Evaluation of Antioxidants Using Electrochemical Sensors: A Bibliometric Analysis
Abstract
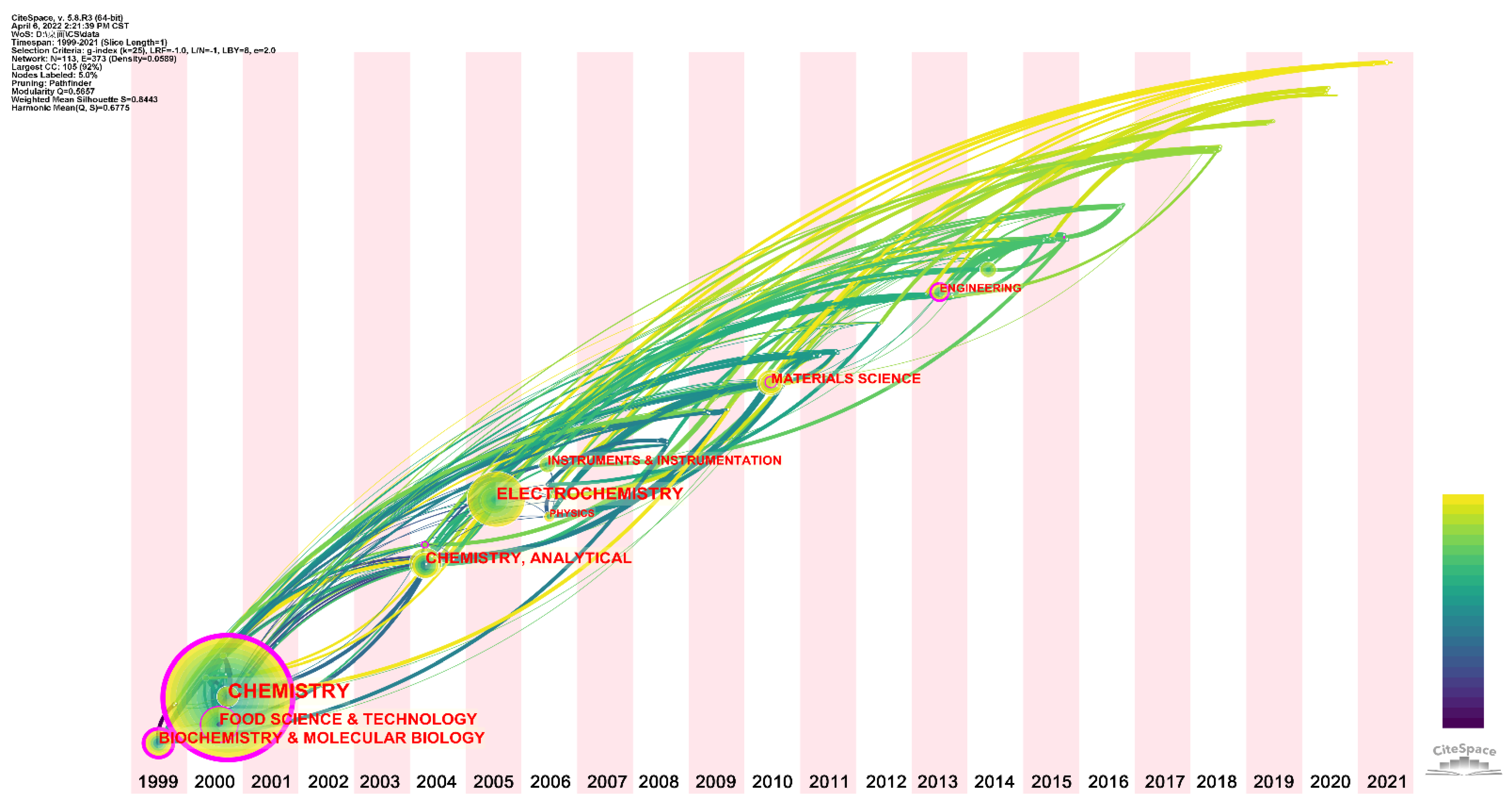
:1. Introduction
2. Developments in the Research Field
2.1. Literature Development Trends
2.2. Journals, Cited Journals, and Research Subjects
2.3. Geographic Distribution
3. Keyword Analysis and Evolution of the Field
| Cluster ID | Articles | Silhouette | Keywords | References |
|---|---|---|---|---|
| 0 | 51 | 0.977 | Antioxidant; Oxidative stress; Disease; Electrochemical detection; Liquid chromatography; DNA damage | [41,42,43,44,45,46,47,116,117,118,119,120,121] |
| 1 | 40 | 0.910 | Sensor; Antioxidant activity; Performance liquid chromatography; Catechin; Phenolic acid; Capillary electrophoresis | [48,49,50,51,52,53,54,55,56,57,58,59,122,123,124,125,126,127,128,129,130] |
| 2 | 31 | 0.909 | Nanoparticle; Electrode; Voltammetric determination; Biosensor; Electrochemical sensor; Film | [60,61,62,63,64,65,66,81,131,132,133,134,135,136] |
| 3 | 29 | 0.793 | Phenolic compound; Cyclic voltammetry; Assay; Ascorbic acid; DPPH; Glassy carbon electrode | [67,68,69,137,138,139,140,141,142,143,144,145,146,147] |
| 4 | 29 | 0.915 | Polyphenol; Antioxidant capacity; Sample; Wine; HPLC | [52,70,71,72,73,74,148,149,150,151,152,153] |
| 5 | 26 | 0.948 | Acid; Product; Iron; Neocuproine; Damage; Aromatic hydroxylation | [75,76,77,78,79,154] |
| 6 | 26 | 0.947 | Antioxidant capacity; Flavonoid; Nanotube; Adsorption; Electron transfer; Protein | [60,70,80,81,82,83,84,85,86,87,97,132,137,138,150,155,156,157,158,159,160,161,162,163,164,165] |
| 7 | 26 | 0.976 | Oxidation; Behavior; Red wine; Anthocyanin; Expression; Storage | [12,81,88,89,90,91,92,93,166,167,168] |
| 8 | 25 | 0.869 | Derivative; Caffeic acid; Electrochemical method; Energy; Aqueous solution; Ferulic acid | [169,170,171,172,173,174,175] |
| 9 | 22 | 0.943 | Capacity; Extract; Vitamin C; Media; Protection; Constituent | [58,67,94,95,96,97,98,99,176,177,178,179,180] |
| 10 | 20 | 0.965 | Nitric oxide; Lipid peroxidation; Alzheimers disease | [100,101] |
| 11 | 17 | 0.992 | Antioxidant property; In vitro; Biological activity; Structural characterization; DNA binding | [102,103,104] |
| 12 | 17 | 0.983 | Mechanism; Graphene oxide; Q(10); Sensitive detection; Inhibition; Carbon electrode | [80,105,106,107,181] |
| 13 | 16 | 0.896 | Food; Total antioxidant capacity; Tea; Detector | [108,109,110,182,183,184] |
| 14 | 15 | 0.949 | Hydrogen peroxide; In vivo; Scavenging assay | [22,111,112] |
| 15 | 11 | 0.988 | Antibacterial activity; By product; Antimicrobial activity; Molecular structure | [113,114] |
| 16 | 6 | 0.986 | Fruit; Antibacterial | [115] |
- (1)
- Cyclic voltammetry and differential pulse voltammetry are the electrochemical techniques most commonly used by electrochemical sensors to analyze the antioxidant capacity of target samples.
- (2)
- The electrochemical behavior of antioxidants can be used to understand the mechanism of these substances during redox.
- (3)
- The primary sources of antioxidants are plants.
- (4)
- Boron doped diamond electrodes, screen printed electrodes, and glassy carbon electrodes are most commonly used as working electrodes for electrochemical sensors.
- (5)
- Carbon nanotubes are the most commonly used nanomaterials for electrode surface modification.
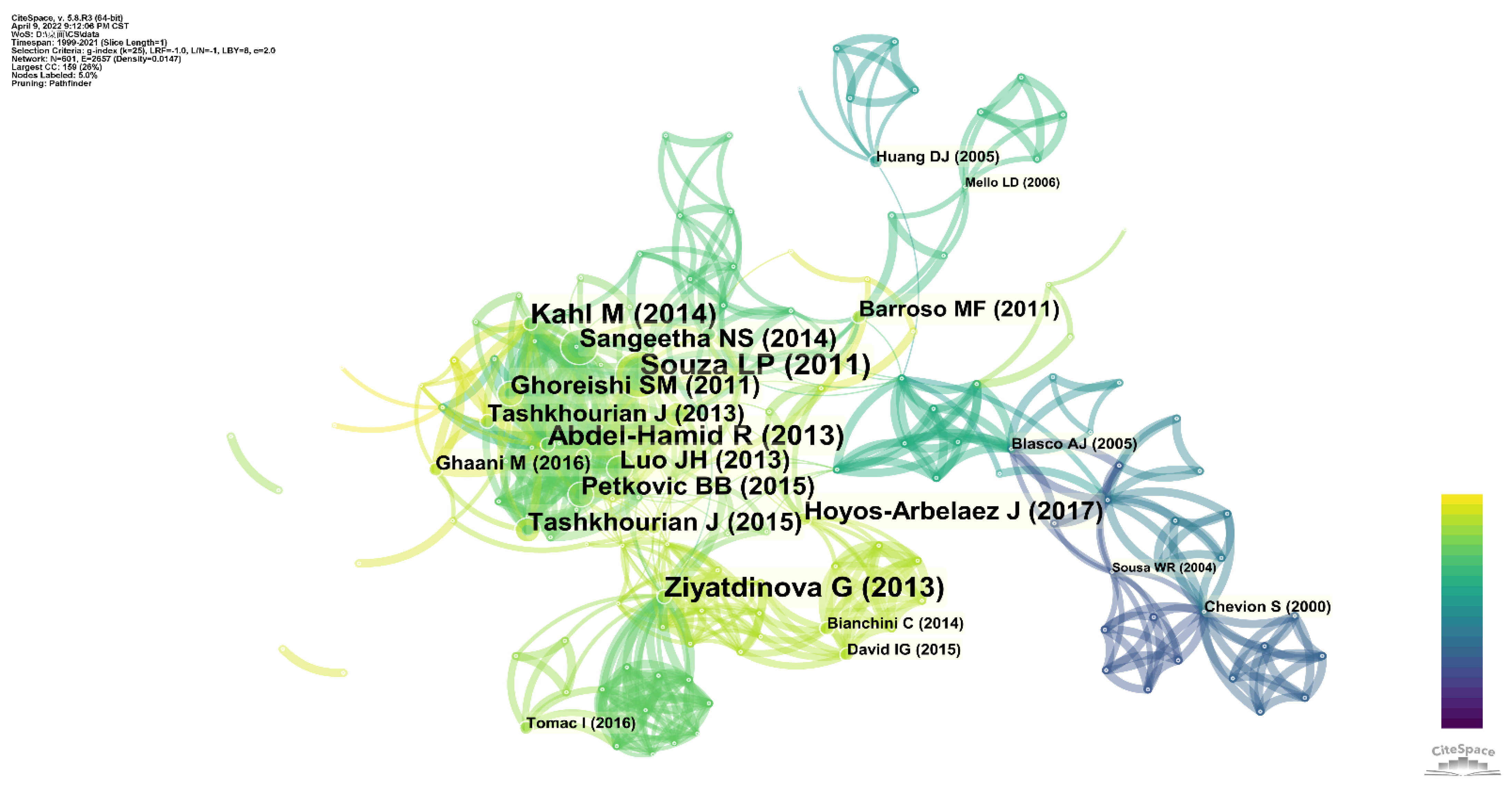
4. Co-Citation Analysis
5. Conclusions
- (1)
- The topic started in 1999 and did not attract much attention until 2010. After 2010, the topic became popular, and the trend continues today. This means that the topic has received much attention so far. At the same time, the problems faced by this theme have not been solved perfectly so far.
