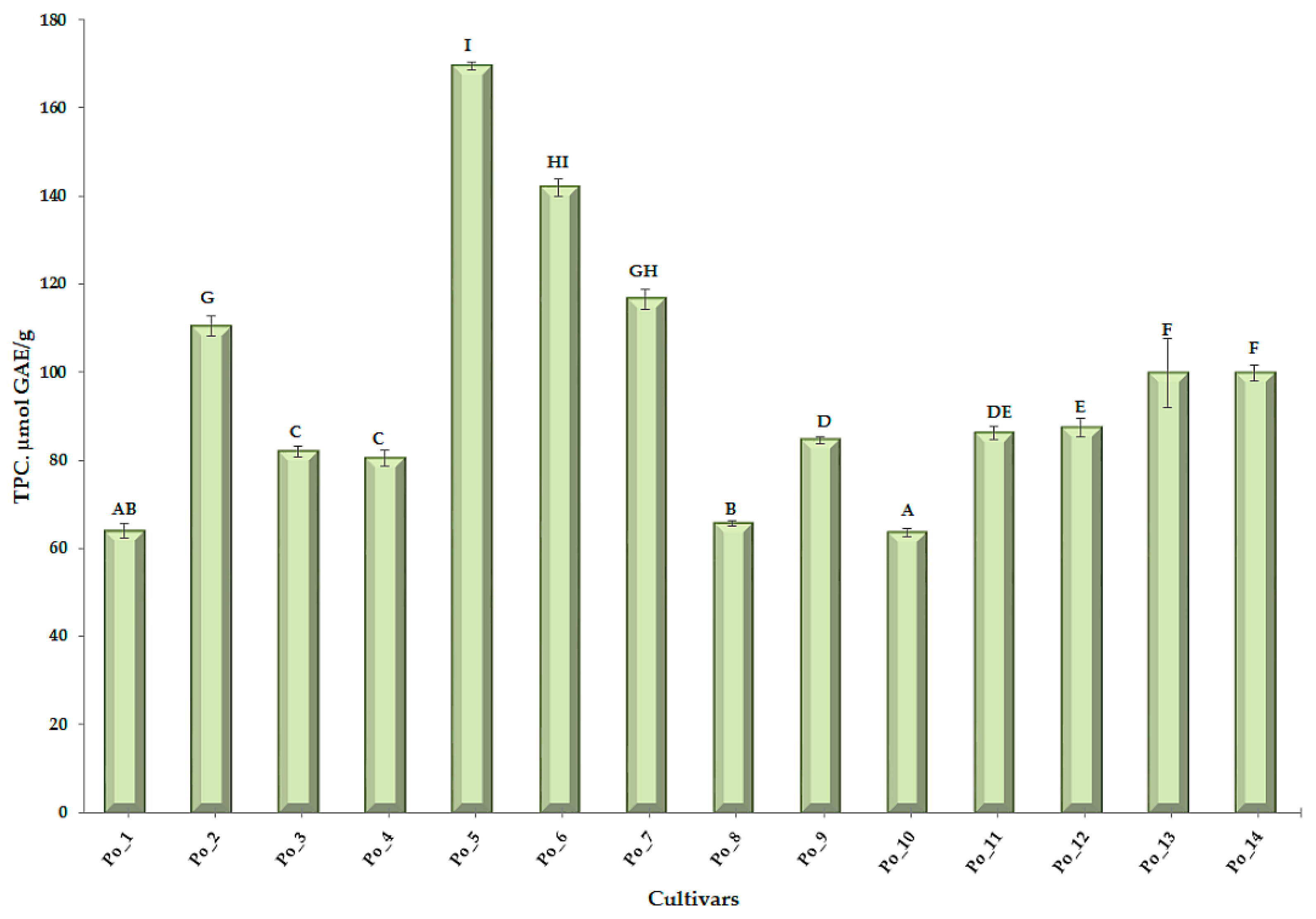
3.1.2. Total Polyphenol Content (TPC)
3.1.3. Antioxidant Capacity (Ferric Reducing Ability of Plasma—FRAP)
3.2. FT-NIR Measurements
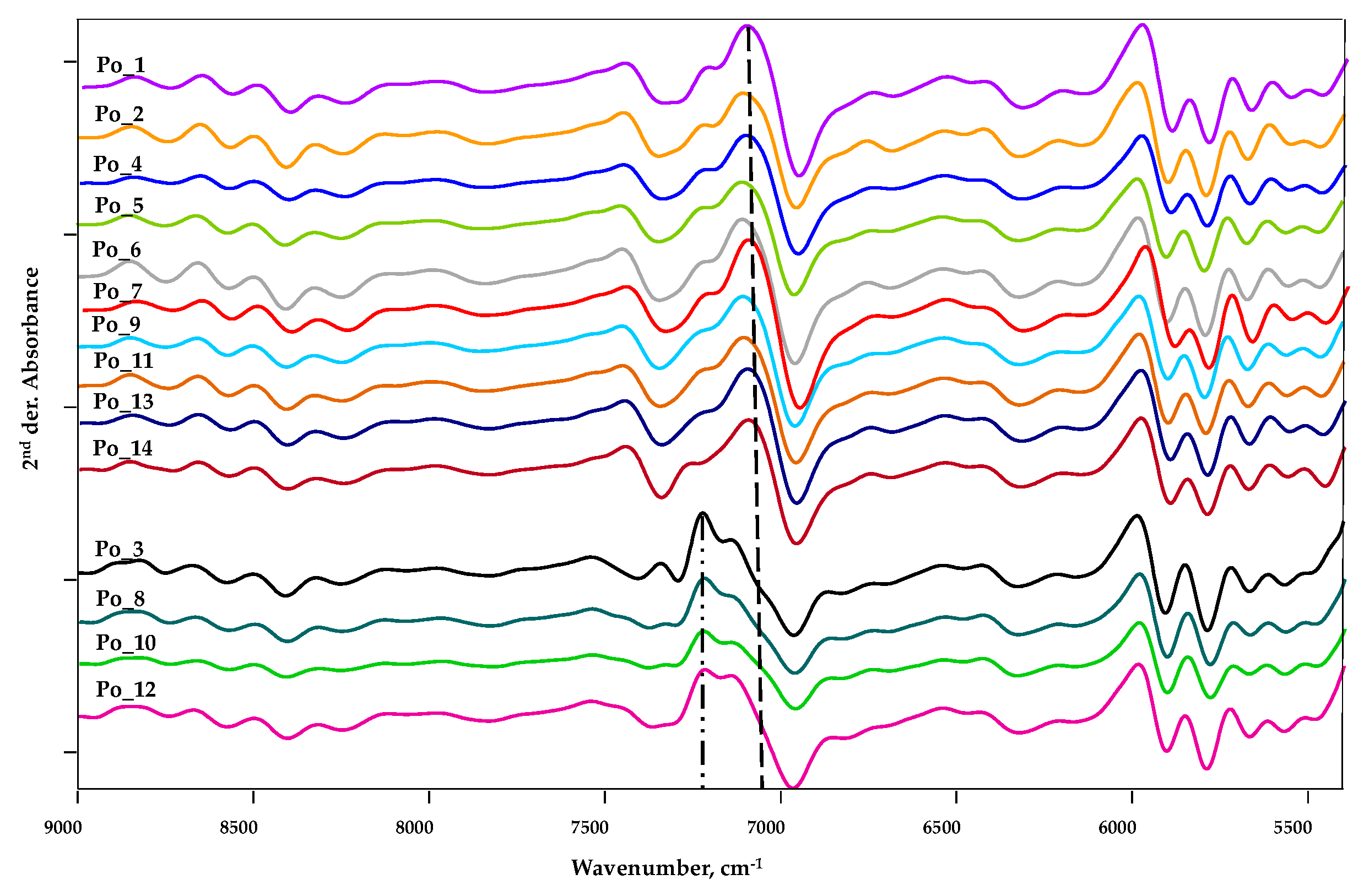
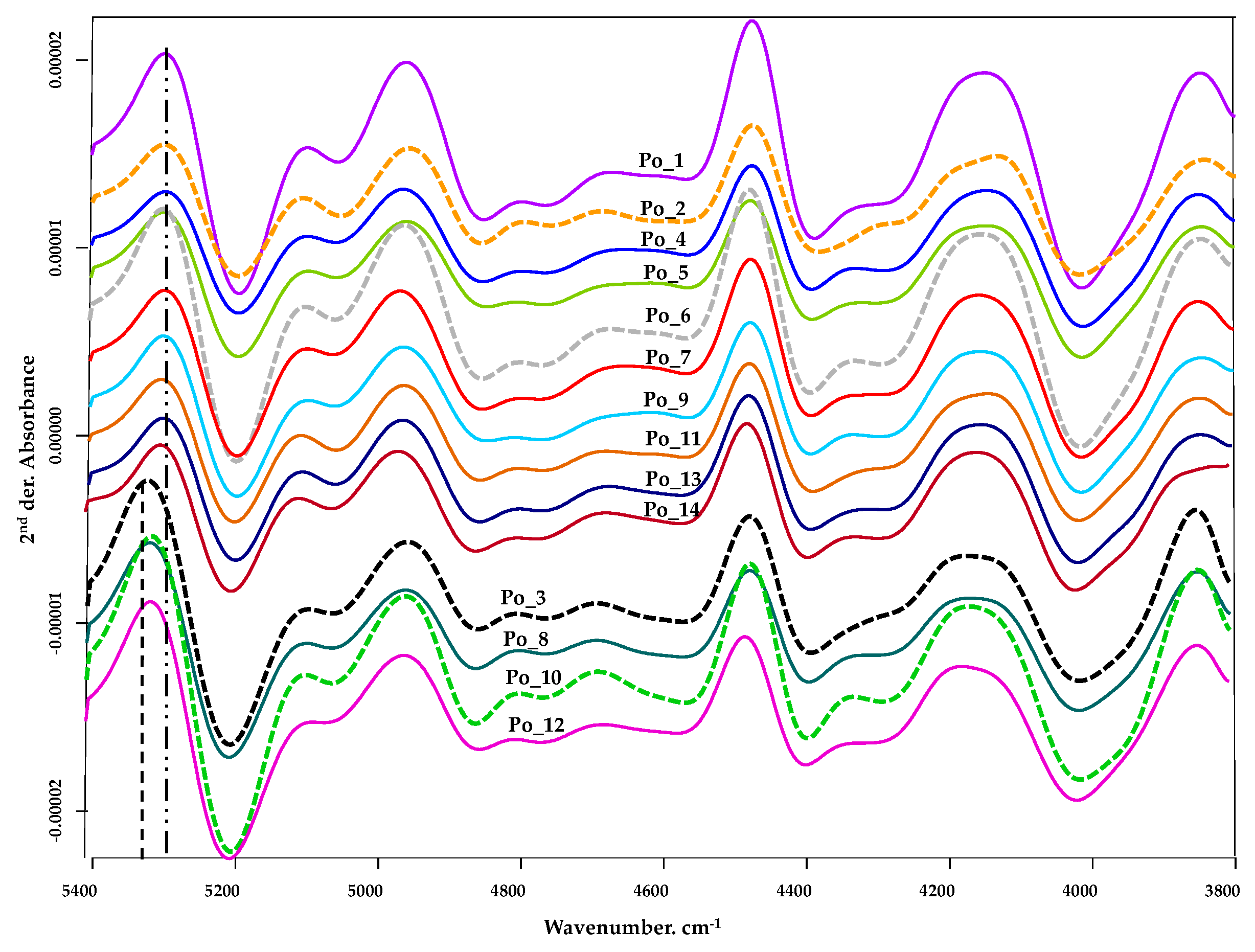
3.2.1. Spectrum Profiling
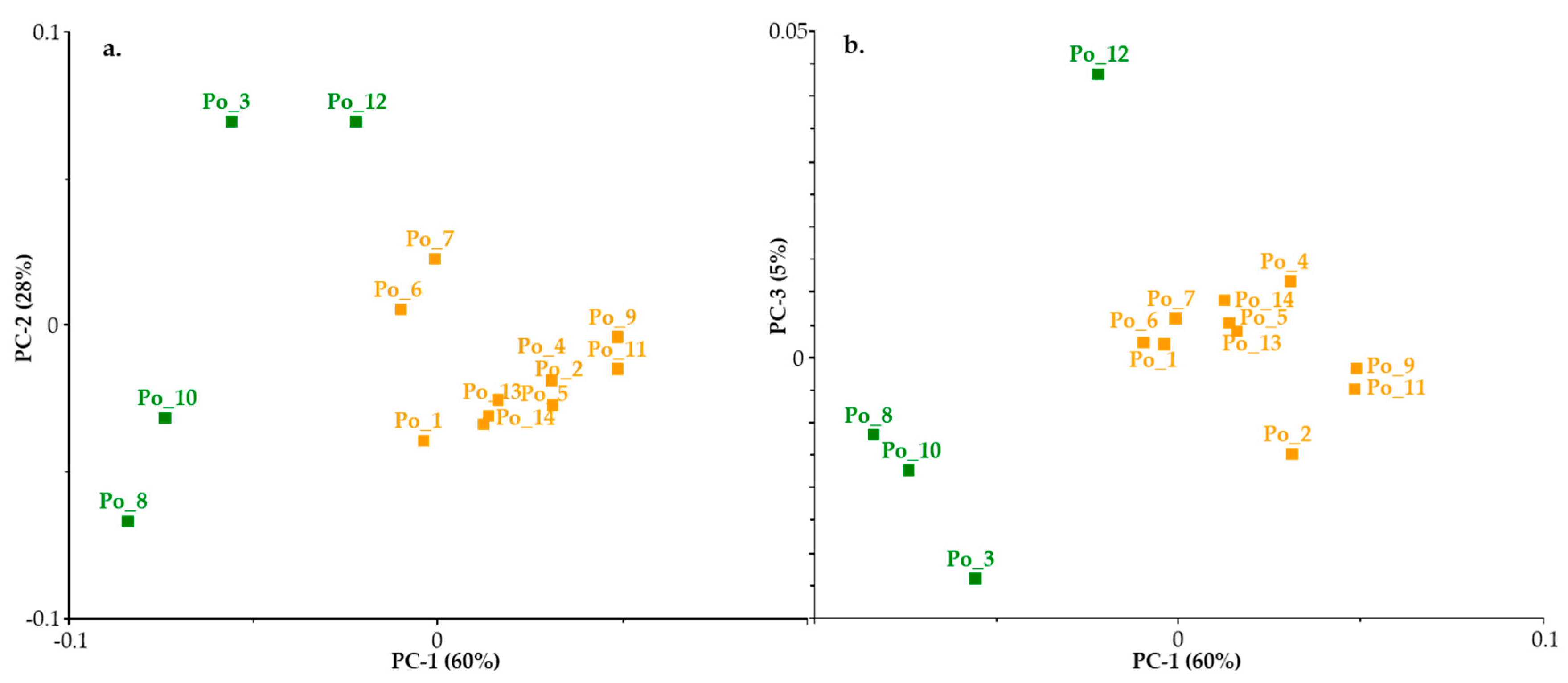
3.2.2. Chemometric Evaluation of NIR spectra
- Principal Component Analysis (PCA)
- Linear Discriminant Analysis (LDA)
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roncero-Ramos, I.; Delgado-Andrade, C. The beneficial role of edible mushrooms in human health. Curr. Opin. Food Sci. 2017, 14, 122–128. [Google Scholar] [CrossRef]
- Chirinang, P.; Intarapichet, K.-O. Amino acids and antioxidant properties of oyster mushrooms. Sci. Asia 2009, 35, 326. [Google Scholar] [CrossRef]
- Reis, F.S.; Barros, L.; Martins, A.; Ferreira, I.C.F.R. Chemical composition and nutritional value of the most widely appreciated cultivated mushrooms: An inter-species comparative study. Food Chem. Toxicol. 2012, 50, 191–197. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Wang, A.; Liu, L.; Tian, G.; Wei, S.; Xu, F. Evaluation of nutritional values of shiitake mushroom (Lentinus edodes) stipes. J. Food Meas. Charact. 2018, 12, 2012–2019. [Google Scholar] [CrossRef]
- Akindahunsi, A.A.; Oyetayo, F.L. Nutrient and antinutrient distribution of edible mushroom, Pleurotus tuber-regium (fries) singer. LWT-Food Sci. Technol. 2006, 39, 548–553. [Google Scholar] [CrossRef]
- Vetter, J. Biological values of cultivated mushrooms—A review. Acta Aliment. 2019, 48, 229–240. [Google Scholar] [CrossRef]
