Inertial-Sensor-Based Monitoring of Sample Entropy and Peak Frequency Changes in Treadmill Walking during Recovery after Total Knee Arthroplasty
Abstract
:1. Introduction
1.1. Using IMUs in Gait Rehabilitation Following TKA
1.2. Using Non-Linear Movement Parameters in Gait Rehabilitation Following TKA
1.3. Sample Entropy (SEn) and Peak Frequency in Power Spectrum Density (PSD) Analysis
1.4. Aims of Present Study
1.5. Empirical Background of the Hypotheses of the Present Study
1.6. Aims and Predictions
1.7. Main Contributions of the Present Study
- Comparison of clinical gait performance scores obtained from standardized tests to kinematic and dynamic features of treadmill walking during rehabilitation after TKA.
- Identification of a typical “bad habit” adopted in response to knee osteoarthritis and maintained following TKA.
- Evaluation of the use of IMUs and sophisticated dynamic gait analyses as a potential means to enrich the diagnostic and monitoring activities by physical therapists treating TKA patients.
2. Materials and Methods
2.1. Participants
2.2. Clinical Tests
2.3. Experimental Tasks
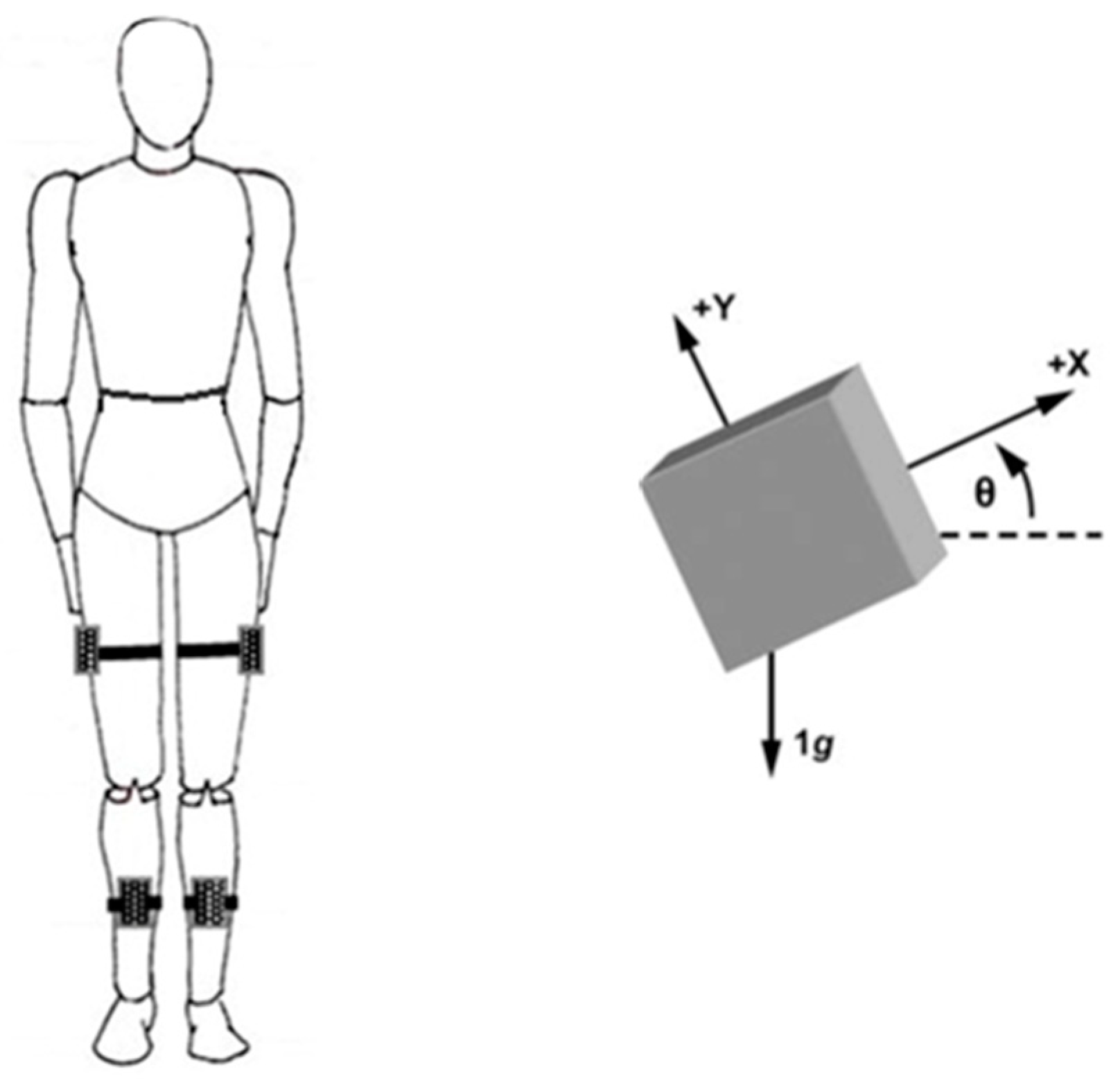
2.4. Recording System
2.5. Data Analysis
2.5.1. Data Preprocessing
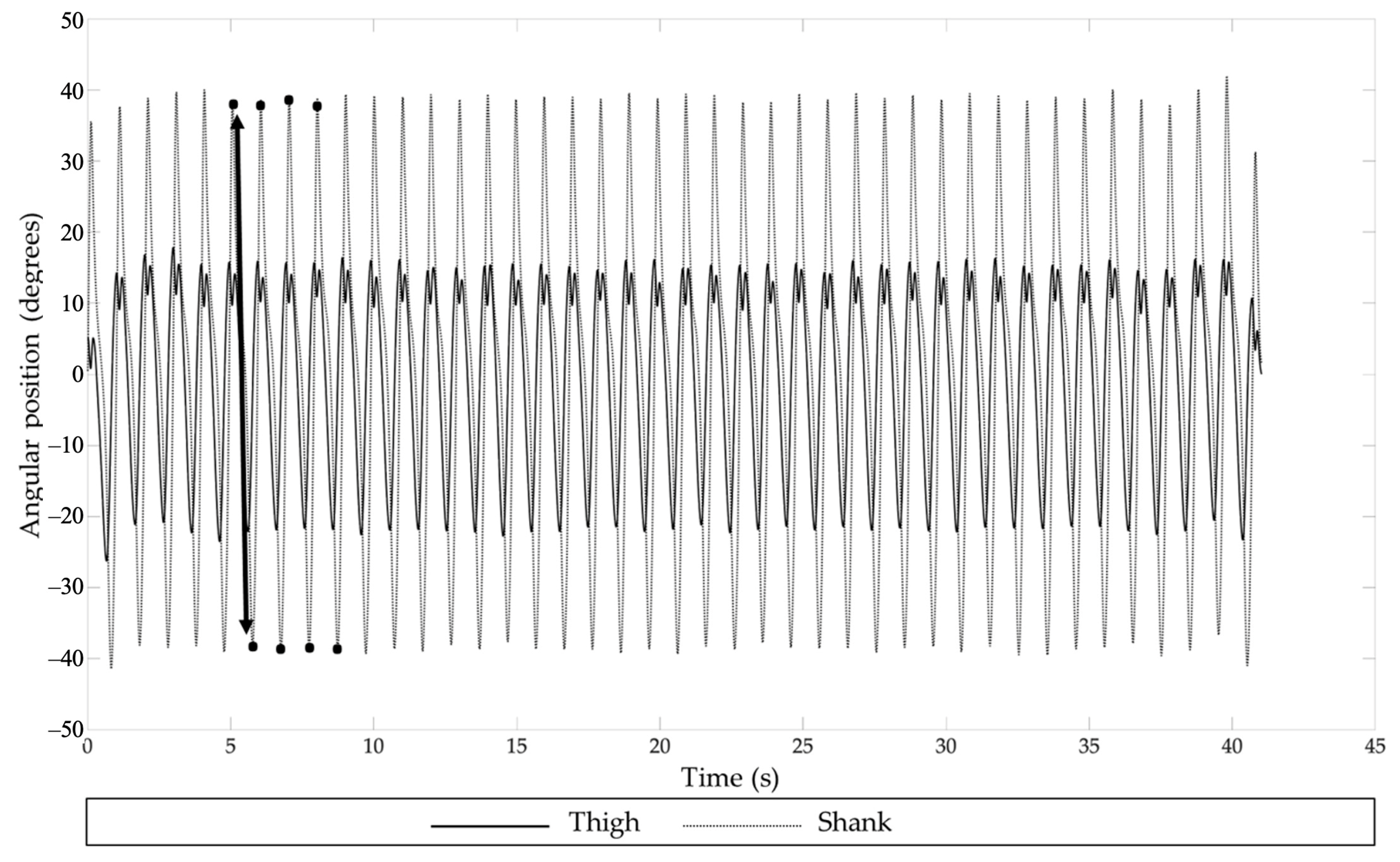
2.5.2. Kinematics
2.6. Statistical Analysis
3. Results
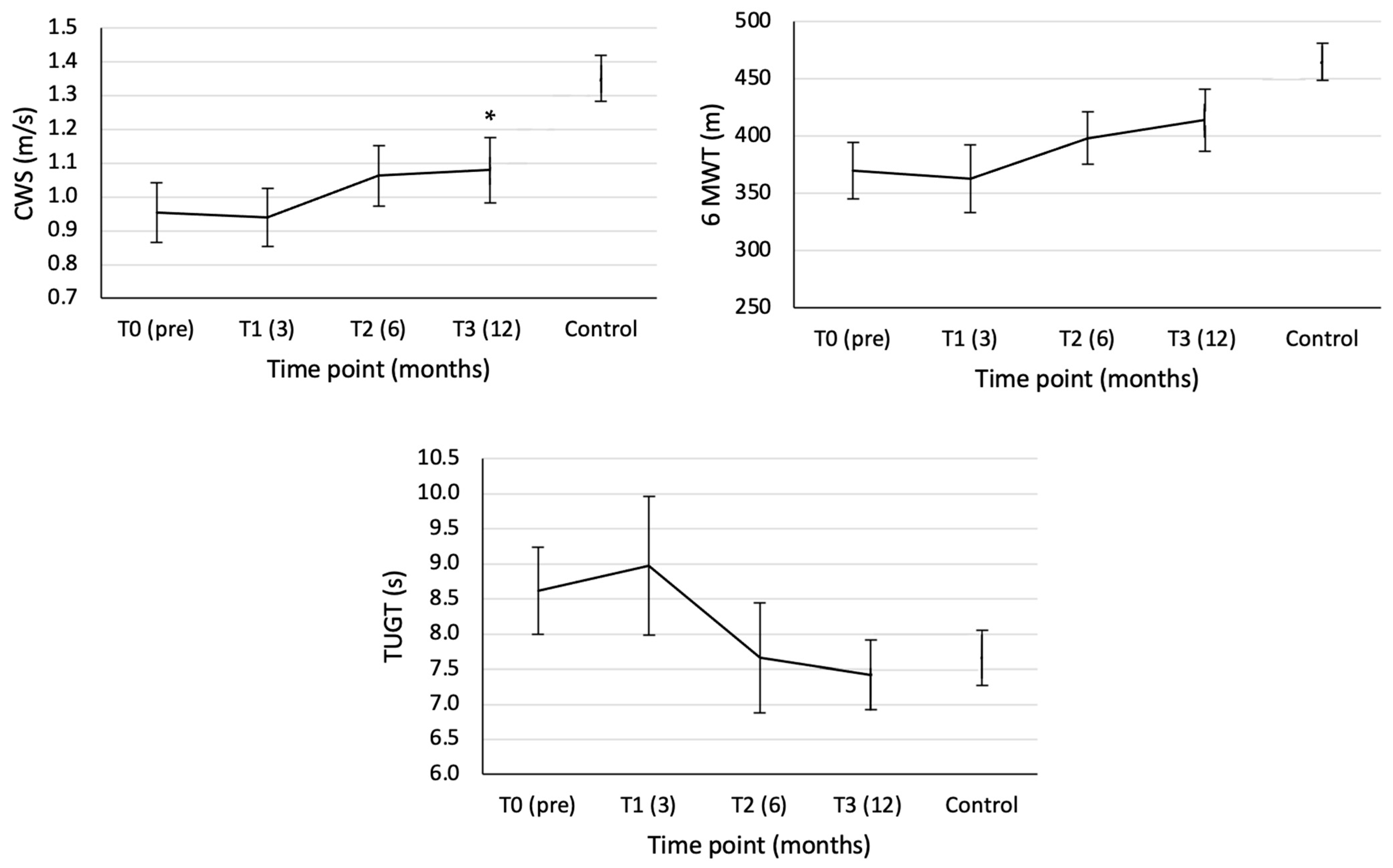
3.1. Clinical Tests
3.2. Treadmill Walking
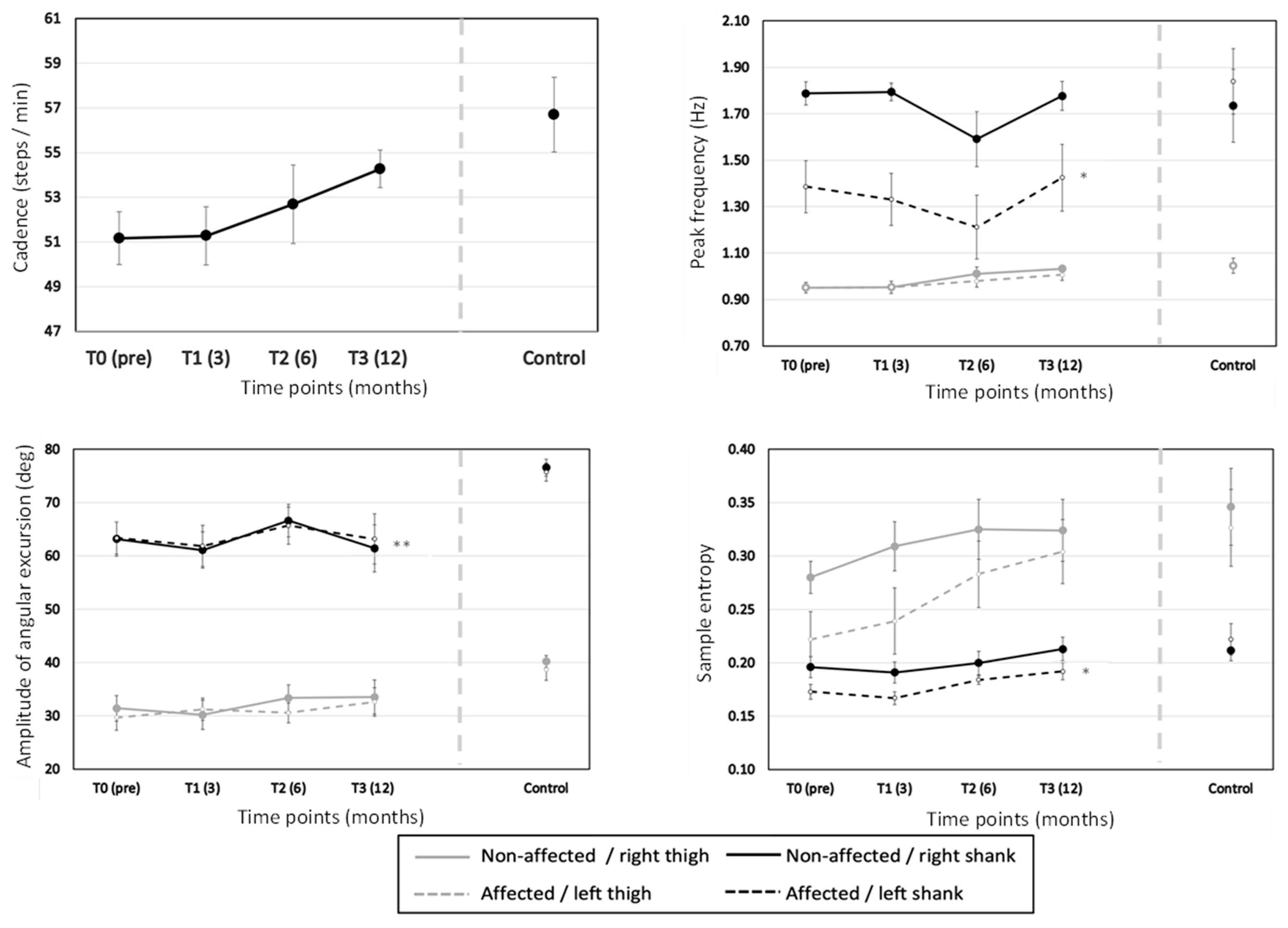
3.2.1. Effect of Time
Effect of TKA
Effect of Segment
TKA vs. Control Group
4. Discussion
4.1. Recovery after TKA
4.2. Health and Clinical Implications
4.3. Limitations and Further Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Glossary
| TKA | total knee arthroplasty |
| KO | knee osteoarthritis |
| SEn | sample entropy |
| PSD | power spectral density |
| 6 MWT | 6 minute walking test |
| TUGT | timed up-and-go test |
| CWS | comfortable walking speed |
References
- Zeni, J.A.; Flowers, P.; Bade, M.; Cheuy, V.; Stevens-Lapsley, J.; Snyder-Mackler, L. Stiff Knee Gait May Increase Risk of Second Total Knee Arthroplasty. J. Orthop. Res. 2019, 37, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Fallah-Yakhdani, H.R.; Abbasi-Bafghi, H.; Meijer, O.G.; Bruijn, S.M.; Van Den Dikkenberg, N.; Benedetti, M.-G.; Van Dieën, J.H. Determinants of Co-Contraction during Walking before and after Arthroplasty for Knee Osteoarthritis. Clin. Biomech. 