- (2)
- In the early stages of this topic, electrochemistry was a quantitative analysis technique, often used in conjunction with chromatographic and spectroscopic techniques for the separation and quantitative detection of complex samples. After that, cyclic voltammetry technology began to attract attention. The electrochemical behavior of antioxidants was used to measure their redox mechanism and quantitatively analyze their antioxidant capacity.
- (3)
- Most antioxidant capacity investigations focus on a specific antioxidant or a group of structurally similar molecules, such as flavonoids and polyphenols. However, due to the availability of plants as an important means for human antioxidant intake, many papers also use electrochemical sensing technology to determine plant samples or plant extracts directly. In addition, antioxidant properties derived from plant extracts can also protect metal materials from corrosion.
- (4)
- Since antioxidants tend to have significant electrochemical redox properties, commonly used commercial electrodes are already capable of direct detection. Carbon paste electrodes, screen printing electrodes, glassy carbon electrodes, and boron-doped diamond electrodes are the most commonly used working electrodes for analysis. However, advances in materials science have greatly improved the performance of electrochemical sensors. Nanomaterial modifications on the surface of the working electrode can improve the sensing performance remarkably. Among them, carbon nanotubes are the materials most used for electrode surface modification in this topic.
- (5)
- Because the oxidants damage DNA, the immobilized DNA on the electrochemical sensor’s surface can be used to measure the antioxidant capacity of antioxidants. The extent to which DNA has been damaged has been an indicator of such electrochemical DNA sensors.
- (1)
- Direct electrochemical sensors mainly rely on antioxidants’ electrochemical oxidation and reduction behavior. This method helps determine a particular antioxidant, but if the sensor is dealing with a complex sample, the electrochemical behavior is difficult to identify accurately. This is because complex samples contain a series of electrochemically active molecules whose presence can interfere with the measured current value of the target molecule. Therefore, it is a challenge to improve the specificity of direct electrochemical sensors to determine antioxidant capacity.
- (2)
- Choosing suitable electrode modification material is also a significant challenge. The current trend is to modify the electrode by using binary, ternary, or even multiple nanocomposites. Although nanomaterials have excellent properties, the synergistic effect between multiple materials cannot be explained theoretically. Therefore, the performance stability of these nanocomposites has been a limitation to their widespread use. At the same time, the raw materials of some nanocomposites’ preparation are costly and do not have the prospect of mass synthesis.
- (3)
- Although DNA biosensors are methodologically attractive, specially designed DNA requires higher prices. At the same time, such biosensors will be significantly affected by the external environment, so how to ensure their stability is also a meaningful direction.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hunyadi, A. The Mechanism (s) of Action of Antioxidants: From Scavenging Reactive Oxygen/Nitrogen Species to Redox Signaling and the Generation of Bioactive Secondary Metabolites. Med. Res. Rev. 2019, 39, 2505–2533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Munteanu, I.G.; Apetrei, C. Analytical Methods Used in Determining Antioxidant Activity: A Review. Int. J. Mol. Sci. 2021, 22, 3380. [Google Scholar] [CrossRef] [PubMed]
- Martinello, M.; Mutinelli, F. Antioxidant Activity in Bee Products: A Review. Antioxidants 2021, 10, 71. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Q.; Wei, B.; Wang, S.; Ke, S.; Chen, J.; Zhang, H.; Wang, H. The Antioxidant Activity of Polysaccharides Derived from Marine Organisms: An Overview. Mar. Drugs 2019, 17, 674. [Google Scholar] [CrossRef] [Green Version]
- Wojtunik-Kulesza, K.; Oniszczuk, A.; Oniszczuk, T.; Combrzyński, M.; Nowakowska, D.; Matwijczuk, A. Influence of in Vitro Digestion on Composition, Bioaccessibility and Antioxidant Activity of Food Polyphenols—A Non-Systematic Review. Nutrients 2020, 12, 1401. [Google Scholar] [CrossRef]
- Ji, M.; Gong, X.; Li, X.; Wang, C.; Li, M. Advanced Research on the Antioxidant Activity and Mechanism of Polyphenols from Hippophae Species—A Review. Molecules 2020, 25, 917. [Google Scholar] [CrossRef] [Green Version]
- Chrysargyris, A.; Mikallou, M.; Petropoulos, S.; Tzortzakis, N. Profiling of Essential Oils Components and Polyphenols for Their Antioxidant Activity of Medicinal and Aromatic Plants Grown in Different Environmental Conditions. Agronomy 2020, 10, 727. [Google Scholar] [CrossRef]
- Chen, X.; Liang, L.; Han, C. Borate Suppresses the Scavenging Activity of Gallic Acid and Plant Polyphenol Extracts on DPPH Radical: A Potential Interference to DPPH Assay. Lwt 2020, 131, 109769. [Google Scholar] [CrossRef]
- Hano, C.; Tungmunnithum, D. Plant Polyphenols, More than Just Simple Natural Antioxidants: Oxidative Stress, Aging and Age-Related Diseases. Medicines 2020, 7, 26. [Google Scholar] [CrossRef]
- Wojdyło, A.; Oszmiański, J. Antioxidant Activity Modulated by Polyphenol Contents in Apple and Leaves during Fruit Development and Ripening. Antioxidants 2020, 9, 567. [Google Scholar] [CrossRef]
- Krakowska-Sieprawska, A.; Rafińska, K.; Walczak-Skierska, J.; Buszewski, B. The Influence of Plant Material Enzymatic Hydrolysis and Extraction Conditions on the Polyphenolic Profiles and Antioxidant Activity of Extracts: A Green and Efficient Approach. Molecules 2020, 25, 2074. [Google Scholar] [CrossRef] [PubMed]
- Correa, C.R.; Li, L.; Aldini, G.; Carini, M.; Chen, C.-Y.O.; Chun, H.-K.; Cho, S.-M.; Park, K.-M.; Russell, R.M.; Blumberg, J.B. Composition and Stability of Phytochemicals in Five Varieties of Black Soybeans (Glycine Max). Food Chem. 2010, 123, 1176–1184. [Google Scholar] [CrossRef]
- Kerasioti, E.; Apostolou, A.; Kafantaris, I.; Chronis, K.; Kokka, E.; Dimitriadou, C.; Tzanetou, E.N.; Priftis, A.; Koulocheri, S.D.; Haroutounian, S.A. Polyphenolic Composition of Rosa Canina, Rosa Sempervivens and Pyrocantha Coccinea Extracts and Assessment of Their Antioxidant Activity in Human Endothelial Cells. Antioxidants 2019, 8, 92. [Google Scholar] [CrossRef] [Green Version]
- Olszowy, M. What Is Responsible for Antioxidant Properties of Polyphenolic Compounds from Plants? Plant Physiol. Biochem. 2019, 144, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Kang, Q.; Chen, S.; Li, S.; Wang, B.; Liu, X.; Hao, L.; Lu, J. Comparison on Characterization and Antioxidant Activity of Polysaccharides from Ganoderma Lucidum by Ultrasound and Conventional Extraction. Int. J. Biol. Macromol. 2019, 124, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Maleh, H.; Orooji, Y.; Karimi, F.; Alizadeh, M.; Baghayeri, M.; Rouhi, J.; Tajik, S.; Beitollahi, H.; Agarwal, S.; Gupta, V.K. A Critical Review on the Use of Potentiometric Based Biosensors for Biomarkers Detection. Biosens. Bioelectron. 2021, 184, 113252. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Khataee, A.; Karimi, F.; Baghayeri, M.; Fu, L.; Rouhi, J.; Karaman, C.; Karaman, O.; Boukherroub, R. A Green and Sensitive Guanine-Based DNA Biosensor for Idarubicin Anticancer Monitoring in Biological Samples: A Simple and Fast Strategy for Control of Health Quality in Chemotherapy Procedure Confirmed by Docking Investigation. Chemosphere 2021, 291, 132928. [Google Scholar] [CrossRef]
- Börner, K.; Chen, C.; Boyack, K.W. Visualizing Knowledge Domains. Annu. Rev. Inf. Sci. Technol. 2003, 37, 179–255. [Google Scholar] [CrossRef]
- Chen, C. CiteSpace II: Detecting and Visualizing Emerging Trends and Transient Patterns in Scientific Literature. J. Am. Soc. Inf. Sci. Technol. 2006, 57, 359–377. [Google Scholar] [CrossRef] [Green Version]
- Chen, C. Searching for Intellectual Turning Points: Progressive Knowledge Domain Visualization. Proc. Natl. Acad. Sci. USA 2004, 101, 5303–5310. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.; Ibekwe-SanJuan, F.; Hou, J. The Structure and Dynamics of Cocitation Clusters: A Multiple-perspective Cocitation Analysis. J. Am. Soc. Inf. Sci. Technol. 2010, 61, 1386–1409. [Google Scholar] [CrossRef] [Green Version]