- Ashraf Khan, A.; Gani, A.; Masoodi, F.A.; Mushtaq, U.; Silotry Naik, A. Structural, rheological, antioxidant, and functional properties of β–glucan extracted from edible mushrooms Agaricus bisporus, Pleurotus ostreatus and Coprinus attrimentarius. Bioact. Carbohydrates Diet. Fibre 2017, 11, 67–74. [Google Scholar] [CrossRef]
- Bekiaris, G.; Tagkouli, D.; Koutrotsios, G.; Kalogeropoulos, N.; Zervakis, G.I. Pleurotus mushrooms content in glucans and ergosterol assessed by ATR-FTIR spectroscopy and multivariate analysis. Foods 2020, 9, 535. [Google Scholar] [CrossRef]
- Kozarski, M.; Klaus, A.; Jakovljevic, D.; Todorovic, N.; Vunduk, J.; Niksic, M.; Vrvic, M.M.; Griensven, L. Van Antioxidants of Edible Mushrooms. Molecules 2015, 20, 19489–19525. [Google Scholar] [CrossRef] [Green Version]
- Ramírez, A.C.; Santoyo, S.; Reglero, G.; Soler-rivas, C. Radical scavenging activities, endogenous oxidative enzymes and total phenols in edible mushrooms commonly consumed in Europe. J. Sci. Food Agric. 2007, 87, 2272–2278. [Google Scholar] [CrossRef]
- Roncero-Ramos, I.; Mendiola-Lanao, M.; Pérez-Clavijo, M.; Delgado-Andrade, C. Effect of different cooking methods on nutritional value and antioxidant activity of cultivated mushrooms. Int. J. Food Sci. Nutr. 2017, 68, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Manley, M. Near-infrared spectroscopy and hyperspectral imaging: Non-destructive analysis of biological materials. Chem. Soc. Rev. 2014, 43, 8200–8214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Workman, J.; Weyer, L. Practical Guide and Spectral Atlas for Interpretive Near-Infrared Spectroscopy, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2012; ISBN 9781574447842. [Google Scholar]