2012, 27, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Fallah Yakhdani, H.R.; Bafghi, H.A.; Meijer, O.G.; Bruijn, S.M.; van den Dikkenberg, N.; Stibbe, A.B.; van Royen, B.J.; van Dieën, J.H. Stability and Variability of Knee Kinematics during Gait in Knee Osteoarthritis before and after Replacement Surgery. Clin. Biomech. 2010, 25, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.J.; Lloyd, D.G.; Wood, D.J. A Kinematic and Kinetic Analysis of Walking after Total Knee Arthroplasty with and without Patellar Resurfacing. Clin. Biomech. 2006, 21, 379–386. [Google Scholar] [CrossRef]
- Roggio, F.; Bianco, A.; Palma, A.; Ravalli, S.; Maugeri, G.; di Rosa, M.; Musumeci, G. Technological Advancements in the Analysis of Human Motion and Posture Management through Digital Devices. World J. Orthop. 2021, 12, 467–484. [Google Scholar] [CrossRef]
- Staab, W.; Hottowitz, R.; Sohns, C.; Sohns, J.M.; Gilbert, F.; Menke, J.; Lotz, J. Accelerometer and Gyroscope Based Gait Analysis Using Spectral Analysis of Patients with Osteoarthritis of the Knee. J. Phys. Ther. Sci. 2014, 26, 997–1002. [Google Scholar] [CrossRef]
- Emmerzaal, J.; Corten, K.; van der Straaten, R.; de Baets, L.; van Rossom, S.; Timmermans, A.; Jonkers, I.; Vanwanseele, B. Movement Quality Parameters During Gait Assessed by a Single Accelerometer in Subjects with Osteoarthritis and Following Total Joint Arthroplasty. Sensors 2022, 22, 2955. [Google Scholar] [CrossRef]
- Harbourne, R.T.; Stergiou, N. Movement Variability and the Use of Nonlinear Tools: Principles to Guide Physical Therapist Practice. Phys. Ther. 2009, 89, 267–282. [Google Scholar] [CrossRef]
- Hadamus, A.; Błażkiewicz, M.; Kowalska, A.J.; Wydra, K.T.; Grabowicz, M.; Łukowicz, M.; Białoszewski, D.; Marczyński, W. Nonlinear and Linear Measures in the Differentiation of Postural Control in Patients after Total Hip or Knee Replacement and Healthy Controls. Diagnostics 2022, 12, 1595. [Google Scholar] [CrossRef]
- Hamill, J.; Palmer, C.; Van Emmerik, R.E.A. Coordinative Variability and Overuse Injury. Sport. Med. Arthrosc. Rehabil. Ther. Technol. 2012, 4, 45. [Google Scholar] [CrossRef]