- Tan, D.; Manchester, L.C.; Reiter, R.J.; Qi, W.; Zhang, M.; Weintraub, S.T.; Cabrera, J.; Sainz, R.M.; Mayo, J.C. Identification of Highly Elevated Levels of Melatonin in Bone Marrow: Its Origin and Significance. Biochim. Et Biophys. Acta (BBA)-Gen. Subj. 1999, 1472, 206–214. [Google Scholar] [CrossRef]
- Chevion, S.; Chevion, M. Antioxidant Status and Human Health: Use of Cyclic Voltammetry for the Evaluation of the Antioxidant Capacity of Plasma and of Edible Plants. Ann. N. Y. Acad. Sci. 2000, 899, 308–325. [Google Scholar] [CrossRef] [PubMed]
- Chevion, S.; Hofmann, M.; Ziegler, R.; Chevion, M.; Nawroth, P. The Antioxidant Properties of Thioctic Acid: Characterization by Cyclic Voltammetry. IUBMB Life 1997, 41, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Chevion, S.; Berry, E.M.; Kitrossky, N.; Kohen, R. Evaluation of Plasma Low Molecular Weight Antioxidant Capacity by Cyclic Voltammetry. Free Radic. Biol. Med. 1997, 22, 411–421. [Google Scholar] [CrossRef]
- Chevion, S.; Or, R.; Berry, E.M. The Antioxidant Status of Patients Subjected to Today Body Irradiation. IUBMB Life 1999, 47, 1019–1027. [Google Scholar] [CrossRef]
- Chevion, S.; Roberts, M.A.; Chevion, M. The Use of Cyclic Voltammetry for the Evaluation of Antioxidant Capacity. Free Radic. Biol. Med. 2000, 28, 860–870. [Google Scholar] [CrossRef]
- Elangovan, V.; Shohami, E.; Gati, I.; Kohen, R. Increased Hepatic Lipid Soluble Antioxidant Capacity as Compared to Other Organs of Streptozotocin-Induced Diabetic Rats: A Cyclic Voltammetry Study. Free Radic. Res. 2000, 32, 125–134. [Google Scholar] [CrossRef]
- Fu, L.; Mao, S.; Chen, F.; Zhao, S.; Su, W.; Lai, G.; Yu, A.; Lin, C.-T. Graphene-Based Electrochemical Sensors for Antibiotic Detection in Water, Food and Soil: A Scientometric Analysis in CiteSpace (2011–2021). Chemosphere 2022, 297, 134127. [Google Scholar] [CrossRef]
- Shen, Y.; Mao, S.; Chen, F.; Zhao, S.; Su, W.; Fu, L.; Zare, N.; Karimi, F. Electrochemical Detection of Sudan Red Series Azo Dyes: Bibliometrics Based Analysis. Food Chem. Toxicol. 2022, 163, 112960. [Google Scholar] [CrossRef]
- Rangaraj, V.M.; Rambabu, K.; Banat, F.; Mittal, V. Effect of Date Fruit Waste Extract as an Antioxidant Additive on the Properties of Active Gelatin Films. Food Chem. 2021, 355, 129631. [Google Scholar] [CrossRef] [PubMed]
- Daneshzadeh, M.S.; Abbaspour, H.; Amjad, L.; Nafchi, A.M. An Investigation on Phytochemical, Antioxidant and Antibacterial Properties of Extract from Eryngium Billardieri F. Delaroche. J. Food Meas. Charact. 2020, 14, 708–715. [Google Scholar] [CrossRef]
- Lim, S.; Choi, A.-H.; Kwon, M.; Joung, E.-J.; Shin, T.; Lee, S.-G.; Kim, N.-G.; Kim, H.-R. Evaluation of Antioxidant Activities of Various Solvent Extract from Sargassum Serratifolium and Its Major Antioxidant Components. Food Chem. 2019, 278, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Waheed, A.; Mansha, M.; Ullah, N. Nanomaterials-Based Electrochemical Detection of Heavy Metals in Water: Current Status, Challenges and Future Direction. TrAC Trends Anal. Chem. 2018, 105, 37–51. [Google Scholar] [CrossRef]
- Baig, N.; Sajid, M.; Saleh, T.A. Recent Trends in Nanomaterial-Modified Electrodes for Electroanalytical Applications. TrAC Trends Anal. Chem. 2019, 111, 47–61. [Google Scholar] [CrossRef]
- Ikram, M.; Park, T.J.; Ali, T.; Kim, M.O. Antioxidant and Neuroprotective Effects of Caffeine against Alzheimer’s and Parkinson’s Disease: Insight into the Role of Nrf-2 and A2AR Signaling. Antioxidants 2020, 9, 902. [Google Scholar] [CrossRef]
- Forman, H.J.; Zhang, H. Targeting Oxidative Stress in Disease: Promise and Limitations of Antioxidant Therapy. Nat. Rev. Drug Discov. 2021, 20, 689–709. [Google Scholar] [CrossRef]
- Oszmiański, J.; Lachowicz, S.; Gamsjäger, H. Phytochemical Analysis by Liquid Chromatography of Ten Old Apple Varieties Grown in Austria and Their Antioxidative Activity. Eur. Food Res. Technol. 2020, 246, 437–448. [Google Scholar] [CrossRef]
- Cantalapiedra, A.; Gismera, M.J.; Sevilla, M.T.; Procopio, J.R. Sensitive and Selective Determination of Phenolic Compounds from Aromatic Plants Using an Electrochemical Detection Coupled with HPLC Method. Phytochem. Anal. 2014, 25, 247–254. [Google Scholar]
- Zhang, P.; Chun, Z.; Shao, Q.; Fu, L.; Luo, Y.; Gu, D.; Chen, R. Evaluation of the Phytochemicals and Antioxidant Activity of Lophatherum Gracile Brongn Based on Chemical Fingerprinting by HPLC with Electrochemical Detection. J. Sep. Sci. 2021, 44, 3777–3788. [Google Scholar]
- Silva, D.G.H.; Belini Junior, E.; Carrocini, G.C.D.S.; Torres, L.D.S.; Ricci Júnior, O.; Lobo, C.L.D.C.; Bonini-Domingos, C.R.; de Almeida, E.A. Genetic and Biochemical Markers of Hydroxyurea Therapeutic Response in Sickle Cell Anemia. BMC Med. Genet. 2013, 14, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rose, S.; Melnyk, S.; Pavliv, O.; Bai, S.; Nick, T.; Frye, R.; James, S. Evidence of Oxidative Damage and Inflammation Associated with Low Glutathione Redox Status in the Autism Brain. Transl. Psychiatry 2012, 2, e134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Zheng, Q.; Pan, J.; Zheng, R. Oxidative Damage of Biomolecules in Mouse Liver Induced by Morphine and Protected by Antioxidants. Basic Clin. Pharmacol. Toxicol. 2004, 95, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Chaves, S.; Canário, S.; Carrasco, M.P.; Mira, L.; Santos, M.A. Hydroxy (Thio) Pyrone and Hydroxy (Thio) Pyridinone Iron Chelators: Physico-Chemical Properties and Anti-Oxidant Activity. J. Inorg. Biochem. 2012, 114, 38–46. [Google Scholar] [CrossRef]
- Sobrova, P.; Vasatkova, A.; Skladanka, J.; Beklova, M.; Zeman, L.; Kizek, R.; Adam, V. Study of Deoxynivalenol Effect on Metallothionein and Glutathione Levels, Antioxidant Capacity, and Glutathione-S-Transferase and Liver Enzymes Activity in Rats. Chem. Pap. 2012, 66, 1092–1102. [Google Scholar] [CrossRef]
- Menke, T.; Niklowitz, P.; Wiesel, T.; Andler, W. Antioxidant Level and Redox Status of Coenzyme Q10 in the Plasma and Blood Cells of Children with Diabetes Mellitus Type 1. Pediatr. Diabetes 2008, 9, 540–545. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Milbury, P.E.; Lapsley, K.; Blumberg, J.B. Flavonoids from Almond Skins Are Bioavailable and Act Synergistically with Vitamins C and E to Enhance Hamster and Human LDL Resistance to Oxidation. J. Nutr. 2005, 135, 1366–1373. [Google Scholar] [CrossRef]
- Kilinc, E. Significance of Chromatographic and Voltammetric Data for the Classification of Green Teas in Türkiye: A Principle Component Analysis Approach. J. Liq. Chromatogr. Relat. Technol. 2008, 32, 221–241. [Google Scholar] [CrossRef]
- Wang, X.-G.; Li, J.; Fan, Y.-J. Fast Detection of Catechin in Tea Beverage Using a Poly-Aspartic Acid Film Based Sensor. Microchim. Acta 2010, 169, 173–179. [Google Scholar] [CrossRef]
- Maximino, M.D.; Martin, C.S.; Paulovich, F.V.; Alessio, P. Layer-by-layer Thin Film of Iron Phthalocyanine as a Simple and Fast Sensor for Polyphenol Determination in Tea Samples. J. Food Sci. 2016, 81, C2344–C2351. [Google Scholar] [CrossRef]
- Tomac, I.; Jakobek, L.; Seruga, M. Chromatographic and Voltammetric Characterization of Chlorogenic Acids in Coffee Samples. Croat. Chem. Acta 2018, 91, 501–512. [Google Scholar] [CrossRef]
- Alpar, N.; Yardım, Y.; Şentürk, Z. Selective and Simultaneous Determination of Total Chlorogenic Acids, Vanillin and Caffeine in Foods and Beverages by Adsorptive Stripping Voltammetry Using a Cathodically Pretreated Boron-Doped Diamond Electrode. Sens. Actuators B Chem. 2018, 257, 398–408. [Google Scholar] [CrossRef]
- Rodríguez-Gómez, R.; Vanheuverzwjin, J.; Souard, F.; Delporte, C.; Stevigny, C.; Stoffelen, P.; De Braekeleer, K.; Kauffmann, J.-M. Determination of Three Main Chlorogenic Acids in Water Extracts of Coffee Leaves by Liquid Chromatography Coupled to an Electrochemical Detector. Antioxidants 2018, 7, 143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blanc, N.; Hauchard, D.; Audibert, L.; Gall, E.A. Radical-Scavenging Capacity of Phenol Fractions in the Brown Seaweed Ascophyllum Nodosum: An Electrochemical Approach. Talanta 2011, 84, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.W.; Kassim, M.J. A Correlation Study on the Phenolic Profiles and Corrosion Inhibition Properties of Mangrove Tannins (Rhizophora apiculata) as Affected by Extraction Solvents. Corros. Sci. 2011, 53, 569–574. [Google Scholar] [CrossRef]
