Comparison of QEEG Findings before and after Onset of Post-COVID-19 Brain Fog Symptoms
Abstract
:1. Introduction
2. Materials and Methods
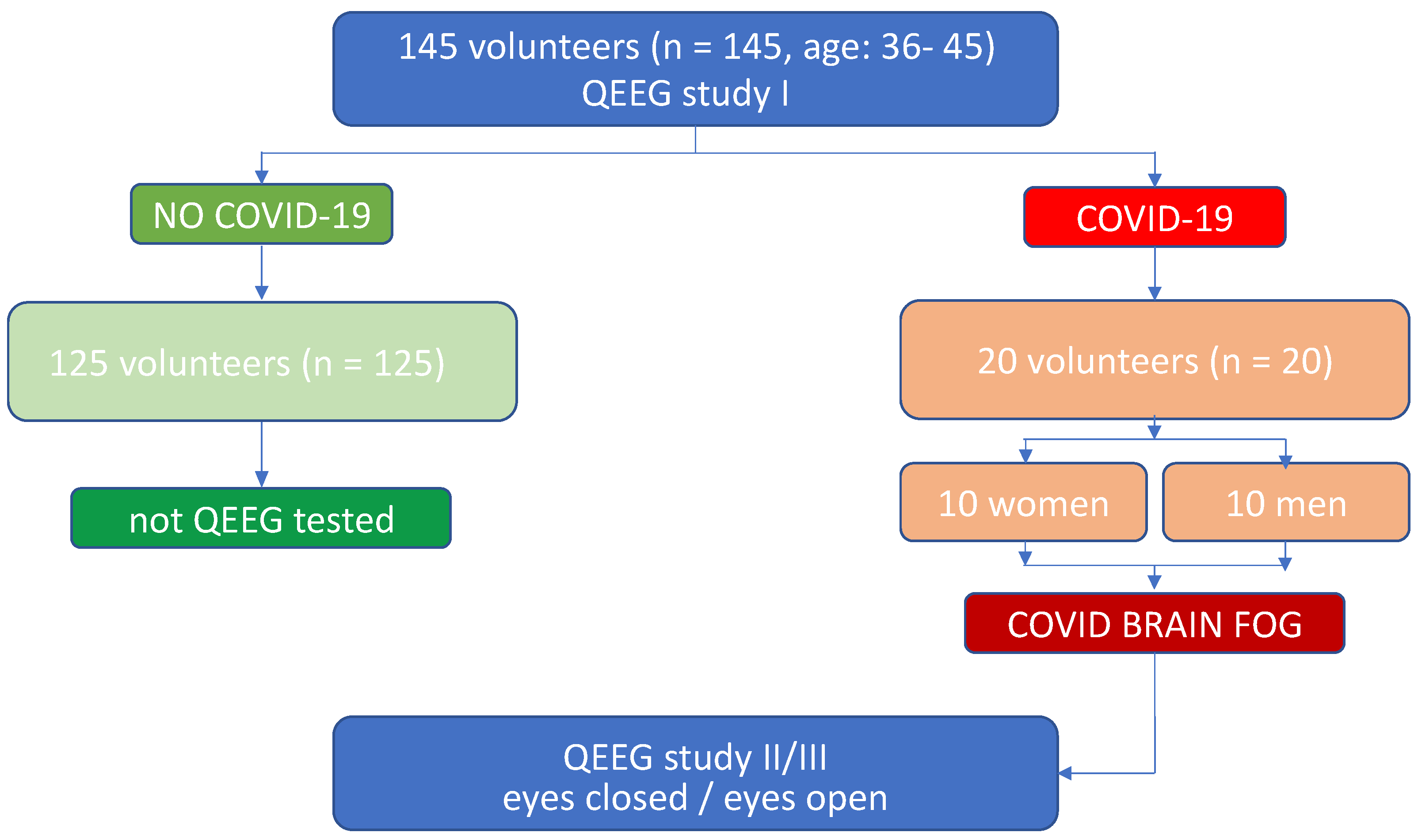
2.1. Participants
2.2. Experimental Design
2.3. Measures for Identifying Brain Fog
2.4. QEEG Procedure
2.5. Linking of Baseline to Experimental Subjects
3. Statistical Analyses
4. Results
5. Discussion of Results and Conclusions
- Relative increase of Theta, Alpha and SMR frequencies in the right hemisphere as compared to the left hemisphere.