- Bosga, J.; Hullegie, W.; van Cingel, R.; Meulenbroek, R.G.J. Solution Space: Monitoring the Dynamics of Motor Rehabilitation. Physiother. Theory Pract. 2018, 35, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Richman, J.S.; Moorman, J.R. Physiological Time-Series Analysis Using Approximate Entropy and Sample Entropy. Am. J. Physiol. Heart Circ. Physiol. 2000, 278, H2039–H2049. [Google Scholar] [CrossRef] [PubMed]
- Wagenaar, R.C.; van Emmerik, R.E.A. Resonant Frequencies of Arms and Legs Identify Different Walking Patterns. J. Biomech. 2000, 33, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Lamoth, C.J.C.; Meijer, O.G.; Wuisman, P.I.J.M.; van Dieën, J.H.; Levin, M.F.; Beek, P.J. Pelvis-Thorax Coordination in the Transverse Plane During Walking in Persons with Nonspecific Low Back Pain. Spine 2002, 27, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Al-Amri, M.; Nicholas, K.; Button, K.; Sparkes, V.; Sheeran, L.; Davies, J.L. Inertial Measurement Units for Clinical Movement Analysis: Reliability and Concurrent Validity. Sensors 2018, 18, 719. [Google Scholar] [CrossRef]
- Zhang, J.T.; Novak, A.C.; Brouwer, B.; Li, Q. Concurrent Validation of Xsens MVN Measurement of Lower Limb Joint Angular Kinematics. Physiol. Meas. 2013, 34, 63–69. [Google Scholar] [CrossRef]
- Astephen, J.L.; Deluzio, K.J.; Caldwell, G.E.; Dunbar, M.J. Biomechanical Changes at the Hip, Knee, and Ankle Joints during Gait Are Associated with Knee Osteoarthritis Severity. J. Orthop. Res. 2008, 26, 332–341. [Google Scholar] [CrossRef]
- Dixon, S.J.; Hinman, R.S.; Creaby, M.W.; Kemp, G.; Crossley, K.M. Knee Joint Stiffness during Walking in Knee Osteoarthritis. Arthritis Care Res. 2010, 62, 38–44. [Google Scholar] [CrossRef]
- Zeni, J.A.; Higginson, J.S. Differences in Gait Parameters between Healthy Subjects and Persons with Moderate and Severe Knee Osteoarthritis: A Result of Altered Walking Speed? Clin. Biomech. 2009, 24, 372–378. [Google Scholar] [CrossRef]
- Roelofsen, E.G.J.; van Cingel, R.; Pronk, Y.; Staal, J.B.; Nijhuis-van der Sanden, M.W.G.; Meulenbroek, R.G.J. Leg-Amplitude Differentiation Guided by Haptic and Visual Feedback to Detect Alterations in Motor Flexibility Due to Total Knee Replacement. Hum. Mov. Sci. 2020, 71, 102623. [Google Scholar] [CrossRef]