- Oliveira-Alves, S.C.; Vendramini-Costa, D.B.; Cazarin, C.B.B.; Júnior, M.R.M.; Ferreira, J.P.B.; Silva, A.B.; Prado, M.A.; Bronze, M.R. Characterization of Phenolic Compounds in Chia (Salvia hispanica L.) Seeds, Fiber Flour and Oil. Food Chem. 2017, 232, 295–305. [Google Scholar] [CrossRef]
- Gavahian, M.; Chu, Y.-H. Ohmic Accelerated Steam Distillation of Essential Oil from Lavender in Comparison with Conventional Steam Distillation. Innov. Food Sci. Emerg. Technol. 2018, 50, 34–41. [Google Scholar] [CrossRef]
- Hompesch, R.W.; García, C.D.; Weiss, D.J.; Vivanco, J.M.; Henry, C.S. Analysis of Natural Flavonoids by Microchip-Micellar Electrokinetic Chromatography with Pulsed Amperometric Detection. Analyst 2005, 130, 694–700. [Google Scholar] [CrossRef]
- García, M.; Escarpa, A. Microchip Electrophoresis–Copper Nanowires for Fast and Reliable Determination of Monossacharides in Honey Samples. Electrophoresis 2014, 35, 425–432. [Google Scholar] [CrossRef]
- Wang, X.; Tan, W.; Wang, Y.; Wu, D.; Kong, Y. Electrosynthesis of Poly (m-Phenylenediamine) on the Nanocomposites of Palygorskite and Ionic Liquid for Electrocatalytic Sensing of Gallic Acid. Sens. Actuators B Chem. 2019, 284, 63–72. [Google Scholar] [CrossRef]
- Koçak, Ç.C.; Karabiberoğlu, Ş.U.; Dursun, Z. Highly Sensitive Determination of Gallic Acid on Poly (l-Methionine)-Carbon Nanotube Composite Electrode. J. Electroanal. Chem. 2019, 853, 113552. [Google Scholar] [CrossRef]
- David, I.; Popa, D.; Buleandra, M.; Moldovan, Z.; Iorgulescu, E.; Badea, I. Cheap Pencil Graphite Electrodes for Rapid Voltammetric Determination of Chlorogenic Acid in Dietary Supplements. Anal. Methods 2016, 8, 6537–6544. [Google Scholar] [CrossRef]
- Della Pelle, F.; Rojas, D.; Scroccarello, A.; Del Carlo, M.; Ferraro, G.; Di Mattia, C.; Martuscelli, M.; Escarpa, A.; Compagnone, D. High-Performance Carbon Black/Molybdenum Disulfide Nanohybrid Sensor for Cocoa Catechins Determination Using an Extraction-Free Approach. Sens. Actuators B Chem. 2019, 296, 126651. [Google Scholar] [CrossRef]
- Marx, Í.M.; Rodrigues, N.; Dias, L.G.; Veloso, A.C.; Pereira, J.A.; Drunkler, D.A.; Peres, A.M. Assessment of Table Olives’ Organoleptic Defect Intensities Based on the Potentiometric Fingerprint Recorded by an Electronic Tongue. Food Bioprocess Technol. 2017, 10, 1310–1323. [Google Scholar] [CrossRef] [Green Version]
- Mukdasai, S.; Uppachai, P.; Srijaranai, S. A Novel Colorimetric Paper Sensor Based on the Layer-by-Layer Assembled Multilayers of Surfactants for the Sensitive and Selective Determination of Total Antioxidant Capacity. RSC Adv. 2019, 9, 28598–28608. [Google Scholar] [CrossRef] [Green Version]
- Ciou, D.-S.; Wu, P.-H.; Huang, Y.-C.; Yang, M.-C.; Lee, S.-Y.; Lin, C.-Y. Colorimetric and Amperometric Detection of Urine Creatinine Based on the ABTS Radical Cation Modified Electrode. Sens. Actuators B Chem. 2020, 314, 128034. [Google Scholar] [CrossRef]
- Brito, R.E.; Mellado, J.R.; Maldonado, P.; Montoya, M.R.; Palma, A.; Morales, E. Elucidation of the Electrochemical Oxidation Mechanism of the Antioxidant Sesamol on a Glassy Carbon Electrode. J. Electrochem. Soc. 2014, 161, G27. [Google Scholar] [CrossRef]
- Marano, S.; Minnelli, C.; Ripani, L.; Marcaccio, M.; Laudadio, E.; Mobbili, G.; Amici, A.; Armeni, T.; Stipa, P. Insights into the Antioxidant Mechanism of Newly Synthesized Benzoxazinic Nitrones: In Vitro and In Silico Studies with DPPH Model Radical. Antioxidants 2021, 10, 1224. [Google Scholar] [CrossRef]
- Martinez, S.; Valek, L.; Rešetić, J.; Ružić, D.F. Cyclic Voltammetry Study of Plasma Antioxidant Capacity–Comparison with the DPPH and TAS Spectrophotometric Methods. J. Electroanal. Chem. 2006, 588, 68–73. [Google Scholar] [CrossRef]
- Kahoun, D.; Řezková, S.; Veškrnová, K.; Královský, J.; Holčapek, M. Determination of Phenolic Compounds and Hydroxymethylfurfural in Meads Using High Performance Liquid Chromatography with Coulometric-Array and UV Detection. J. Chromatogr. A 2008, 1202, 19–33. [Google Scholar] [CrossRef]
- Volikakis, G.J.; Efstathiou, C.E. Fast Screening of Total Flavonols in Wines, Tea-Infusions and Tomato Juice by Flow Injection/Adsorptive Stripping Voltammetry. Anal. Chim. Acta 2005, 551, 124–131. [Google Scholar] [CrossRef]
- Hájek, T.; Škeříková, V.; Česla, P.; Vyňuchalová, K.; Jandera, P. Multidimensional LC× LC Analysis of Phenolic and Flavone Natural Antioxidants with UV-electrochemical Coulometric and MS Detection. J. Sep. Sci. 2008, 31, 3309–3328. [Google Scholar] [CrossRef] [PubMed]
- Kalinke, C.; de Oliveira, P.R.; Bonet San Emeterio, M.; González-Calabuig, A.; del Valle, M.; Salvio Mangrich, A.; Humberto Marcolino Junior, L.; Bergamini, M.F. Voltammetric Electronic Tongue Based on Carbon Paste Electrodes Modified with Biochar for Phenolic Compounds Stripping Detection. Electroanalysis 2019, 31, 2238–2245. [Google Scholar] [CrossRef]
- Mehmeti, E.; Stanković, D.M.; Ortner, A.; Zavašnik, J.; Kalcher, K. Highly Selective Electrochemical Determination of Phlorizin Using Square Wave Voltammetry at a Boron-Doped Diamond Electrode. Food Anal. Methods 2017, 10, 3747–3752. [Google Scholar] [CrossRef] [Green Version]
- Özyürek, M.; Bektaşoğlu, B.; Güçlü, K.; Apak, R. Hydroxyl Radical Scavenging Assay of Phenolics and Flavonoids with a Modified Cupric Reducing Antioxidant Capacity (CUPRAC) Method Using Catalase for Hydrogen Peroxide Degradation. Anal. Chim. Acta 2008, 616, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Bektaşoğlu, B.; Celik, S.E.; Özyürek, M.; Güçlü, K.; Apak, R. Novel Hydroxyl Radical Scavenging Antioxidant Activity Assay for Water-Soluble Antioxidants Using a Modified CUPRAC Method. Biochem. Biophys. Res. Commun. 2006, 345, 1194–1200. [Google Scholar] [CrossRef]
- De la Vega-Hernández, K.; Antuch, M.; Cuesta-Rubio, O.; Núñez-Figueredo, Y.; Pardo-Andreu, G.L. Discerning the Antioxidant Mechanism of Rapanone: A Naturally Occurring Benzoquinone with Iron Complexing and Radical Scavenging Activities. J. Inorg. Biochem. 2017, 170, 134–147. [Google Scholar] [CrossRef]
- Gorjanović, S.; Pastor, F.T.; Vasic, R.; Novaković, M.; Simonović, M.; Milić, S.; Suznjevic, D. Electrochemical versus Spectrophotometric Assessment of Antioxidant Activity of Hop (Humulus lupulus L.) Products and Individual Compounds. J. Agric. Food Chem. 2013, 61, 9089–9096. [Google Scholar] [CrossRef]
- Adamsen, C.E.; Hansen, M.L.; Møller, J.K.; Skibsted, L.H. Studies on the Antioxidative Activity of Red Pigments in Italian-Type Dry-Cured Ham. Eur. Food Res. Technol. 2003, 217, 201–206. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, S.; Muguruma, H.; Osakabe, N.; Inoue, H.; Ohsawa, T. Electrochemical Determination with a Long-Length Carbon Nanotube Electrode of Quercetin Glucosides in Onion, Apple Peel, and Tartary Buckwheat. Food Chem. 2019, 300, 125189. [Google Scholar] [CrossRef]
- Abdel-Hamid, R.; Newair, E.F. Voltammetric Determination of Polyphenolic Content in Pomegranate Juice Using a Poly (Gallic Acid)/Multiwalled Carbon Nanotube Modified Electrode. Beilstein J. Nanotechnol. 2016, 7, 1104–1112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreno, M.; Arribas, A.S.; González, L.; Bermejo, E.; Zapardiel, A.; Chicharro, M. Flow Injection Analysis with Amperometric Detection of Polyphenols at Carbon Nanotube/Polyvinylpyrrolidone-Modified Electrodes as Classification Tool for White Wine Varieties. Microchem. J. 2021, 162, 105836. [Google Scholar] [CrossRef]
- Ziyatdinova, G.; Guss, E.; Morozova, E.; Budnikov, H.; Davletshin, R.; Vorobev, V.; Osin, Y. Simultaneous Voltammetric Determination of Gallic and Ellagic Acids in Cognac and Brandy Using Electrode Modified with Functionalized SWNT and Poly (Pyrocatechol Violet). Food Anal. Methods 2019, 12, 2250–2261. [Google Scholar] [CrossRef]
- Su, Y.; Xie, Q.; Yang, Q.; Tu, X.; Cao, Z.; Jia, X.; Su, Z.; Zhang, Y.; Meng, W.; Yao, S. Electrochemical Quartz Crystal Impedance and Fluorescence Quenching Studies on the Binding of Carbon Nanotubes (CNTs)-adsorbed and Solution Rutin with Hemoglobin. Biotechnol. Prog. 2007, 23, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Liu, Y.; Shi, X.; Yang, X.; Bentley, W.E.; Payne, G.F. Biomimetic Approach to Confer Redox Activity to Thin Chitosan Films. Adv. Funct. Mater. 2010, 20, 2683–2694. [Google Scholar] [CrossRef]
- Chon, K.; Salhi, E.; Von Gunten, U. Combination of UV Absorbance and Electron Donating Capacity to Assess Degradation of Micropollutants and Formation of Bromate during Ozonation of Wastewater Effluents. Water Res. 2015, 81, 388–397. [Google Scholar] [CrossRef]