- Remarkable increase in Beta 2 versus SMR in both hemispheres.
- Increase in Beta 1 in the left hemisphere.
- Reduction in SMR values
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Program STOP-COVID (the First Polish Program for the Assessment of Complications after COVID-19 in People Who Have Not Been Hospitalized due to Coronavirus). Available online: http://www.stop-covid.pl/ (accessed on 28 August 2022).
- Horesh, D.; Brown, A.D. Traumatic stress in the age of COVID-19: A call to close critical gaps and adapt to new realities. Psychol. Trauma Theory Res. Pract. Policy 2020, 12, 331–335. [Google Scholar] [CrossRef]
- Leigh-Hunt, N.; Bagguley, D.; Bash, K.; Turner, V.; Turnbull, S.; Valtorta, N.; Caan, W. An overview of systematic reviews on the public health consequences of social isolation and loneliness. Public Health 2017, 152, 157–171. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Pan, R.; Wan, X.; Tan, Y.; Xu, L.; Ho, C.S.; Ho, R.C. Immediate Psychological Responses and Associated Factors during the Initial Stage of the 2019 Coronavirus Disease (COVID-19) Epidemic among the General Population in China. Int. J. Environ. Res. Public Health 2020, 17, 1729. [Google Scholar] [CrossRef] [PubMed]
- de Figueiredo, C.S.; Sandre, P.C.; Portugal, L.C.L.; Mázala-De-Oliveira, T.; da Silva Chagas, L.; Raony, Í.; Ferreira, E.S.; Giestal-De-Araujo, E.; dos Santos, A.A.; Bomfim, P.O.-S. COVID-19 pandemic impact on children and adolescents’ mental health: Biological, environmental, and social factors. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2020, 106, 110171. [Google Scholar] [CrossRef] [PubMed]
- Ren, F.-F.; Guo, R.-J. Public Mental Health in Post-COVID-19 Era. Psychiatr. Danub. 2020, 32, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Hellmuth, J.; Barnett, T.A.; Asken, B.M.; Kelly, J.D.; Torres, L.; Stephens, M.L.; Greenhouse, B.; Martin, J.N.; Chow, F.C.; Deeks, S.G.; et al. Persistent COVID-19 associated neurocogni-tive symptoms in non-hospitalized patients. J. Neurovirol. 2021, 27, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Bliddal, S.; Banasik, K.; Pedersen, O.B.; Nissen, J.; Cantwell, L.; Schwinn, M.; Tulstrup, M.; Westergaard, D.; Ullum, H.; Brunak, S.; et al. Acute and persistent symptoms in non-hospitalized PCR-confirmed COVID-19 patients. Sci. Rep. 2021, 11, 1–11. [Google Scholar] [CrossRef]
- Correia, A.O.; Feitosa, P.W.G.; Moreira, J.L.D.S.; Nogueira, S.R.; Fonseca, R.B.; Nobre, M.E.P. Neurological manifestations of COVID-19 and other coronaviruses: A systematic review. Neurol. Psychiatry Brain Res. 2020, 37, 27–32. [Google Scholar] [CrossRef]
- Kopańska, M.; Batoryna, M.; Bartman, P.; Szczygielski, J.; Banaś-Ząbczyk, A. Disorders of the Cholinergic System in COVID-19 Era—A Review of the Latest Research. Int. J. Mol. Sci. 2022, 23, 672. [Google Scholar] [CrossRef]
- Han, Q.; Zheng, B.; Daines, L.; Sheikh, A. Long-Term Sequelae of COVID-19: A Systematic Review and Meta-Analysis of One-Year Follow-Up Studies on Post-COVID Symptoms. Pathogens 2022, 11, 269. [Google Scholar] [CrossRef]
