Performance of Oral Cavity Sensors: A Systematic Review
Abstract
:1. Introduction
- What sensor technologies have been tested in the human mouth?
- Which fields of study develop sensor technologies for the mouth?
- What is the performance of these sensors in terms of accuracy and/or agreement?
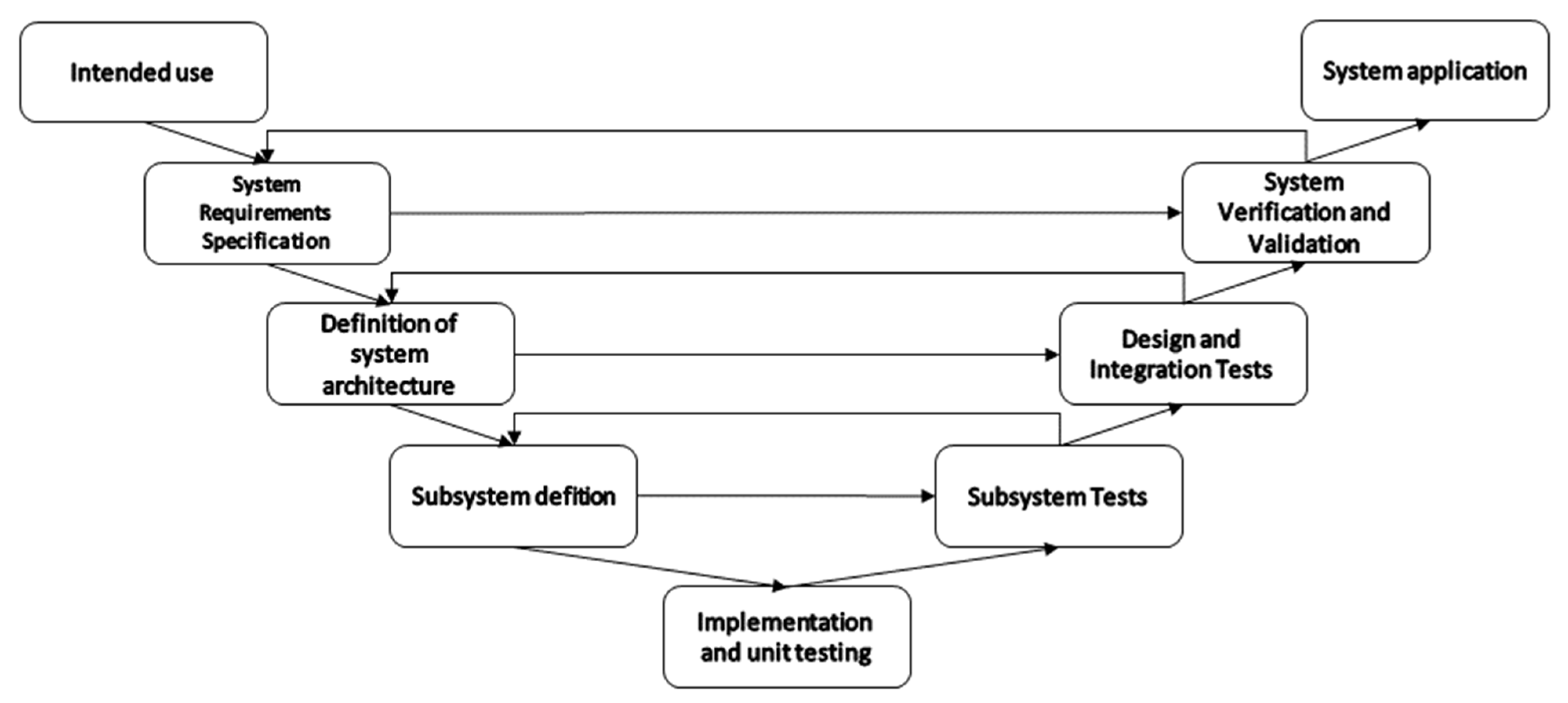
2. Methods
3. Results
3.1. Quality Assessment
3.2. Fields of Study
3.3. Sensor Technologies
3.4. Study Settings
3.5. Agreement and Accuracy
3.5.1. Dentistry
3.5.2. Neurology
Physical and Rehabilitation Medicine
Internal Medicine
Obstetrics
Aerospace Medicine
4. Discussion
4.1. Summary of Evidence
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Neuman, M.R. Physical Sensors for Biomedical Applications; CRC Press: Boca Raton, FL, USA, 2018; ISBN 9781351084086. [Google Scholar]
- Jones, D.P. Biomedical Sensors; Sensor Technology Series; Momentum Press: New York, NY, USA, 2010; ISBN 9781606500569. [Google Scholar]
- Paulovich, F.V.; de Oliveira, M.C.F.; Oliveira, O.N. A Future with Ubiquitous Sensing and Intelligent Systems. ACS Sens. 2018, 3, 1433–1438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mosca, A.C.; Chen, J. Food-Saliva Interactions: Mechanisms and Implications. Trends Food Sci. Technol. 2017, 66, 125–134. [Google Scholar] [CrossRef]
- Brodsky, L. Chapter 35—Structure and Development of the Upper Respiratory System in Infants and Children. In Pediatric Critical Care; Fuhrman, B.P., Zimmerman, J.J.B.T.-P.C.C., Eds.; Mosby: Saint Louis, MO, USA, 2011; pp. 485–489. ISBN 978-0-323-07307-3. [Google Scholar]
- McDowell, J. Encyclopedia of Human Body Systems; Greenwood: Westport, CT, USA, 2010; ISBN 9780313391750. [Google Scholar]
- Glandular Mechanisms of Salivary Secretion, 1st ed.; Garrett, J.R.; Ekström, J.; Anderson, L.C.; Sharpe, P.T. (Eds.) S. Karger Publishers: Basel, Switzerland, 1998; ISBN 3805566301. [Google Scholar]
- Olsen, I.; Singhrao, S.K.; Potempa, J. Citrullination as a Plausible Link to Periodontitis, Rheumatoid Arthritis, Atherosclerosis and Alzheimer’s Disease. J. Oral Microbiol. 2018, 10, 1487742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kharlamova, N.; Jiang, X.; Sherina, N.; Potempa, B.; Israelsson, L.; Quirke, A.-M.; Eriksson, K.; Yucel-Lindberg, T.; Venables, P.J.; Potempa, J.; et al. Antibodies to Porphyromonas Gingivalis Indicate Interaction Between Oral Infection, Smoking, and Risk Genes in Rheumatoid Arthritis Etiology. Arthritis Rheumatol. 2016, 68, 604–613. [Google Scholar] [CrossRef] [Green Version]
- Pfaffe, T.; Cooper-White, J.; Beyerlein, P.; Kostner, K.; Punyadeera, C. Diagnostic Potential of Saliva: Current State and Future Applications. Clin. Chem. 2011, 57, 675–687. [Google Scholar] [CrossRef] [Green Version]
- Malon, R.S.P.; Sadir, S.; Balakrishnan, M.; Córcoles, E.P. Saliva-Based Biosensors: Noninvasive Monitoring Tool for Clinical Diagnostics. Biomed. Res. Int. 2014, 2014, 1–20. [Google Scholar] [CrossRef] [Green Version]
- Windmiller, J.R.; Wang, J. Wearable Electrochemical Sensors and Biosensors: A Review. Electroanalysis 2013, 25, 29–46. [Google Scholar] [CrossRef]
- Aguirre, A.; Testa-Weintraub, L.A.; Banderas, J.A.; Haraszthy, G.G.; Reddy, M.S.; Levine, M.J. Sialochemistry: A Diagnostic Tool? Crit. Rev. Oral Biol. Med. 1993, 4, 343–350. [Google Scholar] [CrossRef]
- Yuk, H.; Lu, B.; Zhao, X. Hydrogel Bioelectronics. Chem. Soc. Rev. 2019, 48, 1642–1667. [Google Scholar] [CrossRef] [Green Version]