- Kandasamy, S.; Chinnappan, S.; Thangaswamy, S.; Balakrishnan, S.; Khalifa, A.Y.Z. Assessment of Antioxidant, Antibacterial Activities and Bioactive Compounds of the Wild Edible Mushroom Pleurotus sajor-caju. Int. J. Pept. Res. Ther. 2020, 26, 1575–1581. [Google Scholar] [CrossRef]
- Yan, J.; Zhu, L.; Qu, Y.; Qu, X.; Mu, M.; Zhang, M.; Muneer, G.; Zhou, Y.; Sun, L. Analyses of active antioxidant polysaccharides from four edible mushrooms. Int. J. Biol. Macromol. 2019, 123, 945–956. [Google Scholar] [CrossRef]
- Zhang, Y.; Dai, L.; Kong, X.; Chen, L. Characterization and in vitro antioxidant activities of polysaccharides from Pleurotus ostreatus. Int. J. Biol. Macromol. 2012, 51, 259–265. [Google Scholar] [CrossRef]
- Kortei, N.; Wiafe-Kwagyan, M. Comparative Appraisal of the Total Phenolic Content, Flavonoids, Free Radical Scavenging Activity and Nutritional Qualities of Pleurotus ostreatus (EM-1) and Pleurotus eous (P-31) Cultivated on Rice (Oryzae sativa) Straw in Ghana. J. Adv. Biol. Biotechnol. 2015, 3, 153–164. [Google Scholar] [CrossRef]
- Jeena, G.S.; Punetha, H.; Prakash, O.; Chandra, M.; Kushwaha, K.P.S. Study on in vitro antioxidant potential of some cultivated pleurotus species (oyster mushroom). Indian J. Nat. Prod. Resour. 2014, 5, 56–61. [Google Scholar]
- Yim, H.S.; Chye, F.Y.; Tan, C.T.; Ng, Y.C.; Ho, C.W. Antioxidant activities and total phenolic content of aqueous extract of Pleurotus ostreatus (cultivated oyster mushroom). Malays. J. Nutr. 2010, 16, 281–291. [Google Scholar]