- Mao, L.; Jin, H.; Wang, M.; Hu, Y.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D.; et al. Neurologic Manifestations of Hospitalized Patients With Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020, 77, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Sharifian-Dorche, M.; Huot, P.; Osherov, M.; Wen, D.; Saveriano, A.; Giacomini, P.S.; Antel, J.P.; Mowla, A. Neurological complications of coronavirus infection; a comparative review and lessons learned during the COVID-19 pandemic. J. Neurol. Sci. 2020, 417, 117085. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.-C.; Ko, W.-C.; Lee, P.-I.; Jean, S.-S.; Hsueh, P.-R. Extra-respiratory manifestations of COVID-19. Int. J. Antimicrob. Agents 2020, 56, 106024. [Google Scholar] [CrossRef] [PubMed]
- Callan, C.; Ladds, E.; Husain, L.; Pattinson, K.; Greenhalgh, T. ‘I can’t cope with multiple inputs’: A qualitative study of the lived experience of ‘brain fog’ after COVID-19. BMJ Open 2022, 12, e056366. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, K.; Lin, Y.; Prewitt, K.-R.M.; Potter, D.A. Multidisciplinary Approach to Brain Fog and Related Persisting Symptoms Post COVID-19. J. Health Serv. Psychol. 2022, 1–8. [Google Scholar] [CrossRef]
- Matias-Guiu, J.A.; Delgado-Alonso, C.; Yus, M.; Polidura, C.; Gómez-Ruiz, N.; Valles-Salgado, M.; Ortega-Madueño, I.; Cabrera-Martín, M.N.; Matias-Guiu, J. “Brain Fog” by COVID-19 or Alzheimer’s Disease? A Case Report. Front. Psychol. 2021, 12, 724022. [Google Scholar] [CrossRef]
- Ortelli, P.; Ferrazzoli, D.; Sebastianelli, L.; Maestri, R.; Dezi, S.; Spampinato, D.; Saltuari, L.; Alibardi, A.; Kofler, M.; Quartarone, A.; et al. Fatigue and “brain fog” in the aftermath of mild COVID-19: A neuropsychological and TMS study. J. Neurol. Sci. 2021, 429, 119854. [Google Scholar] [CrossRef]
- Hugon, J.; Msika, E.-F.; Queneau, M.; Farid, K.; Paquet, C. Long COVID: Cognitive complaints (brain fog) and dysfunction of the cingulate cortex. J. Neurol. 2021, 269, 44–46. [Google Scholar] [CrossRef]
- Cecchetti, G.; Agosta, F.; Canu, E.; Basaia, S.; Barbieri, A.; Cardamone, R.; Bernasconi, M.P.; Castelnovo, V.; Cividini, C.; Cursi, M.; et al. Cognitive, EEG, and MRI features of COVID-19 survivors: A 10-month study. J. Neurol. 2022, 269, 3400–3412. [Google Scholar] [CrossRef]
- van der Hiele, K.; Vein, A.; Reijntjes, R.; Westendorp, R.; Bollen, E.; van Buchem, M.; van Dijk, J.; Middelkoop, H. EEG correlates in the spectrum of cognitive decline. Clin. Neurophysiol. 2007, 118, 1931–1939. [Google Scholar] [CrossRef]
- Vespignani, H.; Colas, D.; Lavin, B.S.; Soufflet, C.; Maillard, L.; Pourcher, V.; Paccoud, O.; Medjebar, S.; Mba, P.F. Report on Electroencephalographic Findings in Critically Ill Patients with COVID-19. Ann. Neurol. 2020, 88, 626–630. [Google Scholar] [CrossRef]
- Antony, A.R.; Haneef, Z. Systematic review of EEG findings in 617 patients diagnosed with COVID-19. Seizure 2020, 83, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Altuna, M.; Sánchez-Saudinós, M.; Lleó, A. Cognitive symptoms after COVID-19. Neurol. Perspect. 2021, 1, S16–S24. [Google Scholar] [CrossRef]
