Current Technologies for Detection of COVID-19: Biosensors, Artificial Intelligence and Internet of Medical Things (IoMT): Review
Abstract
:1. Introduction
1.1. Comparison with Similar Studies
1.2. Scope
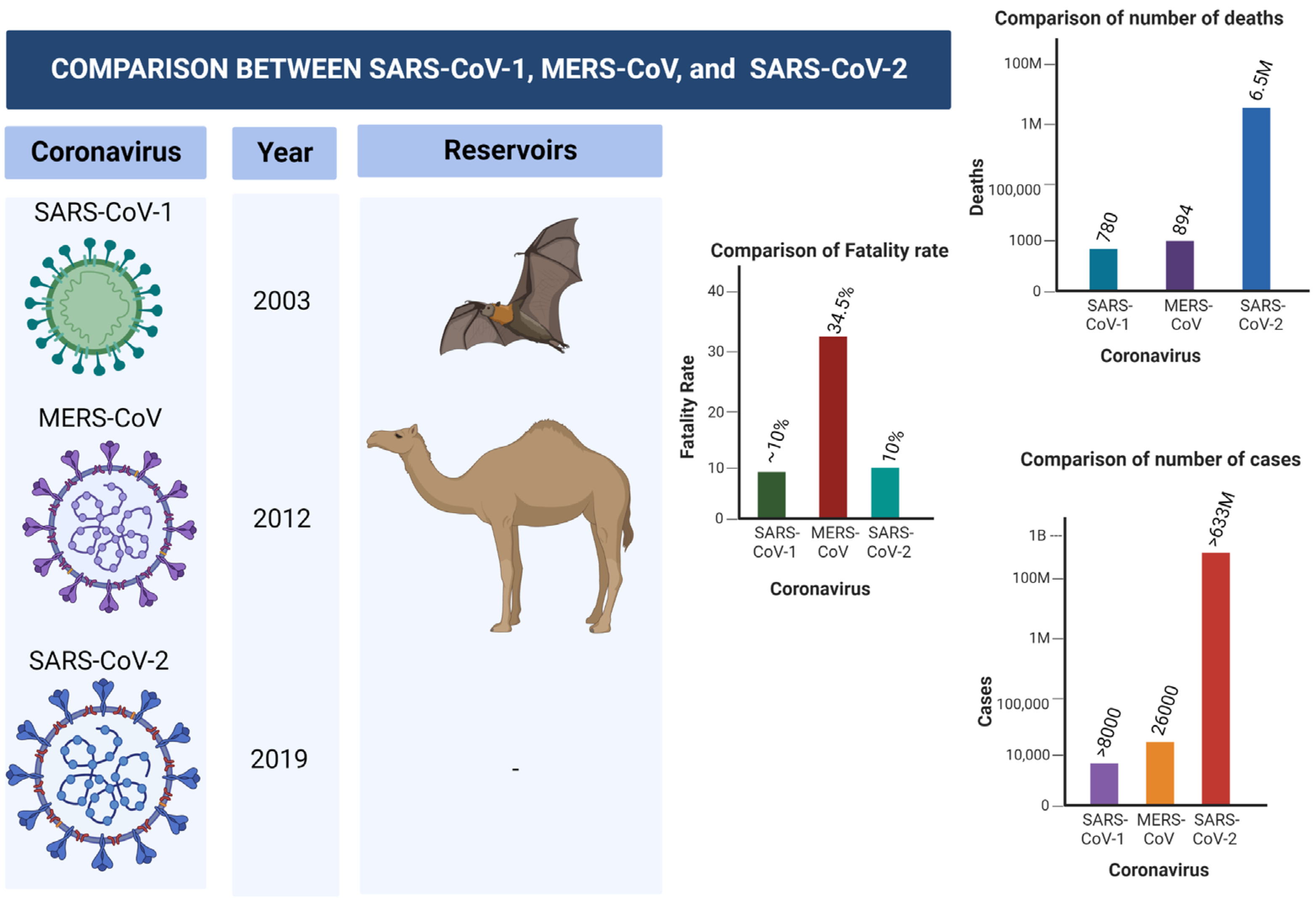
2. COVID-19
2.1. Transmission of SARS-CoV-2
2.2. Symptoms of SARS-CoV-2
3. Molecular Diagnosis of COVID-19
3.1. Laboratory Assays
3.1.1. Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
3.1.2. Antibody-Based Method
3.1.3. Antigen-Based Method
3.2. Strengths and Weakness of Molecular Testing
3.3. Application of Biosensors for the Detection of SARS-CoV-2
4. Computer-Aided Diagnosis (CAD) and Internet of Medical Things (IoMT)
4.1. Artificial Intelligence (AI) and Machine Learning (ML)
4.1.1. Supervised Machine Learning
4.1.2. Unsupervised Machine Learning
4.2. Deep Learning (DL)
4.3. Internet of Medical Things (IoMT)
4.3.1. Advantage of IoT-Based Systems: How IoT Is Shaping Clinical Diagnosis
4.3.2. Disadvantage of IoT-Based Systems
4.3.3. Deployment of IoT-Based Systems
5. Diagnostic Imaging
5.1. Radiographic Imaging of COVID-19
5.1.1. X-ray Imaging
5.1.2. CT Scan Imaging
5.2. Radiographic Dataset
- (A)
- JP Cohen COVID-19 Xray Dataset
- (B)
- COVID-19 Radiography Dataset
- (C)
- COVIDx Dataset
- (D)
- HCV-UFPR COVID-19 Dataset
- (E)
- SARS-CoV-2 CT Scan Dataset
- (F)
- Chest X-ray
- (G)
- ChestX-ray8
5.3. AI-Powered Detection of COVID-19 from Radiographic Imaging
5.3.1. AI-Powered Detection of COVID-19 from X-ray Images
- (A)
- Binary
- (B)
- Ternary
- (C)
- Quaternary
5.3.2. AI-Powered Detection of COVID-19 from CT Scans
5.3.3. IoT-Enabled Devices for Detection of COVID-19
6. Open Research Issue
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviations | Full-Meaning |
| AI | Artificial Intelligence |
| ANN | Artificial Neural Network |
| AP | Anterior–posterior |
| AUC | Area Under the Curve |
| AUNPS | Gold Nanoparticles |
| AUSPE | Gold Screen Printed Electrode |
| CAD | Computer Aided Detection/Diagnosis |
| CNN | Convolutional Neural Network |
| COVID-19 | Coronavirus Disease 2019 |
| CRISPR | Clustered Regularly Interspace Short Palindromic Repeat |
| CT | Computed Tomography |
| CXR | Chest X-ray |
| DL | Deep Learning |
| DNA | Deoxyribonucleic Acid |
| ELISA | Enzyme-linked Immunosorbent Assay |
| FET | Field-effect Transistor |
| Fg | Femptogram |
| fM | Fento Molar |
| FR-CNN | Faster Region CNN |
| GBM | Gradient Boosting Machine |
| HRP | Horseradish Peroxidase |
| IoT | Internet of Things |
| IoMT | Internet of Medical Things |
| IT | Information Technology |
| LOD | Limit of Detection |
| LSTM | Long-Short Term Memory |
| MERS-CoV | Middle East Respiratory Syndrome Coronavirus |
| ML | Machine Learning |
| MRI | Magnetic Resonance Imaging |
| NA | Nucleic Acid |
| NPs | Nanoparticles |
| PA | Posterior–anterior |
| PET | Position Emission Tomography |
| Pfu | Plaque-forming unit |
| POC | Point of Care |
| RML | Reinforcement Machine Learning |
| RNA | Ribonucleic Acid |
| ROIs | Region of Interests |
| RPA | Recombinase Polymerase Amplification |
| RT-PCR | Reverse Transcription-Polymerase Chain Reaction |
| SARS-CoV-1 | Severe Acute Respiratory Coronavirus-1 |
| SARS-CoV-2 | Severe Acute Respiratory Coronavirus-2 |
| SML | Supervised Machine Learning |
| SPECT | Single Position Emission Computed Tomography |
| SSDNA | Single Strand Deoxyribonucleic Acid |
| SVM | Support Vector Machine |
| TL | Transfer Learning |
| µL | Micro Litre |
| UML | Unsupervised Machine Learning |
| WE | Working Electrode |
| WNN | Wavelet Neural Network |
| WOA | Whale Optimization Algorithms |
References