- Bindhu, J.; Das, A. Evaluation of Edible Oyster Mushroom (Pleurotus ostreatus) on Oxidative Stress and Neurological Cognitive Disorder in Streptozotocin –Diabetic Rats. Clin. Investig. (Lond.) 2018, 07, 43–52. [Google Scholar] [CrossRef] [Green Version]
- Manzi, P.; Gambelli, L.; Marconi, S.; Vivanti, V.; Pizzoferrato, L. Nutrients in edible mushrooms: An inter-species comparative study. Food Chem. 1999, 65, 477–482. [Google Scholar] [CrossRef]
- Oyetayo, O.V.; Ariyo, O.O. Micro and Macronutrient Properties of Pleurotus ostreatus (Jacq.:Fries) Cultivated on Different Wood Substrates. Jordan J. Biol. Sci. 2013, 6, 223–226. [Google Scholar] [CrossRef] [Green Version]
- Mendez, L.A.; Sandoval Castro, C.A.; Belmar Casso, R.; Capetillo Leal, C.M. Effect of substrate and harvest on the amino acid profile of Oyster mushroom (Pleurotus ostreatus). J. Food Compos. Anal. 2005, 18, 447–450. [Google Scholar] [CrossRef]
- Sopanrao PATIL, S.; Abrar AHMED, S.; Manoharrao TELANG, S.; Mushtaq Vaseem BAIG, M. the Nutritional Value of Pleurotus Ostreatus (Jacq.:Fr.) Kumm Cultivated on Different Lignocellulosic Agro-Wastes. Innov. Rom. Food Biotechnol. 2010, 7, 66–76. [Google Scholar]
- Mattila, P.; Salo-Väänänen, P.; Könkö, K.; Aro, H.; Jalava, T. Basic composition and amino acid contents of mushrooms cultivated in Finland. J. Agric. Food Chem. 2002, 50, 6419–6422. [Google Scholar] [CrossRef]
- Bach, F.; Helm, C.V.; Bellettini, M.B.; Maciel, G.M.; Haminiuk, C.W.I. Edible mushrooms: A potential source of essential amino acids, glucans and minerals. Int. J. Food Sci. Technol. 2017, 52, 2382–2392. [Google Scholar] [CrossRef]