- Bade, M.J.; Kohrt, W.M.; Stevens-Lapsley, J.E. Outcomes Before and After Total Knee Arthroplasty Compared to Healthy Adults. J. Orthop. Sport. Phys. Ther. 2010, 40, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Ko, V.; Naylor, J.M.; Harris, I.A.; Crosbie, J.; Yeo, A. The Six-Minute Walk Test Is an Excellent Predictor of Functional Ambulation after Total Knee Arthroplasty. BMC Musculoskelet. Disord. 2013, 14, 145. [Google Scholar] [CrossRef] [PubMed]
- Tochigi, Y.; Segal, N.A.; Vaseenon, T.; Brown, T.D. Entropy Analysis of Tri-Axial Leg Acceleration Signal Waveforms for Measurement of Decrease of Physiological Variability in Human Gait. J. Orthop. Res. 2012, 30, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Lingard, E.A.; Sledge, C.B.; Learmonth, I.D. Patient Expectations Regarding Total Knee Arthroplasty: Differences Among the United States, United Kingdom, and Australia. J. Bone Jt. Surg. 2006, 88, 1201–1207. [Google Scholar]
- Lange, T.; Schmitt, J.; Kopkow, C.; Rataj, E.; Günther, K.P.; Lützner, J. What Do Patients Expect from Total Knee Arthroplasty? A Delphi Consensus Study on Patient Treatment Goals. J. Arthroplast. 2017, 32, 2093–2099. [Google Scholar] [CrossRef]
- de Groot, I.B.; Bussmann, H.J.; Stam, H.J.; Verhaar, J.A. Small Increase of Actual Physical Activity 6 Months after Total Hip or Knee Arthroplasty. Clin. Orthop. Relat. Res. 2008, 466, 2201–2208. [Google Scholar] [CrossRef]
- Berghmans, D.D.P.; Lenssen, A.F.; Emans, P.J.; de Bie, R.A. Functions, Disabilities and Perceived Health in the First Year after Total Knee Arthroplasty; a Prospective Cohort Study. BMC Musculoskelet. Disord. 2018, 19, 250. [Google Scholar] [CrossRef]
- Bourne, R.B.; Chesworth, B.M.; Davis, A.M.; Mahomed, N.N.; Charron, K.D.J. Patient Satisfaction after Total Knee Arthroplasty: Who Is Satisfied and Who Is Not? Clin. Orthop. Relat. Res. 2010, 468, 57–63. [Google Scholar] [CrossRef]
- Lim, H.A.; Song, E.K.; Seon, J.K.; Park, K.S.; Shin, Y.J.; Yang, H.Y. Causes of Aseptic Persistent Pain after Total Knee Arthroplasty. CiOS Clin. Orthop. Surg. 2017, 9, 50–56. [Google Scholar] [CrossRef]
- Dalury, D.F.; Pomeroy, D.L.; Gorab, R.S.; Adams, M.J. Why Are Total Knee Arthroplasties Being Revised? J. Arthroplast. 2013, 28, 120–121. [Google Scholar] [CrossRef]
- Walter, C.B.; Swinnen, S.P. The Formation and Dissolution of “Bad Habits” during the Acquisition of Coordination Skills. In Interlimb Coordination: Neural, Dynamical and, Cognitive Constraints; Academic Press: Cambridge, MA, USA, 1994; pp. 491–513. ISBN 0126792704. [Google Scholar]