- Güner, H.R.; Hasanoğlu, İ.; Aktaş, F. COVID-19: Prevention and control measures in community. Turk. J. Med. Sci. 2020, 50, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Ai, T.; Yang, Z.; Hou, H.; Zhan, C.; Chen, C.; Lv, W.; Tao, Q.; Sun, Z.; Xia, L. Correlation of chest CT and RT-PCR testing in coronavirus disease 2019 (COVID-19) in China: A report of 1014 cases. Rad 2020, 296, E32–E40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamza, A.; Khan, M.A.; Wang, S.H.; Alhaisoni, M.; Alharbi, M.; Hussein, H.S.; Alshazly, H.; Kim, Y.J.; Cha, J. COVID-19 classification using chest X-ray images based on fusion-assisted deep Bayesian optimization and Grad-CAM visualization. Front. Public Health 2022, 10, 1046296. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Alhaisoni, M.; Nazir, M.; Alqahtani, A.; Binbusayyis, A.; Alsubai, S.; Nam, Y.; Kang, B.G. A Healthcare System for COVID19 Classification Using Multi-Type Classical Features Selection. Comput. Mater. Contin. 2023, 74, 1393–1412. [Google Scholar] [CrossRef]
- Arun, K.R.; Elizabeth, T.R.; Sukumaran, A.; Paul, J.K.; Vasudevan, D.M. COVID-19: Current trends in invitro diagnostics. Indian J. Clin. Biochem. 2020, 35, 285–289. [Google Scholar] [CrossRef]
- Yüce, M.; Filiztekin, E.; Özkaya, K.G. COVID-19 diagnosis—A review of current methods. Biosens. Bioelectron. 2021, 172, 112752. [Google Scholar] [CrossRef]
- Afzal, A. Molecular diagnostic technologies for COVID-19: Limitations and challenges. J. Adv. Res. 2020, 26, 149–159. [Google Scholar] [CrossRef]
- Liu, G.; Rusling, J.F. COVID-19 antibody tests and their limitations. ACS Sens. 2021, 6, 593–612. [Google Scholar] [CrossRef]
- Quraishi, M.; Upadhyay, S.K.; Nigam, A. COVID-19 Diagnostics: A Panoramic View on Its Present Scenario, Challenges and Solutions. India Sect. B Biol. Sci. 2022, 92, 709–721. [Google Scholar] [CrossRef]
- Doi, K. Computer-aided diagnosis in medical imaging: Historical review, current status and future potential. Comput. Med. Imaging Graph. 2007, 31, 198–211. [Google Scholar] [CrossRef]
- Van Ginneken, B.; Schaefer-Prokop, C.M.; Prokop, M. Computer-aided diagnosis: How to move from the laboratory to the clinic. Rad 2011, 261, 719–732. [Google Scholar] [CrossRef]
- Yassin, N.I.; Omran, S.; El Houby, E.M.; Allam, H. Machine learning techniques for breast cancer computer aided diagnosis using different image modalities: A systematic review. Comput. Methods Programs Biomed. 2018, 156, 25–45. [Google Scholar] [CrossRef]
- Ahmad, O.F.; Soares, A.S.; Mazomenos, E.; Brandao, P.; Vega, R.; Seward, E.; Stoyanov, D.; Chand, M.; Lovat, L.B. Artificial intelligence and computer-aided diagnosis in colonoscopy: Current evidence and future directions. Lancet Gastroenterol. Hepatol. 2019, 4, 71–80. [Google Scholar] [CrossRef] [Green Version]
- Litjens, G.; Debats, O.; Barentsz, J.; Karssemeijer, N.; Huisman, H. Computer-aided detection of prostate cancer in MRI. IEEE Trans. Med. Imaging 2014, 33, 1083–1092. [Google Scholar] [CrossRef]
- Iqbal, S.; Ghani Khan, M.U.; Saba, T.; Mehmood, Z.; Javaid, N.; Rehman, A.; Abbasi, R. Deep learning model integrating features and novel classifiers fusion for brain tumor segmentation. Microsc. Res. Tech. 2019, 82, 1302–1315. [Google Scholar] [CrossRef]
- Ibrahim, A.U.; Al-Turjman, F.; Ozsoz, M.; Serte, S. Computer aided detection of tuberculosis using two classifiers. Biomed. Eng./Biomed. Tech. 2022, 67, 513–524. [Google Scholar] [CrossRef]
- Umar Ibrahim, A.; Ozsoz, M.; Serte, S.; Al-Turjman, F.; Habeeb Kolapo, S. Convolutional neural network for diagnosis of viral pneumonia and COVID-19 alike diseases. Expert Syst. 2022, 39, e12705. [Google Scholar] [CrossRef]
- Ibrahim, A.U.; Ozsoz, M.; Serte, S.; Al-Turjman, F.; Yakoi, P.S. Pneumonia classification using deep learning from chest X-ray images during COVID-19. Cognit. Comput. 2021, 1, 1–3. [Google Scholar] [CrossRef]
- Arshad, M.; Khan, M.A.; Tariq, U.; Armghan, A.; Alenezi, F.; Younus Javed, M.; Aslam, S.M.; Kadry, S. A computer-aided diagnosis system using deep learning for multiclass skin lesion classification. Comput. Intell. Neurosci. 2021, 2021, 9619079. [Google Scholar] [CrossRef]
- Razdan, S.; Sharma, S. Internet of Medical Things (IoMT): Overview, emerging technologies, and case studies. IETE Tech. Rev. 2022, 39, 775–788. [Google Scholar] [CrossRef]