- Nishizawa, M. Soft, Wet and Ionic Microelectrode Systems. Bull. Chem. Soc. Jpn. 2018, 91, 1141–1149. [Google Scholar] [CrossRef]
- Jinno, H.; Fukuda, K.; Xu, X.; Park, S.; Suzuki, Y.; Koizumi, M.; Yokota, T.; Osaka, I.; Takimiya, K.; Someya, T. Stretchable and Waterproof Elastomer-Coated Organic Photovoltaics for Washable Electronic Textile Applications. Nat. Energy 2017, 2, 780–785. [Google Scholar] [CrossRef]
- Yuk, H.; Zhang, T.; Lin, S.; Parada, G.A.; Zhao, X. Tough Bonding of Hydrogels to Diverse Non-Porous Surfaces. Nat. Mater. 2016, 15, 190–196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitsubayashi, K.; Arakawa, T. Cavitas Sensors: Contact Lens Type Sensors & Mouthguard Sensors. Electroanalysis 2016, 28, 1170–1187. [Google Scholar] [CrossRef]
- Wang, J.; Yu, J.; Wang, T.; Li, C.; Wei, Y.; Deng, X.; Chen, X. Emerging Intraoral Biosensors. J. Mater. Chem. B 2020, 8, 3341–3356. [Google Scholar] [CrossRef] [PubMed]
- Clinical Evaluation of Medical Devices—Principles and Case Studies; Becker, K.M.; Whyte, J.J. (Eds.) Humana Press: Totowa, NJ, USA, 2006; ISBN 978-1-58829-422-7. [Google Scholar]
- North, B. The Growing Role of Human Factors and Usability Engineering for Medical Devices. What’s Required in the New Regulatory Landscape. Available online: http://www.bsigroup.com/LocalFiles/en-GB/Medical-devices/whitepapers/The growing role of human factors and usability engineering for medical devices.pdf (accessed on 16 April 2021).
- Branaghan, R.J.; O’Brian, J.S.; Hildebrand, E.A.; Foster, L.B. Humanizing Healthcare—Human Factors for Medical Device Design; Springer International Publishing: Cham, Switzerland, 2021; ISBN 978-3-030-64432-1. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Healthcare Interventions: Explanation and Elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef] [Green Version]
- Tang, W.; Hu, J.; Zhang, H.; Wu, P.; He, H. Kappa Coefficient: A Popular Measure of Rater Agreement. Shanghai Arch Psychiatry 2015, 27, 62–67. [Google Scholar] [CrossRef]
- Ranganathan, P.; Pramesh, C.; Aggarwal, R. Common Pitfalls in Statistical Analysis: Measures of Agreement. Perspect. Clin. Res. 2017, 8, 187–191. [Google Scholar] [CrossRef]
- Watson, P.F.; Petrie, A. Method Agreement Analysis: A Review of Correct Methodology. Theriogenology 2010, 73, 1167–1179. [Google Scholar] [CrossRef] [Green Version]
- Specialist Unit for Review Evidence Questions to Assist with the Critical Appraisal of Qualitative Studies. Available online: http://www.cardiff.ac.uk/specialist-unit-for-review-evidence/resources/critical-appraisal-%0Ahttp://www.cardiff.ac.uk/__data/assets/pdf_file/0006/212766/SURE_RCTs-and-other-experimental-studies_Checklist_2015-update.pdf (accessed on 14 March 2021).
- Whiting, P.F.; Weswood, M.E.; Rutjes, A.W.S.; Reitsma, J.B.; Bossuyt, P.N.M.; Kleijnen, J. Evaluation of QUADAS, a Tool for the Quality Assessment of Diagnostic Accuracy Studies. BMC Med. Res. Methodol. 2006, 6, 1–8. [Google Scholar] [CrossRef]
- WMA Declaration of Helsinki–Ethical Principles for Medical Research Involving Human Subjects. Available online: https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects/ (accessed on 16 April 2021).
- American Medical Association FREIDA’s Specialty Guide—American Medical Association. Available online: https://freida.ama-assn.org/specialty (accessed on 16 April 2021).
- Dental Specialties—American Dental Association. Available online: https://www.ada.org/en/ncrdscb/dental-specialties (accessed on 16 April 2021).
