Fabrication and Characterization of Iridium Oxide pH Microelectrodes Based on Sputter Deposition Method
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Instruments
2.3. Preparation of Iridium Oxide pH Microelectrode
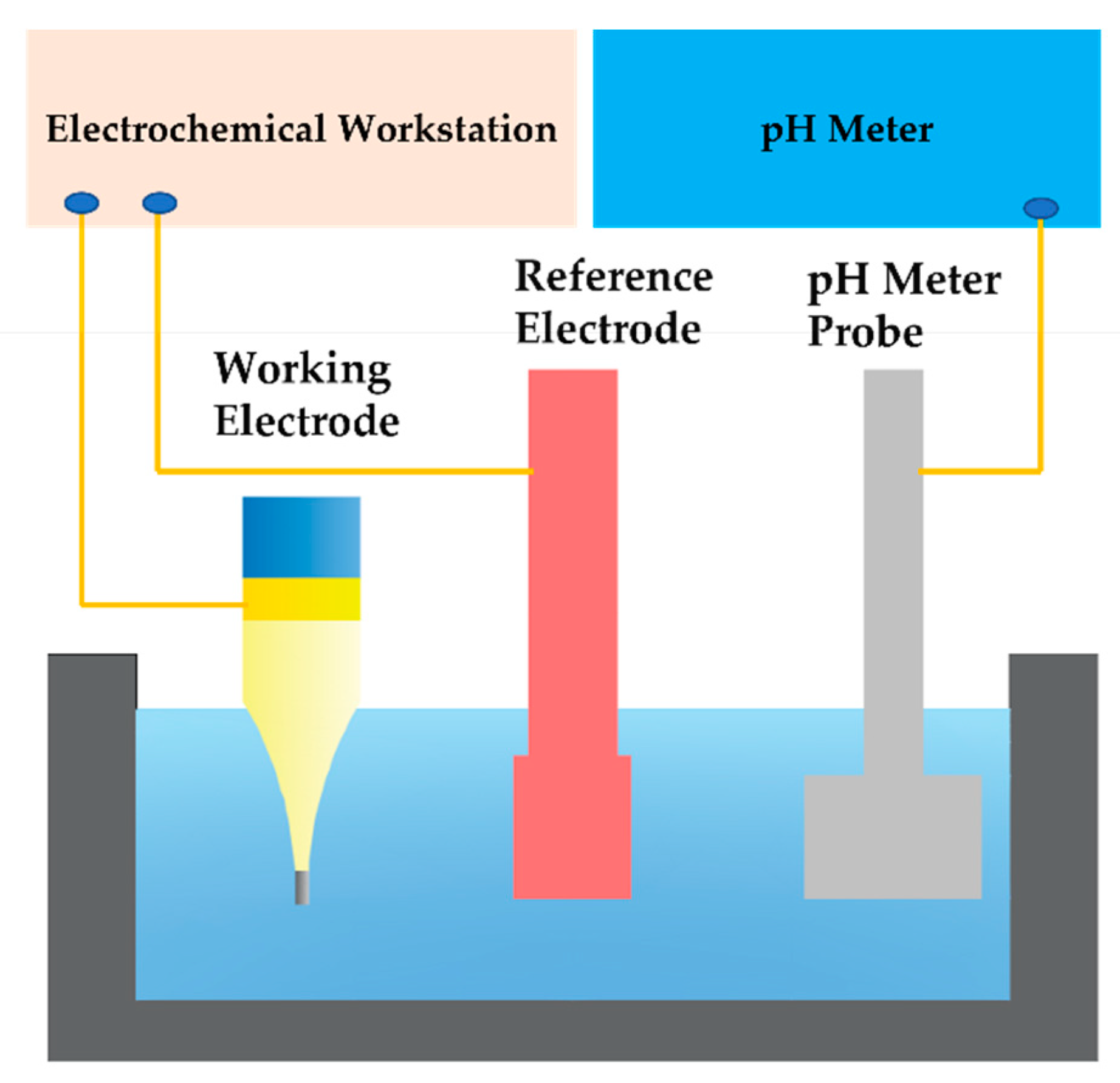
2.4. Electrochemical Testing
3. Results and Discussion
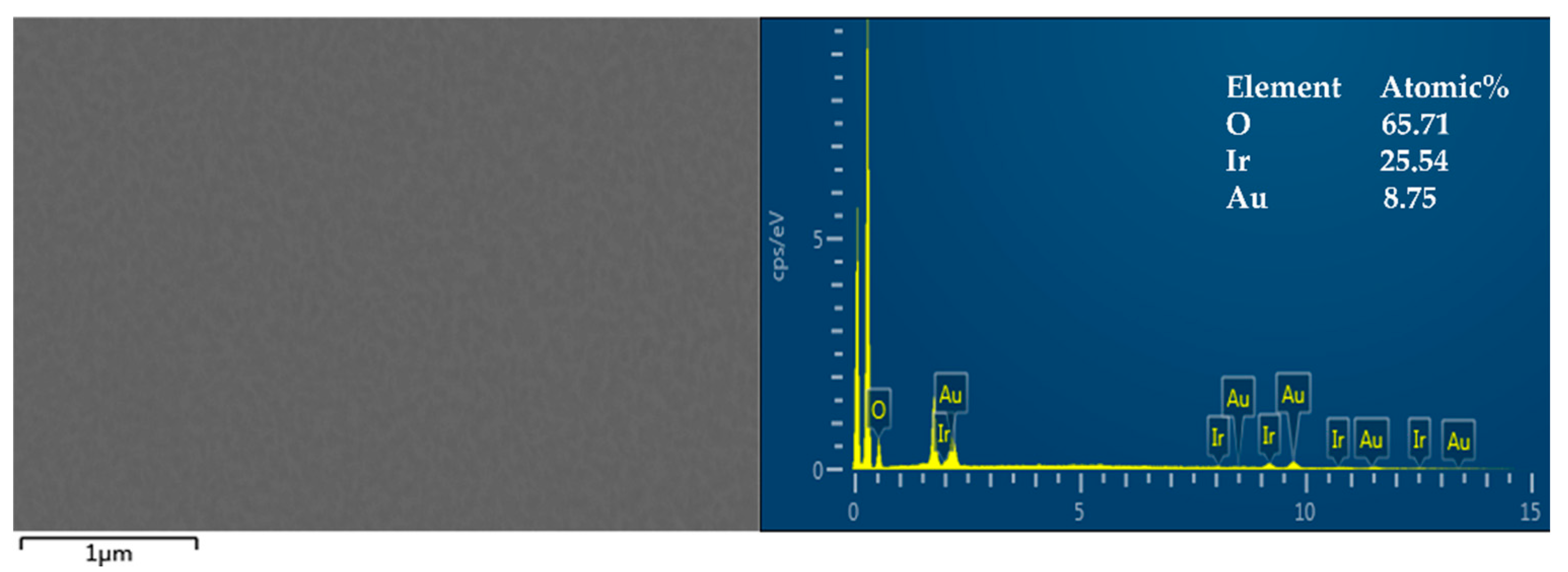
3.1. Preparation of Iridium Oxide Layer
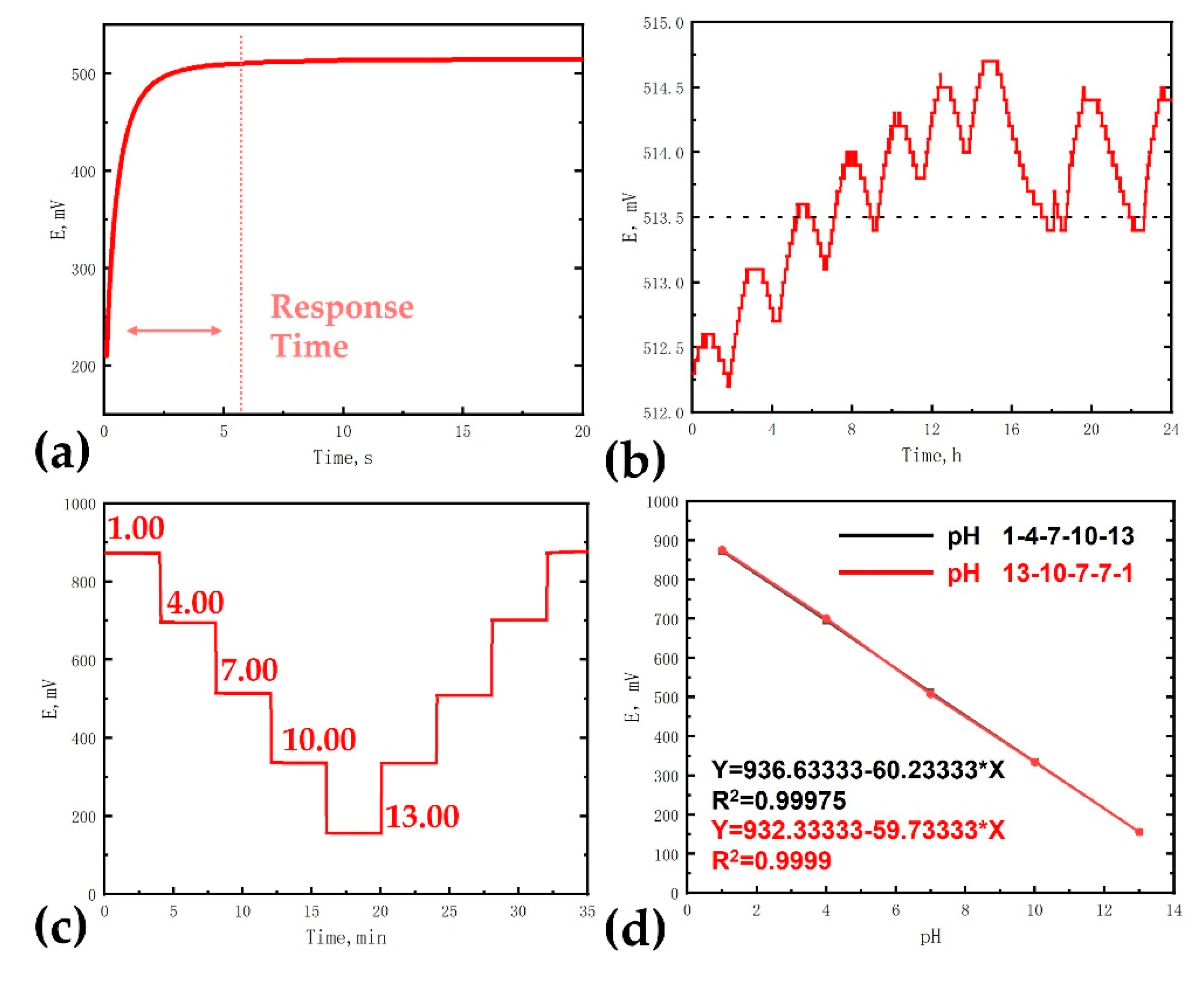
3.2. PH Sensing of Iridium Oxide Microelectrodes
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhou, Z.; Li, J.; Pan, D.; Wei, H.; Wang, C.; Pan, F.; Xia, J.; Ma, S. pH electrodes based on iridium oxide films for marine monitoring. Trends Environ. Anal. Chem. 2020, 25, e83. [Google Scholar] [CrossRef]
- Ganesh Kumar Mani, M.M.Y.Y.; Tsuchiya, A.K. ZnO-Based Microfluidic pH Sensor: A Versatile Approach for Quick Recognition of Circulating Tumor Cells in Blood. ACS Appl. Mater. Interfaces 2017, 9, 5193–5203. [Google Scholar] [CrossRef] [PubMed]
- Venn, A.A.; Tambutte, E.; Holcomb, M.; Laurent, J.; Allemand, D.; Tambutte, S. Impact of seawater acidification on pH at the tissue-skeleton interface calcification in reef corals. Proc. Natl. Acad. Sci. USA 2013, 110, 1634–1639. [Google Scholar] [CrossRef] [Green Version]