- Milić, S.Z.; Potkonjak, N.I.; Gorjanović, S.Ž.; Veljović-Jovanović, S.D.; Pastor, F.T.; Sužnjević, D.Ž. A Polarographic Study of Chlorogenic Acid and Its Interaction with Some Heavy Metal Ions. Electroanalysis 2011, 23, 2935–2940. [Google Scholar] [CrossRef]
- Martinez, S.; Valek, L.; Petrović, Ž.; Metikoš-Huković, M.; Piljac, J. Catechin Antioxidant Action at Various PH Studied by Cyclic Voltammetry and PM3 Semi-Empirical Calculations. J. Electroanal. Chem. 2005, 584, 92–99. [Google Scholar] [CrossRef]
- Wang, X.; Ma, W.; Ying, Y.; Liang, J.; Long, Y. Bis-Coenzyme Q0: Synthesis, Characteristics, and Application. Chem.-Asian J. 2011, 6, 1064–1073. [Google Scholar] [CrossRef]
- Arribas, A.S.; Moreno, M.; González, L.; Blazquez, N.; Bermejo, E.; Zapardiel, A.; Chicharro, M. A Comparative Study of Carbon Nanotube Dispersions Assisted by Cationic Reagents as Electrode Modifiers: Preparation, Characterization and Electrochemical Performance for Gallic Acid Detection. J. Electroanal. Chem. 2020, 857, 113750. [Google Scholar] [CrossRef]
- Kotani, A.; Odagiri, M.; Takamura, K.; Kusu, F. Voltammetric Behavior of Trolox in the Presence of Amino Acid in Unbuffered Dimethylsulfoxide. J. Electroanal. Chem. 2008, 624, 323–326. [Google Scholar] [CrossRef]
- Romero, I.; Domínguez, I.; Morales-Diaz, N.; Escribano, M.I.; Merodio, C.; Sanchez-Ballesta, M.T. Regulation of Flavonoid Biosynthesis Pathway by a Single or Dual Short-Term CO2 Treatment in Black Table Grapes Stored at Low Temperature. Plant Physiol. Biochem. 2020, 156, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Pigani, L.; Simone, G.V.; Foca, G.; Ulrici, A.; Masino, F.; Cubillana-Aguilera, L.; Calvini, R.; Seeber, R. Prediction of Parameters Related to Grape Ripening by Multivariate Calibration of Voltammetric Signals Acquired by an Electronic Tongue. Talanta 2018, 178, 178–187. [Google Scholar] [CrossRef] [PubMed]
- El moussaoui, A.; Kadiri, M.; Bourhia, M.; Agour, A.; Salamatullah, A.M.; Alzahrani, A.; Alyahya, H.K.; Albadr, N.A.; Chedadi, M.; Sfaira, M.; et al. Promising Antioxidant and Anticorrosion Activities of Mild Steel in 1.0 M Hydrochloric Acid Solution by Withania frutescens L. Essential Oil. Front. Chem. 2021, 9, 739273. [Google Scholar] [CrossRef] [PubMed]
- Emori, W.; Bassey, V.M.; Louis, H.; Okonkwo, P.C.; Zhao, S.; Wei, K.; Okafor, P.C.; Wan, J.; Cheng, C.-R. Anticorrosion and Dispersive Adsorption Studies of Natural Andrographolide on Carbon Steel in Acid-Chloride Environments. Bioelectrochemistry 2021, 141, 107840. [Google Scholar] [CrossRef]
- Vrsalović, L.; Gudić, S.; Kliškić, M.; Oguzie, E.; Carev, L. Inhibition of Copper Corrosion in NaCl Solution by Caffeic Acid. Int. J. Electrochem. Sci 2016, 11, 459–474. [Google Scholar]
- Gudić, S.; Vrsalović, L.; Kliškić, M.; Jerković, I.; Radonić, A.; Zekić, M. Corrosion Inhibition of AA 5052 Aluminium Alloy in NaCl Solution by Different Types of Honey. Int. J. Electrochem. Sci. 2016, 11, 998–1011. [Google Scholar]
- Motshakeri, M.; Travas-Sejdic, J.; Phillips, A.R.; Kilmartin, P.A. Rapid Electroanalysis of Uric Acid and Ascorbic Acid Using a Poly (3, 4-Ethylenedioxythiophene)-Modified Sensor with Application to Milk. Electrochim. Acta 2018, 265, 184–193. [Google Scholar] [CrossRef]
- Sebarchievici, I.; Lascu, A.; Fagadar-Cosma, G.; Palade, A.; Fringu, I.; Birdeanu, M.; Taranu, B.; Fagadar-Cosma, E. Optical and Electrochemical-Mediated Detection of Ascorbic Acid Using Manganese Porphyrin and Its Gold Hybrids. Comptes Rendus Chim. 2018, 21, 327–338. [Google Scholar] [CrossRef]
- Nia, A.B.; Van Schooten, F.; Schilderman, P.; De Kok, T.; Haenen, G.; Van Herwijnen, M.; Van Agen, E.; Pachen, D.; Kleinjans, J. A Multi-Biomarker Approach to Study the Effects of Smoking on Oxidative DNA Damage and Repair and Antioxidative Defense Mechanisms. Carcinogenesis 2001, 22, 395–401. [Google Scholar] [CrossRef] [Green Version]
- James, S.J.; Rose, S.; Melnyk, S.; Jernigan, S.; Blossom, S.; Pavliv, O.; Gaylor, D.W. Cellular and Mitochondrial Glutathione Redox Imbalance in Lymphoblastoid Cells Derived from Children with Autism. FASEB J. 2009, 23, 2374–2383. [Google Scholar] [CrossRef] [PubMed]
- Feroci, G.; Fini, A. Voltammetric Investigation of the Interactions between Superoxide Ion and Some Sulfur Amino Acids. Inorg. Chim. Acta 2007, 360, 1023–1031. [Google Scholar] [CrossRef]
- İnan, A.; Ikiz, M.; Tayhan, S.E.; Bilgin, S.; Genç, N.; Sayın, K.; Ceyhan, G.; Köse, M.; Dağ, A.; İspir, E. Antiproliferative, Antioxidant, Computational and Electrochemical Studies of New Azo-Containing Schiff Base Ruthenium (II) Complexes. New J. Chem. 2018, 42, 2952–2963. [Google Scholar] [CrossRef]
- İnan, A.; Sünbül, A.B.; Ikiz, M.; Tayhan, S.E.; Bilgin, S.; Elmastaş, M.; Sayın, K.; Ceyhan, G.; Köse, M.; İspir, E. Half-Sandwich Ruthenium (II) Arene Complexes Bearing the Azo-Azomethine Ligands: Electrochemical, Computational, Antiproliferative and Antioxidant Properties. J. Organomet. Chem. 2018, 870, 76–89. [Google Scholar] [CrossRef]
- Criado, S.; Allevi, C.; Ceballos, C.; García, N.A. Visible-Light Promoted Degradation of the Commercial Antioxidants Butylated Hydroxyanisole (BHA) and Butylated Hydroxytoluene (BHT): A Kinetic Study. Redox Rep. 2007, 12, 282–288. [Google Scholar] [CrossRef]
- Gulaboski, R.; Markovski, V.; Jihe, Z. Redox Chemistry of Coenzyme Q—A Short Overview of the Voltammetric Features. J. Solid State Electrochem. 2016, 20, 3229–3238. [Google Scholar] [CrossRef] [Green Version]
- Poon, J.-F.; Farmer, L.A.; Haidasz, E.A.; Pratt, D.A. Temperature-Dependence of Radical-Trapping Activity of Phenoxazine, Phenothiazine and Their Aza-Analogues Clarifies the Way Forward for New Antioxidant Design. Chem. Sci. 2021, 12, 11065–11079. [Google Scholar] [CrossRef]
- Kanyanee, T.; Fuekhad, P.; Grudpan, K. Micro Coulometric Titration in a Liquid Drop. Talanta 2013, 115, 258–262. [Google Scholar] [CrossRef]
- García, M.; Escarpa, A. A Class-Selective and Reliable Electrochemical Monosaccharide Index in Honeys, as Determined Using Nickel and Nickel-Copper Nanowires. Anal. Bioanal. Chem. 2012, 402, 945–953. [Google Scholar] [CrossRef]
- Wang, Y.; Yao, G.; Zhu, P.; Hu, X. Indirect Biamperometric Determination of O-Phenylenediamine in Lab-on-Valve Format Using Reversible Indicating Redox System. Analyst 2011, 136, 829–834. [Google Scholar] [CrossRef]
- Emir, G.; Dilgin, Y.; Apak, R. A New Redox Mediator (Cupric-Neocuproine Complex)-Modified Pencil Graphite Electrode for the Electrocatalytic Oxidation of H2O2: A Flow Injection Amperometric Sensor. ChemElectroChem 2020, 7, 649–658. [Google Scholar] [CrossRef]
- Miyasaka, T.; Endo, K.; Mochizuki, S.; Sakai, K. Superoxide Sensors. Sens. Lett. 2006, 4, 144–154. [Google Scholar] [CrossRef]
- Raj, K.M.; Vivekanand, B.; Nagesh, G.; Mruthyunjayaswamy, B. Synthesis, Spectroscopic Characterization, Electrochemistry and Biological Evaluation of Some Binuclear Transition Metal Complexes of Bicompartmental ONO Donor Ligands Containing Benzo [b] Thiophene Moiety. J. Mol. Struct. 2014, 1059, 280–293. [Google Scholar]
- Choudhary, V.K.; Bhatt, A.K.; Sharma, N. Theoretical and Spectroscopic Evidence on a New Triphenyltin (IV) 3, 5-Dinitrosalicylhydroxamate Complex: Synthesis, Structural Characterization, and Biological Screening. J. Coord. Chem. 2020, 73, 947–968. [Google Scholar] [CrossRef]
- Saraf, N.; Barkam, S.; Peppler, M.; Metke, A.; Vázquez-Guardado, A.; Singh, S.; Emile, C.; Bico, A.; Rodas, C.; Seal, S. Microsensor for Limonin Detection: An Indicator of Citrus Greening Disease. Sens. Actuators B Chem. 2019, 283, 724–730. [Google Scholar] [CrossRef]
- Niklowitz, P.; Andler, W.; Menke, T. Coenzyme Q10 Concentration in Plasma and Blood Cells: What about Diurnal Changes? Biofactors 2006, 28, 47–54. [Google Scholar] [CrossRef]
- Muthurajan, T.; Rammanohar, P.; Rajendran, N.P.; Sethuraman, S.; Krishnan, U.M. Evaluation of a Quercetin–Gadolinium Complex as an Efficient Positive Contrast Enhancer for Magnetic Resonance Imaging. RSC Adv. 2015, 5, 86967–86979. [Google Scholar] [CrossRef]
- Pastuszko, A.; Niewinna, K.; Czyz, M.; Jóźwiak, A.; Małecka, M.; Budzisz, E. Synthesis, X-Ray Structure, Electrochemical Properties and Cytotoxic Effects of New Arene Ruthenium (II) Complexes. J. Organomet. Chem. 2013, 745, 64–70. [Google Scholar] [CrossRef]
- Daniel, S.; Limson, J.L.; Dairam, A.; Watkins, G.M.; Daya, S. Through Metal Binding, Curcumin Protects against Lead-and Cadmium-Induced Lipid Peroxidation in Rat Brain Homogenates and against Lead-Induced Tissue Damage in Rat Brain. J. Inorg. Biochem. 2004, 98, 266–275. [Google Scholar] [CrossRef]