- Bonakdarchian, M.; Askari, N.; Askari, M. Effect of Face Form on Maximal Molar Bite Force with Natural Dentition. Arch. Oral Biol. 2009, 54, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Ferrario, V.F.; Sforza, C.; Serrao, G.; Dellavia, C.; Tartaglia, G.M. Single Tooth Bite Forces in Healthy Young Adults. J. Oral. Rehabil. 2004, 31, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Lear, C.S.C.; Catz, J.; Grossman, R.C.; Flanagan, J.B.; Moorrees, C.F.A. Measurement of Lateral Muscle Forces on the Dental Arches. Arch. Oral Biol. 1965, 10, 669–690. [Google Scholar] [CrossRef]
- Lundgren, D.; Laurell, L. Occlusal Forces in Prosthetically Restored Dentitions: A Methodological Study. J. Oral Rehabil. 1984, 11, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Shellhart, W.C.; Moawad, M.I.; Paterson, R.L.; Matheny, J. Lip Adaptation to Simulated Dental Arch Expansion. Part 1: Reliability and Precision of Two Lip Pressure Measurement Mechanisms. Angle Orthod. 1996, 66, 249–254. [Google Scholar] [PubMed]
- Gibbs, C.H.; Anusavice, K.J.; Young, H.M.; Jones, J.S.; Esquivel-Upshaw, J.F. Maximum Clenching Force of Patients with Moderate Loss of Posterior Tooth Support: A Pilot Study. J. Prosthet. Dent. 2002, 88, 498–502. [Google Scholar] [CrossRef]
- O’Hare, E.; Cogan, J.A.; Dillon, F.; Lowery, M.; O’Cearbhaill, E.D.; O’Hare, E.; Cogan, J.A.; Dillon, F.; Lowery, M.; O’Cearbhaill, E.D.; et al. An Intraoral Non-Occlusal MEMS Sensor for Bruxism Detection. IEEE Sens. J. 2022, 22, 153–161. [Google Scholar] [CrossRef]
- Ponukumati, A.S.; Wu, X.; Kahng, P.W.; Skinner, J.; Paydarfar, J.A.; Halter, R.J. A System for Characterizing Intraoperative Force Distribution during Operative Laryngoscopy. IEEE Trans. Biomed. Eng. 2020, 67, 2616–2627. [Google Scholar] [CrossRef]
- Mansour, R.M. Piezoelectric Transducers for Oral Force Monitoring. J. Med. Eng. Technol. 1977, 1, 95–97. [Google Scholar] [CrossRef]
- Searl, J.P. Comparison of Transducers and Intraoral Placement Options for Measuring Lingua-Palatal Contact Pressure during Speech. J. Speech Lang. Hear. Res. 2003, 46, 1444–1456. [Google Scholar] [CrossRef]
- Luffingham, J.K. A Technique for the Measurement of Soft Tissue Pressures Acting upon Teeth. Arch. Oral Biol. 1968, 13, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Manning, M.; Casey, V.; Conway, R.; Saunders, J.; Perry, A. A Study of Healthy Adults’ Oro-Lingual Effort During Swallowing Using OroPress, A New Portable Wireless Measurement Tool. Dysphagia 2016, 31, 442–451. [Google Scholar] [CrossRef] [PubMed]
- McCormack, J.; Casey, V.; Conway, R.; Saunders, J.; Perry, A. OroPress a New Wireless Tool for Measuring Oro-Lingual Pressures: A Pilot Study in Healthy Adults. J. Neuroeng. Rehabil. 2015, 12, 32. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, C.P.; Glantz, P.O.J.; Svensson, S.A.; Bergmark, A. A Novel Sensor for Bite Force Determinations. Dent. Mater. 2003, 19, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Levartovsky, S.; Peleg, G.; Matalon, S.; Tsesis, I.; Rosen, E. Maximal Bite Force Measured via Digital Bite Force Transducer in Subjects with or without Dental Implants—A Pilot Study. Appl. Sci. 2022, 12, 1544. [Google Scholar] [CrossRef]
- Khan, A.A.; McCreary, B.; Owatz, C.B.; Schindler, W.G.; Schwartz, S.A.; Keiser, K.; Hargreaves, K.M. The Development of a Diagnostic Instrument for the Measurement of Mechanical Allodynia. J. Endod. 2007, 33, 663–666. [Google Scholar] [CrossRef]
- Mcauliffe, P.; Kim, J.H.; Diamond, D.; Lau, K.T.; O’Connell, B.C. A Sleep Bruxism Detection System Based on Sensors in a Splint—Pilot Clinical Data. J. Oral Rehabil. 2015, 42, 34–39. [Google Scholar] [CrossRef] [Green Version]
- Iwasaki, M.; Maeda, I.; Kokubo, Y.; Tanaka, Y.; Ueno, T.; Ohara, Y.; Motokawa, K.; Hayakawa, M.; Shirobe, M.; Edahiro, A.; et al. Standard Values and Concurrent Validity of a Newly Developed Occlusal Force-Measuring Device among Community-Dwelling Older Adults: The Otassha Study. Int. J. Environ. Res. Public Health 2022, 19, 5588. [Google Scholar] [CrossRef]
- Janani, K.; Palanivelu, A.; Sandhya, R. Diagnostic Accuracy of Dental Pulse Oximeter with Customized Sensor Holder, Thermal Test and Electric Pulp Test for the Evaluation of Pulp Vitality: An In Vivo Study. Braz. Dent. Sci. 2020, 23. [Google Scholar] [CrossRef] [Green Version]
- Janani, K.; Ajitha, P.; Sandhya, R.; Subbaiyan, H.; Jose, J. Efficiency of New Custom-Made Pulse Oximeter Sensor Holder in Assessment of Actual Pulp Status. J. Fam. Med. Prim. Care 2020, 9, 3333–3337. [Google Scholar] [CrossRef]
- Pozzobon, M.H.; de Sousa Vieira, R.; Alves, A.M.H.; Reyes-Carmona, J.; Teixeira, C.S.; de Souza, B.D.M.; Felippe, W.T. Assessment of Pulp Blood Flow in Primary and Permanent Teeth Using Pulse Oximetry. Dent. Traumatol. 2011, 27, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Gopikrishna, V.; Tinagupta, K.; Kandaswamy, D. Evaluation of Efficacy of a New Custom-Made Pulse Oximeter Dental Probe in Comparison With the Electrical and Thermal Tests for Assessing Pulp Vitality. J. Endod. 2007, 33, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Soltanzadeh, R.; Moussavi, Z. Design an Oral Photoplethysmogram for Deriving Peripheral Oxygen Saturation Level. J. Med. Devices Trans. ASME 2015, 9, 020922. [Google Scholar] [CrossRef]
- Aziman, C.; Hellén-Halme, K.; Shi, X.-Q. A Comparative Study on Image Quality of Two Digital Intraoral Sensors. Dentomaxillofacial Radiol. 2019, 48, 20190063. [Google Scholar] [CrossRef] [PubMed]