- Zervakis, G.I.; Bekiaris, G.; Tarantilis, P.A.; Pappas, C.S. Rapid strain classification and taxa delimitation within the edible mushroom genus Pleurotus through the use of diffuse reflectance infrared Fourier transform (DRIFT) spectroscopy. Fungal Biol. 2012, 116, 715–728. [Google Scholar] [CrossRef]
- Jerley, A.A.; Rani, J.J.P.; Rajalakshmi, M.; Gnanam, S.T. Impact of Mushroom Pleurotus Ostreatus on Synthesis of Chitosan & Its Effects in Antioxidant & Antibacterial Activity. JETIR 2020, 7, 399–415. [Google Scholar]
- Radzki, W.; Kalbarczyk, J. Water soluble polysaccharides content in three species of edible and medicinal mushrooms: Lentinula edodes, Pleurotus ostreatus, Agaricus blazei. Herba Pol. 2010, 56, 31–38. [Google Scholar]
- Das, B.; Rajkonwar, J.; Jagannath, A.; Raul, P.K.; Deb, U. Infra-red spectra of different species of cultivated oyster mushrooms: Possible tool for identifying bioactive compounds and establishing taxonomic linkage. Def. Life Sci. J. 2020, 5, 118–124. [Google Scholar] [CrossRef]
- Stamets, P. Growing Gourmet and Medical Mushrooms; Ten Speed Press: New York, NY, USA, 2000; ISBN 978-1-60774-138-1. [Google Scholar]
- UPOV. International Union for the Protection of New Varieties of Plants. Oyster Mushroom; King Oyster Mushroom; Lung Oyster Mushroom; UPOV: Geneva, Switzerland, 2003; pp. 1–35. [Google Scholar]
- Singleton, V.L.; Rossi, J.A.J. Colorimetry to total phenolics with phosphomolybdic acid reagents. Am. J. Enol. Vinic. 1965, 16, 144–158. [Google Scholar]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ostertagová, E.; Ostertag, O.; Kováč, J. Methodology and application of the Kruskal-Wallis test. Appl. Mech. Mater. 2014, 611, 115–120. [Google Scholar] [CrossRef]
