Effect of Ethanol Consumption on the Accuracy of a Glucose Oxidase-Based Subcutaneous Glucose Sensor in Subjects with Type 1 Diabetes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Processing
2.3. Statistical Analysis
3. Results
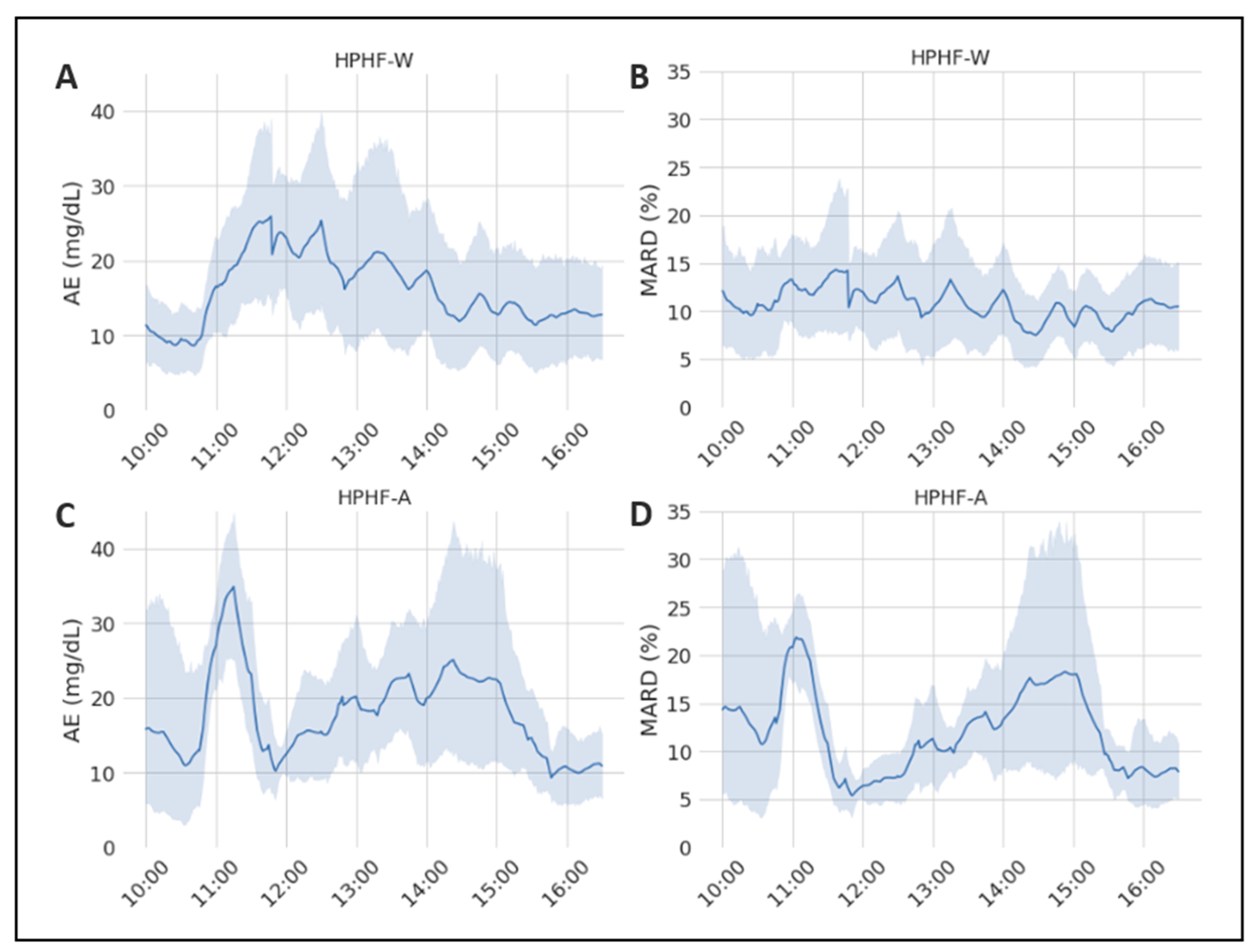
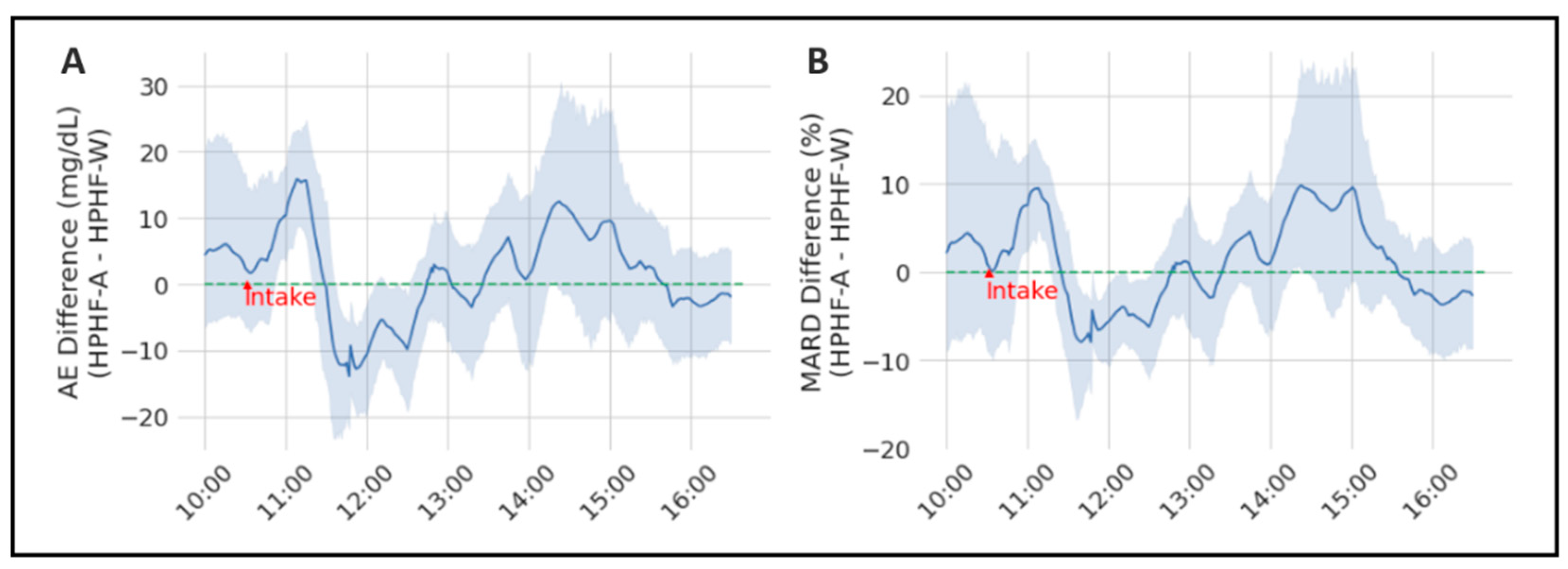
3.1. Evaluation of the Sensor-Accuracy Differences between Studies
3.2. Evaluation of the Relationship between Alcohol and pH Levels and Sensor Accuracy
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lin, R.; Brown, F.; James, S.; Jones, J.; Ekinci, E. Continuous glucose monitoring: A review of the evidence in type 1 and 2 diabetes mellitus. Diabet. Med. 2021, 38, e14528. [Google Scholar] [CrossRef]
- Lee, I.; Probst, D.; Klonoff, D.; Sode, K. Continuous glucose monitoring systems—Current status and future perspectives of the flagship technologies in biosensor research. Biosens. Bioelectron. 2021, 181, 113054. [Google Scholar] [CrossRef] [PubMed]
- Heller, A.; Feldman, B. Electrochemical glucose sensors and their applications in diabetes management. Chem. Rev. 2008, 108, 2482–2505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boehm, R.; Donovan, J.; Sheth, D.; Durfor, A.; Roberts, J.; Isayeva, I. In Vitro Sugar Interference Testing with Amperometric Glucose Oxidase Sensors. J. Diabetes Sci. Technol. 2019, 13, 82–95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Enter, B.J.; von Hauff, E. Challenges and perspectives in continuous glucose monitoring. Chem. Commun. 2018, 54, 5032–5045. [Google Scholar] [CrossRef]
- Price, C.; Ditton, G.; Russell, G.B.; Aloi, J. Reliability of Inpatient CGM: Comparison to Standard of Care. J. Diabetes Sci. Technol. 2021, 19322968211062168. [Google Scholar] [CrossRef]
- Galindo, R.J.; Aleppo, G.; Klonoff, D.C.; Spanakis, E.K.; Agarwal, S.; Vellanki, P.; Pasquel, F.J. Implementation of continuous glucose monitoring in the hospital: Emergent considerations for remote glucose monitoring during the COVID-19 pandemic. J. Diabetes Sci. Technol. 2020, 14, 822–832. [Google Scholar] [CrossRef]
- Barnard, K.; Sinclair, J.M.A.; Lawton, J.; Young, A.J.; Holt, R.I.G. Alcohol-associated risks for young adults with Type 1 diabetes: A narrative review. Diabet. Med. J. Br. Diabet. Assoc. 2012, 29, 434–440. [Google Scholar] [CrossRef] [Green Version]