- Jeba Kumar, R.J.; Roopa Jayasingh, J.; Telagathoti, D.B. Intelligent Transit Healthcare Schema Using Internet of Medical Things (IoMT) Technology for Remote Patient Monitoring. In Internet of Medical Things; Springer: Cham, Switzerland, 2021; pp. 17–33. [Google Scholar]
- Dwivedi, R.; Mehrotra, D.; Chandra, S. Potential of Internet of Medical Things (IoMT) applications in building a smart healthcare system: A systematic review. J. Oral Biol. Craniofacial Res. 2021, 12, 302–318. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Nehra, M.; Kumar, R.; Dilbaghi, N.; Hu, T.; Kumar, S.; Kaushik, A.; Li, C.Z. Internet of medical things (IoMT)-integrated biosensors for point-of-care testing of infectious diseases. Biosens. Bioelectron. 2021, 179, 113074. [Google Scholar] [CrossRef] [PubMed]
- Manickam, P.; Mariappan, S.A.; Murugesan, S.M.; Hansda, S.; Kaushik, A.; Shinde, R.; Thipperudraswamy, S.P. Artificial intelligence (AI) and internet of medical things (IoMT) assisted biomedical systems for intelligent healthcare. Biosens 2022, 12, 562. [Google Scholar] [CrossRef] [PubMed]
- Samson, R.; Navale, G.R.; Dharne, M.S. Biosensors: Frontiers in rapid detection of COVID-19. Biotech 2020, 10, 385. [Google Scholar] [CrossRef] [PubMed]
- Santiago, I. Trends and innovations in biosensors for COVID-19 mass testing. Chem. Biochem. 2020, 21, 2880–2889. [Google Scholar] [CrossRef]
- Falzone, L.; Gattuso, G.; Tsatsakis, A.; Spandidos, D.A.; Libra, M. Current and innovative methods for the diagnosis of COVID 19 infection. Int. J. Mol. Med. 2021, 47, 1–23. [Google Scholar] [CrossRef]
- Huang, S.; Yang, J.; Fong, S.; Zhao, Q. Artificial intelligence in the diagnosis of COVID-19: Challenges and perspectives. Int. J. Biol. Sci. 2021, 17, 1581. [Google Scholar] [CrossRef]
- Ciotti, M.; Ciccozzi, M.; Terrinoni, A.; Jiang, W.C.; Wang, C.B.; Bernardini, S. The COVID-19 pandemic. Crit. Rev. Clin. Lab. Sci. 2020, 57, 365–388. [Google Scholar] [CrossRef]
- Nicholls, J.; Dong, X.P.; Jiang, G.; Peiris, M. SARS: Clinical virology and pathogenesis. Respirology 2003, 8, S6–S8. [Google Scholar] [CrossRef]
- Watanabe, T.; Bartrand, T.A.; Weir, M.H.; Omura, T.; Haas, C.N. Development of a dose-response model for SARS coronavirus. Risk Anal. Int. J. 2010, 30, 1129–1138. [Google Scholar] [CrossRef]
- Oboho, I.K.; Tomczyk, S.M.; Al-Asmari, A.M.; Banjar, A.A.; Al-Mugti, H.; Aloraini, M.S.; Alkhaldi, K.Z.; Almohammadi, E.L.; Alraddadi, B.M.; Gerber, S.I.; et al. 2014 MERS-CoV outbreak in Jeddah—A link to health care facilities. N. Engl. J. Med. 2015, 372, 846–854. [Google Scholar] [CrossRef]
- Ahmadzadeh, J.; Mobaraki, K.; Mousavi, S.J.; Aghazadeh-Attari, J.; Mirza-Aghazadeh-Attari, M.; Mohebbi, I. The risk factors associated with MERS-CoV patient fatality: A global survey. Diagn. Microbiol. Infect. Dis. 2020, 96, 114876. [Google Scholar] [CrossRef]
- Majra, D.; Benson, J.; Pitts, J.; Stebbing, J. SARS-CoV-2 (COVID-19) superspreader events. J. Infect. 2021, 82, 36–40. [Google Scholar] [CrossRef]
- Johansson, M.A.; Quandelacy, T.M.; Kada, S.; Prasad, P.V.; Steele, M.; Brooks, J.T.; Slayton, R.B.; Biggerstaff, M.; Butler, J.C. SARS-CoV-2 transmission from people without COVID-19 symptoms. JAMA Netw. Open 2021, 4, e2035057. [Google Scholar] [CrossRef]
- Uddin, M.; Mustafa, F.; Rizvi, T.A.; Loney, T.; Al Suwaidi, H.; Al-Marzouqi, A.H.; Kamal Eldin, A.; Alsabeeha, N.; Adrian, T.E.; Stefanini, C.; et al. SARS-CoV-2/COVID-19: Viral genomics, epidemiology, vaccines, and therapeutic interventions. Viruses 2020, 12, 526. [Google Scholar] [CrossRef]
- Touma, M. COVID-19: Molecular diagnostics overview. J. Mol. Med. 2020, 98, 947–954. [Google Scholar] [CrossRef]
- Ye, G.; Lin, H.; Chen, S.; Wang, S.; Zeng, Z.; Wang, W.; Zhang, S.; Rebmann, T.; Li, Y.; Pan, Z.; et al. Environmental contamination of SARS-CoV-2 in healthcare premises. J. Infect. 2020, 81, e1–e5. [Google Scholar] [CrossRef]
- Lee, E.Y.; Ng, M.Y.; Khong, P.L. COVID-19 pneumonia: What has CT taught us? Lancet Infect. Dis. 2020, 20, 384–385. [Google Scholar] [CrossRef]
- Lai, C.K.; Lam, W. Laboratory testing for the diagnosis of COVID-19. Biochem. Biophys. Res. Commun. 2021, 538, 226–230. [Google Scholar] [CrossRef]