- Webb, B.A.; Chimenti, M.; Jacobson, M.P. Dysregulated pH: A perfect storm for cancer progression. Nat. Rev. Cancer 2011, 11, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Salvoa, P.; Calisi, N.; Melai, B.; Cortigiani, B.; Mannini, M.; Caneschi, A.; Lorenzetti, G.; Paoletti, C.; Lomonaco, T.; Paolicchi, A. Temperature pH sensors based on graphenic materials. Biosens. Bioelectron. 2017, 91, 870–877. [Google Scholar] [CrossRef] [PubMed]
- Fan, K.; Weisenhorn, P.; Gilbert, J.A.; Shi, Y.; Bai, Y.; Chu, H. Soil pH correlates with the co-occurrence and assemblage process of diazotrophic communities in rhizosphere and bulk soils of wheat fields. Soil Biol. Biochem. 2018, 121, 185–192. [Google Scholar] [CrossRef]
- Abu-Thabit, N.; Umar, Y.; Ratemi, E.; Ahmad, A.; Ahmad Abuilaiwi, F. A Flexible Optical pH Sensor Based on Polysulfone Membranes Coated with pH-Responsive Polyaniline Nanofibers. Sensors 2016, 16, 986. [Google Scholar] [CrossRef] [Green Version]
- Benčina, M. Illumination of the Spatial Order of Intracellular pH by Genetically Encoded pH-Sensitive Sensors. Sensors 2013, 13, 16736–16758. [Google Scholar] [CrossRef] [Green Version]
- Huang, W.; Cao, H.; Deb, S.; Chiao, M.; Chiao, J.C. A flexible pH sensor based on the iridium oxide sensing film. Sens. Actuator A Phys. 2011, 169, 1–11. [Google Scholar] [CrossRef]
- Martinez, C.C.M.; Madrid, R.E.; Felice, C.J. A pH sensor based on a stainless steel electrode electrodeposited with iridium oxide. IEEE Trans. Educ. 2009, 52, 133–136. [Google Scholar]
- Jang, H.; Lee, J. Iridium oxide fabrication and application: A review. J. Energy Chem. 2020, 46, 152–172. [Google Scholar] [CrossRef]