- Roberts, A.J.; Law, J.R.; Suerken, C.K.; Reboussin, B.A.; Lawrence, J.; Taplin, C.E.; Mayer-Davis, E.J.; Pihoker, C. Alcohol consumption patterns in young adults with type 1 diabetes: The SEARCH for diabetes in youth study. Diabetes Res. Clin. Pract. 2020, 159, 107980. [Google Scholar] [CrossRef] [Green Version]
- Basu, A.; Slama, M.Q.; Nicholson, W.T.; Langman, L.; Peyser, T.; Carter, R.; Basu, R. Continuous Glucose Monitor Interference with Commonly Prescribed Medications: A Pilot Study. J. Diabetes Sci. Technol. 2017, 11, 936–941. [Google Scholar] [CrossRef] [Green Version]
- McGuire, L.C. Alcoholic ketoacidosis. Emerg. Med. J. 2006, 23, 417–420. [Google Scholar] [CrossRef] [PubMed]
- García, A.; Moscardó, V.; Ramos-Prol, A.; Díaz, J.; Boronat, M.; Bondia, J.; Rossetti, P. Effect of meal composition and alcohol consumption on postprandial glucose concentration in subjects with type 1 diabetes: A randomized crossover trial. BMJ Open Diabetes Res. Care 2021, 9, e002399. [Google Scholar] [CrossRef] [PubMed]
- Schrangl, P.; Reiterer, F.; Heinemann, L.; Freckmann, G.; Del Re, L. Limits to the evaluation of the accuracy of continuous glucose monitoring systems by clinical trials. Biosensors 2018, 8, 50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilson, D.F.; Matschinsky, F.M. Ethanol metabolism: The good, the bad, and the ugly. Med. Hypotheses 2020, 140, 109638. [Google Scholar] [CrossRef]
- Chen, C.; Xie, Q.; Yang, D.; Xiao, H.; Fu, Y.; Tan, Y.; Yao, S. Recent advances in electrochemical glucose biosensors: A review. RSC Adv. 2013, 3, 4473–4491. [Google Scholar] [CrossRef]
- Sehit, E.; Altintas, Z. Significance of nanomaterials in electrochemical glucose sensors: An updated review (2016–2020). Biosens. Bioelectron. 2020, 159, 112165. [Google Scholar] [CrossRef]
- Hassan, M.H.; Vyas, C.; Grieve, B.; Bartolo, P. Recent advances in enzymatic and non-enzymatic electrochemical glucose sensing. Sensors 2021, 21, 4672. [Google Scholar] [CrossRef]
- Wilson, R.; Turner, A.P.F. Glucose oxidase: An ideal enzyme. Biosens. Bioelectron. 1992, 7, 165–185. [Google Scholar] [CrossRef]