- Shyu, D.; Dorroh, J.; Holtmeyer, C.; Ritter, D.; Upendran, A.; Kannan, R.; Dandachi, D.; Rojas-Moreno, C.; Whitt, S.P.; Regunath, H. Laboratory tests for COVID-19: A review of peer-reviewed publications and implications for clinical use. Missouri Med. 2020, 117, 184. [Google Scholar]
- Nyaruaba, R.; Mwaliko, C.; Hong, W.; Amoth, P.; Wei, H. SARS-CoV-2/COVID-19 laboratory biosafety practices and current molecular diagnostic tools. J. Biosaf. Biosecur. 2021, 3, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Smyrlaki, I.; Ekman, M.; Lentini, A.; Rufino de Sousa, N.; Papanicolaou, N.; Vondracek, M.; Aarum, J.; Safari, H.; Muradrasoli, S.; Rothfuchs, A.G.; et al. Massive and rapid COVID-19 testing is feasible by extraction-free SARS-CoV-2 RT-PCR. Nat. Commun. 2020, 11, 4812. [Google Scholar] [CrossRef] [PubMed]
- Barza, R.; Patel, P.; Sabatini, L.; Singh, K. Use of a simplified sample processing step without RNA extraction for direct SARS-CoV-2 RT-PCR detection. J. Clin. Virol. 2020, 132, 104587. [Google Scholar] [CrossRef] [PubMed]
- Augustine, R.; Das, S.; Hasan, A.; Abdul Salam, S.; Augustine, P.; Dalvi, Y.B.; Varghese, R.; Primavera, R.; Yassine, H.M.; Thakor, A.S.; et al. Rapid antibody-based COVID-19 mass surveillance: Relevance, challenges, and prospects in a pandemic and post-pandemic world. J. Clin. Med. 2020, 9, 3372. [Google Scholar] [CrossRef] [PubMed]
- La Marca, A.; Capuzzo, M.; Paglia, T.; Roli, L.; Trenti, T.; Nelson, S.M. Testing for SARS-CoV-2 (COVID-19): A systematic review and clinical guide to molecular and serological in-vitro diagnostic assays. Reprod. Biomed. Online 2020, 41, 483–499. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Ma, Y.; Si, Y.; Zhu, X.; Zhang, L.; Feng, H.; Tian, D.; Liao, Y.; Liu, T.; Lu, H.; et al. Rapid SARS-CoV-2 antigen detection potentiates early diagnosis of COVID-19 disease. Biosci. Trends 2021, 15, 93–99. [Google Scholar] [CrossRef]
- Li, K.; Tong, C.; Ha, X.; Zeng, C.; Chen, X.; Xu, F.; Yang, J.; Du, H.; Chen, Y.; Cai, J.; et al. Development and clinical evaluation of a rapid antibody lateral flow assay for the diagnosis of SARS-CoV-2 infection. BMC Infect. Dis. 2021, 21, 860. [Google Scholar] [CrossRef]
- Kyosei, Y.; Namba, M.; Yamura, S.; Takeuchi, R.; Aoki, N.; Nakaishi, K.; Watabe, S.; Ito, E. Proposal of de novo antigen test for COVID-19: Ultrasensitive detection of spike proteins of SARS-CoV-2. Diagnostics 2020, 10, 594. [Google Scholar] [CrossRef]
- Pray, I.W. Performance of an antigen-based test for asymptomatic and symptomatic SARS-CoV-2 testing at two university campuses—Wisconsin, September–October 2020. Morb. Mortal. Wkly. Rep. 2021, 69, 1642–1647. [Google Scholar] [CrossRef]
- Jayamohan, H.; Lambert, C.J.; Sant, H.J.; Jafek, A.; Patel, D.; Feng, H.; Beeman, M.; Mahmood, T.; Nze, U.; Gale, B.K. SARS-CoV-2 pandemic: A review of molecular diagnostic tools including sample collection and commercial response with associated advantages and limitations. Anal. Bioanal. Chem. 2021, 413, 49–71. [Google Scholar] [CrossRef]
- Surkova, E.; Nikolayevskyy, V.; Drobniewski, F. False-positive COVID-19 results: Hidden problems and costs. Lancet Respir. Med. 2020, 8, 1167–1168. [Google Scholar] [CrossRef]
- Dao Thi, V.L.; Herbst, K.; Boerner, K.; Meurer, M.; Kremer, L.P.; Kirrmaier, D.; Freistaedter, A.; Papagiannidis, D.; Galmozzi, C.; Stanifer, M.L.; et al. A colorimetric RT-LAMP assay and LAMP-sequencing for detecting SARS-CoV-2 RNA in clinical samples. Sci. Transl. Med. 2020, 12, eabc7075. [Google Scholar] [CrossRef]
- Alafeef, M.; Dighe, K.; Moitra, P.; Pan, D. Rapid, ultrasensitive, and quantitative detection of SARS-CoV-2 using antisense oligonucleotides directed electrochemical biosensor chip. ACS Nano 2020, 14, 17028–17045. [Google Scholar] [CrossRef]
- de Puig, H.; Lee, R.A.; Najjar, D.; Tan, X.; Soenksen, L.R.; Angenent-Mari, N.M.; Donghia, N.M.; Weckman, N.E.; Ory, A.; Ng, C.F.; et al. Minimally instrumented SHERLOCK (miSHERLOCK) for CRISPR-based point-of-care diagnosis of SARS-CoV-2 and emerging variants. Sci. Adv. 2021, 7, eabh2944. [Google Scholar] [CrossRef]