- Benedetti, M.G.; Catani, F.; Bilotta, T.W.; Marcacci, M.; Mariani, E.; Giannini, S. Muscle Activation Pattern and Gait Biomechanics after Total Knee Replacement. Clin. Biomech. 2003, 18, 871–876. [Google Scholar] [CrossRef] [PubMed]
- Stergiou, N.; Harbourne, R.T.; Cavanaugh, J.T. Optimal Movement Variability: A New Theoretical Perspective for Neurologic Physical Therapy. J. Neurol. Phys. Ther. 2006, 30, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Dounskaia, N. The Internal Model and the Leading Joint Hypothesis: Implications for Control of Multi-Joint Movements. Exp. Brain Res. 2005, 166, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Lakens, D. Calculating and Reporting Effect Sizes to Facilitate Cumulative Science: A Practical Primer for t-Tests and ANOVAs. Front. Psychol. 2013, 4, 863. [Google Scholar] [CrossRef] [PubMed]
- Dobson, F.; Hinman, R.S.; Hall, M.; Terwee, C.B.; Roos, E.M.; Bennell, K.L. Measurement Properties of Performance-Based Measures to Assess Physical Function in Hip and Knee Osteoarthritis: A Systematic Review. Osteoarthr. Cartil. 2012, 20, 1548–1562. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.M.; Stratford, P.W.; Wessel, J.; Gollish, J.D.; Penney, D. Assessing Stability and Change of Four Performance Measures: A Longitudinal Study Evaluating Outcome Following Total Hip and Knee Arthroplasty. BMC Musculoskelet. Disord. 2005, 6, 3. [Google Scholar] [CrossRef] [PubMed]
- Parent, E.; Moffet, H. Comparative Responsiveness of Locomotor Tests and Questionnaires Used to Follow-Early Recovery after Total Knee Arthroplasty. Arch. Phys. Med. Rehabil. 2002, 83, 70–80. [Google Scholar] [CrossRef]
- Filimon, F. Are All Spatial Reference Frames Egocentric? Reinterpreting Evidence for Allocentric, Object-Centered, or World-Centered Reference Frames. Front. Hum. Neurosci. 2015, 9, 648. [Google Scholar] [CrossRef]
- van de Ven, W.A.F.; Bosga, J.; Hullegie, W.; Verra, W.C.; Meulenbroek, R.G.J. Increased Speed Elicited More Automatized but Less Predictable Control in Cyclical Arm and Leg Movements. Motor Control 2022, 26, 15–35. [Google Scholar] [CrossRef]
- Fisher, C.J. Using an Accelerometer for Inclination Sensing. AN-1057, Application Note, Analog Devices. 2010, pp. 1–8. Available online: https://www.analog.com/en/app-notes/an-1057.html (accessed on 14 April 2023).