- Marics, G.; Koncz, L.; Eitler, K.; Vatai, B.; Szénási, B.; Zakariás, D.; Mikos, B.; Körner, A.; Toth-Heyn, P. Effects of pH, lactate, hematocrit and potassium level on the accuracy of continuous glucose monitoring (CGM) in pediatric intensive care unit Critical Care. Ital. J. Pediatr. 2015, 41, 1–6. [Google Scholar] [CrossRef] [Green Version]
- van Steen, S.C.J.; Rijkenberg, S.; Limpens, J.; van der Voort, P.H.J.; Hermanides, J.; DeVries, J.H. The clinical benefits and accuracy of continuous glucose monitoring systems in critically ill patients—A systematic scoping review. Sensors 2017, 17, 146. [Google Scholar] [CrossRef] [Green Version]
- Lorencio, C.; Leal, Y.; Bonet, A.; Bondia, J.; Palerm, C.C.; Tache, A.; Sirvent, J.-M.; Vehi, J. Real-time continuous glucose monitoring in an intensive care unit: Better accuracy in patients with septic shock. Diabetes Technol. Ther. 2012, 14, 568–575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wentholt, I.M.; Hoekstra, J.B.; DeVries, J.H. A critical appraisal of the continuous glucose-error grid analysis. Diabetes Care 2006, 29, 1805–1811. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Type of Variable | Variable | Meaning |
|---|---|---|
| Measured variables | PG | Plasma glucose recordings Sampling period: each 15 min |
| CGM | Continuous glucose-monitoring measurements Sampling period: each 5 min | |
| PA | Plasma alcohol recordings Sampling period: Each 30 min until two hours after meal, then each 60 min | |
| pH | Plasma pH recordings Sampling period: Each 30 min until two hours after meal, then each 60 min | |
| study | Type of study Categorical values: {HPHF-A, HPHF-W, LPLF-W} | |
| Calculatedvariables | AE | Absolute error calculated as: |
| MARD | Mean absolute relative difference calculated as: | |
| PA_Level | Category of plasma alcohol level determined as: | |
| pH_Level | Category of plasma pH level determined as: |
| Metrics | Study | N | Mean (Std) | Median [IQR] | Range | Mann-Whitney U Test |
|---|---|---|---|---|---|---|
| AE | HPHF-A | 312 | 17.93 (17.839) | 13.15 [6.55; 22.50] | [0.00; 105.0] | 0.0418 * |
| HPHF-W | 312 | 16.21 (15.988) | 10.80 [4.925; 22.425] | [0.00; 90.6] | ||
| MARD | HPHF-A | 312 | 12.239 (13.635) | 9.177 [5.132; 14.11] | [0.00; 96.82] | 0.248 |