- Dündar, A.; Çiftçi, M.E.; İşman, Ö.; Aktan, A.M. In Vivo Performance of Near-Infrared Light Transillumination for Dentine Proximal Caries Detection in Permanent Teeth. Saudi Dent. J. 2020, 32, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Taravati, S.; Balak, Z.; Rakhshan, V. Diagnostic Accuracy of Periapical Radiography in Detection and Measurement of the External Root Resorption in Primary Molars: A Single-Blind Prospective Clinical Study. Int. J. Dent. 2022, 2022, 1–10. [Google Scholar] [CrossRef]
- Elkhateeb, S.M.; Aloyouny, A.Y.; Omer, M.M.S.; Mansour, S.M. Analysis of Photostimulable Phosphor Image Plate Artifacts and Their Prevalence. World J. Clin. Cases 2022, 10, 437–447. [Google Scholar] [CrossRef]
- Moghadam, A.A.; Nemati, S.; Mirshafa, S.N.; Nikbin, A. Correlation between Digital Intra-Oral System with Pocket Sounding in Detection of Bone Defects. Ann. Dent. Spec. 2017, 5, 110–116. [Google Scholar]
- Natto, Z.S.; Olwi, A.; Abduljawad, F. A Comparison of the Horizontal and Vertical Bitewing Images in Detecting Approximal Caries and Interdental Bone Loss in Posterior Teeth: A Diagnostic Accuracy Randomized Cross over Clinical Trial. J. Dent. Sci. 2022, in press. [Google Scholar] [CrossRef]
- Amini, M.; Hisdal, J.; Gjøvaag, T.; Kapetanovic, N.; Strand, T.E.; Owe, J.O.; Hørthe, J.R.; Mirtaheri, P. Near-Infrared Spectra in Buccal Tissue as a Marker for Detection of Hypoxia. Aerosp. Med. Hum. Perform 2016, 87, 498–504. [Google Scholar] [CrossRef]
- Metzger, Z.; Colson, D.G.; Bown, P.; Weihard, T.; Baresel, I.; Nolting, T. Reflected Near-Infrared Light versus Bite-Wing Radiography for the Detection of Proximal Caries: A Multicenter Prospective Clinical Study Conducted in Private Practices. J. Dent. 2022, 116, 103861. [Google Scholar] [CrossRef] [PubMed]
- Tiba, M.H.; Awad, A.B.; Pennington, A.; Fung, C.M.; Napolitano, L.M.; Park, P.K.; Machado-Aranda, D.A.; Gunnerson, K.J.; Romfh, P.; Ward, K.R. Resonance Raman Spectroscopy Derived Tissue Hemoglobin Oxygen Saturation in Critically Ill and Injured Patients. Shock 2021, 56, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Bhowmik, A.; Ghosh, B.; Pal, M.; Paul, R.R.; Chatterjee, J.; Chakraborty, S. Portable, Handheld, and Affordable Blood Perfusion Imager for Screening of Subsurface Cancer in Resource-Limited Settings. Proc. Natl. Acad. Sci. USA 2022, 119, e2026201119. [Google Scholar] [CrossRef] [PubMed]
- Amin Tily, M.; Al-Nashash, H.; Mir, H. An Intraoral Camera for Supporting Assistive Devices. IEEE Sens. J. 2020, 21, 8553–8563. [Google Scholar] [CrossRef]
- Atieh, M.A.; Alsabeeha, N.H.M.; Payne, A.G.T.; de Silva, R.K.; Schwass, D.S.; Duncan, W.J. The Prognostic Accuracy of Resonance Frequency Analysis in Predicting Failure Risk of Immediately Restored Implants. Clin. Oral Implant. Res. 2014, 25, 29–35. [Google Scholar] [CrossRef]
- Herrero-Climent, M.; Albertini, M.; Rios-Santos, J.-V.; Lázaro-Calvo, P.; Fernández-Palacín, A.; Bullon, P. Resonance Frequency Analysis-Reliability in Third Generation Instruments: Osstell Mentor. Med. Oral Patol. Oral Cir. Bucal 2012, 17, e801–e806. [Google Scholar] [CrossRef] [Green Version]
- Jaramillo, R.; Santos, R.; Lázaro, P.; Romero, M.; Rios-Santos, J.V.; Bullón, P.; Fernández-Palacín, A.; Herrero-Climent, M. Comparative Analysis of 2 Resonance Frequency Measurement Devices: Osstell Mentor and Osstell ISQ. Implant Dent. 2014, 23, 351–356. [Google Scholar] [CrossRef] [Green Version]
- Herrero-Climent, M.; Santos-Garcia, R.; Jaramillo-Santos, R.; Romero-Ruiz, M.M.; Fernandez-Palacin, A.; Lazaro-Calvo, P.; Bullon, P.; Rios-Santos, J.V. Assessment of Osstell Isq’s Reliability for Implant Stability Measurement: A Cross-Sectional Clinical Study. Med. Oral Patol. Oral Cir. Bucal 2013, 18, e877–e882. [Google Scholar] [CrossRef]
- Bertl, M.H.; Weinberger, T.; Schwarz, K.; Gruber, R.; Crismani, A.G. Resonance Frequency Analysis: A New Diagnostic Tool for Dental Ankylosis. Eur. J. Oral Sci. 2012, 120, 255–258. [Google Scholar] [CrossRef]
- Yamada, Y.; Yoshida, N.; Kobayashi, K.; Yamauchi, K. An Application of Magnet and Magnetic Sensor: Measurement System for Tooth Movement. IEEE Trans. Biomed. Eng. 1990, 37, 919–924. [Google Scholar] [CrossRef]
- Yoshida, N.; Koga, Y.; Saimoto, A.; Ishimatsu, T.; Yamada, Y.; Kobayashi, K. Development of a Magnetic Sensing Device for Tooth Displacement under Orthodontic Forces. IEEE Trans. Biomed. Eng. 2001, 48, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, N.; Koga, Y.; Kobayashi, K.; Yamada, Y.; Yoneda, T. A New Method for Qualitative and Quantitative Evaluation of Tooth Displacement under the Application of Orthodontic Forces Using Magnetic Sensors. Med. Eng. Phys. 2000, 22, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Ghovanloo, M. An Arch-Shaped Intraoral Tongue Drive System with Built-in Tongue-Computer Interfacing SoC. Sensors 2014, 14, 21565–21587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woodford, S.C.; Robinson, D.L.; Edelmann, C.; Mehl, A.; Röhrle, O.; Vee Sin Lee, P.; Ackland, D.C. Low-Profile Electromagnetic Field Sensors in the Measurement and Modelling of Three-Dimensional Jaw Kinematics and Occlusal Loading. Ann. Biomed. Eng. 2021, 49, 1561–1571. [Google Scholar] [CrossRef]
- Kirshenblatt, S.; Chen, H.; Dieltjens, M.; Pliska, B.; Almeida, F.R. Accuracy of Thermosensitive Microsensors Intended to Monitor Patient Use of Removable Oral Appliances. J. Can. Dent. Assoc. 2018, 84, i2. [Google Scholar] [PubMed]
- de Almeida e Bueno, L.; Milnthorpe, W.; Bergmann, J.H.M. Determining the Performance of a Temperature Sensor Embedded into a Mouthguard. BDJ Open 2022, 8, 23. [Google Scholar] [CrossRef]
- Brierley, C.A.; Benson, P.E.; Sandler, J. How Accurate Are TheraMon® Microsensors at Measuring Intraoral Wear-Time? Recorded vs. Actual Wear Times in Five Volunteers. J. Orthod. 2017, 44, 241–248. [Google Scholar] [CrossRef]