- Song, Z.; Ma, Y.; Chen, M.; Ambrosi, A.; Ding, C.; Luo, X. Electrochemical biosensor with enhanced antifouling capability for COVID-19 nucleic acid detection in complex biological media. Anal. Chem. 2021, 93, 5963–5971. [Google Scholar] [CrossRef]
- Seo, G.; Lee, G.; Kim, M.J.; Baek, S.H.; Choi, M.; Ku, K.B.; Lee, C.S.; Jun, S.; Park, D.; Kim, H.G.; et al. Rapid detection of COVID-19 causative virus (SARS-CoV-2) in human nasopharyngeal swab specimens using field-effect transistor-based biosensor. ACS Nano 2020, 14, 5135–5142. [Google Scholar] [CrossRef] [Green Version]
- Tian, J.; Liang, Z.; Hu, O.; He, Q.; Sun, D.; Chen, Z. An electrochemical dual-aptamer biosensor based on metal-organic frameworks MIL-53 decorated with Au@ Pt nanoparticles and enzymes for detection of COVID-19 nucleocapsid protein. Electrochim. Acta 2021, 387, 138553. [Google Scholar] [CrossRef]
- Büyüksünetçi, Y.T.; Çitil, B.E.; Anık, Ü. An impedimetric approach for COVID-19 detection. Analyst 2022, 147, 130–138. [Google Scholar] [CrossRef]
- Cady, N.C.; Tokranova, N.; Minor, A.; Nikvand, N.; Strle, K.; Lee, W.T.; Page, W.; Guignon, E.; Pilar, A.; Gibson, G.N. Multiplexed detection and quantification of human antibody response to COVID-19 infection using a plasmon enhanced biosensor platform. Biosens. Bioelectron. 2021, 171, 112679. [Google Scholar] [CrossRef]
- Kim, H.E.; Schuck, A.; Lee, S.H.; Lee, Y.; Kang, M.; Kim, Y.S. Sensitive electrochemical biosensor combined with isothermal amplification for point-of-care COVID-19 tests. Biosens. Bioelectron. 2021, 182, 113168. [Google Scholar] [CrossRef]
- Chan, H.P.; Hadjiiski, L.M.; Samala, R.K. Computer-aided diagnosis in the era of deep learning. Med. Phys. 2020, 47, e218–e227. [Google Scholar] [CrossRef] [PubMed]
- Kok, J.N.; Boers, E.J.; Kosters, W.A.; Van der Putten, P.; Poel, M. Artificial intelligence: Definition, trends, techniques, and cases. Artif. Intell. 2009, 1, 270–299. [Google Scholar]
- Zhou, L.; Pan, S.; Wang, J.; Vasilakos, A.V. Machine learning on big data: Opportunities and challenges. Neurocomputing 2017, 237, 350–361. [Google Scholar] [CrossRef] [Green Version]
- Ahuja, R.; Chug, A.; Gupta, S.; Ahuja, P.; Kohli, S. Classification and clustering algorithms of machine learning with their applications. In Nature-Inspired Computation in Data Mining and Machine Learning; Springer: Cham, Switzerland, 2020; pp. 225–248. [Google Scholar]
- Alzubi, J.; Nayyar, A.; Kumar, A. Machine learning from theory to algorithms: An overview. J. Phys. Conf. Ser. 2018, 1142, 012012. [Google Scholar] [CrossRef]
- Lillicrap, T.P.; Santoro, A. Backpropagation through time and the brain. Curr. Opin. Neurobiol. 2019, 55, 82–89. [Google Scholar] [CrossRef]
- Morales, E.F.; Escalante, H.J. A brief introduction to supervised, unsupervised, and reinforcement learning. In Biosignal Processing and Classification Using Computational Learning and Intelligence; Academic Press: Cambridge, MA, USA, 2022; pp. 111–129. [Google Scholar]
- Jiang, T.; Gradus, J.L.; Rosellini, A.J. Supervised machine learning: A brief primer. Behav. Ther. 2020, 51, 675–687. [Google Scholar] [CrossRef]
- Ying, X. An overview of overfitting and its solutions. J. Phys. Conf. Ser. 2019, 1168, 022022. [Google Scholar] [CrossRef]
- Fatima, M.; Pasha, M. Survey of machine learning algorithms for disease diagnostic. J. Intell. Learn. Syst. Appl. 2017, 9, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Jordan, M.I.; Mitchell, T.M. Machine learning: Trends, perspectives, and prospects. Science 2015, 349, 255–260. [Google Scholar] [CrossRef]
- Ding, C.; He, X. K-means clustering via principal component analysis. In Proceedings of the Twenty-First International Conference on Machine Learning, Banff, AB, Canada, 4 July 2004; p. 29. [Google Scholar]