- van Galen, G.P.; van Doorn, R.R.A.; Schomaker, L.R.B. Effects of Motor Programming on the Power Spectral Density Function of Finger and Wrist Movements. J. Exp. Psychol. Hum. Percept. Perform. 1990, 16, 755–765. [Google Scholar] [CrossRef]
- Hanlon, M.; Anderson, R. Real-Time Gait Event Detection Using Wearable Sensors. Gait Posture 2009, 30, 523–527. [Google Scholar] [CrossRef] [PubMed]
- Rácz, K.; Kiss, R.M. Marker Displacement Data Filtering in Gait Analysis: A Technical Note. Biomed. Signal Process. Control. 2021, 70, 102974. [Google Scholar] [CrossRef]
- Wang, Z.; Ji, R. Estimate Spatial-Temporal Parameters of Human Gait Using Inertial Sensors. In Proceedings of the 2015 IEEE International Conference on Cyber Technology in Automation, Control and Intelligent Systems, IEEE-CYBER 2015, Shenyang, China, 8–12 June 2015; pp. 1883–1888. [Google Scholar] [CrossRef]
- Roelofsen, E.G.J.; Brown, D.D.; Nijhuis-van der Sanden, M.W.G.; Staal, J.B.; Meulenbroek, R.G.J. Does Motor Expertise Facilitate Amplitude Differentiation of Lower Limb-Movements in an Asymmetrical Bipedal Coordination Task? Hum. Mov. Sci. 2018, 59, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Yentes, J.M.; Hunt, N.; Schmid, K.K.; Kaipust, J.P.; McGrath, D.; Stergiou, N. The Appropriate Use of Approximate Entropy and Sample Entropy with Short Data Sets. Ann. Biomed. Eng. 2013, 41, 349–365. [Google Scholar] [CrossRef]
- Royal Dutch Society for Physical Therapy (KNGF). KNGF-Richtlijn Artrose Heup-Knie; Royal Dutch Society for Physical Therapy: Amersfoort, The Netherlands, 2018. [Google Scholar]
- Bertram, J.E.A.; Ruina, A. Multiple Walking Speed-Frequency Relations Are Predicted by Constrained Optimization. J. Theor. Biol. 2001, 209, 445–453. [Google Scholar] [CrossRef]
- Levinger, P.; Lai, D.T.; Begg, R.; Menz, H.; Feller, J.; Webster, K. Fourier Analysis of Tibia Acceleration in Subjects with Knee Osteoarthritis: Preliminary Results. In Proceedings of the 2008 International Conference on Intelligent Sensors, Sensor Networks and Information Processing, Sydney, Australia, 15–18 December 2008; IEEE: Piscataway, NJ, USA, 2008; pp. 315–320. [Google Scholar]
- Acasio, J.C.; Butowicz, C.M.; Golyski, P.R.; Nussbaum, M.A.; Hendershot, B.D. Associations between Trunk Postural Control in Walking and Unstable Sitting at Various Levels of Task Demand. J. Biomech. 2018, 75, 181–185. [Google Scholar] [CrossRef]
- Georgoulis, A.D.; Moraiti, C.O.; Ristanis, S.; Stergiou, N. A Novel Approach to Measure Variability in the Anterior Cruciate Ligament Deficient Knee during Walking: The Use of the Approximate Entropy in Orthopaedics. J. Clin. Monit. Comput. 2006, 20, 11–18. [Google Scholar] [CrossRef]
- Breugem, S.J.M.; Haverkamp, D. Anterior Knee Pain after a Total Knee Arthroplasty: What Can Cause This Pain? World J. Orthop. 2014, 5, 163–170. [Google Scholar] [CrossRef]
- Smith, A.J.; Lloyd, D.G.; Wood, D.J. Pre-Surgery Knee Joint Loading Patterns during Walking Predict the Presence and Severity of Anterior Knee Pain after Total Knee Arthroplasty. J. Orthop. Res. 2004, 22, 260–266. [Google Scholar] [CrossRef]
- Bonacci, J.; Fox, A.; Hall, M.; Fuller, J.T.; Vicenzino, B. Effect of Gait Retraining on Segment Coordination and Joint Variability in Individuals with Patellofemoral Pain. Clin. Biomech. 2020, 80, 105179. [Google Scholar] [CrossRef]
- Mentiplay, B.F.; Banky, M.; Clark, R.A.; Kahn, M.B.; Williams, G. The Effect of Walking Speed on Lower Limb Angular Velocity. Arch. Phys. Med. Rehabil. 2018, 65, 190–196. [Google Scholar] [CrossRef]
- Grimmer, M.; Schmidt, K.; Duarte, J.E.; Neuner, L.; Koginov, G.; Riener, R. Stance and Swing Detection Based on the Angular Velocity of Lower Limb Segments during Walking. Front. Neurorobotics 2019, 13, 57. [Google Scholar] [CrossRef] [PubMed]
