It is impossible to imagine today’s everyday life without functional coatings modifying and enhancing the surface properties of different materials. Specially tailored for specific applications they serve a broad range of functionalities, for example the improvement of historic urban districts [
1], drag reduction by means of riblets in the aircraft industry [
2,
3], increased biological compatibility in medical technologies [
4,
5] and corrosion protection in the light metal industry. One specific type of the latter used in this study are so called conversion coatings which can be applied as part of a pre-treatment process, thus providing excellent protection against environmental degradation and improving the adhesion of subsequently applied coatings at the same time. One noteworthy and, in the last decade, widely used form of passivation is the highly corrosion resistant chromate conversion coating (CCC) consisting of a backbone of trivalent Cr(III) and hexavalent Cr(VI) chromium [
6,
7]. Because of the toxic and carcinogenic attributes of the latter, Europe’s REACH agreements 2017 (Regulation on Registration, Evaluation, Authorisation and Restriction of Chemicals) restricted the use of conversion coatings containing Cr(VI) in general. While those restrictions have led to further investigations of suitable alternatives, one of the most promising substitutes based on Cr(III) and Zr(IV) (SurTec
® 650-chromitAL TCP., SurTec International GmbH, Bensheim, Germany) is used exemplarily in this study. According to the literature, several papers have already been published using SurTec
® 650 as passivation agent on different aluminium alloys backing our choice [
8,
9,
10,
11,
12,
13]. Additionally, researchers like Kim et al. applied this agent on the commonly used AA3003 (here on aluminium foil) which serves as our substrate of choice [
14]. While most publications focus on an improved understanding of the chemical formation process, the optimization of treatment conditions in terms of increased corrosion protection or use in situ spectroscopic ellipsometry (SE) as a tool for studying the growth kinetics of TCP coatings [
15,
16], this study primarily focuses on coating thickness measurements. Note that it is a challenging task for SE as well as for reflectometry to investigate rough samples in general. Exemplarily, Lehmann et al. addressed this problem by presenting a new optical roughness model for thin films with a peak-to-valley distances between 10
and 60
, which still is significantly smaller than our cold-rolled and coated industrial specimens ranging in the micron regime [
17]. At this point it is noteworthy that Siah et al. showed that it is possible to extract film thicknesses and refractive indices only at discrete wavelengths due to depolarization effects when investigating thin films deposited on rough Si-wafer [
18].
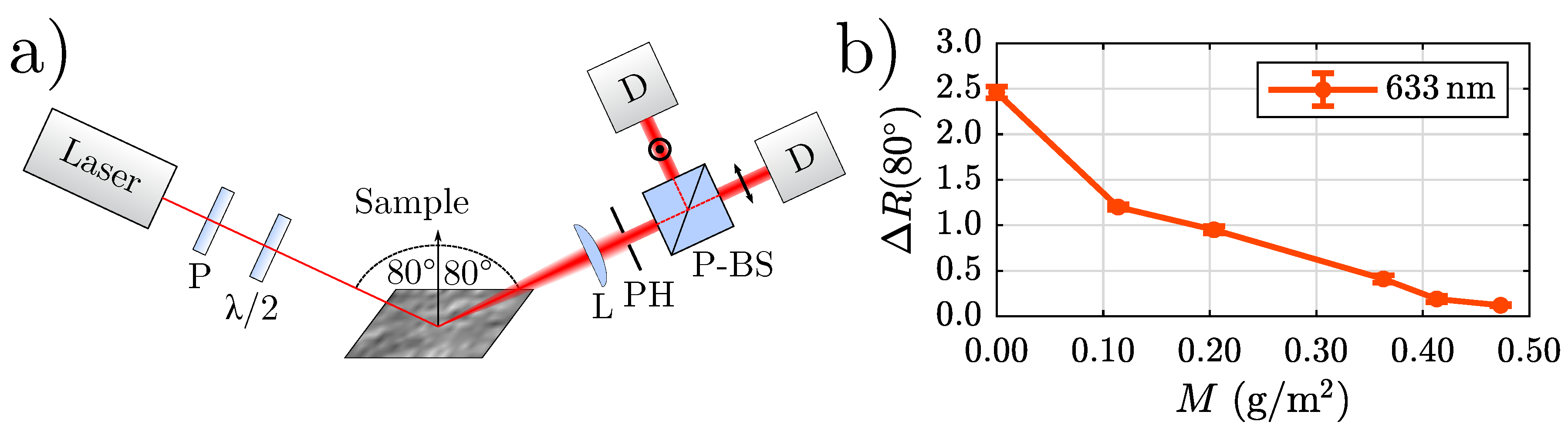
An optical method capable of detecting and distinguishing between different thicknesses of trivalent chromium conversion (TCC) coatings on rough cold-rolled AA3003 aluminium substrates has already been presented in the literature by us showing that our suggested measurand
correlates strongly with the estimated coating weight
M [
19]. This finding is briefly reviewed in
Figure 1. The left panel of
Figure 1 schematically depicts the sensor concept: The light beam of a helium-neon laser (
,
) is guided grazingly (angle of incidence amounts to
with respect to the surface normal) onto the sample. The polarizer (P) only transmits linearly polarized light and the
/2 waveplate rotates the polarization direction to
. The specularly reflected light is focused with a lens (L,
) and guided through a pinhole (PH). Using a polarizing beam splitter (P-BS), the light beam is separated into two perpendicularly polarized parts and detected by two Si-photodiodes (D, OSD50). It is important that the detection takes place in specular direction, meaning
.
Figure 1b shows the result of a measurement performed with the setup depicted in
Figure 1a. Six cold-rolled AA3003 samples with different coating weights
M have been investigated. Our measurand
decreases strongly with increasing coating weight.
In this publication we address the underlying physical origin of the previously found correlation.