- Forbes, J.M.; Ke, B.-X.; Nguyen, T.-V.; Henstridge, D.C.; Penfold, S.A.; Laskowski, A.; Sourris, K.C.; Groschner, L.N.; Cooper, M.E.; Thorburn, D.R. Deficiency in Mitochondrial Complex I Activity Due to Ndufs6 Gene Trap Insertion Induces Renal Disease. Antioxid. Redox Signal. 2013, 19, 331–343. [Google Scholar] [CrossRef]
- Guette-Marquet, S.; Roques, C.; Bergel, A. Catalysis of the Electrochemical Oxygen Reduction Reaction (ORR) by Animal and Human Cells. PLoS ONE 2021, 16, e0251273. [Google Scholar] [CrossRef] [PubMed]
- Bhoopalan, V.; Han, S.G.; Shah, M.M.; Thomas, D.M.; Bhalla, D.K. Tobacco Smoke Modulates Ozone-Induced Toxicity in Rat Lungs and Central Nervous System. Inhal. Toxicol. 2013, 25, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, Z.; Li, G. Preparation of Sulfonated Graphene/Polypyrrole Solid-Phase Microextraction Coating by in Situ Electrochemical Polymerization for Analysis of Trace Terpenes. J. Chromatogr. A 2014, 1346, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Panya, A.; Kittipongpittaya, K.; Laguerre, M.; Bayrasy, C.; Lecomte, J.; Villeneuve, P.; McClements, D.J.; Decker, E.A. Interactions between α-Tocopherol and Rosmarinic Acid and Its Alkyl Esters in Emulsions: Synergistic, Additive, or Antagonistic Effect? J. Agric. Food Chem. 2012, 60, 10320–10330. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Li, X.; Fan, L.; Lu, X.; Kan, X. “Sign-on/off” Sensing Interface Design and Fabrication for Propyl Gallate Recognition and Sensitive Detection. Biosens. Bioelectron. 2016, 86, 741–747. [Google Scholar] [CrossRef]
- Wang, C.; Zhu, F.; Yu, Z.; Zhou, X.; Cheng, W.; Yang, F.; Zhang, X. A Poly (3,4-Ethylenedioxythiophene)/Carbon Nanotube Hybrid Film for Electrocatalytic Determination of Tertiary Butylhydroquinone. Analyst 2021, 146, 6846–6851. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Karaman, C.; Karaman, O.; Karimi, F.; Vasseghian, Y.; Fu, L.; Baghayeri, M.; Rouhi, J.; Senthil Kumar, P.; Show, P.-L.; et al. Nanochemistry Approach for the Fabrication of Fe and N Co-Decorated Biomass-Derived Activated Carbon Frameworks: A Promising Oxygen Reduction Reaction Electrocatalyst in Neutral Media. J. Nanostruct. Chem. 2022. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Beitollahi, H.; Kumar, P.S.; Tajik, S.; Jahani, P.M.; Karimi, F.; Karaman, C.; Vasseghian, Y.; Baghayeri, M.; Rouhi, J. Recent Advances in Carbon Nanomaterials-Based Electrochemical Sensors for Food Azo Dyes Detection. Food Chem. Toxicol. 2022, 164, 112961. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Ayati, A.; Ghanbari, S.; Orooji, Y.; Tanhaei, B.; Karimi, F.; Alizadeh, M.; Rouhi, J.; Fu, L.; Sillanpää, M. Recent Advances in Removal Techniques of Cr (VI) Toxic Ion from Aqueous Solution: A Comprehensive Review. J. Mol. Liq. 2021, 329, 115062. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Ayati, A.; Davoodi, R.; Tanhaei, B.; Karimi, F.; Malekmohammadi, S.; Orooji, Y.; Fu, L.; Sillanpää, M. Recent Advances in Using of Chitosan-Based Adsorbents for Removal of Pharmaceutical Contaminants: A Review. J. Clean. Prod. 2021, 291, 125880. [Google Scholar] [CrossRef]
- Magro, M.; Baratella, D.; Colò, V.; Vallese, F.; Nicoletto, C.; Santagata, S.; Sambo, P.; Molinari, S.; Salviulo, G.; Venerando, A. Electrocatalytic Nanostructured Ferric Tannate as Platform for Enzyme Conjugation: Electrochemical Determination of Phenolic Compounds. Bioelectrochemistry 2020, 132, 107418. [Google Scholar] [CrossRef] [PubMed]
- Zagrean-Tuza, C.; Mot, A.C.; Chmiel, T.; Bende, A.; Turcu, I. Sugar Matters: Sugar Moieties as Reactivity-Tuning Factors in Quercetin O-Glycosides. Food Funct. 2020, 11, 5293–5307. [Google Scholar] [CrossRef]
- Eguílaz, M.; Gutierrez, A.; Gutierrez, F.; González-Domínguez, J.M.; Anson-Casaos, A.; Hernandez-Ferrer, J.; Ferreyra, N.F.; Martinez, M.T.; Rivas, G. Covalent Functionalization of Single-Walled Carbon Nanotubes with Polytyrosine: Characterization and Analytical Applications for the Sensitive Quantification of Polyphenols. Anal. Chim. Acta 2016, 909, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Uliana, C.V.; Garbellini, G.S.; Yamanaka, H. Electrochemical Investigations on the Capacity of Flavonoids to Protect DNA against Damage Caused by Textile Disperse Dyes. Sens. Actuators B Chem. 2014, 192, 188–195. [Google Scholar] [CrossRef]
- Kumar, D.R.; Sayed, M.S.; Baynosa, M.L.; Shim, J.-J. 5-Amino-2-Mercapto-1, 3, 4-Thiadiazole Coated Nitrogen-Doped-Carbon Sphere Composite for the Determination of Phenolic Compounds. Microchem. J. 2020, 157, 105023. [Google Scholar] [CrossRef]
- Bardyn, M.; Maye, S.; Lesch, A.; Delobel, J.; Tissot, J.; Cortés-Salazar, F.; Tacchini, P.; Lion, N.; Girault, H.H.; Prudent, M. The Antioxidant Capacity of Erythrocyte Concentrates Is Increased during the First Week of Storage and Correlated with the Uric Acid Level. Vox Sang. 2017, 112, 638–647. [Google Scholar] [CrossRef] [Green Version]
- Hájková, A.; Barek, J.; Vyskočil, V. Electrochemical DNA Biosensor for Detection of DNA Damage Induced by Hydroxyl Radicals. Bioelectrochemistry 2017, 116, 1–9. [Google Scholar] [CrossRef]
- Zhou, X.; Cheng, W.; Compton, R.G. Contrasts between Single Nanoparticle and Ensemble Electron Transfer: Oxidation and Reduction of DPPH Nanoparticles in Aqueous Media. ChemElectroChem 2015, 2, 691–699. [Google Scholar] [CrossRef]
- Morozova, K.; Aprea, E.; Cantini, C.; Migliorini, M.; Gasperi, F.; Scampicchio, M. Determination of Bitterness of Extra Virgin Olive Oils by Amperometric Detection. Electroanalysis 2016, 28, 2196–2204. [Google Scholar] [CrossRef]
- Dossi, N.; Toniolo, R.; Impellizzieri, F.; Tubaro, F.; Bontempelli, G.; Terzi, F.; Piccin, E. A Paper-Based Platform with a Pencil-Drawn Dual Amperometric Detector for the Rapid Quantification of Ortho-Diphenols in Extravirgin Olive Oil. Anal. Chim. Acta 2017, 950, 41–48. [Google Scholar] [CrossRef]
- Deng, J.; Liu, Q.; Zhang, C.; Cao, W.; Fan, D.; Yang, H. Extraction Optimization of Polyphenols from Waste Kiwi Fruit Seeds (Actinidia Chinensis Planch.) and Evaluation of Its Antioxidant and Anti-Inflammatory Properties. Molecules 2016, 21, 832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erady, V.; Mascarenhas, R.J.; Satpati, A.K.; Bhakta, A.K.; Mekhalif, Z.; Delhalle, J.; Dhason, A. Carbon Paste Modified with Bi Decorated Multi-Walled Carbon Nanotubes and CTAB as a Sensitive Voltammetric Sensor for the Detection of Caffeic Acid. Microchem. J. 2019, 146, 73–82. [Google Scholar] [CrossRef]
- Erady, V.; Mascarenhas, R.J.; Satpati, A.K.; Detriche, S.; Mekhalif, Z.; Dalhalle, J.; Dhason, A. Sensitive Detection of Ferulic Acid Using Multi-Walled Carbon Nanotube Decorated with Silver Nano-Particles Modified Carbon Paste Electrode. J. Electroanal. Chem. 2017, 806, 22–31. [Google Scholar] [CrossRef]
- Lesch, A.; Cortés-Salazar, F.; Amstutz, V.; Tacchini, P.; Girault, H.H. Inkjet Printed Nanohydrogel Coated Carbon Nanotubes Electrodes for Matrix Independent Sensing. Anal. Chem. 2015, 87, 1026–1033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stanković, D.M.; Ognjanović, M.; Martin, F.; Švorc, Ľ.; Mariano, J.F.; Antić, B. Design of Titanium Nitride-and Wolfram Carbide-Doped RGO/GC Electrodes for Determination of Gallic Acid. Anal. Biochem. 2017, 539, 104–112. [Google Scholar] [CrossRef]
- Munoz-Espada, A.; Wood, K.; Bordelon, B.; Watkins, B. Anthocyanin Quantification and Radical Scavenging Capacity of Concord, Norton, and Marechal Foch Grapes and Wines. J. Agric. Food Chem. 2004, 52, 6779–6786. [Google Scholar] [CrossRef]
- Hoyos-Arbeláez, J.; García, G.R.; Arévalo, F.J.; Vázquez, M.; Fernández, H.; Granados, S.G. Electrochemical Determination of Mangiferin Using Glassy Carbon Electrodes Modified with Carbonaceous Nanomaterials. J. Electroanal. Chem. 2018, 808, 1–7. [Google Scholar] [CrossRef]
- Spissu, Y.; Barberis, A.; Bazzu, G.; D’hallewin, G.; Rocchitta, G.; Serra, P.A.; Marceddu, S.; Vineis, C.; Garroni, S.; Culeddu, N. Functionalization of Screen-Printed Sensors with a High Reactivity Carbonaceous Material for Ascorbic Acid Detection in Fresh-Cut Fruit with Low Vitamin C Content. Chemosensors 2021, 9, 354. [Google Scholar] [CrossRef]
- Chen, B.-Y.; Liao, J.-H.; Hsueh, C.-C.; Qu, Z.; Hsu, A.-W.; Chang, C.-T.; Zhang, S. Deciphering Biostimulation Strategy of Using Medicinal Herbs and Tea Extracts for Bioelectricity Generation in Microbial Fuel Cells. Energy 2018, 161, 1042–1054. [Google Scholar] [CrossRef]