- Wurdeman, S.R.; Huben, N.B.; Stergiou, N. Variability of Gait Is Dependent on Direction of Progression: Implications for Active Control. J. Biomech. 2012, 45, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Noble, P.C.; Conditt, M.A.; Cook, K.F.; Mathis, K.B. Patient Expectations Affect Satisfaction with Total Knee Arthroplasty. Clin. Orthop. Relat. Res. 2006, 452, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Kolasinski, S.L.; Neogi, T.; Hochberg, M.C.; Oatis, C.; Guyatt, G.; Block, J.; Callahan, L.; Copenhaver, C.; Dodge, C.; Felson, D.; et al. 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Management of Osteoarthritis of the Hand, Hip, and Knee. Arthritis Rheumatol. 2020, 72, 220–233. [Google Scholar] [CrossRef]
- Marcolin, G.; Petrone, N.; Moro, T.; Battaglia, G.; Bianco, A.; Paoli, A. Selective Activation of Shoulder, Trunk, and Arm Muscles: A Comparative Analysis of Different Push-up Variants. J. Athl. Train. 2015, 50, 1126–1132. [Google Scholar] [CrossRef]
- Lyman, S.; Hidaka, C.; Fields, K.; Islam, W.; Mayman, D. Monitoring Patient Recovery after THA or TKA Using Mobile Technology. HSS J. 2020, 16, 358–365. [Google Scholar] [CrossRef]
- Constantinescu, D.; Pavlis, W.; Rizzo, M.; Berge, D.V.; Barnhill, S.; Hernandez, V.H. The Role of Commercially Available Smartphone Apps and Wearable Devices in Monitoring Patients after Total Knee Arthroplasty: A Systematic Review. EFORT Open Rev. 2022, 7, 481–490. [Google Scholar] [CrossRef]
| TKA Group (T0) (5 Males; 6 Females) (M ± SD) | TKA Group (T3) (4 Males; 5 Females) (M ± SD) | Control Group (5 Males; 6 Females) (M ± SD) | p-Value T0 vs. Control | p-Value T3 vs. Control | |
|---|---|---|---|---|---|
| Age (years) | 64.00 ± 5.00 | 64.33 ± 5.39 | 61.00 ± 4.67 | 0.161 | 0.155 |
| Height (meters) | 1.74 ± 0.11 | 1.74 ± 0.11 | 1.73 ± 0.09 | 0.832 | 0.828 |
| Mass (kilograms) | 94.00 ± 16.96 | 95.00 ± 16.36 | 81.91 ± 16.92 | 0.110 | 0.098 |
| Comorbidities | None | None | None | ||
| Number of evaluations | 1 | 4 | 1 |
| Clinical Tests | Time Point (Months) | Test Statistics | Polynomial Contrasts | |||
|---|---|---|---|---|---|---|
| T0 (pre) | T1 (3) | T2 (6) | T3 (12) | |||
| M | M | M | M | |||
| (SE) | (SE) | (SE) | (SE) | |||
| CWS (m/s) | 0.954 (0.089) | 0.940 (0.086) | 1.063 (0.090) | 1.080 (0.097) | F(3,24) = 3.889, p = 0.021, ηp2 = 0.327 | Linear: F(1,8) = 5.131, p = 0.053, ηp2 = 0.391 |
| 6 MWT (m) | 369.66 (24.63) | 362.89 (29.67) | 398.28 (23.01) | 414.06 (27.20) | F(3,24) = 5.126, p = 0.007, ηp2 = 0.391 | Linear: F(1,8) = 6.299, p = 0.036, ηp2 = 0.441 |
| TUGT (s) | 8.61 (0.62) | 8.97 (0.99) | 7.66 (0.78) | 7.42 (0.50) | F(3,24) = 2.394, p = 0.093, ηp2 = 0.230 | Linear: F(1,8) = 4.631, p = 0.064, ηp2 = 0.367 |
| Parameters | Time Point (Months) | Test Statistics | Polynomial Contrasts | |||
|---|---|---|---|---|---|---|
| T0 (pre) | T1 (3) | T2 (6) | T3 (12) | |||
| M | M | M | M | |||
| (SE) | (SE) | (SE) | (SE) | |||
| Cadence (steps per min) | 51.0 (1.20) | 51.0 (1.20) | 52.8 (1.80) | 54.6 (0.60) | F(3,24) = 1.658, p = 0.203, ηp2 = 0.172 | Linear: F(1,8) = 7.774, p = 0.024, ηp2 = 0.493 |
| Amplitude of angular excursion (degrees) | 46.91 (2.50) | 46.10 (2.88) | 49.05 (2.55) | 47.69 (3.53) | F(3,24) = 1.207, p = 0.328, ηp2 = 0.131 | Linear: F(1,8) = 0.572, p = 0.471, ηp2 = 0.067 |
| Sen | 0.22 (0.01) | 0.23 (0.01) | 0.25 (0.01) | 0.26 (0.01) | F(3,24) = 8.476, p < 0.001, ηp2 = 0.514 | Linear: F(1,8) = 40.394, p < 0.001, ηp2 = 0.835 |
| Peak frequency (Hz) | 1.27 (0.04) | 1.26 (0.03) | 1.20 (0.06) | 1.31 (0.04) | F(3,24) = 1.960, p = 0.147, ηp2 = 0.197 | Linear: F(1,8) = 0.288, p = 0.606, ηp2 = 0.035 |
| Parameters | TKA Leg | Non-TKA Leg | Test Statistics |
|---|---|---|---|
| M | M | ||
| (SE) | (SE) | ||
| Amplitude of angular excursion (degrees) | 47.27 (3.00) | 47.60 (2.49) | TKA: F(1,8) = 0.117, p = 0.742, ηp2 = 0.014 TKA×Time: F(3,24) = 2.482, p = 0.085, ηp2 = 0.237 |