- Gjerde, K.; Lehmann, S.; Naterstad, I.F.; Berge, M.E.; Johansson, A. Reliability of an Adherence Monitoring Sensor Embedded in an Oral Appliance Used for Treatment of Obstructive Sleep Apnoea. J. Oral. Rehabil. 2018, 45, 110–115. [Google Scholar] [CrossRef]
- Morán-Navarro, R.; Courel-Ibáñez, J.; Martínez-Cava, A.; Conesa-Ros, E.; Sánchez-Pay, A.; Mora-Rodriguez, R.; Pallarés, J.G. Validity of Skin, Oral and Tympanic Temperatures During Exercise in the Heat: Effects of Wind and Sweat. Ann. Biomed. Eng. 2019, 47, 317–331. [Google Scholar] [CrossRef]
- Hutton, S.; Probst, E.; Kenyon, C.; Morse, D.; Friedman, B.; Arnold, K.; Helsley, L. Accuracy of Different Temperature Devices in the Postpartum Population. JOGNN J. Obstet. Gynecol. Neonatal Nurs. 2009, 38, 42–49. [Google Scholar] [CrossRef]
- Park, S.; Kim, J.-H.; Lee, M.-K.; Ihm, J.-J.; Ahn, J.-S.; Park, J.-K.; Seo, D.-G. In Vivo Real-time Temperature Measurement on the Surface of Intact and Gold-restored Teeth during Consumption of Hot and Cold Drinks. Eur. J. Oral Sci. 2022, 130, e12870. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.S. Temperature Measurement in Critical Care Adults: A Comparison of Thermometry and Measurement Routes. Biol. Res. Nurs. 2004, 6, 117–125. [Google Scholar] [CrossRef]
- Gabler, L.F.; Huddleston, S.H.; Dau, N.Z.; Lessley, D.J.; Arbogast, K.B.; Thompson, X.; Resch, J.E.; Crandall, J.R. On-Field Performance of an Instrumented Mouthguard for Detecting Head Impacts in American Football. Ann. Biomed. Eng. 2020, 48, 2599–2612. [Google Scholar] [CrossRef] [PubMed]
- Kieffer, E.E.; Begonia, M.T.; Tyson, A.M.; Rowson, S. A Two-Phased Approach to Quantifying Head Impact Sensor Accuracy: In-Laboratory and On-Field Assessments. Ann. Biomed. Eng. 2020, 48, 2613–2625. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.C.; Nangia, V.; Bui, K.; Hammoor, B.; Kurt, M.; Hernandez, F.; Kuo, C.; Camarillo, D.B. In Vivo Evaluation of Wearable Head Impact Sensors. Ann. Biomed. Eng. 2016, 44, 1234–1245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rich, A.M.; Filben, T.M.; Miller, L.E.; Tomblin, B.T.; van Gorkom, A.R.; Hurst, M.A.; Barnard, R.T.; Kohn, D.S.; Urban, J.E.; Stitzel, J.D. Development, Validation and Pilot Field Deployment of a Custom Mouthpiece for Head Impact Measurement. Ann. Biomed. Eng. 2019, 47, 2109–2121. [Google Scholar] [CrossRef]
- Kuo, C.; Wu, L.; Loza, J.; Senif, D.; Anderson, S.C.; Camarillo, D.B. Comparison of Video-Based and Sensor-Based Head Impact Exposure. PLoS ONE 2018, 13, 1–20. [Google Scholar] [CrossRef] [Green Version]
- King, D.; Hume, P.A.; Brughelli, M.; Gissane, C. Instrumented Mouthguard Acceleration Analyses for Head Impacts in Amateur Rugby Union Players over a Season of Matches. Am. J. Sport. Med. 2015, 43, 614–624. [Google Scholar] [CrossRef]
- Goodin, P.; Gardner, A.J.; Dokani, N.; Nizette, B.; Ahmadizadeh, S.; Edwards, S.; Iverson, G.L. Development of a Machine-Learning-Based Classifier for the Identification of Head and Body Impacts in Elite Level Australian Rules Football Players. Front. Sport. Act. Living 2021, 3, 725245. [Google Scholar] [CrossRef]
- Fu, L.; Ling, C.; Jin, Z.; Luo, J.; Palma-Chavez, J.; Wu, Z.; Zhou, J.J.; Zhou, J.J.; Donovan, B.; Qi, B.; et al. Photoacoustic Imaging of Posterior Periodontal Pocket Using a Commercial Hockey-Stick Transducer. J. Biomed. Opt. 2022, 27, 056005. [Google Scholar] [CrossRef]
- Heppt, W.J.; Issing, W.J. Assessment of Tumorous Mandibular Involvement by Transcutaneous Ultrasound and Flexible Endosonography. J. Cranio-Maxillofac. Surg. 1993, 21, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Salmon, B.; le Denmat, D. Intraoral Ultrasonography: Development of a Specific High-Frequency Probe and Clinical Pilot Study. Clin. Oral Investig. 2012, 16, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Bertram, S.; Emshoff, R. Sonography of Periimplant Buccal Bone Defects in Periodontitis Patients: A Pilot Study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2008, 105, 99–103. [Google Scholar] [CrossRef]
- Yesuratnam, A.; Wiesenfeld, D.; Tsui, A.; Iseli, T.A.; Hoorn, S.V.; Ang, M.T.; Guiney, A.; Phal, P.M. Preoperative Evaluation of Oral Tongue Squamous Cell Carcinoma with Intraoral Ultrasound and Magnetic Resonance Imaging—Comparison with Histopathological Tumour Thickness and Accuracy in Guiding Patient Management. Int. J. Oral. Maxillofac. Surg. 2014, 43, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Costantino, T.G.; Satz, W.A.; Dehnkamp, W.; Goett, H. Randomized Trial Comparing Intraoral Ultrasound to Landmark-Based Needle Aspiration in Patients with Suspected Peritonsillar Abscess. Clin. Otolaryngol. 2013, 38, 244. [Google Scholar] [CrossRef]
- Caltenco, H.A.; Lontis, E.R.; Boudreau, S.A.; Bentsen, B.; Struijk, J.; Andreasen Struijk, L.N.S. Tip of the Tongue Selectivity and Motor Learning in the Palatal Area. IEEE Trans. Biomed. Eng. 2012, 59, 174–182. [Google Scholar] [CrossRef]
- Caltenco, H.A.; Lontis, E.R.; Bentsen, B.; Andreasen Struijk, L.N.S. The Impact of Function Location on Typing and Pointing Tasks With an Intraoral Tongue-Computer Interface. Int. J. Hum. Comput. Interact. 2014, 30, 267–277. [Google Scholar] [CrossRef]
- Mohammadi, M.; Knoche, H.; Gaihede, M.; Bentsen, B.; Andreasen Struijk, L.N.S. A High-Resolution Tongue-Based Joystick to Enable Robot Control for Individuals with Severe Disabilities. IEEE Int. Conf. Rehabil. Robot. 2019, 2019, 1043–1048. [Google Scholar] [CrossRef]
- Andreasen Struijk, L.N.S.; Bentsen, B.; Gaihede, M.; Lontis, E.R. Error-Free Text Typing Performance of an Inductive Intra-Oral Tongue Computer Interface for Severely Disabled Individuals. IEEE Trans. Neural Syst. Rehabil. Eng. 2017, 25, 2094–2104. [Google Scholar] [CrossRef]