- Ringnér, M. What is principal component analysis? Nat. Biotechnol. 2008, 26, 303–304. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, P.; Soares, A.; Castanho, A.; Almeida, A.S.; Oliveira, J.; Brites, C. Dataset of Near-infrared spectroscopy measurement for amylose determination using PLS algorithms. Data Br. 2017, 15, 389–396. [Google Scholar] [CrossRef]
- Tharwat, A.; Gaber, T.; Ibrahim, A.; Hassanien, A.E. Linear discriminant analysis: A detailed tutorial. AI Commun. 2017, 30, 169–190. [Google Scholar] [CrossRef] [Green Version]
- Beć, K.B.; Huck, C.W. Breakthrough potential in near-infrared spectroscopy: Spectra simulation. A review of recent developments. Front. Chem. 2019, 7, 1–22. [Google Scholar] [CrossRef] [Green Version]
- Mikola, E.; Geösel, A.; Stefanovits-Bányai, É.; Fodor, M. Quantitative determination of macro components and classification of some cultivated mushrooms using near- infrared spectroscopy. J. Food Process. Preserv. 2020, 1–11. [Google Scholar] [CrossRef]
- Rácz, A.; Héberger, K.; Fodor, M. Quantitative determination and classification of energy drinks using near-infrared spectroscopy. Anal. Bioanal. Chem. 2016, 408, 6403–6411. [Google Scholar] [CrossRef] [Green Version]





| Examined property | Method | References |
|---|---|---|
| Free radical scavenging activity | DPPH value | [2,7,14,15,16,17,18,19], |
| FRAP value | [2,19,20] | |
| Total polyphenol content | Folin Ciocalteu method | [2,17,18,19] |
| Active antioxidant polysaccharides | HPLC | [15] |