- Kurzweil, P. Metal Oxides and Ion-Exchanging Surfaces as pH Sensors in Liquids: State-of-the-Art and Outlook. Sensors 2009, 9, 4955–4985. [Google Scholar] [CrossRef] [Green Version]
- Mingels, R.H.G.; Kalsi, S.; Cheong, Y.; Morgan, H. Iridium and Ruthenium oxide miniature pH sensors: Long-term performance. Sens. Actuators B Chem. 2019, 297, 126779. [Google Scholar] [CrossRef]
- Yao, X.; Vepsäläinen, M.; Isa, F.; Martin, P.; Munroe, P.; Bendavid, A. Advanced RuO2 Thin Films for pH Sensing Application. Sensors 2020, 20, 6432. [Google Scholar] [CrossRef]
- Pan, Y.; Sun, Z.; He, H.; Li, Y.; You, L.; Zheng, H. An improved method of preparing iridium oxide electrode based on carbonate-melt oxidation mechanism. Sens. Actuators B Chem. 2018, 261, 316–324. [Google Scholar] [CrossRef]
- Khalil, M.; Wang, S.; Yu, J.; Lee, R.L.; Liu, N. Electrodeposition of Iridium Oxide Nanoparticles for pH Sensing Electrodes. J. Electrochem. Soc. 2016, 163, B485–B490. [Google Scholar] [CrossRef]
- Manjakkal, L.; Szwagierczak, D.; Dahiya, R. Metal oxides based electrochemical pH sensors: Current progress and future perspectives. Prog. Mater. Sci. 2020, 109, 100635. [Google Scholar] [CrossRef]
- Naroa, U.; Natalia, A.; Andrey, B.; Francesc-Xavier, M.; Eva, B. Miniaturized metal oxide pH sensors for bacteria detection. Talanta 2016, 147, 364–369. [Google Scholar]
- Xi, Y.; Ji, B.; Guo, Z.; Li, W.; Liu, J. Fabrication and Characterization of Micro-Nano Electrodes for Implantable BCI. Micromachines 2019, 10, 242. [Google Scholar] [CrossRef] [Green Version]