- Luo, X.; Gutierrez Pulido, H.V.; Rutkove, S.; Sanchez, B. A Bioimpedance-Based Device to Assess the Volume Conduction Properties of the Tongue in Neurological Disorders Affecting Bulbar Function. IEEE Open J. Eng. Med. Biol. 2021, 2, 278–285. [Google Scholar] [CrossRef]
- Menon, D.; Mansfield, P.; Cordice, D.; Studer, C.; O’Leary, M.; Sheean, G.; Bril, V. Pilot Study of a Novel Transmembranous Electromyography Device for Assessment of Oral Cavity and Oropharyngeal Muscles. Muscle Nerve 2022, 65, 303–310. [Google Scholar] [CrossRef]
- Jiang, B.; Kim, J.; Park, H. Palatal Electrotactile Display Outperforms Visual Display in Tongue Motor Learning. IEEE Trans. Neural Syst. Rehabil. Eng. 2022, 30, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Castillo, Y.; Blanco-Almazan, D.; Whitney, J.; Mersky, B.; Jane, R. Characterization of a Tooth Microphone Coupled to an Oral Appliance Device: A New System for Monitoring OSA Patients. In Proceedings of the 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Jeju Island, Korea, 11–15 July 2017; Volume 2017, pp. 1543–1546. [Google Scholar]
- de Almeida e Bueno, L.; Kwong, M.T.; Milnthorpe, W.R.F.; Cheng, R.; Bergmann, J.H.M. Applying Ubiquitous Sensing to Estimate Perceived Exertion Based on Cardiorespiratory Features. Sport. Eng. 2021, 24, 9. [Google Scholar] [CrossRef]
- Watanabe, T.; Kobayashi, K.; Suzuki, T.; Oizumi, M.; Clark, G.T. A Preliminary Report on Continuous Recording of Salivary PH Using Telemetry in an Edentulous Patient. Int. J. Prosthodont. 1999, 12, 313–317. [Google Scholar] [PubMed]
- Makary, C.; Rebaudi, A.; Sammartino, G.; Naaman, N. Implant Primary Stability Determined by Resonance Frequency Analysis: Correlation With Insertion Torque, Histologic Bone Volume, and Torsional Stability at 6 Weeks. Implant Dent. 2012, 21, 474–480. [Google Scholar] [CrossRef]
- Patil, S.R.; Maragathavalli, G.; Ramesh, D.N.S.V.; Naidu, G.S.; Alam, M.K.; AlZoubi, I.A. The Reliability of a New Device for Measuring the Maximum Bite Force. Biomed. Res. Int. 2022, 2022, 1–6. [Google Scholar] [CrossRef]
- Kelsey, C.C.; Reid, F.D.; Coplowitz, J.A. A Method of Measuring Pressures against Tissues Supporting Functioning Complete Dentures. J. Prosthet. Dent. 1976, 35, 376–383. [Google Scholar] [CrossRef]
- Mokhtar, E.A.; Rattan, V.; Rai, S.; Jolly, S.S.; Lal, V. Analysis of Maximum Bite Force and Chewing Efficiency in Unilateral Temporomandibular Joint Ankylosis Cases Treated with Buccal Fat Pad Interpositional Arthroplasty. Br. J. Oral Maxillofac. Surg. 2022, 60, 313–319. [Google Scholar] [CrossRef]
- Castroflorio, T.; Icardi, K.; Becchino, B.; Merlo, E.; Debernardi, C.; Bracco, P.; Farina, D. Reproducibility of Surface EMG Variables in Isometric Sub-Maximal Contractions of Jaw Elevator Muscles. J. Electromyogr. Kinesiol. 2006, 16, 498–505. [Google Scholar] [CrossRef]
- Mountain, G.; Wood, D.; Toumba, J. Bite Force Measurement in Children with Primary Dentition. Int. J. Paediatr. Dent. 2011, 21, 112–118. [Google Scholar] [CrossRef]
- Ogata, K.; Nishigawa, G.; Aoki, T.; Maeda, Y.; Okuno, Y. Lateral Forces Exerted on the Abutment Tooth of Complete Mandibular Overdentures. J. Oral Rehabil. 1988, 15, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Luffingham, J.K. Pressure Exerted on Teeth by the Lips and Cheeks. D. Pract. 1968, 19, 61–64. [Google Scholar] [CrossRef]
- Hadley, A.J.; Krival, K.R.; Ridgel, A.L.; Hahn, E.C.; Tyler, D.J. Neural Network Pattern Recognition of Lingual–Palatal Pressure for Automated Detection of Swallow. Dysphagia 2015, 30, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Konaka, K.; Kondo, J.; Hirota, N.; Tamine, K.; Hori, K.; Ono, T.; Maeda, Y.; Sakoda, S.; Naritomi, H. Relationship between Tongue Pressure and Dysphagia in Stroke Patients. Eur. Neurol. 2010, 64, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Martinez, G.; Ventura, L.; Cugusi, L.; Cadeddu, F.; Limoncino, M.; Deriu, F.; Monticone, M.; Manca, A. Healthy Women and Men Do Not Show Differences in Tongue Strength and Regular Effort Saliva Swallows as Assessed by Piezo-Resistive Sensors: Results from a Reproducibility Study. Dysphagia 2022, 37, 1217–1225. [Google Scholar] [CrossRef]
- Waltzman, D.; Sarmiento, K.; Devine, O.; Zhang, X.; DePadilla, L.; Kresnow, M.; Borradaile, K.; Hurwitz, A.; Jones, D.; Goyal, R.; et al. Head Impact Exposures Among Youth Tackle and Flag American Football Athletes. Sport. Health A Multidiscip. Approach 2021, 13, 454–462. [Google Scholar] [CrossRef]
- Kitagawa, H.; Scheetz, J.P.; Farman, A.G. Comparison of Complementary Metal Oxide Semiconductor and Charge-Coupled Device Intraoral X-Ray Detectors Using Subjective Image Quality. Dentomaxillofac. Radiol. 2003, 32, 408–411. [Google Scholar] [CrossRef]
- van Assche, N.; Jacobs, R.; Coucke, W.; van Steenberghe, D.; Quirynen, M. Radiographic Detection of Artificial Intra-Bony Defects in the Edentulous Area. Clin. Oral. Implant. Res. 2009, 20, 273–279. [Google Scholar] [CrossRef]
- Vandenberghe, B.; Jacobs, R.; Yang, J. Diagnostic Validity (or Acuity) of 2D CCD versus 3D CBCT-Images for Assessing Periodontal Breakdown. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2007, 104, 395–401. [Google Scholar] [CrossRef]
- Aps, J.K.M.; Lim, L.Z.; Tong, H.J.; Kalia, B.; Chou, A.M. Diagnostic Efficacy of and Indications for Intraoral Radiographs in Pediatric Dentistry: A Systematic Review. Eur. Arch. Paediatr. Dent. 2020, 21, 429–462. [Google Scholar] [CrossRef]
- Honda, E.; Prince, J.L.; Fontanella, V.R.C. State-of-the-Art Digital Imaging in Dentistry: Advanced Research of MRI, CT, CBCT, and Digital Intraoral Imaging. Biomed. Res. Int. 2018, 2018, 1. [Google Scholar] [CrossRef] [PubMed]