| Amino acid content | HPLC with fluorescence detector | [2,21,22,23,24,25,26] |
| Glucans and ergosterol | ATR-FTIR | [7,8] |
| Strain classification and taxa delimitation | FT-IR | [27] |
| Identifying bioactive compounds | ATR-FTIR | [27] |
| Antioxidant activity | DPPH value and FTIR | [28] |
| Water soluble polysaccharides | FTIR | [29] |
| Phylogenetic relatedness—DNA | FTIR | [30] |
| Wavenumber, cm−1 | Spectra Structure | Material Type |
|---|---|---|
| 7353 | C–H methyl second combination band | Aliphatic hydrocarbons |
| 7300 | Combination of first overtone of methyl C–H stretching and CH3 bending | Aromatic hydrocarbons |
| 7194 | Combination of first overtone of methyl C–H stretching and CH3 bending | Aliphatic hydrocarbons |
| 7168 | C–H methylene second combination band | Aliphatic hydrocarbons |
| 7163 | Combination of first overtone of methyl C–H stretching and CH3 bending | Aliphatic hydrocarbons |
| 7200–7100 | First overtone of O–H stretching modes | Alcohols |
| 7140–6940 | First overtone of O–H stretching modes from phenols | Phenolic O–H |
| 7085–7067 | Combination of first overtone of methylene C–H stretching and methylene C–H bending modes | Aliphatic hydrocarbons |
| 7057 | Combination of first overtone of C–H and aryl C–H | Aromatic hydrocarbons |
| Wavenumber, cm−1 | Spectra Structure | Material Type |
|---|---|---|
| 4400 | CONH2 specifically due to peptide β-sheet structures | Proteins |
| 4365–4370 | CONH2 specifically due to the α-helix peptide structure | Proteins |
| 4360 | First overtone of CH stretching and C–H bending modes | C–H aryl |