- Fetz, E.E. Volitional control of neural activity: Implications for brain-computer interfaces. J. Physiol. 2007, 579, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Jurewicz, K.; Paluch, K.; Kublik, E.; Rogala, J.; Mikicin, M.; Wróbel, A. EEG-neurofeedback training of Beta band (12–22 Hz) affects Alpha and Beta frequencies—A controlled study of a healthy population. Neuropsychologia 2018, 108, 13–24. [Google Scholar] [CrossRef]
- Zamysłowski, S. Schemes of EEG electrode placement in humans. In Licensing training for a biofeedback specialist and therapist, Edition II; Kubik A, Ed.; Polish Society of Clinical Neurophysiology: Warsaw, Poland, 2015; pp. 47–51. [Google Scholar]
- Gudmundsson, S.; Runarsson, T.P.; Sigurdsson, S.; Eiriksdottir, G.; Johnsen, K. Reliability of quantitative EEG features. Clin. Neurophysiol. 2007, 118, 2162–2171. [Google Scholar] [CrossRef]
- Marzbani, H.; Marateb, H.R.; Mansourian, M. Methodological Note: Neurofeedback: A Comprehensive Review on System Design, Methodology and Clinical Applications. Basic Clin. Neurosci. J. 2016, 7, 143–158. [Google Scholar] [CrossRef]
- Pati, S.; Toth, E.; Chaitanya, G. Quantitative EEG markers to prognosticate critically ill patients with COVID-19: A retrospective cohort study. Clin. Neurophysiol. 2020, 131, 1824–1826. [Google Scholar] [CrossRef]
- Li, G.; Huang, S.; Xu, W.; Jiao, W.; Jiang, Y.; Gao, Z.; Zhang, J. The impact of mental fatigue on brain activity: A comparative study both in resting state and task state using EEG. BMC Neurosci. 2020, 21, 1–9. [Google Scholar] [CrossRef]
- Park, W.; Cho, M.; Park, S. Effects of Electroencephalogram Biofeedback on Emotion Regulation and Brain Homeostasis of Late Adolescents in the COVID-19 Pandemic. J. Korean Acad. Nurs. 2022, 52, 36–51. [Google Scholar] [CrossRef]
- Kopańska, M.; Ochojska, D.; Dejnowicz-Velitchkov, A.; Banaś-Ząbczyk, A. Quantitative Electroencephalography (QEEG) as an Innovative Diagnostic Tool in Mental Disorders. Int. J. Environ. Res. Public Health. 2022, 19, 2465. [Google Scholar] [CrossRef] [PubMed]
- Oaklander, A.L.; Mills, A.J.; Kelley, M.; Toran, L.S.; Smith, B.; Dalakas, M.C.; Nath, A. Peripheral Neuropathy Evaluations of Patients With Prolonged Long COVID. Neurol.-Neuroimmunol. 2022, 9, e1146. [Google Scholar] [CrossRef] [PubMed]
- Younger, D.S. Post-acute sequelae of SARS-CoV-2 infection (PASC): Peripheral, autonomic, and central nervous system fea-tures in a child. Neurol. Sci. 2021, 42, 3959–3963. [Google Scholar] [CrossRef] [PubMed]
- Abrams, R.M.C.; Simpson, D.M.; Navis, A.; Jette, N.; Zhou, L.; Shin, S.C. Small fiber neuropathy associated with SARS-CoV-2 infection. Muscle Nerve. 2021, 65, 440–444. [Google Scholar] [CrossRef] [PubMed]
- Pastor, J.; Vega-Zelaya, L.; Abad, E.M. Specific EEG Encephalopathy Pattern in SARS-CoV-2 Patients. J. Clin. Med. 2020, 9, 1545. [Google Scholar] [CrossRef]
- Johns, C.; Tooley, K.M.; Traxler, M.J. Discourse Impairments Following Right Hemisphere Brain Damage: A Critical Review. Lang. Linguistics Compass 2008, 2, 1038–1062. [Google Scholar] [CrossRef]
- Tyng, C.M.; Amin, H.U.; Saad, M.N.M.; Malik, A.S. The Influences of Emotion on Learning and Memory. Front. Psychol. 2017, 8, 1454. [Google Scholar] [CrossRef]