- McHugh, M.; McCaffery, F.; Casey, V. Barriers to Adopting Agile Practices When Developing Medical Device Software. In International Conference on Software Process Improvement and Capability Determination, Palma de Mallorca, 29–31 May 2012; Springer: Berlin/Heidelberg, Germany, 2012; pp. 141–147. ISBN 9783642304385. [Google Scholar]
- Khan, A.A.; Akram, M.U.; Salam, A.A.; Butt, W.H. An Enhanced Agile-V Model for System Engineering in Complex Medical Device Development. In Proceedings of the 2022 2nd International Conference on Digital Futures and Transformative Technologies (ICoDT2), Islamabad, Pakistan, 24 May 2022; pp. 1–6. [Google Scholar]



| Item | Description | Papers (#/%) |
|---|---|---|
| 1 | Does the study address a clearly focused question? | 72/88% |
| 2 | Do the authors discuss how they decided which method to use? | 68/83% |
| 3 | Is there sufficient detail on the methods used? | 69/84% |
| 4 | Is the method of data collection well described? | 68/83% |
| 5 | Was there a comparison with an appropriate reference standard? | 46/56% |
| 6 | Are the results presented in a complete and comprehensible way? | 67/82% |
| 7 | Are we sure about the results, consequences and cost of alternatives performed? | 66/80% |
| 8 | Can the results be applied to the population of interest? | 70/85% |
| 9 | Can the method be applied to the population of interest? | 79/96% |
| 10 | Are the explanations for the results plausible and coherent? | 75/91% |
| 11 | Are the results of the study compared with those from other studies? | 71/87% |
| 12 | Did the authors identify any limitations? | 63/77% |
| 13 | Was ethical approval sought? | 59/72% |
| Force/Pressure Sensors | ||||
|---|---|---|---|---|
| Technology | Papers (#/%) | Applications | Performance Metrics | Technical Considerations |
| Strain gauge-based | 13/15% | Measuring bite force, mechanical pain threshold, lateral forces on teeth, oro-lingual pressures, chewing efficiency, detection of swallow | Calibration error, measurement error, accuracy, true positive rate and false positive rate, intraclass correlation coefficient, kappa analysis | Readings affected by sensors size [33,34], encapsulation [34], response time [35], measurement direction [35,36], quantity [36], and placement [36], also by oral temperature variation [33,37] and subjects’ anatomy [38] |
| Piezoelectrical | 4/5% | Measuring bite force, forces between a laryngoscope and soft tissue during surgery, oro-lingual pressures, diagnose sleep bruxism | Accuracy, F1-score, sensitivity, specificity, positive predictability, negative predictability, calibration error, dynamic validation error, measurement drift error, standard error of measurement, minimal detectable change, intraclass correlation coefficient | Readings affected by sensor size [39], output variation [40], measuring direction [41], quantity, placement [40,42] and loading hysteresis [40] as also by oral temperature variation [42] and subjects’ anatomy [40] |
| Diaphragm | 5/6% | Measuring bite force, lateral forces on teeth, oro-lingual pressures | Calibration error, intraclass correlation coefficient, method error, Cronbach’s alpha | Readings affected by sensor connections [43], placement [42], and size [43], as also by oral temperature variation [37,42,43]. Sensors affect subjects [42,43]. Sensors are adequate to measure soft tissue contact and bolus pressure [44,45] |
| Force sensing resistor | 2/2% | Measuring bite force, diagnose dysphagia in stroke patients | Intraclass correlation coefficient, sensitivity, specificity | Readings affected by non-linearity and load rate-dependent properties of sensor [46] |
| Hydraulic | 2/2% | Measuring bite force | Intraclass correlation coefficient | The bite element is soft, improving safety and comfort for recording bite force [47,48] |
| Polymer composite | 1/1% | Diagnose sleep bruxism | Sensitivity, specificity | Sensor size and oral liquids are key design factors. Sensor’s polymer hysteresis and sensitivity limit measurement of actual force [49] |
| Capacitive | 1/1% | Classify oral hypofunction | Sensitivity, specificity | Sensor thickness is a key design factor [50] |
| Optical/Image Sensors | ||||
| Photoplethysmography | 5/6% | Measuring blood SpO2, diagnose dental pulp vitality | Accuracy, sensitivity, specificity, negative predictive value, positive predictive value. | Readings affected by background absorption due to venous blood and tissue constituents [51], sensor type and holding probe [51,52,53,54,55], as well as age, gender, and general health of the patient [52] |
| X-ray radiography | 6/7% | Assess dental health, measure external root resorption, diagnose alveolar bone defect and caries | Percentage error, absolute error, kappa analysis, Cronbach’s alpha, intraclass correlation coefficient | Readings can be affected by sensor technology [56,57], configuration [58,59], size [57,60], and positioning [61]. Examiner experience [61], and definition of diagnostically acceptable images [56] and absolute ground truth [58] can bias results |
| Near-infrared spectroscopy | 3/4% | Measuring blood SpO2, diagnose caries | Root mean squared error | Readings affected by probe fixation [62], motion artefacts [62] and saliva accumulation [62]. Definition of diagnostically acceptable images and absolute ground truth [63], examiner experience [57], and access to intact proximal surfaces [63] can bias results |
| Resonance Raman spectroscopy | 1/1% | Measuring tissue blood SpO2 | Clarke Error Grid, kappa analysis | Readings affected by probe fixation, motion artefacts and saliva accumulation. Comparison with diagnostically acceptable ground truth is difficult [64] |