| 4348 | Second overtone of C–H stretching mode | Amides |
| 4333 | Combination of first overtone of CH2 asymmetric stretching and CH2 bending modes | Aliphatic hydrocarbons |
| 4314 | Combination of first overtone of CH2 asymmetric stretching and CH2 bending modes | Aliphatic hydrocarbons |
| 4261 | Combination of first overtone of CH2 symmetric stretching and CH2 bending modes | Aliphatic hydrocarbons |
| 4232 | Combination of first overtone of CH2 symmetric stretching and CH2 bending modes | Aliphatic hydrocarbons |
| 4190 | C–C bending and C–H stretching modes | C–H Aryl |
| 4155 | C–H stretching and C–H bending modes | C–H Aryl |
| 4091 | C–C stretching and C–H stretching modes | C–H Aryl |
| 4090 | CONH2 specifically due to the α-helix peptide structure | Proteins |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fodor, M.; Mikola, E.E.; Geösel, A.; Stefanovits-Bányai, É.; Mednyánszky, Z. Application of Near-Infrared Spectroscopy to Investigate Some Endogenic Properties of Pleurotus ostreatus Cultivars. Sensors 2020, 20, 6632. https://doi.org/10.3390/s20226632
Fodor M, Mikola EE, Geösel A, Stefanovits-Bányai É, Mednyánszky Z. Application of Near-Infrared Spectroscopy to Investigate Some Endogenic Properties of Pleurotus ostreatus Cultivars. Sensors. 2020; 20(22):6632. https://doi.org/10.3390/s20226632
Chicago/Turabian StyleFodor, Marietta, Erika Etelka Mikola, András Geösel, Éva Stefanovits-Bányai, and Zsuzsanna Mednyánszky. 2020. "Application of Near-Infrared Spectroscopy to Investigate Some Endogenic Properties of Pleurotus ostreatus Cultivars" Sensors 20, no. 22: 6632. https://doi.org/10.3390/s20226632