- Eckhardt, C.M.; Madjarova, S.J.; Williams, R.J.; Ollivier, M.; Karlsson, J.; Pareek, A.; Nwachukwu, B.U. Unsupervised machine learning methods and emerging applications in healthcare. Knee Surg. Sports Traumatol. Arthrosc. 2022. [Google Scholar] [CrossRef]
- Saxe, A.; Nelli, S.; Summerfield, C. If deep learning is the answer, what is the question? Nat. Rev. Neurosci. 2021, 22, 55–67. [Google Scholar] [CrossRef]
- O’Mahony, N.; Campbell, S.; Carvalho, A.; Harapanahalli, S.; Hernandez, G.V.; Krpalkova, L.; Riordan, D.; Walsh, J. Deep learning vs. traditional computer vision. In Proceedings of the Science and Information Conference, Las Vegas, NV, USA, 25 April 2019; pp. 128–144. [Google Scholar]
- Pathak, A.R.; Pandey, M.; Rautaray, S. Application of deep learning for object detection. Procedia Comput. Sci. 2018, 132, 1706–1717. [Google Scholar] [CrossRef]
- Dhiyya, A.J. Architecture of IoMT in Healthcare. Internet Med. Things Healthc. Transform. 2022, 161–172. [Google Scholar] [CrossRef]
- Doi, K. Diagnostic imaging over the last 50 years: Research and development in medical imaging science and technology. Phys. Med. Biol. 2006, 51, R5. [Google Scholar] [CrossRef] [Green Version]
- Ndiaye, M.; Oyewobi, S.S.; Abu-Mahfouz, A.M.; Hancke, G.P.; Kurien, A.M.; Djouani, K. IoT in the wake of COVID-19: A survey on contributions, challenges and evolution. IEEE Access 2020, 8, 186821–186839. [Google Scholar] [CrossRef]
- Akhbarifar, S.; Javadi, H.H.; Rahmani, A.M.; Hosseinzadeh, M. A secure remote health monitoring model for early disease diagnosis in cloud-based IoT environment. Pers. Ubiquitous Comput. 2020, 16, 1–7. [Google Scholar] [CrossRef]
- Šimon, M.; Huraj, L.; Horák, T. DDoS reflection attack based on IoT: A case study. In Proceedings of the Computer Science On-line Conference, Poznań, Poland, 9–12 September 2018; pp. 44–52. [Google Scholar]
- Huraj, L.; Horak, T.; Strelec, P.; Tanuska, P. Mitigation against DDoS Attacks on an IoT-Based Production Line Using Machine Learning. Appl. Sci. 2021, 11, 1847. [Google Scholar] [CrossRef]
- Horak, T.; Strelec, P.; Huraj, L.; Tanuska, P.; Vaclavova, A.; Kebisek, M. The vulnerability of the production line using industrial IoT systems under ddos attack. Electronics 2021, 10, 381. [Google Scholar] [CrossRef]
- Mujawar, M.A.; Gohel, H.; Bhardwaj, S.K.; Srinivasan, S.; Hickman, N.; Kaushik, A. Nano-enabled biosensing systems for intelligent healthcare: Towards COVID-19 management. Mater. Today Chem. 2020, 17, 100306. [Google Scholar] [CrossRef]
- Fujita, H. AI-based computer-aided diagnosis (AI-CAD): The latest review to read first. Radiol. Phys. Technol. 2020, 13, 6–19. [Google Scholar] [CrossRef]
- Bhardwaj, V.; Joshi, R.; Gaur, A.M. IoT-based smart health monitoring system for COVID-19. SN Comput. Sci. 2022, 3, 137. [Google Scholar] [CrossRef] [PubMed]
- Iglehart, J.K. The new era of medical imaging—Progress and pitfalls. N. Engl. J. Med. 2006, 354, 2822–2828. [Google Scholar] [CrossRef] [PubMed]
- Syed, H.H.; Khan, M.A.; Tariq, U.; Armghan, A.; Alenezi, F.; Khan, J.A.; Rho, S.; Kadry, S.; Rajinikanth, V. A rapid artificial intelligence-based computer-aided diagnosis system for COVID-19 classification from CT images. Behav. Neurol. 2021, 2021, 2560388. [Google Scholar] [CrossRef] [PubMed]
- Raoof, S.; Feigin, D.; Sung, A.; Raoof, S.; Irugulpati, L.; Rosenow, E.C., III. Interpretation of plain chest roentgenogram. Chest 2012, 141, 545–558. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Çallı, E.; Sogancioglu, E.; van Ginneken, B.; van Leeuwen, K.G.; Murphy, K. Deep learning for chest X-ray analysis: A survey. Med. Image Anal. 2021, 72, 102125. [Google Scholar] [CrossRef] [PubMed]
- Hayden, G.E.; Wrenn, K.W. Chest radiograph vs. computed tomography scan in the evaluation for pneumonia. J. Emerg. Med. 2009, 36, 266–270. [Google Scholar] [CrossRef]
- Cohen, J.P.; Morrison, P.; Dao, L.; Roth, K.; Duong, T.Q.; Ghassemi, M. COVID-19 image data collection: Prospective predictions are the future. arXiv 2020, arXiv:2006.11988. Available online: https://github.com/ieee8023/covid-chestxray-dataset (accessed on 7 December 2022).