| SEn | 0.22 (0.01) | 0.26 (0.02) | TKA: F(1,8) = 7.173, p = 0.028, ηp2 = 0.473 TKA×Time: F(3,24) = 1.016, p = 0.403, ηp2 = 0.113 |
| Peak Frequency (Hz) | 1.16 (0.06) | 1.36 (0.03) | TKA: F(1,8) = 11.356, p = 0.010, ηp2 = 0.587 TKA×Time: F(3,12.207) = 0.184, p = 0.777, ηp2 = 0.022 |
| Parameters | Thigh | Shank | Test Statistics |
|---|---|---|---|
| M | M | ||
| (SE) | (SE) | ||
| Amplitude of angular excursion (degrees) | 31.58 (2.14) | 63.29 (3.34) | Segment: F(1, 8) = 521.344, p < 0.001, ηp2 = 0.985 Segment×Time: F(3,24) = 3.938, p = 0.020, ηp2 = 0.330 Segment×TKA: F(1,8) = 0.329, p = 0.582, ηp2 = 0.040 Segment×TKA×Time: F(3,24) = 0.785, p = 0.514, ηp2 = 0.089 |
| SEn | 0.29 (0.02) | 0.19 (0.01) | Segment: F(1,8) = 20.743, p = 0.002, ηp2 = 0.722 Segment×Time: F(3,24) = 3.781, p = 0.024, ηp2 = 0.321 Segment×TKA: F(1,8) = 2.188, p = 0.177, ηp2 = 0.215 Segment×TKA×Time: F(3,24) = 1.342, p = 0.284, ηp2 = 0.144 |
| Peak Frequency (Hz) | 0.98 (0.01) | 1.54 (0.06) | Segment: F(1,8) = 91.086, p < 0.001, ηp2 = 0.919 Segment×Time: F(3,24) = 3.317, p = 0.037, ηp2 = 0.293 Segment×TKA: F(1,8) = 10.565, p = 0.012, ηp2 = 0.569 Segment×TKA×Time: F(3,24) = 0.865, p = 0.473, ηp2 = 0.098 |
| Parameters | ||||
|---|---|---|---|---|
| Amplitude of Angular | SEn | Peak Frequency (Hz) | ||
| Time Points | Segment | Excursion (Degrees) | ||
| (Months) | M (SE) | M (SE) | M (SE) | |
| T0 (pre) | Non-TKA thigh | 31.43 (2.34) | 0.28 (0.03) | 0.95 (0.02) |
| Non-TKA shank | 63.16 (3.20) | 0.20 (0.01) | 1.79 (0.05) | |
| TKA thigh | 29.66 (2.41) | 0.22 (0.02) | 0.95 (0.02) | |
| TKA shank | 63.37 (3.02) | 0.17 (0.01) | 1.39 (0.11) | |
| T1 (3) | Non-TKA thigh | 30.19 (2.09) | 0.31 (0.03) | 0.95 (0.03) |
| Non-TKA shank | 61.08 (3.42) | 0.19 (0.01) | 1.79 (0.04) | |
| TKA thigh | 31.24 (2.76) | 0.24 (0.02) | 0.95 (0.03) | |
| TKA shank | 61.85 (3.87) | 0.17 (0.01) | 1.33 (0.11) | |
| T2 (6) | Non-TKA thigh | 33.37 (1.86) | 0.33 (0.03) | 1.01 (0.03) |
| Non-TKA shank | 66.63 (3.05) | 0.20 (0.01) | 1.59 (0.12) | |
| TKA thigh | 30.56 (2.49) | 0.28 (0.03) | 0.98 (0.03) | |
| TKA shank | 65.66 (3.48) | 0.18 (0.004) | 1.21 (0.14) | |
| T3 (12) | Non-TKA thigh | 33.53 (2.67) | 0.32 (0.03) | 1.03 (0.02) |
| Non-TKA shank | 61.42 (4.41) | 0.21 (0.01) | 1.78 (0.06) | |
| TKA thigh | 32.64 (3.19) | 0.30 (0.03) | 1.01 (0.01) | |
| TKA shank | 63.16 (4.73) | 0.19 (0.01) | 1.43 (0.14) | |
| Controls | Right thigh | 40.21 (2.01) | 0.35 (0.04) | 1.05 (0.03) |
| Right shank | 76.56 (1.58) | 0.21 (0.01) | 1.74 (0.16) | |
| Left thigh | 38.69 (1.10) | 0.33 (0.04) | 1.05 (0.03) | |
| Left shank | 75.72 (1.70) | 0.22 (0.02) | 1.84 (0.14) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
van de Ven, W.A.F.; Bosga, J.; Hullegie, W.; Verra, W.C.; Meulenbroek, R.G.J. Inertial-Sensor-Based Monitoring of Sample Entropy and Peak Frequency Changes in Treadmill Walking during Recovery after Total Knee Arthroplasty. Sensors 2023, 23, 4968. https://doi.org/10.3390/s23104968
van de Ven WAF, Bosga J, Hullegie W, Verra WC, Meulenbroek RGJ. Inertial-Sensor-Based Monitoring of Sample Entropy and Peak Frequency Changes in Treadmill Walking during Recovery after Total Knee Arthroplasty. Sensors. 2023; 23(10):4968. https://doi.org/10.3390/s23104968
Chicago/Turabian Stylevan de Ven, Werner A. F., Jurjen Bosga, Wim Hullegie, Wiebe C. Verra, and Ruud G. J. Meulenbroek. 2023. "Inertial-Sensor-Based Monitoring of Sample Entropy and Peak Frequency Changes in Treadmill Walking during Recovery after Total Knee Arthroplasty" Sensors 23, no. 10: 4968. https://doi.org/10.3390/s23104968
APA Stylevan de Ven, W. A. F., Bosga, J., Hullegie, W., Verra, W. C., & Meulenbroek, R. G. J. (2023). Inertial-Sensor-Based Monitoring of Sample Entropy and Peak Frequency Changes in Treadmill Walking during Recovery after Total Knee Arthroplasty. Sensors, 23(10), 4968. https://doi.org/10.3390/s23104968