- Pan, R.; Xu, M.; Jiang, D.; Burgess, J.D.; Chen, H. Nanokit for single-cell electrochemical analyses. Proc. Natl. Acad. Sci. USA 2016, 113, 11436–11440. [Google Scholar] [CrossRef] [Green Version]
- Zhao, H.; Ye, D.; Mao, X.; Li, F.; Xu, J.; Li, M.; Zuo, X. Stepping gating of ion channels on nanoelectrode via DNA hybridization for label-free DNA detection. Biosens. Bioelectron. 2019, 133, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Kang, X.; Liu, J.; Tian, H.; Yang, B.; NuLi, Y.; Yang, C. Optimization and electrochemical characterization of RF-sputtered iridium oxide microelectrodes for electrical stimulation. J. Micromech. Microeng. 2014, 24, 25015. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xi, Y.; Guo, Z.; Wang, L.; Xu, Q.; Ruan, T.; Liu, J. Fabrication and Characterization of Iridium Oxide pH Microelectrodes Based on Sputter Deposition Method. Sensors 2021, 21, 4996. https://doi.org/10.3390/s21154996
Xi Y, Guo Z, Wang L, Xu Q, Ruan T, Liu J. Fabrication and Characterization of Iridium Oxide pH Microelectrodes Based on Sputter Deposition Method. Sensors. 2021; 21(15):4996. https://doi.org/10.3390/s21154996
Chicago/Turabian StyleXi, Ye, Zhejun Guo, Longchun Wang, Qingda Xu, Tao Ruan, and Jingquan Liu. 2021. "Fabrication and Characterization of Iridium Oxide pH Microelectrodes Based on Sputter Deposition Method" Sensors 21, no. 15: 4996. https://doi.org/10.3390/s21154996