- Wang, L.L.; Lo, K.; Chandrasekhar, Y.; Reas, R.; Yang, J.; Eide, D.; Funk, K.; Kinney, R.; Liu, Z.; Merrill, W.; et al. Cord-19: The COVID-19 open research dataset. arXiv 2020. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7251955/ (accessed on 7 December 2022).
- Luz, E.; Silva, P.; Silva, R.; Silva, L.; Guimarães, J.; Miozzo, G.; Moreira, G.; Menotti, D. Towards an effective and efficient deep learning model for COVID-19 patterns detection in X-ray images. Res. Biomed. Eng. 2021, 38, 149–162. [Google Scholar] [CrossRef]
- Soares, E.; Angelov, P.; Biaso, S.; Froes, M.H.; Abe, D. SARS-CoV-2 CT-scan dataset: A large dataset of real patients CT scans for SARS-CoV-2 identification. medRxiv 2020, 1–8. [Google Scholar] [CrossRef]
- Kermany, D.S.; Goldbaum, M.; Cai, W.; Valentim, C.C.; Liang, H.; Baxter, S.L.; McKeown, A.; Yang, G.; Wu, X.; Yan, F.; et al. Identifying medical diagnoses and treatable diseases by image-based deep learning. Cell 2018, 172, 1122–1131. [Google Scholar] [CrossRef]
- Wang, X.; Peng, Y.; Lu, L.; Lu, Z.; Bagheri, M.; Summers, R.M. Chestx-ray8: Hospital-scale chest x-ray database and benchmarks on weakly-supervised classification and localization of common thorax diseases. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, Honolulu, HI, USA, 21–26 July 2017. [Google Scholar]
- Gayathri, J.L.; Abraham, B.; Sujarani, M.S.; Nair, M.S. A computer-aided diagnosis system for the classification of COVID-19 and non-COVID-19 pneumonia on chest X-ray images by integrating CNN with sparse autoencoder and feed forward neural network. Comput. Biol. Med. 2022, 141, 105134. [Google Scholar]
- Nayak, S.R.; Nayak, D.R.; Sinha, U.; Arora, V.; Pachori, R.B. Application of deep learning techniques for detection of COVID-19 cases using chest X-ray images: A comprehensive study. Biomed. Signal Process. Control 2021, 64, 102365. [Google Scholar] [CrossRef]
- Narin, A.; Kaya, C.; Pamuk, Z. Automatic detection of coronavirus disease (covid-19) using x-ray images and deep convolutional neural networks. Pattern Anal. Appl. 2021, 24, 1207–1220. [Google Scholar] [CrossRef]
- Naseer, A.; Tamoor, M.; Azhar, A. Computer-aided COVID-19 diagnosis and a comparison of deep learners using augmented CXRs. J. X-ray Sci. Technol. 2022, 1–21. [Google Scholar] [CrossRef]
- Aziz, S.; Khan, M.U.; Rehman, A.; Tariq, Z.; Iqtidar, K. Computer-aided diagnosis of COVID-19 disease from chest x-ray images integrating deep feature extraction. Expert Syst. 2022, 39, e12919. [Google Scholar] [CrossRef]
- Ouchicha, C.; Ammor, O.; Meknassi, M. CVDNet: A novel deep learning architecture for detection of coronavirus (COVID-19) from chest x-ray images. Chaos Solitons Fractals 2020, 140, 10245. [Google Scholar] [CrossRef]
- Shorfuzzaman, M.; Masud, M.; Alhumyani, H.; Anand, D.; Singh, A. Artificial neural network-based deep learning model for COVID-19 patient detection using X-ray chest images. J. Healthc. Eng. 2021, 2021, 5513679. [Google Scholar] [CrossRef]
- Li, H.; Zeng, N.; Wu, P.; Clawson, K. Cov-Net: A computer-aided diagnosis method for recognizing COVID-19 from chest X-ray images via machine vision. Expert Syst. Appl. 2022, 207, 118029. [Google Scholar] [CrossRef]
- Hira, S.; Bai, A.; Hira, S. An automatic approach based on CNN architecture to detect COVID-19 disease from chest X-ray images. Appl. Intell. 2021, 51, 2864–2889. [Google Scholar] [CrossRef]
- Chaddad, A.; Hassan, L.; Desrosiers, C. Deep CNN models for predicting COVID-19 in CT and x-ray images. J. Med. Imaging 2021, 8, 014502. [Google Scholar] [CrossRef]
- Mishra, N.K.; Singh, P.; Joshi, S.D. Automated detection of COVID-19 from CT scan using convolutional neural network. Biocybern. Biomed. Eng. 2021, 41, 572–588. [Google Scholar] [CrossRef] [PubMed]
- Katar, O.; Duman, E. Deep Learning Based COVID-19 Detection with A Novel CT Images Dataset: EFSCH-19. Avrupa Bilim ve Teknoloji Dergisi 2021, 29, 150–155. [Google Scholar] [CrossRef]
- Kogilavani, S.V.; Prabhu, J.; Sandhiya, R.; Kumar, M.S.; Subramaniam, U.; Karthick, A.; Muhibbullah, M.; Imam, S.B. COVID-19 detection based on lung CT scan using deep learning techniques. Comput. Math. Methods Med. 2022, 2022, 7672196. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Jiang, W.; Qiu, X. Deep learning for COVID-19 detection based on CT images. Sci. Rep. 2021, 11, 14353. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.; Chung, H.; Kang, W.S.; Kim, K.W.; Shin, Y.; Kang, S.J.; Lee, J.H.; Kim, Y.J.; Kim, N.Y.; Jung, H.; et al. COVID-19 pneumonia diagnosis using a simple 2D deep learning framework with a single chest CT image: Model development and validation. J. Med. Internet Res. 2020, 22, e19569. [Google Scholar] [CrossRef]