| Infrared camera | 1/1% | Diagnose subsurface cancer | Accuracy | Readings affected by temperature, moist, airflow and sensor stability [65] |
| Endoscope camera | 1/1% | Human–computer interface for severely paralyzed individuals | Percentage error | Readings affected by environmental light intensity, system cables and size [66] |
| Magnetic Sensors | ||||
| Mechanical resonance analysis | 5/6% | Measuring implant stability, predict failure of implants, diagnose dental ankylosis | Intraclass correlation coefficient, sensitivity, specificity | Readings affected by anatomical site [67], implant system [67,68,69,70], time between assessments [67], and dental damage [71] |
| Magnetic field sensors | 5/6% | Human–computer interface for severely paralyzed individuals, measuring chewing force, and tooth displacement. | Accuracy, root mean squared error, calibration error | Readings affected by head movement [72], shapes of magnets [72], arrangement of sensors [72,73,74,75], tooth contact [72], oral temperature variation [72,73], face anatomy [72], and kinematics measurement [76] |
| Temperature sensors | ||||
| Silicon-based | 4/5% | Measuring oral appliance use time, intraoral temperature | Intraclass correlation coefficient and Bland–Altman analysis | Readings affected by embedding material [77,78], sensor dimensions and placement [77], measuring setting [77,78,79,80], oral temperature variation [77,78,79,80], air flow [77,78,79,80], humidity [77,78,79,80], and time to steady-state reading [78]. |
| Thermocouple | 3/4% | Measuring body temperature, temperature changes in teeth | Bland–Altman analysis and mean error, calibration error | Readings affected by application setting [81,82,83], air flow [81,82], oral temperature variation [81,82,83], humidity, and sensor size [83] |
| Thermistor | 2/2% | Measuring body temperature | Bland–Altman analysis | Readings affected by application setting [81], air flow [81,82], oral temperature variation [81,82], and sensor encapsulation [84] |
| Heat-sensitive plastic | 1/1% | Measuring body temperature | Bland–Altman analysis | Readings affected by application setting, oral temperature variation [82] |
| Inertial Sensors | ||||
| MEMS accelerometer and gyroscope | 8/9% | Identify and measure head impacts | Accuracy, Sensitivity, specificity, true positive rate, true negative rate, F1 score, concordance correlation coefficient, positive predictive rate, per cent and normalized root mean squared error of bench test. | Readings affected by sensing threshold [85,86], mandible movements and variations [85,87,88,89], saliva [90], and remotion of mouthguards from the mouth [86]. Video validation is time-consuming and biased by reviewers [85,86,88,91], camera angles [91], image quality [89,91] and sports dynamics [88,90,91] |
| Sonography | ||||
| Endosonography | 6/7% | Assess cancer invasion, measure cancer tumour thickness, assess oral soft tissue, bone loss, abscesses, and cellulitis | Sensitivity, specificity, predictive value, Bland–Altman analysis and positive predictive value, intraclass correlation coefficient, method error, accuracy, kappa analysis | Readings affected by sensor size [92], field of exploration [93,94,95,96] and frequency [93]. Clinicians’ experience can bias results [95,97] |
| Inductive sensor | ||||
| Inductance resonating sensor | 4/5% | Human–computer interface for severely paralyzed individuals | Percentage error, accuracy, root mean squared error. | Readings affected by connections [98], coil pads shape [98], coil pad layout [98,99,100], activation method [101], involuntary activations [99], oral temperature variation [99] |
| Bioelectrical sensors | ||||
| Bioimpedance | 1/1% | Assess the conduction properties of the tongue | Accuracy | Readings affected by electrode polarization impedance. Needs validation as a diagnostic biomarker [102] |
| Electromyography | 1/1% | Assess oral-cavity and oropharyngeal muscles for neuromuscular pathology | Kappa analysis | Readings affected by access to subcutaneous muscles, movement, and comfort [103] |
| Electro tactile | 1/1% | Rehabilitation of tongue motor control | Error score | Readings affected by sensor size and placement, tongue impedance and patient and sensor sensitivity to stimulation [104] |
| Microphone | ||||
| MEMS | 2/2% | Diagnose obstructive sleep apnea, estimate perceived exertion | True positives, false positives, false negatives, root-mean-squared error, normalised root-mean-squared error | Intraoral breathing audio may have a higher signal-to-noise ratio than tracheal sensors [105]. Coughing, speech, movement noise and spurious breathing events need to be filtered out [106]. |
| Biochemical sensor | ||||
| pH sensor | 1/1% | Measuring salivary pH | Calibration error | Long-term measurement is challenging [107] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Almeida e Bueno, L.; Kwong, M.T.; Bergmann, J.H.M. Performance of Oral Cavity Sensors: A Systematic Review. Sensors 2023, 23, 588. https://doi.org/10.3390/s23020588
de Almeida e Bueno L, Kwong MT, Bergmann JHM. Performance of Oral Cavity Sensors: A Systematic Review. Sensors. 2023; 23(2):588. https://doi.org/10.3390/s23020588
Chicago/Turabian Stylede Almeida e Bueno, Leonardo, Man Ting Kwong, and Jeroen H. M. Bergmann. 2023. "Performance of Oral Cavity Sensors: A Systematic Review" Sensors 23, no. 2: 588. https://doi.org/10.3390/s23020588
APA Stylede Almeida e Bueno, L., Kwong, M. T., & Bergmann, J. H. M. (2023). Performance of Oral Cavity Sensors: A Systematic Review. Sensors, 23(2), 588. https://doi.org/10.3390/s23020588