- Ricci, A.; Parpinello, G.P.; Teslić, N.; Kilmartin, P.A.; Versari, A. Suitability of the Cyclic Voltammetry Measurements and DPPH• Spectrophotometric Assay to Determine the Antioxidant Capacity of Food-Grade Oenological Tannins. Molecules 2019, 24, 2925. [Google Scholar] [CrossRef] [Green Version]
- Mosleh, M.; Ghoreishi, S.M.; Masoum, S.; Khoobi, A. Determination of Quercetin in the Presence of Tannic Acid in Soft Drinks Based on Carbon Nanotubes Modified Electrode Using Chemometric Approaches. Sens. Actuators B Chem. 2018, 272, 605–611. [Google Scholar] [CrossRef]
- Wang, L.; Malpass-Evans, R.; Carta, M.; McKeown, N.B.; Reeksting, S.B.; Marken, F. Catechin or Quercetin Guests in an Intrinsically Microporous Polyamine (PIM-EA-TB) Host: Accumulation, Reactivity, and Release. RSC Adv. 2021, 11, 27432–27442. [Google Scholar] [CrossRef]
- Kaškonienė, V.; Ruočkuvienė, G.; Kaškonas, P.; Akuneca, I.; Maruška, A. Chemometric Analysis of Bee Pollen Based on Volatile and Phenolic Compound Compositions and Antioxidant Properties. Food Anal. Methods 2015, 8, 1150–1163. [Google Scholar] [CrossRef]
- Chen, S.-S.; Huang, W.J.; Chang, L.S.; Wei, Y.-H. Attenuation of Oxidative Stress after Varicocelectomy in Subfertile Patients with Varicocele. J. Urol. 2008, 179, 639–642. [Google Scholar] [CrossRef] [PubMed]
- Banu, K.; Shimura, T.; Sadeghi, S. Selective Detection and Recovery of Gold at Tannin-Immobilized Non-Conducting Electrode. Anal. Chim. Acta 2015, 853, 207–213. [Google Scholar] [CrossRef] [Green Version]
- Đurđić, S.; Stanković, V.; Vlahović, F.; Ognjanović, M.; Kalcher, K.; Veličković, T.Ć.; Mutić, J.; Stanković, D.M. Laccase Polyphenolic Biosensor Supported on MnO2@ GNP Decorated SPCE: Preparation, Characterization, and Analytical Application. J. Electrochem. Soc. 2021, 168, 037510. [Google Scholar] [CrossRef]
- Di Fusco, M.; Tortolini, C.; Deriu, D.; Mazzei, F. Laccase-Based Biosensor for the Determination of Polyphenol Index in Wine. Talanta 2010, 81, 235–240. [Google Scholar] [CrossRef]
- Rosado, T.; Henriques, I.; Gallardo, E.; Duarte, A. Determination of Melatonin Levels in Different Cherry Cultivars by High-Performance Liquid Chromatography Coupled to Electrochemical Detection. Eur. Food Res. Technol. 2017, 243, 1749–1757. [Google Scholar] [CrossRef]
- Tsiakiri, E.; Sompatzi, E.; Voukia, F.; Sotiropoulos, S.; Pantazaki, A. Biocatalytic and Bioelectrolytic Decolorization of Simulated Melanoidin Wastewaters by Saccharomyces Cerevisiae Cells Suspended and Conjugated on Silica and Alumina. J. Environ. Chem. Eng. 2020, 8, 104078. [Google Scholar] [CrossRef]
- Lima, K.C.; Fernandes, R.N.; dos Santos, C.C.; Damos, F.S.; Rita de Cássia, S.L. Determination of 3,4,5-Trihydroxybenzoic Acid Exploiting a Visible-Light-Driven Photoelectrochemical Platform: Application in Wine and Tea Samples. J. Braz. Chem. Soc. 2022, 33, 425–436. [Google Scholar] [CrossRef]
- Manasa, G.; Mascarenhas, R.J.; Bhakta, A.K.; Mekhalif, Z. Nano-Graphene-Platelet/Brilliant-Green Composite Coated Carbon Paste Electrode Interface for Electrocatalytic Oxidation of Flavanone Hesperidin. Microchem. J. 2021, 160, 105768. [Google Scholar] [CrossRef]
- Zielińska, D.; Nagels, L.; Piskuła, M. Determination of Quercetin and Its Glucosides in Onion by Electrochemical Methods. Anal. Chim. Acta 2008, 617, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Uddin, W.; Hu, G.; Sun, X.; Ullah, S.; Sardar, S.; Wangning, Z.; Nawabi, M.Y. Determination of Morin by Using a Briggs-Rauscher Oscillator. Int. J. Electrochem. Sci. 2018, 13, 598–609. [Google Scholar] [CrossRef]
- Eremia, S.A.; Radu, G.; Litescu, S. Monitoring of Rosmarinic Acid Accumulation in Sage Cell Cultures Using Laccase Biosensor. Phytochem. Anal. 2013, 24, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Aderyani, S.; Flouda, P.; Shah, S.; Green, M.; Lutkenhaus, J.; Ardebili, H. Simulation of Cyclic Voltammetry in Structural Supercapacitors with Pseudocapacitance Behavior. Electrochim. Acta 2021, 390, 138822. [Google Scholar] [CrossRef]
- Wang, L.; Aversa, R.; Houa, Z.; Tian, J.; Liang, S.; Ge, S.; Chen, Y.; Perrotta, V.; Apicella, A.; Apicella, D. Bioresorption Control and Biological Response of Magnesium Alloy AZ31 Coated with Poly-β-Hydroxybutyrate. Appl. Sci. 2021, 11, 5627. [Google Scholar] [CrossRef]
- Capecchi, E.; Piccinino, D.; Tomaino, E.; Bizzarri, B.M.; Polli, F.; Antiochia, R.; Mazzei, F.; Saladino, R. Lignin Nanoparticles Are Renewable and Functional Platforms for the Concanavalin a Oriented Immobilization of Glucose Oxidase–Peroxidase in Cascade Bio-Sensing. RSC Adv. 2020, 10, 29031–29042. [Google Scholar] [CrossRef]
- Erol, A.; Akpınar, F.; Muti, M. Electrochemical Determination of Anticancer Drug Bendamustine and Its Interaction with Double Strand DNA in the Absence and Presence of Quercetin. Colloids Surf. B Biointerfaces 2021, 205, 111884. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Milbury, P.E.; Kwak, H.-K.; Collins, F.W.; Samuel, P.; Blumberg, J.B. Avenanthramides and Phenolic Acids from Oats Are Bioavailable and Act Synergistically with Vitamin C to Enhance Hamster and Human LDL Resistance to Oxidation. J. Nutr. 2004, 134, 1459–1466. [Google Scholar] [CrossRef] [Green Version]
- Djaković, S.; Maračić, S.; Lapić, J.; Kovalski, E.; Hildebrandt, A.; Lang, H.; Vrček, V.; Raić-Malić, S.; Cetina, M. Triazole-Tethered Ferrocene-Quinoline Conjugates: Solid-State Structure Analysis, Electrochemistry and Theoretical Calculations. Struct. Chem. 2021, 32, 2291–2301. [Google Scholar] [CrossRef]
- Giacomelli, C.; Miranda, F.; da Silva Miranda, F.; Gonçalves, N.S.; Spinelli, A. Antioxidant Activity of Phenolic and Related Compounds: A Density Functional Theory Study on the O–H Bond Dissociation Enthalpy. Redox Rep. 2004, 9, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Villaño, D.; Fernández-Pachón, M.; Troncoso, A.; Garcıa-Parrilla, M. The Antioxidant Activity of Wines Determined by the ABTS+ Method: Influence of Sample Dilution and Time. Talanta 2004, 64, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Kahl, M.; Golden, T.D. Electrochemical Determination of Phenolic Acids at a Zn/Al Layered Double Hydroxide Film Modified Glassy Carbon Electrode. Electroanalysis 2014, 26, 1664–1670. [Google Scholar] [CrossRef]
- Diamantis, D.A.; Oblukova, M.; Chatziathanasiadou, M.V.; Gemenetzi, A.; Papaemmanouil, C.; Gerogianni, P.S.; Syed, N.; Crook, T.; Galaris, D.; Deligiannakis, Y. Bioinspired Tailoring of Fluorogenic Thiol Responsive Antioxidant Precursors to Protect Cells against H2O2-Induced DNA Damage. Free Radic. Biol. Med. 2020, 160, 540–551. [Google Scholar] [CrossRef] [PubMed]
- Boček, I.; Starčević, K.; Jovanović, I.N.; Vianello, R.; Hranjec, M. Novel Imidazo [4, 5-b] Pyridine Derived Acrylonitriles: A Combined Experimental and Computational Study of Their Antioxidative Potential. J. Mol. Liq. 2021, 342, 117527. [Google Scholar] [CrossRef]
- Lavelli, V.; Peri, C.; Rizzolo, A. Antioxidant Activity of Tomato Products as Studied by Model Reactions Using Xanthine Oxidase, Myeloperoxidase, and Copper-Induced Lipid Peroxidation. J. Agric. Food Chem. 2000, 48, 1442–1448. [Google Scholar] [CrossRef]
- Chau, F.-T.; Chan, H.-Y.; Cheung, C.-Y.; Xu, C.-J.; Liang, Y.; Kvalheim, O.M. Recipe for Uncovering the Bioactive Components in Herbal Medicine. Anal. Chem. 2009, 81, 7217–7225. [Google Scholar] [CrossRef]
- Jiang, L.; Li, X.; Wang, D. Development of a Rapid Method for the Evaluation of DPPH Radical Scavenging Activity of Ginger (Zingiber Officinale) Foods Based on Cyclic Voltammetry. Food Anal. Methods 2017, 10, 1419–1429. [Google Scholar] [CrossRef]
- Sužnjević, D.; Petrović, M.; Pastor, F.T.; Veljović, M.; Zlatanović, S.; Antić, M.; Gorjanović, S. Reduction of Hg2+ by Individual Phenolics and Complex Samples and Its Application in Polarographic Antioxidant Assay. J. Electrochem. Soc. 2015, 162, H428. [Google Scholar] [CrossRef]
- Nikolaevskaya, E.N.; Kansuzyan, A.V.; Filonova, G.E.; Zelenova, V.A.; Pechennikov, V.M.; Krylova, I.V.; Egorov, M.P.; Jouikov, V.V.; Syroeshkin, M.A. Germanium Dioxide and the Antioxidant Properties of Catechols. Eur. J. Inorg. Chem. 2019, 2019, 676–681. [Google Scholar] [CrossRef]
- Lee, M.-L.; Li, Y.-H.; Yeh, J.-W.; Shih, H.C. Improvement in Safety and Cycle Life of Lithium-Ion Batteries by Employing Quercetin as an Electrolyte Additive. J. Power Sources 2012, 214, 251–257. [Google Scholar] [CrossRef]