- Choi, M.-J.; Park, W.-J. The Effects of Neurofeedback Training on Physical, Psychoemotional Stress Response and Self-Regulation for Late Adolescence: A Non-Randomized Trial. J. Korean Acad. Nurs. 2018, 48, 208–220. [Google Scholar] [CrossRef]
- Batail, J.M.; Bioulac, S.; Cabestaing, F.; Daudet, C.; Drapier, D.; Fouillen, M.; Fovet, T.; Hakoun, A.; Jardri, R.; Jeunet, C.; et al. EEG neurofeedback research: A fertile ground for psychiatry? L’encephale. 2019, 45, 245–255. [Google Scholar] [CrossRef]
- Markiewicz, R. The use of EEG Biofeedback/Neurofeedback in psychiatric rehabilitation. Psychiatr. Pol. 2017, 51, 1095–1106. [Google Scholar] [CrossRef]
- Kanda, P.A.D.M.; Anghinah, R.; Smidth, M.T.; Silva, J.M. The clinical use of quantitative EEG in cognitive disorders. Dement. Neuropsychol. 2009, 3, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Kopańska, M.; Banaś-Ząbczyk, A.; Łagowska, A.; Kuduk, B.; Szczygielski, J. Changes in EEG Recordings in COVID-19 Patients as a Basis for More Accurate QEEG Diagnostics and EEG Neurofeedback Therapy: A Systematic Review. J. Clin. Med. 2021, 10, 1300. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Chen, K.; Zou, J.; Han, P.; Hao, J.; Han, Z. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front. Med. 2020, 14, 185–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Mode | Pre-COVID | Post-COVID | p | |
|---|---|---|---|---|
| C3, eyes open | mean ± SD | 15.06 ± 0.83 | 13.58 ± 3.52 | p = 0.073 |
| Median | 15.11 | 12.85 | ||
| Quartiles | 14.46–15.5 | 10.46–16.61 | ||
| C4, eyes open | mean ± SD | 15.49 ± 0.85 | 16.66 ± 3.84 | p = 0.24 |
| Median | 15.69 | 15.44 | ||
| Quartiles | 14.76–15.84 | 13.57–21.45 | ||
| C3, eyes closed | mean ± SD | 14.35 ± 0.56 | 14.38 ± 3 | p = 0.927 |
| Median | 14.39 | 14.26 | ||
| Quartiles | 14.05–14.65 | 11.89–17.36 | ||
| C4, eyes closed | mean ± SD | 14.94 ± 0.92 | 16.41 ± 3.21 | p = 0.067 |
| Median | 14.76 | 15.44 | ||
| Quartiles | 14.59–15.31 | 13.78–19.08 | ||
| Mode | Pre-COVID | Post-COVID | p | |
|---|---|---|---|---|
| C3, eyes open | mean ± SD | 8.29 ± 0.59 | 8.55 ± 1.52 | p = 0.452 |
| Median | 8.36 | 9.06 | ||
| Quartiles | 7.7–8.78 | 7.02–9.87 | ||
| C4, eyes open | mean ± SD | 8.49 ± 0.32 | 10.54 ± 1.93 | p = 0.001 * |
| Median | 8.46 | 10.88 | ||
| Quartiles | 8.32–8.69 | 8.25–12.52 | ||
| C3, eyes closed | mean ± SD | 7.59 ± 0.41 | 9.55 ± 0.98 | p < 0.001 * |
| Median | 7.54 | 9.86 | ||
| Quartiles | 7.34–7.7 | 9.13–10.29 | ||
| C4, eyes closed | mean ± SD | 7.89 ± 0.75 | 11.04 ± 1.41 | p < 0.001 * |
| Median | 7.94 | 11.12 | ||
| Quartiles | 7.6–8.43 | 10.4–12.52 | ||
| Mode | Pre-COVID | Post-COVID | p | |
|---|---|---|---|---|
| C3, eyes open | mean ± SD | 6.74 ± 0.74 | 6.84 ± 2.42 | p = 0.538 |
| Median | 7.08 | 6.42 | ||
| Quartiles | 6.19–7.18 | 6.06–6.61 | ||
| C4, eyes open | mean ± SD | 6.58 ± 0.59 | 8.95 ± 2.64 | p = 0.001 * |
| Median | 6.49 | 8.54 | ||
| Quartiles | 6.08–6.99 | 7.71–9.82 | ||
| C3, eyes closed | mean ± SD | 6.02 ± 0.73 | 6.34 ± 0.35 | p = 0.042 * |
| Median | 5.85 | 6.4 | ||
| Quartiles | 5.43–6.38 | 6.04–6.58 | ||