| HPHF-W | 312 | 11.018 (9.152) | 8.149 [4.184; 16.02] | [0.00; 49.06] |
| Metrics | Difference (HPHF-A—HPHF-W) Mean [CI95%] | Wilcoxon Paired Test (p-Value) |
|---|---|---|
| AE | 1.71 [1.03; 2.39] | <0.001 * |
| MARD | 1.22 [0.76; 1.68] | 0.080 |
| Level Value | N | Mean (Std) | Median[IQR] | Range | Mann–Whitney U Test | ||
|---|---|---|---|---|---|---|---|
| AE | pH_Level | Low | 83 | 17.63 (13.37) | 15.40 [8.45; 20.75] | [0.00; 58.80] | 0.028 * |
| High | 277 | 16.08 (16.19) | 11.80 [5.13; 22.00] | [0.00; 104.50] | |||
| PA_Level | Low | 277 | 15.29 (15.09) | 11.15 [5.05; 20.20] | [0.00; 99.40] | <0.001 * | |
| High | 83 | 20.25 (16.65) | 17.10 [8.80; 24.68] | [0.50; 104.50] | |||
| MARD | pH_Level | Low | 83 | 12.04 (10.53) | 9.48 [5.64; 15.19] | [0.00; 78.18] | 0.091 |
| High | 277 | 11.47 (11.61) | 8.49 [3.70; 15.71] | [0.00; 91.19] | |||
| PA_Level | Low | 277 | 11.22 (10.77) | 8.58 [3.91; 15.03] | [0.00; 91.19] | 0.097 | |
| High | 83 | 12.85 (13.15) | 9.24 [5.96; 16.45] | [0.00; 84.94] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moscardó, V.; Garcia, A.; Bondia, J.; Diaz, J.; Ramos-Prol, A.; Rossetti, P. Effect of Ethanol Consumption on the Accuracy of a Glucose Oxidase-Based Subcutaneous Glucose Sensor in Subjects with Type 1 Diabetes. Sensors 2022, 22, 3101. https://doi.org/10.3390/s22093101
Moscardó V, Garcia A, Bondia J, Diaz J, Ramos-Prol A, Rossetti P. Effect of Ethanol Consumption on the Accuracy of a Glucose Oxidase-Based Subcutaneous Glucose Sensor in Subjects with Type 1 Diabetes. Sensors. 2022; 22(9):3101. https://doi.org/10.3390/s22093101
Chicago/Turabian StyleMoscardó, Vanessa, Alia Garcia, Jorge Bondia, Julián Diaz, Agustín Ramos-Prol, and Paolo Rossetti. 2022. "Effect of Ethanol Consumption on the Accuracy of a Glucose Oxidase-Based Subcutaneous Glucose Sensor in Subjects with Type 1 Diabetes" Sensors 22, no. 9: 3101. https://doi.org/10.3390/s22093101
APA StyleMoscardó, V., Garcia, A., Bondia, J., Diaz, J., Ramos-Prol, A., & Rossetti, P. (2022). Effect of Ethanol Consumption on the Accuracy of a Glucose Oxidase-Based Subcutaneous Glucose Sensor in Subjects with Type 1 Diabetes. Sensors, 22(9), 3101. https://doi.org/10.3390/s22093101