- Barquero-Quirós, M.; Arcos-Martínez, M.J. Effect of Nanoparticles on Modified Screen Printed Inhibition Superoxide Dismutase Electrodes for Aluminum. Sensors 2016, 16, 1588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mittal, A.; Göke, F.; Flint, R.; Loveday, B.P.; Thompson, N.; Delahunt, B.; Kilmartin, P.A.; Cooper, G.J.; MacDonald, J.; Hickey, A. The Redox Status of Experimental Hemorrhagic Shock as Measured by Cyclic Voltammetry. Shock 2010, 33, 460–466. [Google Scholar] [CrossRef]
- Wang, P.; Chen, H.; Tian, J.; Dai, Z.; Zou, X. Electrochemical Evaluation of DNA Methylation Level Based on the Stoichiometric Relationship between Purine and Pyrimidine Bases. Biosens. Bioelectron. 2013, 45, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Chevion, S.; Moran, D.S.; Heled, Y.; Shani, Y.; Regev, G.; Abbou, B.; Berenshtein, E.; Stadtman, E.R.; Epstein, Y. Plasma Antioxidant Status and Cell Injury after Severe Physical Exercise. Proc. Natl. Acad. Sci. USA 2003, 100, 5119–5123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karimi-Maleh, H.; Karimi, F.; Fu, L.; Sanati, A.L.; Alizadeh, M.; Karaman, C.; Orooji, Y. Cyanazine Herbicide Monitoring as a Hazardous Substance by a DNA Nanostructure Biosensor. J. Hazard. Mater. 2022, 423, 127058. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Alizadeh, M.; Orooji, Y.; Karimi, F.; Baghayeri, M.; Rouhi, J.; Tajik, S.; Beitollahi, H.; Agarwal, S.; Gupta, V.K.; et al. Guanine-Based DNA Biosensor Amplified with Pt/SWCNTs Nanocomposite as Analytical Tool for Nanomolar Determination of Daunorubicin as an Anticancer Drug: A Docking/Experimental Investigation. Ind. Eng. Chem. Res. 2021, 60, 816–823. [Google Scholar] [CrossRef]
- Sousa, W.R.; da Rocha, C.; Cardoso, C.L.; Silva, D.H.S.; Zanoni, M.V.B. Determination of the Relative Contribution of Phenolic Antioxidants in Orange Juice by Voltammetric Methods. J. Food Compos. Anal. 2004, 17, 619–633. [Google Scholar] [CrossRef]
- Blasco, A.J.; Rogerio, M.C.; González, M.C.; Escarpa, A. “Electrochemical Index” as a Screening Method to Determine “Total Polyphenolics” in Foods: A Proposal. Anal. Chim. Acta 2005, 539, 237–244. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The Chemistry behind Antioxidant Capacity Assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Barroso, M.F.; de-los-Santos-Álvarez, N.; Delerue-Matos, C.; Oliveira, M.B.P.P. Towards a Reliable Technology for Antioxidant Capacity and Oxidative Damage Evaluation: Electrochemical (Bio)Sensors. Biosens. Bioelectron. 2011, 30, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mello, L.D.; Hernandez, S.; Marrazza, G.; Mascini, M.; Kubota, L.T. Investigations of the Antioxidant Properties of Plant Extracts Using a DNA-Electrochemical Biosensor. Biosens. Bioelectron. 2006, 21, 1374–1382. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.P.; Calegari, F.; Zarbin, A.J.G.; Marcolino-Júnior, L.H.; Bergamini, M.F. Voltammetric Determination of the Antioxidant Capacity in Wine Samples Using a Carbon Nanotube Modified Electrode. J. Agric. Food Chem. 2011, 59, 7620–7625. [Google Scholar] [CrossRef] [PubMed]
- Sangeetha, N.S.; Narayanan, S.S. A Novel Bimediator Amperometric Sensor for Electrocatalytic Oxidation of Gallic Acid and Reduction of Hydrogen Peroxide. Anal. Chim. Acta 2014, 828, 34–45. [Google Scholar] [CrossRef]
- Ghoreishi, S.M.; Behpour, M.; Khayatkashani, M.; Motaghedifard, M.H. Simultaneous Determination of Ellagic and Gallic Acid in Punica Granatum, Myrtus Communis and Itriphal Formulation by an Electrochemical Sensor Based on a Carbon Paste Electrode Modified with Multi-Walled Carbon Nanotubes. Anal. Methods 2011, 3, 636–645. [Google Scholar] [CrossRef] [PubMed]
- Tashkhourian, J.; Ana, S.F.N.; Hashemnia, S.; Hormozi-Nezhad, M.R. Construction of a Modified Carbon Paste Electrode Based on TiO2 Nanoparticles for the Determination of Gallic Acid. J. Solid State Electrochem. 2013, 17, 157–165. [Google Scholar] [CrossRef]
- Luo, J.H.; Li, B.L.; Li, N.B.; Luo, H.Q. Sensitive Detection of Gallic Acid Based on Polyethyleneimine-Functionalized Graphene Modified Glassy Carbon Electrode. Sens. Actuators B Chem. 2013, 186, 84–89. [Google Scholar] [CrossRef]
- Tashkhourian, J.; Nami-Ana, S.F. A Sensitive Electrochemical Sensor for Determination of Gallic Acid Based on SiO2 Nanoparticle Modified Carbon Paste Electrode. Mater. Sci. Eng. C 2015, 52, 103–110. [Google Scholar] [CrossRef]
- Petković, B.B.; Stanković, D.; Milčić, M.; Sovilj, S.P.; Manojlović, D. Dinuclear Copper(II) Octaazamacrocyclic Complex in a PVC Coated GCE and Graphite as a Voltammetric Sensor for Determination of Gallic Acid and Antioxidant Capacity of Wine Samples. Talanta 2015, 132, 513–519. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ziyatdinova, G.; Aytuganova, I.; Nizamova, A.; Budnikov, H. Differential Pulse Voltammetric Assay of Coffee Antioxidant Capacity with MWNT-Modified Electrode. Food Anal. Methods 2013, 6, 1629–1638. [Google Scholar] [CrossRef]
- Tomac, I.; Šeruga, M. Electrochemical Properties of Chlorogenic Acids and Determination of Their Content in Coffee Using Differential Pulse Voltammetry. Int. J. Electrochem. Sci 2016, 11, 2854–2876. [Google Scholar] [CrossRef]
- Bianchini, C.; Curulli, A.; Pasquali, M.; Zane, D. Determination of Caffeic Acid in Wine Using PEDOT Film Modified Electrode. Food Chem. 2014, 156, 81–86. [Google Scholar] [CrossRef] [PubMed]
- David, I.G.; Bizgan, A.-M.C.; Popa, D.E.; Buleandra, M.; Moldovan, Z.; Badea, I.A.; Tekiner, T.A.; Basaga, H.; Ciucu, A.A. Rapid Determination of Total Polyphenolic Content in Tea Samples Based on Caffeic Acid Voltammetric Behaviour on a Disposable Graphite Electrode. Food Chem. 2015, 173, 1059–1065. [Google Scholar] [CrossRef] [PubMed]








| No. | Freq | Cited Journal |
|---|---|---|
| 1 | 165 | Journal of Agricultural and Food Chemistry |
| 2 | 154 | Food Chemistry |
| 3 | 107 | Analytica Chimica Acta |
| 4 | 102 | Talanta |
| 5 | 102 | Journal of Electroanalytical Chemistry |
| 6 | 94 | Electroanalysis |
| 7 | 91 | Electrochimica Acta |
| 8 | 87 | Free Radical Biology and Medicine |
| 9 | 74 | Analytical Chemistry |
| 10 | 70 | Sensors and Actuators B: Chemical |
| No. | Freq | Centrality | Keywords | No. | Freq | Centrality | Keywords |
|---|---|---|---|---|---|---|---|
| 1 | 87 | 0.60 | Antioxidant capacity | 11 | 15 | 0.01 | Polyphenol |
| 2 | 28 | 0.16 | Capacity | 12 | 15 | 0.02 | Electrode |
| 3 | 23 | 0.05 | Oxidation | 13 | 14 | 0.11 | Behavior |
| 4 | 22 | 0.00 | Sensor | 14 | 13 | 0.02 | Voltammetric determination |
| 5 | 20 | 0.07 | Phenolic compound | 15 | 13 | 0.08 | Oxidative stress |
| 6 | 19 | 0.29 | Antioxidant activity | 16 | 12 | 0.18 | Mechanism |
| 7 | 17 | 0.27 | Flavonoid | 17 | 12 | 0.09 | Biosensor |
| 8 | 16 | 0.21 | Acid | 18 | 12 | 0.05 | Electrochemical sensor |
| 9 | 16 | 0.15 | Antioxidant | 19 | 10 | 0.03 | Derivative |
| 10 | 15 | 0.04 | Nanoparticle | 20 | 10 | 0.13 | Extract |
| Keywords | Strength | Begin | End | 1999–2021 |
|---|---|---|---|---|
| Disease | 3.14 | 2004 | 2013 | ▂▂▂▂▂▃▃▃▃▃▃▃▃▃▃▂▂▂▂▂▂▂▂ |
| Performance liquid chromatography | 2.80 | 2005 | 2010 | ▂▂▂▂▂▂▃▃▃▃▃▃▂▂▂▂▂▂▂▂▂▂▂ |
| Electrochemical detection | 3.11 | 2009 | 2012 | ▂▂▂▂▂▂▂▂▂▂▃▃▃▃▂▂▂▂▂▂▂▂▂ |
| Assay | 3.17 | 2010 | 2014 | ▂▂▂▂▂▂▂▂▂▂▂▃▃▃▃▃▂▂▂▂▂▂▂ |
| Sample | 3.55 | 2015 | 2018 | ▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▃▃▃▃▂▂▂ |
| Glassy carbon electrode | 2.60 | 2016 | 2017 | ▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▃▃▂▂▂▂ |
| Nanoparticle | 3.28 | 2018 | 2021 | ▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▃▃▃▃ |
| Food | 2.93 | 2018 | 2021 | ▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▃▃▃▃ |
| Vitamin c | 2.74 | 2018 | 2019 | ▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▃▃▂▂ |
| Electrochemical sensor | 3.07 | 2019 | 2021 | ▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▃▃▃ |
| Oxidation | 2.87 | 2019 | 2021 | ▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▂▃▃▃ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, Y.; Karimi-Maleh, H.; Fu, L. Evaluation of Antioxidants Using Electrochemical Sensors: A Bibliometric Analysis. Sensors 2022, 22, 3238. https://doi.org/10.3390/s22093238
Zheng Y, Karimi-Maleh H, Fu L. Evaluation of Antioxidants Using Electrochemical Sensors: A Bibliometric Analysis. Sensors. 2022; 22(9):3238. https://doi.org/10.3390/s22093238
Chicago/Turabian StyleZheng, Yuhong, Hassan Karimi-Maleh, and Li Fu. 2022. "Evaluation of Antioxidants Using Electrochemical Sensors: A Bibliometric Analysis" Sensors 22, no. 9: 3238. https://doi.org/10.3390/s22093238