| C4, eyes closed | mean ± SD | 6.36 ± 0.78 | 7.95 ± 1.31 | p = 0.001 * |
| Median | 6.17 | 8.04 | ||
| Quartiles | 5.83–6.99 | 7.33–8.81 | ||
| Mode | Pre-COVID | Post-COVID | p | |
|---|---|---|---|---|
| C3, eyes open | mean ± SD | 4.33 ± 0.2 | 3.19 ± 0.23 | p < 0.001 * |
| Median | 4.38 | 3.16 | ||
| Quartiles | 4.2–4.45 | 3.05–3.19 | ||
| C4, eyes open | mean ± SD | 4.3 ± 0.33 | 4.53 ± 0.69 | p = 0.332 |
| Median | 4.23 | 4.48 | ||
| Quartiles | 4.05–4.4 | 4.23–4.61 | ||
| C3, eyes closed | mean ± SD | 4.69 ± 0.64 | 4.2 ± 0.43 | p = 0.011 * |
| Median | 4.44 | 4.16 | ||
| Quartiles | 4.28–4.99 | 4.05–4.19 | ||
| C4, eyes closed | mean ± SD | 5.01 ± 0.64 | 4.63 ± 0.46 | p = 0.017 * |
| Median | 4.85 | 4.56 | ||
| Quartiles | 4.4–5.73 | 4.31–4.73 | ||
| Mode | Pre-COVID | Post-COVID | p | |
|---|---|---|---|---|
| C3, eyes open | mean ± SD | 4.53 ± 0.33 | 4.32 ± 0.52 | p = 0.191 |
| Median | 4.4 | 4.58 | ||
| Quartiles | 4.25–4.77 | 3.74–4.73 | ||
| C4, eyes open | mean ± SD | 4.45 ± 0.33 | 5.23 ± 0.72 | p = 0.001 * |
| Median | 4.46 | 5.36 | ||
| Quartiles | 4.39–4.53 | 4.47–5.68 | ||
| C3, eyes closed | mean ± SD | 4.48 ± 0.28 | 4.53 ± 0.45 | p = 0.823 |
| Median | 4.36 | 4.71 | ||
| Quartiles | 4.25–4.61 | 4.47–4.76 | ||
| C4, eyes closed | mean ± SD | 4.55 ± 0.29 | 4.93 ± 0.49 | p = 0.014 * |
| Median | 4.51 | 5 | ||
| Quartiles | 4.45–4.62 | 4.47–5.39 | ||
| Mode | Pre-COVID | Post-COVID | p | |
|---|---|---|---|---|
| C3, eyes open | mean ± SD | 5.01 ± 0.25 | 6.8 ± 1.08 | p < 0.001 * |
| Median | 5.06 | 7.09 | ||
| Quartiles | 4.78–5.12 | 5.59–7.59 | ||
| C4, eyes open | mean ± SD | 4.91 ± 0.58 | 8.33 ± 1.3 | p < 0.001 * |
| Median | 4.64 | 8.7 | ||
| Quartiles | 4.38–5.43 | 6.69–8.94 | ||
| C3, eyes closed | mean ± SD | 4.48 ± 0.53 | 6.54 ± 0.97 | p < 0.001 * |
| Median | 4.4 | 6.59 | ||
| Quartiles | 4–4.94 | 5.59–7.5 | ||
| C4, eyes closed | mean ± SD | 4.92 ± 0.62 | 7.33 ± 0.95 | p < 0.001 * |
| Median | 4.97 | 7.08 | ||
| Quartiles | 4.36–5.43 | 6.69–7.74 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopańska, M.; Ochojska, D.; Muchacka, R.; Dejnowicz-Velitchkov, A.; Banaś-Ząbczyk, A.; Szczygielski, J. Comparison of QEEG Findings before and after Onset of Post-COVID-19 Brain Fog Symptoms. Sensors 2022, 22, 6606. https://doi.org/10.3390/s22176606
Kopańska M, Ochojska D, Muchacka R, Dejnowicz-Velitchkov A, Banaś-Ząbczyk A, Szczygielski J. Comparison of QEEG Findings before and after Onset of Post-COVID-19 Brain Fog Symptoms. Sensors. 2022; 22(17):6606. https://doi.org/10.3390/s22176606
Chicago/Turabian StyleKopańska, Marta, Danuta Ochojska, Renata Muchacka, Agnieszka Dejnowicz-Velitchkov, Agnieszka Banaś-Ząbczyk, and Jacek Szczygielski. 2022. "Comparison of QEEG Findings before and after Onset of Post-COVID-19 Brain Fog Symptoms" Sensors 22, no. 17: 6606. https://doi.org/10.3390/s22176606
APA StyleKopańska, M., Ochojska, D., Muchacka, R., Dejnowicz-Velitchkov, A., Banaś-Ząbczyk, A., & Szczygielski, J. (2022). Comparison of QEEG Findings before and after Onset of Post-COVID-19 Brain Fog Symptoms. Sensors, 22(17), 6606. https://doi.org/10.3390/s22176606