- Iskanderani, A.I.; Mehedi, I.M.; Aljohani, A.J.; Shorfuzzaman, M.; Akther, F.; Palaniswamy, T.; Latif, S.A.; Latif, A.; Alam, A. Artificial intelligence and medical internet of things framework for diagnosis of coronavirus suspected cases. J. Healthc. Eng. 2021, 2021, 3277988. [Google Scholar] [CrossRef]
- Kini, A.S.; Gopal Reddy, A.N.; Kaur, M.; Satheesh, S.; Singh, J.; Martinetz, T.; Alshazly, H. Ensemble deep learning and internet of things-based automated COVID-19 diagnosis framework. Contrast Med. Mol. Imaging 2022, 2022, 7377502. [Google Scholar] [CrossRef]
- Le, D.N.; Parvathy, V.S.; Gupta, D.; Khanna, A.; Rodrigues, J.J.; Shankar, K. IoT enabled depthwise separable convolution neural network with deep support vector machine for COVID-19 diagnosis and classification. Int. J. Mach. Learn. Cybern. 2021, 12, 3235–3248. [Google Scholar] [CrossRef]
- Ahmed, I.; Ahmad, A.; Jeon, G. An IoT-based deep learning framework for early assessment of COVID-19. IEEE Internet Things J. 2020, 8, 15855–15862. [Google Scholar] [CrossRef]
- Rehman, A.; Sadad, T.; Saba, T.; Hussain, A.; Tariq, U. Real-time diagnosis system of COVID-19 using X-ray images and deep learning. Professional 2021, 23, 57–62. [Google Scholar] [CrossRef]
- Punitha, S.; Al-Turjman, F.; Stephan, T. A novel e-healthcare diagnosing system for COVID-19 via whale optimization algorithm. J. Exp. Theor. Artif. Intell. 2022. [Google Scholar] [CrossRef]
- Alzubaidi, L.; Fadhel, M.A.; Al-Shamma, O.; Zhang, J.; Santamaría, J.; Duan, Y.; Oleiwi, R.S. Towards a better understanding of transfer learning for medical imaging: A case study. Appl. Sci. 2020, 10, 4523. [Google Scholar] [CrossRef]
- Kora, P.; Ooi, C.P.; Faust, O.; Raghavendra, U.; Gudigar, A.; Chan, W.Y.; Meenakshi, K.; Swaraja, K.; Plawiak, P.; Acharya, U.R. Transfer learning techniques for medical image analysis: A review. Biocybern. Biomed. Eng. 2021, 42, 79–107. [Google Scholar] [CrossRef]
- Gavrilov, A.D.; Jordache, A.; Vasdani, M.; Deng, J. Preventing model overfitting and underfitting in convolutional neural networks. Int. J. Softw. Sci. Comput. Intell. 2018, 10, 19–28. [Google Scholar] [CrossRef]
- Chlap, P.; Min, H.; Vandenberg, N.; Dowling, J.; Holloway, L.; Haworth, A. A review of medical image data augmentation techniques for deep learning applications. J. Med. Imaging Radiol. Oncol. 2021, 65, 545–563. [Google Scholar] [CrossRef]
- Milyaev, S.; Laptev, I. Towards reliable object detection in noisy images. Pattern Recognit. Image Anal. 2017, 27, 713–722. [Google Scholar] [CrossRef]
- Dong, X.; Yu, Z.; Cao, W.; Shi, Y.; Ma, Q. A survey on ensemble learning. Front. Comput. Sci. 2020, 14, 241–258. [Google Scholar] [CrossRef]
- Shui-Hua, W.; Khan, M.A.; Govindaraj, V.; Fernandes, S.L.; Zhu, Z.; Yu-Dong, Z. Deep rank-based average pooling network for COVID-19 recognition. Comput. Mater. Contin. 2022, 2797–2813. [Google Scholar] [CrossRef]
- Ibrahim, A.U.; Al-Turjman, F.; Sa’id, Z.; Ozsoz, M. Futuristic CRISPR-based biosensing in the cloud and internet of things era: An overview. Multimed. Tools Appl. 2022, 81, 35143–35171. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Irkham, I.; Ibrahim, A.U.; Nwekwo, C.W.; Al-Turjman, F.; Hartati, Y.W. Current Technologies for Detection of COVID-19: Biosensors, Artificial Intelligence and Internet of Medical Things (IoMT): Review. Sensors 2023, 23, 426. https://doi.org/10.3390/s23010426
Irkham I, Ibrahim AU, Nwekwo CW, Al-Turjman F, Hartati YW. Current Technologies for Detection of COVID-19: Biosensors, Artificial Intelligence and Internet of Medical Things (IoMT): Review. Sensors. 2023; 23(1):426. https://doi.org/10.3390/s23010426
Chicago/Turabian StyleIrkham, Irkham, Abdullahi Umar Ibrahim, Chidi Wilson Nwekwo, Fadi Al-Turjman, and Yeni Wahyuni Hartati. 2023. "Current Technologies for Detection of COVID-19: Biosensors, Artificial Intelligence and Internet of Medical Things (IoMT): Review" Sensors 23, no. 1: 426. https://doi.org/10.3390/s23010426







